Abstract
Calcitonin gene-related peptide (CGRP) and substance P (SP) play an important role in the development of pain and hyperalgesia. Experimental models have demonstrated that nitroglycerin (NTG)—a nitric oxide donor—provokes a hyperalgesic state, probably via the activation of second-order neurons in the nucleus trigeminalis caudalis. In order to gain further insight into the role of CGRP and SP in different types of experimental pain, we evaluated and compared changes in immunoreactivity (-ir) for these two neuropeptides at different levels of the central nervous system [nucleus trigeminalis caudalis (NTC) and dorsal horns of the lumbar spinal cord] in two animal models of hyperalgesia: systemic NTG administration and formalin test. Following NTG administration, CGRP-ir decreased steadily in the NTC, whereas SP-ir increased transiently. In the lumbar dorsal horns, NTG induced a decrease in SP-ir 1 h after its administration. Formalin injection induced an ipsilateral increase in both CGRP and SP immunostaining at 1 and 2 h in the lumbar dorsal horns. In the NTC, a significant decrease in CGRP-ir was observed at 1 h. The changes in the staining intensities were paralleled by changes in the numbers of CGRP and of SP varicosities in both the NTC and the lumbar dorsal horns. These findings show specific changes in CGRP and SP at different levels of the central nervous system in the different models of pain. In the case of the formalin test, the changes involve both neuropeptides synchronously and to the same extent, whereas in the case of NTG administration, CGRP seems to play a more prevalent and long-lasting role, particularly at the NTC level.
Keywords
Introduction
Several lines of evidence indicate that calcitonin gene-related peptide (CGRP) and substance P (SP), together with serotonin, bradykinin, nitric oxide (NO) and prostaglandins (PGs), contribute to the generation of hyperalgesia (1, 2). NO plays a pivotal role in pain processing at spinal level, where it regulates the release of neuropeptides (3, 4). It has also been hypothesized that CGRP and SP are implicated in the pathogenesis of migraine attacks (5–7). In particular, CGRP is a potent vasodilator that is contained in and released from sensory nerve endings during migraine attacks (8). CGRP modulates nociceptive transmission in the trigeminovascular system (9), whereas SP seems to mediate an increase in vascular permeability (10). Nitroglycerin (NTG) is a NO donor that produces rapid vasodilation and delayed spontaneous-like headaches in migraine sufferers (11, 12). In rats, NTG induces a hyperalgesic condition (13, 14) associated with the activation of nuclei involved in nociception, the nucleus trigeminalis caudalis (NTC) in primis, as detected with c-fos immunoreactivity (15–17). The biochemical effects of NTG administration on the brain, which range from neuronal activation of specific nuclei to a condition of hyperalgesia, have been taken as a specific animal model of migraine (18). The formalin test is a tool widely used for studying inflammatory or tonic pain (19). Subcutaneous injection of 1% formalin in the rat hindpaw provokes nociceptive behaviour, which is an expression of peripherally and centrally mediated sensitization phenomena (20). Immunohistochemistry is a technique widely used to investigate the presence of neuropeptides involved in nociception in the spinal dorsal horn (21, 22). The aim of this study was to evaluate and compare changes in CGRP and SP immunoreactivity in the NTC and in the lumbar dorsal horns in two different animal models of pain (systemic NTG administration and formalin test) in order to gain new insight into the role of these two neuropeptides in the mediation of different pain conditions.
Materials and methods
Animals
Adult male Sprague-Dawley rats weighing 180–220 g were used in this experimental study. The rats were housed, in threes, in plastic boxes with water and food available ad libitum. They were kept on a 12:12 h light–dark cycle at the constant temperature of 22 °C. The experimental protocols were approved by the ethics committee of the IRCCS ‘C. Mondino Institute of Neurology’ Foundation.
