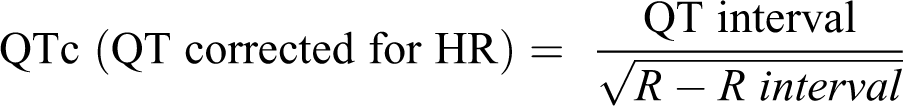Abstract
Arsenic trioxide (As2O3) is utilized for treating patients suffering from hematological malignancies particularly acute promyelocytic leukemia. Unfortunately, the extensive application of this chemotherapeutic agent has been limited due to its adverse effects such as cardiotoxicity. Ellagic acid, as a phenolic compound, has shown to exert antioxidant, anti-inflammatory, antifibrotic, and antiatherogenic properties. It is also capable of protecting against drug toxicity. In this study, we evaluated whether ellagic acid can protect against As2O3-induced heart injury in rats. Thirty-two male Wistar rats were randomly divided into four treatment groups, that is, control (0.2 mL of normal saline, intraperitoneally (ip)), As2O3 (5 mg/kg, ip), As2O3 plus ellagic acid, and ellagic acid (30 mg/kg, orally) groups. The drugs were administered daily for 10 days and pretreatment with ellagic acid was performed 1 h prior to As2O3 injection. Cardiotoxicity was characterized by electrocardiological, biochemical, and histopathological evaluations. Our results showed that ellagic acid pretreatment significantly ameliorated As2O3-induced increase in glutathione peroxidase activity and malondialdehyde concentration (p < 0.05 and p < 0.001, respectively) and also diminished QTc prolongation (p < 0.0001) and cardiac tissue damages. Pretreatment with ellagic acid also lowered the increased troponin I (p < 0.0001) and creatine kinase isoenzyme MB (p < 0.01) levels in response to As2O3. In conclusion, results of this study demonstrated that ellagic acid has beneficial cardioprotective effects against As2O3 toxicity. It is suggested that the protective effects were mediated by antioxidant properties of ellagic acid.
Introduction
Arsenic has been utilized as an effective drug in the treatment of diseases such as syphilis, tuberculosis, malarial fever, and sleeping sickness in ancient China for about 2500 years. 1
In 2000, arsenic trioxide (As2O3) was approved by the US Food and Drug Administration for the management of patients suffering from acute promyelocytic leukemia (APL). 2 As2O3 has also been demonstrated to be effective in preclinical and clinical studies in the treatment of a wide spectrum of hematological neoplasia such as acute myelogenous leukemia and multiple myeloma as well as some solid tumors such as esophageal and ovarian carcinomas. 2
Unfortunately, the extensive use of this drug has been confined due to its cardiotoxicity, including QTc prolongation, torsades de pointes, endothelial dysfunction, and sudden death. 1,3,4 The main mechanism of pathological adverse effects of arsenic results from oxidative stress and generation of reactive oxygen species (ROS) such as superoxide anion, hydrogen peroxide, and hydroxyl and peroxyl radicals. 3,5 Oxidative stress causes harmful alterations in critical macromolecules such as DNA, proteins, and lipids. 6,7 In addition, ROS induce alterations in nuclear factor kappa B (NF-κB), nuclear factor erythroid 2-related factor 2 (Nrf2), and activator protein 1, which are important transcription factors involved in intracellular antioxidant defense. 3,8,9
Natural products with antioxidant properties have shown beneficial effects against the oxidative damages induced by As2O3. 10 Ellagic acid (2,3,7,8-tetrahydroxy-cheromeno [5,4,3-cde]chromene-5,10-dione), as a phenolic compound, is found in blackberries, raspberries, strawberries, cranberries, grapes, pomegranate, and walnuts. 11,12 It has been reported to exhibit multiple pharmacological properties such as antibacterial, antiviral, anti-inflammatory, antifibrotic, antiatherogenic, antimutagenic, and immune regulatory. It is also considered as a potent antioxidant agent. 11,13,14
The aim of the current study was to evaluate the possible protective effect of ellagic acid in As2O3-induced cardiotoxicity.
