Abstract
The species belonging to Scrophularia genus grow mainly in Irano-Turanian and Mediterranean regions and have been used as folk remedy for inflammatory-related diseases since ancient times. The present study was aimed to evaluate the anti-inflammatory activity of the extracts of Scrophularia kotschyana as well as the isolated compounds. The aerial parts and the roots of the plant were separately extracted with methanol. Anti-inflammatory activities of both extracts were evaluated with formalin test in mice. As the methanolic extract of the aerial parts significantly (p < .05) inhibited inflammation, it was then submitted to successive solvent extractions with n-hexane, dichloromethane, ethyl acetate and n-butanol to yield subextracts. Anti-inflammatory activities of the subextracts were evaluated within the same test system. Among the subextracts tested, the n-butanol subextract produced a significant (p < .05) anti-inflammatory activity at all doses (5, 10, and 30 mg/kg, ip.). Sequential chromatographic separation of the n-butanol subextract yielded 8-O-acetyl-4′-O-(E)-p-coumaroylharpagide, 8-O-acetyl-4′-O-(Z)-p-coumaroylharpagide, β-sitosterol 3-O-β-glucopyranoside, apigenin 7-O-β-glucopyranoside, apigenin 7-O-rutinoside, luteolin 7-O-β-glucopyranoside and luteolin 7-O-rutinoside. The anti-inflammatory activities of the isolates were evaluated at 5 mg/kg dose. Luteolin 7-O-β-glucopyranoside and apigenin 7-O-rutinoside caused a significant (p < .05) inhibition of oedema formation.
Introduction
Medicinal plants have been used for years in the treatment of various diseases. It is important to investigate the pharmacological effects of medical plants, to prove the traditional usage of them and to establish resources for pharmaceutical raw materials.
Scrophularia L. genus that belongs to Scrophulariaceae family has 270 taxa worldwide and represented by 78 species and subspecies in Turkey.1–3 Scrophularia species which are primarily located in the Irano-Turanian and Mediterranean regions have been used as folk medicine since ancient times. 4
Scrophularia species are used as analgesic, antirheumatic and antiseptic, against bacterial-viral infections such as pharyngitis laryngitis, as wound healer and in the treatment of various skin diseases in some folk medicines. The dried roots of S. buergeriana have been reported to be used in China and Vietnam for its anti-inflammatory effects.4,5 The aerial parts of S. trifoliata, S. alpestris, S. libanotica and S. deserti were used to relieve inflammatory skin diseases in various regions of the world.6,7 The usage of Scrophularia species in Turkey is also similar. It has been reported that the decoction made from the aerial parts of S. libanotica was used against inflamed wounds in Turkey.8,9
In connection with the ethnobotanical uses of Scrophularia species, many pharmacological activity tests and subsequent phytochemical analysis studies have been carried out, and in most of these studies, the compounds responsible for the activity have been identified. In recent studies several phenylethanoid glycosides, iridoid glycosides and saponins have been isolated from some Scrophularia species as bioactive constituents.4,5 In our previous study, we reported in vivo analgesic activities of the MeOH extract prepared from the aerial parts of S. kotschyana and isolated the secondary metabolites which are responsible of the analgesic activity. 10 As a continuation of our previous study, we aimed to evaluate the anti-inflammatory potentials of the extracts, subextracts and the isolated compounds from S. kotschyana by means of in vivo model to find active components through bioassay-guided procedures.
Materials and Methods
Plant material
The plant specimen, S. kotschyana, was collected in May 2013 from Sumela Trabzon, Turkey. The authority to access the plant samples that were used in this study was obtained from Directorate General for Nature Conservation and National Parks of Ministry of Forestry and Water Management of Turkey (Date: 27.02.2013, Number: 72784983-488.04). The plants were identified by Associated Professor Dr Gülin Renda from the Department of Pharmaceutical Botany, Faculty of Pharmacy, Karadeniz Technical University. A voucher specimen (HUEF 15002) is kept in the Herbarium of Hacettepe University Faculty of Pharmacy, Ankara, Turkey.