Drug administration
NTG, dissolved in saline, alcohol and propylene glycol, was injected intraperitoneally at a dose of 10 mg/kg. Previous reports have suggested that this relatively high dose corresponds to the quantity needed to induce the neurochemical changes that could help to clarify the pathogenesis of migraine (18, 23, 24). Other authors who used, in the rat, doses theoretically equivalent to those used in human migraine studies have failed to detect NTG-induced neuronal activation in the NTC (25, 26). The dose of NTG used in the present study is known to induce moderate (−23.3 ± 4.9% vs. baseline) and transient (recovery within 90 min of injection) hypotension, as demonstrated previously (24).
For the formalin test, 100 μl of a 1% formalin solution was injected subcutaneously into the plantar surface of the right hindpaw. The rats submitted to the formalin test were left in a plexiglas cage for 1 h after the formalin injection to allow observation and quantification of their pain-related behaviour.
Every precaution was taken to avoid unnecessary pain to animals at the moment of the injection of either NTG or formalin, but anaesthesia was avoided because of the possible interference with the expected nociceptive response, especially in the case of formalin test.
Twelve groups, each formed by four to six rats, were treated according to the following schedules:
Groups NTG-1 h, NTG-2 h and NTG-4 h: intraperitoneal (i.p.) injection of NTG (Astra Company, Basiglio, Milan, Italy) and perfusion after 1, 2 and 4 h, respectively;
Groups Control-1 h, Control-2 h and Control-4 h: i.p. injection of vehicle (saline, alcohol and propylene glycol) and perfusion after 1, 2 and 4 h, respectively;
Groups Formalin-1 h, Formalin-2 h and Formalin-4 h: intraplantar injection of 100 μl of formalin 1% and perfusion after 1, 2 and 4 h, respectively;
Groups Control-F1 h, Control-F2 h and Control-F4 h: intraplantar injection of 100 μl of saline and perfusion after 1, 2 and 4 h, respectively.
After treatment, all animals were anaesthetized with an overdose of sodium thiopental (200 mg/kg i.p.) and perfused through the ascending aorta with cold saline followed by ice-cold 4% paraformaldehyde solution in 0.1
The areas corresponding to the lower brainstem, cervical (C1-C2) and lumbar (L4-L6) spinal cords were removed and postfixed for 2 h in the same fixative, then placed in sucrose 20% for 72 h. These areas were cut into 50-μm sections on a freezing sliding microtome, placed in six wells containing 0.05
Immunohistochemistry
Rabbit polyclonal antibodies were diluted at 1:1600 for CGRP (Santa Cruz, Santa Cruz, CA, USA) and at 1:5000 for SP (Chemicon, Temecula, CA, USA). Following several rinses in PBS, sections were incubated in a blocking solution (4% normal goat serum) for 30 min and, subsequently, in primary antibodies for 48 h at room temperature in a KPBS solution containing 4% normal goat serum and 0.4% Triton X-100 (TX). After several rinses in a KPBS solution containing 0.04% TX, sections were incubated at room temperature with the secondary biotinylated antibody (Vector Laboratories, Burlingame, CA, USA) for 90 min and subsequently for another period of 90 min with the avidin–biotin complex (Vectastain; Vector Laboratories). Before staining, the sections were rinsed with acetate imidazole (AI) buffer 0.1
In order to avoid variability in the background staining due to the procedure, each treated animal was simultaneously stained with the corresponding control.
Data analysis
A bright-field Zeiss microscope linked via a digital camera to a computer-assisted image-processing system was used to semiquantify the fibre staining intensity for the two neuropeptides in tissue sections taken at the same levels in the NTC and lumbar dorsal horns. Optical density values were taken to indicate the quantities of CGRP and SP present, as suggested by previous reports (27–29).
The optical density of immunoreactive fibres was measured using a 10× objective and a Scion Image Analysis system (Scion, Frederick, MD, USA) by an examiner who was blind to the treatments. Optical density was calculated by correcting the intensity of stained areas for the intensity of the background: to do this, four optical density measurements were taken in regions with no specific staining and their mean was subtracted from the intensity of staining detected in immunoreactive areas, thereby giving the final optical density value. In order to avoid variability of intensity due to the immunostaining procedure, we compared groups of animals that had been stained simultaneously (see paragraph on immunohistochemistry). Optical density was determined by analysing nine adjacent sections per area in each animal.