Methods
Chemicals
Arsenic (III) oxide (As2O3, MW: 197.84) with trace metal basis purity of 99.99% and ellagic acid (C14H6O8, MW: 302.19) with high-performance liquid chromatography (HPLC) purity of ≥95% were purchased from Sigma-Aldrich Chemical Co. (St. Louis, Missouri, USA). Hematoxylin and eosin were also obtained from Sigma-Aldrich. Ketamine and xylazine were provided from Alfasan Co. (Woderen, Holland).
Experimental animals
All the experiments were carried out on 32 male albino Wistar rats weighing from 150 to 200 g purchased from animal house of Ahvaz Jundishapur University of Medical Sciences. The animals were housed under normal laboratory condition of humidity (50%), temperature (22°C ± 2°C) and a 12-h light/dark cycle, and allowed free access to standard rodent chow and water. The procedure was approved by Local Committee of Animal Ethics in Ahvaz Jundishapur University of Medical Sciences (No. IR. AJUMS. REC. 1395.104).
Experimental design
Thirty-two male rats were randomly divided into four treatment groups. Group I: normal control rats were intraperitoneally injected with normal saline daily for a period of 10 days. Group II: rats were intraperitoneally injected with As2O3 (5 mg/kg) daily for a period of 10 days. Group III: rats pretreated with ellagic acid (30 mg/kg) orally by gastric intubation 1 h prior to As2O3 (5 mg/kg) intraperitoneal administration daily for a period of 10 days. Group IV: rats were treated with ellagic acid (30 mg/kg) orally by gastric intubation daily for a period of 10 days. The applied doses and duration of treatments were determined based on previous studies regarding cardiotoxic and cardioprotective effects of As2O3 and ellagic acid, respectively. 3,15 LD50 value of As2O3 in rat was reported to be 14.98 mg/kg. 16
Electrocardiogram recording
At the beginning of the experiment, before any treatment and 48 h after the last dose (12th day), all rats were anesthetized by ketamine HCl (100 mg/kg, i.p.) and xylazine (5 mg/kg, i.p.). Fifteen minutes after anesthesia, standard bipolar limb lead II electrocardiogram (ECG) was recorded in order to measure QT interval. The QT intervals are affected by heart rate (R–R cycle length). Therefore, heart rate modification is required in the analysis of repolarization duration. Bazett’s formula is applied for correcting the QT interval in the ECG for heart rate (R–R interval). 17
Bazett’s formula:
Lead II ECG was recorded by Bio-Amp and monitored by a Power Lab system (ADInstruments, Sydney, Australia).
Biochemical measurements
After recording the ECG, animals were sacrificed under mild anesthesia with ketamine and xylazine and blood samples were obtained by heart puncture. Serums were separated from the blood samples and used for the biochemical measurements. After the blood sampling, hearts were dissected out and excised in ice-cold media. Then they were weighed and 100 mg of heart tissues were homogenized using homogenizer (Heidolph Silent Crusher M, Germany) in 1 mL phosphate-buffered saline (PBS) (50 mM, pH = 7.4) and centrifuged at 4000 rpm for 10 min. The clear supernatant was collected and used for the determination of glutathione peroxidase (GPx) activity and malondialdehyde (MDA) concentration.
Measurement of cardiac biomarkers
Cardiac troponin-I levels in serum were measured by an immunodiagnostic kit purchased from Monobind Inc. (Lake Forest, California, USA). Creatine kinase isoenzyme MB (CK-MB) in the serum was assayed by a standard diagnostic kit from Pars Azmun Co. (Iran). Assays were performed following the kit instructions.
GPx activity
A GPx activity assay kit from ZellBio GmbH (Germany) was used to measure GPx. GPx in the tissue homogenate uses glutathione (GSH) as an electron donor to convert it to oxidized glutathione (GSSG) and the remaining GSH can be reduced to generate a yellow color by reducing 5,5′-dithio-bis-[2-nitrobenzoic acid] (DTNB). In brief, 125 µL of tissue homogenate samples and 40 µL GSH solutions (2 mM) were added to microplate wells. Following 5-min incubation at 37°C, 10 µL of each reaction mixture were mixed with 200 µL DTNB reagent. The GPx activity of the samples is indirectly proportional to color formation of this reaction at 412 nm.