Extraction and isolation
The air-dried aerial parts and roots of S. kotschyana were separated from each. Processes after this stage were carried out on the aerial parts and on the roots separately. Plant materials were powdered. 5 g of each sample was extracted with MeOH (2 × 50 mL) for 1 h (40°C). The extracts were filtered through filter paper, and the organic phases were evaporated to dryness under reduced pressure at 40°C to obtain 1.092 g aerial part extract (yield 21.8%) and 0.335 g root extract (yield 6.7%). Anti-inflammatory activities of both the extracts were then tested with formalin test in mice at the doses of 5, 10 and 30 mg/kg (i.p.).
To obtain the subextracts, air-dried and powdered aerial parts of S. kotschyana (125 g) were extracted three times with methanol (each 3 L) at 40°C. The combined methanolic extracts were evaporated to dryness in vacuo (29.7 g, yield 23.8%). The aerial part extract was dissolved in methanol: water (1:4) mixture (200 mL) and subsequently transferred to the separatory funnel. The residue was partitioned with n-hexane (3 × 200 mL), then with dichloromethane (3 × 200 mL), after that with ethyl acetate (3 × 200 mL) and finally with n-butanol (3 × 200 mL). The organic phases of each extract and the final aqueous phase were evaporated to dryness under reduced pressure to give n-hexane (2.06 g), dichloromethane (4.30 g), ethyl acetate (2.30 g), n-butanol (11.45 g) and remaining H2O (7.91 g) subextracts, respectively.
The n-butanol subextract which was found as the most active extract was further fractionated by using chromatographical techniques, according to the bioassay-guided fractionation procedures. Firstly, it was subjected to vacuum liquid chromatography (VLC) over reversed-phase material (LiChroprep C18), and elution with H2O, followed by increasing concentrations of MeOH in H2O mixtures (0–100% MeOH, in steps of 10% of MeOH, each 100 mL, fraction volumes 100 mL) as eluent, yielded 10 fractions, A (100% H2O; 5.66 g), B (10–20% MeOH; 1.27 g), C (30% MeOH; 582.9 mg), D (40% MeOH; 140.7 mg), E (50% MeOH; 586.0 mg), F (60% MeOH; 602.4 mg), G (80% MeOH; 267.6 mg), H (90% MeOH; 135 mg), I (100% MeOH; 93.2 mg) and J (100% MeOH; 98.1 mg). Purification of the fractions C, E and F was carried out by successive chromatographic analyses. Structure elucidation of the compounds was done on the basis of 1D- and 2D-NMR experiments, HR-MS analysis and by comparison of their spectroscopic data with the literature. Isolation and structure elucidation procedures for the isolated pure compounds (1–6) were reported in our previous study. 10 The chemical structures of the isolated compounds were identified as 8-O-acetyl-4′-O-(E)-p-coumaroylharpagide and 8-O-acetyl-4′-O-(Z)-p-coumaroylharpagide (an inseparable mixture of isomeric compounds) (1), apigenin 7-O-β-glucopyranoside (2), apigenin 7-O-rutinoside (3), luteolin 7-O-β-glucopyranoside (4), luteolin 7-O-rutinoside (5) and β-sitosterol 3-O-β-glucopyranoside (6).
Animals and experimental setup
Male or female Balb C mice weighing 30–35 g were used in groups of eight. All the animals were obtained from KTU CAM (Karadeniz Technical University Surgical and Application Research Center) and were fed ad libitum with a standard diet except during the experimentation period. Mice were maintained in an isolated room at a constant temperature (23 ± 2°C) with a 12 h light/dark programme during the experiments in accordance with European Union regulations. The protocol of the experiment was approved by the Karadeniz Technical University Animal Care and Ethics Committee (2013/9).