In order to avoid differences due to asymmetrical sectioning of the spinal cord, the final optical density values for the two peptides in the animals injected with NTG were obtained by analysing both sides of each section and subsequently calculating the mean value of the two sides. Conversely, in the case of formalin administration, the final optical density values were obtained by analysing and considering separately the tissue sections that were ipsilateral or contralateral to the injection side in each rat.
The immunoreactive varicosities were counted using a 40× objective and a modified version of functions originally designed for autoradiographic grain counting (Scion Image Analysis).
In each animal, a total of nine tissue sections were measured bilaterally both in the NTC and in the dorsal horn. In each section, varicosities were counted within a rectangular area (100 × 60 μm) placed above laminae I and II, equidistant from the dorsal and ventral edges of the section in the NTC and from the dorsal and medial edges of the dorsal horn in the lumbar area (30, 31). The software was set to count only varicosities of a given size (in order to exclude cut axons). In addition, for the determination of the cross-sectional areas, we selected boutons which were in focus and were recognized and measured by the program as single objects.
Analysis of variance (
Results
NTG administration
In the control groups, no change in the intensity of CGRP or SP immunostaining was observed in either laminae I or II of the NTC or in the lumbar dorsal horns at any time point (Table 1).
Effect of nitroglycerin administration on immunoreactivity for calcitonin gene-related peptide (CGRP) and substance P (SP) in the nucleus trigeminalis caudalis

Analysis of variance (
P < 0.05 for NTG vs. control.
Rats were injected intraperitoneally with 10 mg/kg body weight of nitroglycerin (NTG) or with an equivalent volume of vehicle (Control). Data are expressed as mean ± SD of optical densities.
Nucleus trigeminalis caudalis
All the NTG-injected groups (NTG-1 h, NTG-2 h and NTG-4 h) showed a significant decrease (P < 0.05) in CGRP immunoreactivity (-ir) in the superficial laminae I and II of the NTC when compared with controls. Conversely, SP-ir showed a significant increase (P < 0.05) 1 h after NTG administration (Table 1 and Fig. 1A,B,E,F).

(A,B) Expression of calcitonin gene-related peptide (CGRP) in representative sections of the nucleus trigeminalis caudalis in rats treated with vehicle (A) or nitroglycerin (B) 1 h before. (E,F) Expression of substance P (SP) in representative sections of nucleus trigeminalis caudalis of rats treated with vehicle (E) or nitroglycerin (F) 1 h before. Scale bar, 10 μm. (C,D) CGRP+ varicosities in representative sections of the nucleus trigeminalis caudalis of rats injected with vehicle (C) or nitroglycerin (D) and perfused 4 h later. Note that the observed decrease in CGRP staining was associated with a decrease in the number of varicosities. (G,H) SP+ varicosities in representative sections of the nucleus trigeminalis caudalis of rats injected with vehicle (G) or nitroglycerin (H), perfused 1 h later. Note that the observed increase in SP staining was associated with an increase in the number of varicosities. Scale bar, 4 μm.