Glutathione assay
The concentrations of GSH in cardiac tissue homogenates were assayed using a standard diagnostic kit from ZellBio GmbH. The assay is based on the glutathione and DTNB reaction. Ten microliters of prepared samples were transferred to microplate wells and 200 µL of DTNB reagent was added to the wells. After 5-min incubation, absorbance of reaction mixtures was read at 412 nm.
Lipid peroxidation assay
MDA levels were assessed as a marker of lipid peroxidation in biological samples using a diagnostic kit from ZellBio GmbH. In this method, 50 µL of the tissue homogenate samples were mixed with 50 µL phosphoric acid reagent and then following the vortex mixing, 1 mL of thiobarbituric acid (TBA) reagent was added to the samples. The samples were heated in boiling water for 1 h and chilled on ice. Test tubes containing the supernatant were centrifuged at 4000 rpm for 10 min and the absorbance was measured spectrophotometrically at 532 nm and the concentration of MDA was quantified according to the prepared calibration curve using MDA standards.
Histopathology analysis
Heart tissue samples were fixed in formalin solution (10%) for 24 h at 37°C. Then samples were embedded in paraffin and tissue block sections were prepared. Following staining by hematoxylin and eosin, the sections of 5 µm thickness were examined under the light microscope with 10×, 40×, and 100× magnification to observe the histopathological changes.
Statistical analysis
All results were presented as the mean ± standard error of the mean. One-way analysis of variance was utilized to analyze the differences between groups followed by Tukey’s post hoc test for multiple comparisons. p-Values <0.05 were considered statistically significant. Data were analyzed using SPSS software version 22.
Results
ECG results
At the beginning of the study before drug administrations, normal ECG patterns and similar QTc intervals were observed in the rats of all groups. Forty-eight hours after the last dose of treatments, normal control and ellagic acid-treated rats showed a normal ECG pattern, whereas As2O3-treated rats showed a significant (p < 0.0001) increase in QTc interval as compared to the normal control group. As shown in Figure 1, oral pretreatment with ellagic acid (30 mg/kg) prior to As2O3 injection resulted in a significant (p < 0.0001) decrease in QTc interval when compared to the As2O3-treated group.

Values of QTc intervals of rats based on electrocardiogram analysis. Values were recorded before and 48 h after the last dose of treatments (n = 8). Data are expressed as mean ± standard error of the mean (SEM). *p < 0.01 vs. NS, **p < 0.0001 vs. NS, # p < 0.0001 vs. ATO. NS: normal saline, ATO: arsenic trioxide, EA: ellagic acid.
Serum levels of cardiac biomarkers
As2O3 injection at the dose of 5 mg/kg for 10 days resulted in a significant increase in the serum troponin-I and CK-MB levels in comparison to the control group (p < 0.0001). Our results showed that pretreatment with ellagic acid markedly attenuated the increases in serum troponin-I and CK-MB levels compared to the arsenic-administered group (p < 0.0001 and p < 0.01, respectively; Figures 2 and 3).

Serum level of troponin I. Troponin I concentrations were measured using enzyme-linked immunosorbent assay 48 h after the last dose of treatments (n = 8). Data are expressed as mean ± standard error of the mean. *p < 0.0001 vs. NS, # p < 0.0001 vs. ATO. NS: normal saline, ATO: arsenic trioxide, EA: ellagic acid.

Serum levels of creatine kinase isoenzyme MB (CK-MB). Immunoassay of CK-MB was performed 48 h after the last dose of treatments (n = 8). Data are expressed as mean ± standard error of the mean. *p < 0.0001 vs. NS, # p < 0.01 vs. ATO. NS: normal saline, ATO: arsenic trioxide, EA: ellagic acid.
Levels of MDA, GSH, and GPx activity in heart tissues
GPx activity in heart tissue homogenates showed a marked (p < 0.01) increase in As2O3-treated rats, compared to the control group. As shown in Figure 4, pretreatment with ellagic acid (30 mg/kg, orally) in the As2O3-treated rats significantly attenuated the increase in GPx activity (p < 0.05).