All the extracts, subextracts and the compounds 1–5 were dissolved in the vehicle; saline (0.9% sodium chloride). All the extracts were given to the animals at the doses of 5, 10 and 30 mg/kg, beside the compounds were given at the doses of 5 mg/kg. Vehicle (saline) was injected into the control groups at the same volume. Treatments with extract (in saline), compounds (in saline), diclofenac or saline were done by using intraperitoneal injection (i.p.) at a dose of 0.1 mL per 10 g of body weight. Diclofenac Na (10 mg/kg i. p.) was used as the reference. 11
Experimental groups are as follows: control group (n = 8) in which mice were given saline (i.p.) and then formalin test was applied; aerial part methanol extract group (n = 24) in which formalin test was applied after being given aerial part methanol extract at doses of 5 mg/kg, 10 mg/kg and 30 mg/kg (8 animals for each group, i. p.); root methanol extract group (n = 24) in which formalin test was applied after being given root methanol extract at doses of 5 mg/kg, 10 mg/kg and 30 mg/kg (8 animals for each group, i. p.); n-hexane subextract group (n = 8) in which formalin test was applied after being given n-hexane subextract at a dose of 30 mg/kg (i.p.); dichloromethane subextract group (n = 8) in which formalin test was applied after being given dichloromethane subextract at a dose of 30 mg/kg (i.p.); ethyl acetate subextract group in which formalin test (n = 8) was applied after being given ethyl acetate subextract at a dose of 30 mg/kg (i.p.); n-butanol subextract group in which formalin test (n = 24) was applied after being given n-butanol subextract at doses of 5 mg/kg, 10 mg/kg and 30 mg/kg (8 animals for each group, i. p.); water subextract group (n=24) in which formalin test was applied after being given water subextract at doses of 5 mg/kg, 10 mg/kg and 30 mg/kg (8 animals for each group, i. p.); isolated compounds group (n = 40) in which formalin test was applied after being given compounds at doses of 5 mg/kg (8 animals for compound, i. p.); diclofenac Na group (n = 8) in which formalin test was applied after being given diclofenac Na at a dose of 10 mg/kg (i.p.).
Formalin test
The formalin test is a primary behavioural screen method for assaying the antinociceptive activity of compounds in laboratory rodents. Within this method, a small amount of dilute formalin which is injected just under the skin on the dorsal or plantar surface of the hind paw of the rodent causes inflammation and an immediate retraction of the paw followed by a series of behavioural responses. The test was based on that described by Saddi and Abbott
12
in the literature. Mice were placed in transparent acrylic chamber (11 × 11 × 15 cm) and a mirror was placed at the back of the chamber. The animals did not have access to water or food after they had been placed in the observation chamber. Formalin (20 μL, 1% in saline) was injected subcutaneously into the dorsal surface of the right hind paw using a microsyringe. The mouse was then put back in the chamber and the observation period started. The behaviours such as licking, biting and shaking the paw as a response to inflammation were observed for a period of 1 h. The number of movements and the duration of the movements were recorded for 1 minute from the first minute following the injection. Behavioural responses to the formalin-induced inflammation test consist of two different phases: the acute phase (acute early phase: 0–10 min) and the chronic phase (late second phase: 10–45 min). While acute phase scoring is done for every minute, in continuation, observations were made for 1 minute at 5 minutes intervals in the chronic phase. Pain score was determined by measuring the three behavioural categories: (1) the paw has little or no weight placed on it; (2) the paw is elevated; (3) the paw is licked, bitten or shaken. Pain scores were calculated for acute and chronic phases according to the formula given below
13
Formalin-induced paw oedema test
Changes in paw thicknesses of the mice.

Values are expressed as mean ± SD of eight mice.
p < .05 as compared with vehicle control.
% changes between formalin control and treated group.
Changes in paw volumes of the mice. *

Values are expressed as mean ± SD of eight mice.
p < .05 as compared with vehicle control.
% changes between formalin control and treated group.
Statistical analysis of data
Statistical analyses were conducted using SPSS 23 software (IBM, Turkey). The one-way ANOVA test and post hoc Bonferroni test were used in the evaluation of the data which were suitable for normal distribution. The Kruskal–Wallis test and post hoc Mann–Whitney U test were used to evaluate the data in nonnormal distribution. The results are presented as mean ± standard error (SEM). The level of significance was set at p < .05.
Acute toxicity
After the formalin test of extracts and compounds, food and water were allowed ad libitum to animals used in the test, and all of them were observed for 24 h in terms of mortality and behavioural changes for possible toxicity symptoms. 14
Results
In this study, the in vivo anti-inflammatory activity of S. kotschyana has been investigated using a formalin test in mice. The formalin causes inflammation at the affected hind paw and the reductions in oedema evaluated by measuring paw thickness and paw volume. In our study, an increase in hind paw thickness and paw volumes was detected as a result of formalin injection and the changes were evaluated by comparing with the reference drug “diclofenac Na”. Tables 1 and 2 show the changes in paw thicknesses and paw volumes, respectively.