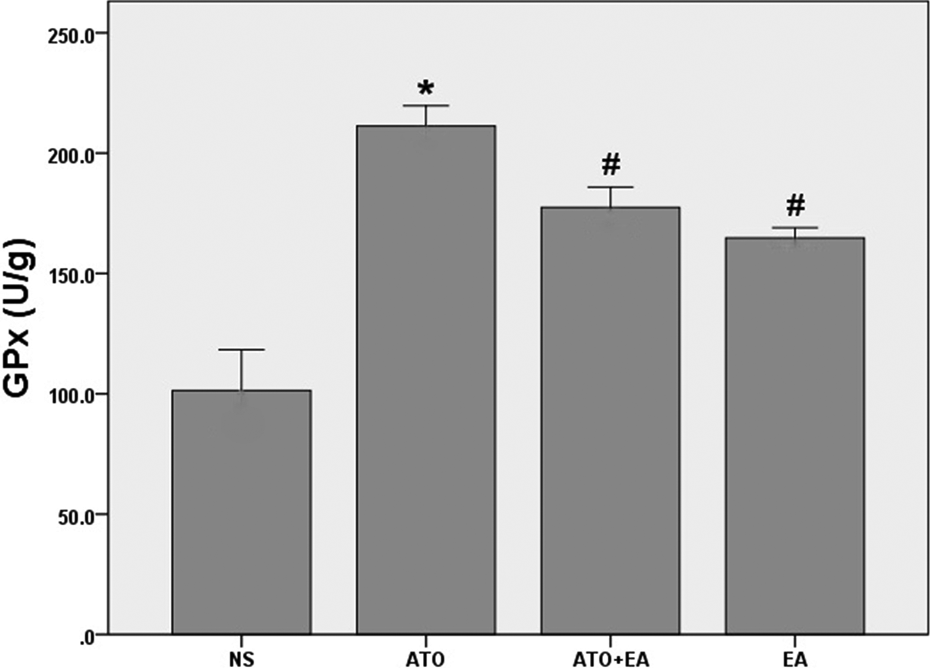
Glutathione peroxidase (GPx) levels in cardiac tissue homogenates. Values of GPx enzyme activity was determined using a colorimetric assay 48 h after the last dose of treatments (n = 8). Data are expressed as mean ± standard error of the mean. *p < 0.01 vs. NS, # p < 0.05 vs. ATO and NS. NS: normal saline, ATO: arsenic trioxide, EA: ellagic acid.
The mean GSH concentrations in tissue homogenates of the control group rats was 136 ± 10 µM. As2O3, ellagic acid, and ellagic acid plus As2O3 treatments did not show statistically significant changes in GSH level. GSH concentration in these treatment groups was found to be 110 ± 5, 140 ± 14, and 130 ± 11 µM, respectively.
As2O3 injection for 10 days enhanced lipid peroxidation in heart tissue of the animals. This was found by a significant increase in MDA levels in tissue homogenates in response to As2O3 treatment (p < 0.01). Ellagic acid when pretreated prior to As2O3 administration effectively (p < 0.001) prevented the enhanced lipid peroxidation in the rat hearts (Figure 5).