Hind paw thicknesses were significantly (p < .05) decreased after the treatment by the root methanol extract at all the doses injected, compared with the control. The change of paw thickness after formalin injection was also significantly (p < .05) affected by the treatment with the MeOH extract prepared from the aerial parts at the dose of 30 mg/kg (Table 1). At the same time, paw volumes were significantly (p < .05) decreased compared with the control group after the treatment with all doses of the aerial part MeOH extract and with the root MeOH extract at the dose of 30 mg/kg (Table 2).
Anti-inflammatory activity was also indicated by the reduction of the total mean time determined for the test groups compared with the control group. Behavioural observations to the formalin-induced inflammation test were scored to calculate the pain scores for acute and chronic phases. Behaviours that are scored for 0–10 min post-injection occur in a biphasic temporal pattern, with an acute early phase, followed by a quiescent period (10–45 min) forming a chronic phase. Early phase licking occurs due to a direct effect of the formalin injection on nociceptors, and the late phase due to a central sensitization and subsequent peripheral inflammation. Both the aerial part and root methanol extracts inhibited both phases of the formalin test showing a pain inhibition (Figure 1). For the pain inhibition, only all doses of aerial parts showed a statistically significant difference (p < .05). Therefore, activity studies were performed on the subextracts obtained from the crude MeOH extract prepared from the aerial parts. Like our previous study on the analgesic activities of the crude extracts of the same species, we evaluated the anti-inflammatory activities of the n-hexane, dichloromethane, ethyl acetate, n-butanol and water subextracts derived from the MeOH extract of aerial parts at the dose of 30 mg/kg.
10
As shown in Figure 2, all of the subextracts obtained from the aerial parts exhibited significant (p < .05) pain inhibition at the dose of 30 mg/kg when compared with saline. In hind paw thicknesses, volume and pain inhibition tests, all the subextracts tend to have positive effects (Tables 1 and 2; Figure 2), but as we compared the results of all tests, we decided to study the effect of lower doses (5 mg/kg and 10 mg/kg) of n-butanol and water subextracts. During this evaluation procedure, the n-butanol and water subextracts at all the doses tested significantly (p < .05) inhibited both phases of the formalin test showing a pain inhibition (Figure 3). Also, both paw volumes and paw thicknesses were significantly (p < .05) decreased when compared with the formalin-injected control after the treatment with the n-butanol and water subextracts at all the doses tested within the tested subextracts (Tables 1 and 2). Formalin-induced paw pain score in both the early first phase (0–10 min) and the late second phase (10–50 min) following injection of saline (as control) and (a) aerial parts (b) roots (5, 10 and 30 mg/kg). Vertical lines show means ± S.E.M (n = 8). *: p < .05, as compared to control. Formalin-induced paw pain score in both the early first phase (0–10 min) and the late second phase (10–50 min) following injection of the aerial parts subextracts of the aerial parts (30 mg/kg). Vertical lines show means ± S.E.M. (n = 8). *:p < .05, as compared to control. Formalin-induced paw pain score in both the early first phase (0–10 min) and the late second phase (10–50 min) following injection of the aerial parts subextracts of the aerial parts (a) n-butanol (b) water (5, 10 and 30 mg/kg). Vertical lines show means ± SEM (n = 8). *:p < .05, as compared to control.


In the continuation of the study, phytochemical studies were carried out on the n-butanol subextract as it was found to be the most active extract. As a result of chromatographic separations, 8-O-acetyl-4′-O-(E)-p-coumaroylharpagide, 8-O-acetyl-4′-O-(Z)-p-coumaroylharpagide, apigenin 7-O-β-glucopyranoside, apigenin 7-O-rutinoside, luteolin 7-O-β-glucopyranoside, luteolin 7-O-rutinoside and β-sitosterol 3-O-β-glucopyranoside were isolated and tested for their in vivo anti-inflammatory potentials. The results of the bioactivity were presented in Tables 1 and 2 and Figure 4. β-sitosterol 3-O-β-glucopyranoside showed anti-inflammatory effect on three tests (data not shown). However, it was not clear whether the compound itself or its dissolvent caused these effects. Luteolin 7-O-β-glucopyranoside and apigenin 7-O-rutinoside showed a significant (p < .01) inhibition of oedema formation and revealed significant pain inhibition at all the doses tested (Figure 4). Formalin-induced paw pain score in both the early first phase (0–10 min) and the late second phase (10–50 min) following injection of compounds 1–6 (5 mg/kg) and diclofenac (10 mg/kg). Vertical lines show means ± SEM (n = 8). *:p < .05, as compared to control.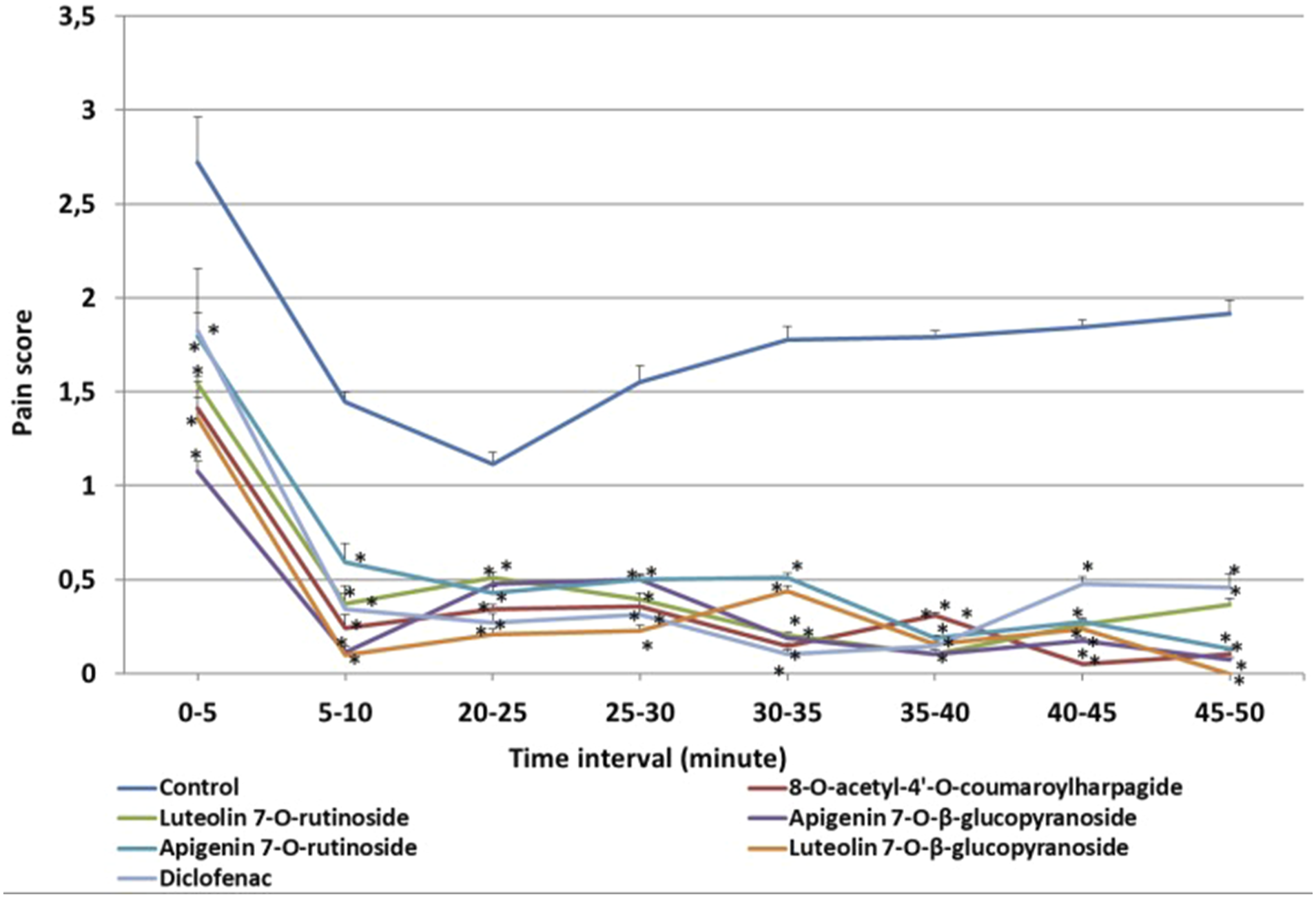
Discussion
Inflammation is defined as a body defense reaction of tissues to injury. 15 Until now, despite the use of various anti-inflammatory drugs such as immunosuppressants, NSAIDs, corticosteroids and antihistamine, potential side effects impose a limitation on their use. The need to find new safe, powerful, less toxic analgesics and anti-inflammatory agents to protect against various chronic diseases is leading us to herbal origins. The formalin test is usually used as a classical method to detect the anti-inflammatory efficacy of drugs. 16 Formalin response shows an early and a late phase and measures pain of both neurogenic (first phase) and inflammatory (second phase) origins. 17 It has been reported that the early phase which was caused by C-fibre activation due to the peripheral stimulus and a direct effect on nociceptors was not inhibited by prostaglandins while the late phase that was dependent on the combination of an inflammatory reaction in the peripheral tissue and functional changes in the dorsal horn of the spinal cord could be inhibited by anti-inflammatory drugs. 18 Scrophularia species have been used for the treatment of inflammatory-related diseases worldwide.