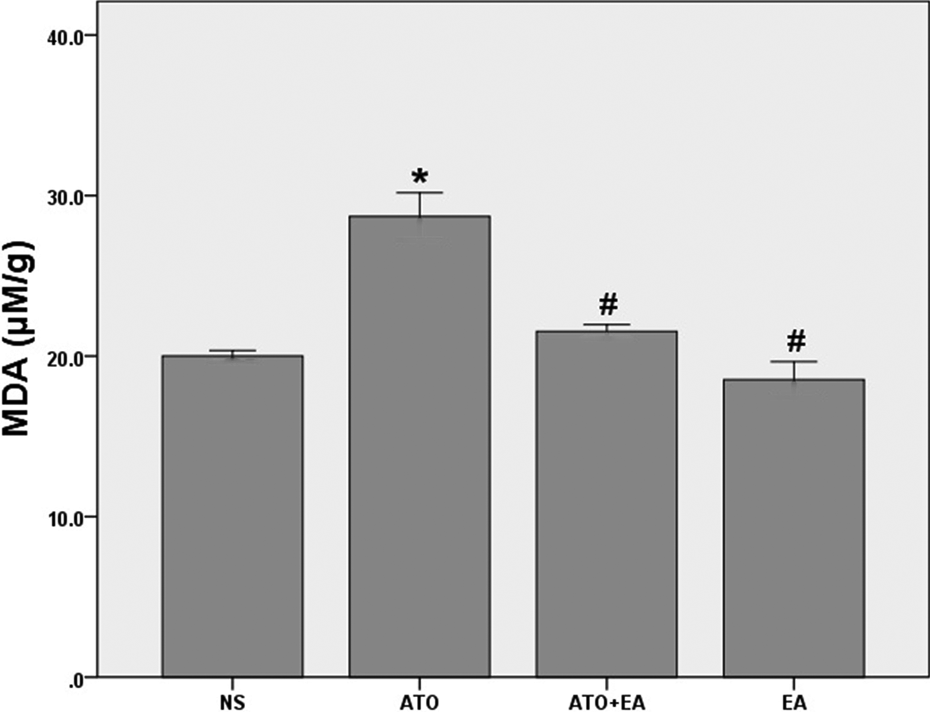
Malondialdehyde (MDA) levels in cardiac tissue homogenates. MDA concentrations were quantified as thiobarbituric acid-reactive substances 48 h after the last dose of treatments (n = 8). Data are expressed as mean ± standard error of the mean. *p < 0.01 vs. NS, # p < 0.001 vs. ATO. NS: normal saline, ATO: arsenic trioxide, EA: ellagic acid.
Effect of ellagic acid on As2O3-induced cardiotoxicity
Results of histopathological examinations of myocardial tissues are shown in Figure 6. Tissue slides prepared from the groups that received normal saline and ellagic acid showed normal cardiac muscle bundles and were devoid of histopathological changes (Figure 6 (a) and (d)). In contrast, myofibrillar loss and moderate myocardial coagulative necrosis were observed in the heart tissue slides of the As2O3-treated rats as compared to the control group (Figure 6(b)). Pretreatment with ellagic acid prevented morphological abnormalities in myocardial tissues of the As2O3-treated rats (Figure 6(c)).

Histopathologic view of cardiac tissue sections following treatments. Paraffin-embedded tissue blocks were stained with H&E and examined with 40× magnification. (a) Normal saline treated, (b) arsenic trioxide (ATO) treated, (c) ATO + ellagic acid (EA) treated, (d) EA treated.
Discussion
Inorganic arsenic exposure provokes ROS formation and cellular oxidative damages. 3,8,9 Arsenic-induced imbalance between generation of oxygen-derived radicals and cellular antioxidant capacity plays a major role in disease progression. 18,19
Arsenic stimulates oxidative stress via ROS production, decrease in antiapoptotic molecules such as B-cell lymphoma 2 (Bcl-2) and Bcl-XL and upregulation of proapoptotic molecules such as Bcl-2-associated X protein (Bax) and BH3 interacting domain death agonist (Bid), release of cytosolic cytochrome c from the mitochondria, and activation of caspases. These events ultimately lead to cardiac cell damage and apoptosis. 3,8,9,20
Natural antioxidant agents acting as effective ROS scavengers have an important role in defense against As2O3-induced free radicals and the ensuing cardiotoxicity. 21,22
In the present study, we aimed to determine the possible protective effect of ellagic acid as an antioxidant compound against the adverse effects of arsenic on ECG, serum cardiac markers, oxidative stress parameters, and histopathological alterations using the experimental model of As2O3-induced cardiotoxicity in rats.