Although a large number of compounds have been isolated from the species of the Scrophulariaceae family, only a few species of the Scrophularia genus have been extensively studied. The major constituents of this genus belong to glycoside esters or phenylpropanoid glycosides saponins and iridoids. Of these, phenylpropanoid glycosides and iridoids which showed significant therapeutic potential have been reported to be major secondary metabolites for the genus Scrophularia. 19 The effectiveness of the Scrophularia species is also related to its relatively higher flavonoid contents. 20 Of course, to confirm the anti-inflammatory and analgesic active components of S. kotschyana, a more in-depth study of these compounds is necessary.
The anti-inflammatory activity of some Scrophularia extracts and the pure compounds was evaluated before. In an anti-inflammatory activity study on 8-O-acetyl harpagide, isolated from S. deserti, the pure substance was tested in a carrageenan-induced paw oedema test at a dose of 10 mg/kg. Phenylbutazone was used at the dose of 100 mg/kg as a positive control. 8-O-acetyl harpagide was found to have moderate activity, and it was suggested that the cinnamoyl unit increased the activity.
21
Analgesic and anti-inflammatory activities of acetyl harpagide and harpagide have also been shown in previous in vitro and in vivo studies.22-24 It was reported that neither harpagide nor harpagoside showed any inhibition on COX-1 or COX-2 at the concentrations between 2.5 and 100 μ
The anti-inflammatory activities of apigenin and luteolin aglycones were tested in various in vitro test systems, and the compounds were found to be active.25-27 Generally, flavonoids have been reported to have beneficial effects in inflammatory conditions due to their radical scavenging activities and interaction with enzyme functions. 28
Safety is an important aspect of natural products. There are studies investigating the cytotoxic properties of some of the isolated compounds against various cancer cells in the literature. There is no previous study on the toxicity of compound 1 up to date. Two of the compounds isolated as a result of our study are derivatives of the most studied flavonoids; luteolin and apigenin. A series of toxicological studies on the ingestion of apigenin were conducted, including acute toxicity, genetic toxicity as well as sub-chronic toxicity experiments. Apigenin was also reported to be non-toxic with no genetic toxicity. Results of the Ames test showed that apigenin was not mutagenic or toxic when tested alone. 29 Anti-genotoxic activity of luteolin-7-O-glucoside, luteolin-7-O-rutinoside and luteolin-7-O-glucuronide was reported previously in the micronucleus test by using human cells. 30 Previous data in the literature has been supported by our results that the administration of all extracts and isolated compounds in our study did not show any symptoms of toxicity in terms of mortality and behavioural changes during the 24-hour observation period.
Based on the results obtained from the study it can be concluded that, the methanol extracts of the roots and aerial parts of S. kotschyana possess significant anti-inflammatory potential. n-butanol subextract was found as the most active subextract. Among the isolated compounds, luteolin 7-O-β-glucopyranoside and apigenin 7-O-rutinoside were found to possess the highest activity among the other compounds. The change in paw volumes and paw thicknesses with the main extracts is higher than the change with luteolin 7-O-β-glucopyranoside. The higher activity obtained with the methanol extract may be the result of the synergic effects of the components in the extract. The present results are the first report of the anti-inflammatory effects of S. kotschyana and its constituents. n-butanol and water subextracts generated a significant reduction at both phases of the formalin test, by suggesting that n-butanol extract has an analgesic effect with inhibitory mechanisms on neurogenic nociception and inflammatory nociception. 10 These results require further study to investigate the possible mechanism of action that may be involved.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by TÜBİTAK (Project number: 113S252).