Our results indicated that QTc interval was prolonged in the ECG examination of As2O3-treated rats. This finding is consistent with an epidemiological study on 630 Taiwanese people residing in an arsenic-exposed area that demonstrated a correlation between QT prolongation and tissue arsenic concentration. 23 In another epidemiological study, the prevalence of QT prolongation in As2O3-exposed people was reported to be 90%. 24 It has been shown that the underlying physiological pathways involving in arsenic cardiotoxicity includes intracellular Ca2+ overload, prolongation of cardiac repolarization, increased calcium currents, and enhanced intracellular ROS production. 20,24 Oral pretreatment by ellagic acid restored this QTc prolongation (p < 0.0001). Various studies have demonstrated that polyphenolic compounds such as curcumin, resveratrol, quercetin, ellagic acid, and caffeic acid possess ROS scavenging properties and, consequently, are capable of preventing morphological alterations in cardiomyocytes and electrophysiological abnormalities. 13,25,26 Ellagic acid has been shown to have positive inotropic and antiarrhythmic effects as well as regulating heart rate and blood pressure. 27,28 Our results confirmed that ellagic acid was able to prevent ECG abnormalities in part by its antioxidant capacity. 25,26 Administration of ellagic acid significantly suppressed the increased serum troponin I and CK-MB concentrations in response to As2O3, suggesting its beneficial protective effect on the heart by restricting the extent of the cardiomyocytes injuries, thereby reducing the release of these cardiac markers from the myocardium.
In the present study, we also found that the combined treatment of ellagic acid and As2O3 markedly inhibited the As2O3-induced increase in GPx activity and lipid peroxidation.
Previous studies have reported arsenic-induced Nrf2 activation. Activation of Nrf2 pathway, a transcription factor that tightly regulates endogenous antioxidant defense, can enhance the gene expression of antioxidant enzymes such as GPx, glutathione S-transferase, catalase, and superoxide dismutase and attenuates arsenic-induced oxidative injury. 9,29,30
Preconditioning with ellagic acid protected animals through its ROS scavenging property and preserved the GPx activity in As2O3-treated rats.
A direct correlation between arsenic levels in tissue homogenate samples and lipid peroxidation has been shown in previous studies. ROS cause oxidation of polyunsaturated fatty acids that result in MDA formation. The formation of MDA is used as an indirect biomarker of oxidative stress in tissues. 8,9,28 Our results showed that ellagic acid may exert its protective effect against arsenic cardiotoxicity by decreasing lipid peroxidation in cardiac tissue.
A recent study reported that chronic consumption of ellagic acid protected against atherosclerosis and endothelial oxidative stress. 13 These effects of ellagic acid were partly attributed to improvements in nitric oxide bioavailability, increased cellular antioxidants, and Nrf2/heme oxygenase-1 activation. 13
Our histopathological observations presented evidences for the beneficial effects of ellagic acid on oxidative stress-induced necrosis of cardiomyocytes. In our study, pretreatment with ellagic acid before As2O3 administration effectively prevented histopathological alterations, exhibiting a potential effect of ellagic acid against chemotherapeutic drug-induced cardiotoxicity.
A similar study showed that ellagic acid exhibited protective effects against isoproterenol-induced pathological alterations in ECG pattern, cardiac biomarkers, oxidative status, and histopathology of myocardium in rats. 25 Our results confirmed these cardioprotective effects of ellagic acid. It was also suggested that ellagic acid has antimutagenic effects which may be beneficial in combination therapy with antineoplastic agents with mutagenic effects such as As2O3. 31
Based on our study and previous reports, the underlying mechanisms of cardioprotective properties of EA can be summarized to metal ion chelating, preventing metal-catalyzed formation of ROS, protecting against lipid peroxidation, lowering caspase-3 activity, protecting against mitochondrial damages, suppressing activity and protein level of NF-κB, and downregulating p-p38 and phosphorylated extracellular Signal-regulated Kinase 1/2 (p-ERK 1/2) expressions. 32 –35
In summary, our findings showed that ellagic acid was effective in alleviating As2O3-induced myocardial injury. Ellagic acid could enhance endogenous antioxidant system against oxidative damage to protect myocardium. Therefore, ellagic acid provides a safe and natural option for the prevention of As2O3-induced cardiotoxicity in APL patients. However, such application of ellagic acid needs further investigation.
Footnotes
Acknowledgments
This article is the thesis of Samaneh Olapour, and authors gratefully acknowledge the help and financial support of Research Affairs of Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran (Grant no. APRC-9503).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support of this research was provided by Jundishapur University of Medical Sciences, Ahvaz, Iran.