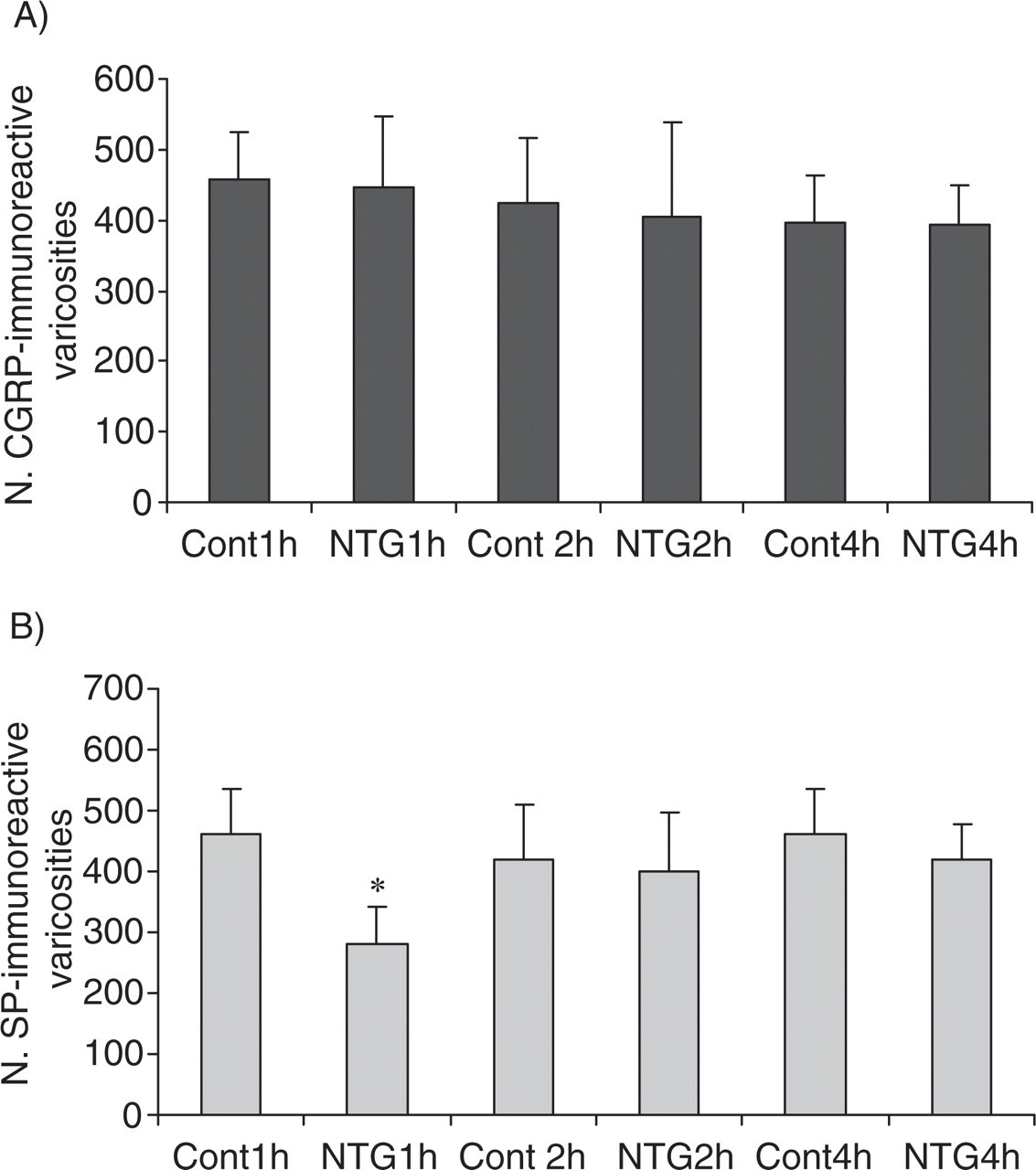
The decrease in the optical density of fibre staining for CGRP was paralleled by a significant decrease in the number of CGRP-immunoreactive varicosities (Fig. 1C,D and Fig. 2A) in all the NTG-treated groups. Conversely, the number of SP-positive varicosities was found to be increased 1 h after NTG administration (Fig. 1G,H and Fig. 2B).

Quantitative analysis of the changes in the number of varicosities immunoreactive for calcitonin gene-related peptide (CGRP) (A) and substance P (SP) (B) within the nucleus trigeminalis caudalis of rats treated with vehicle (Cont) or nitroglycerin (NTG) 1, 2 and 4 h before. Data are expressed as mean ± SD. ∗P < 0.05 vs. controls.
Lumbar dorsal horns
NTG administration did not cause any change in CGRP-ir in the lumbar dorsal horns (Fig. 3A), whereas SP staining intensity was found to be decreased 1 h after NTG administration (Table 2). The decrease in SP-ir observed in this area was associated with a simultaneous decrease in the number of SP-positive varicosities (Fig. 3B).
Effect of nitroglycerin administration on immunoreactivity for calcitonin gene-related peptide (CGRP) and substance P (SP) in the lumbar dorsal horns
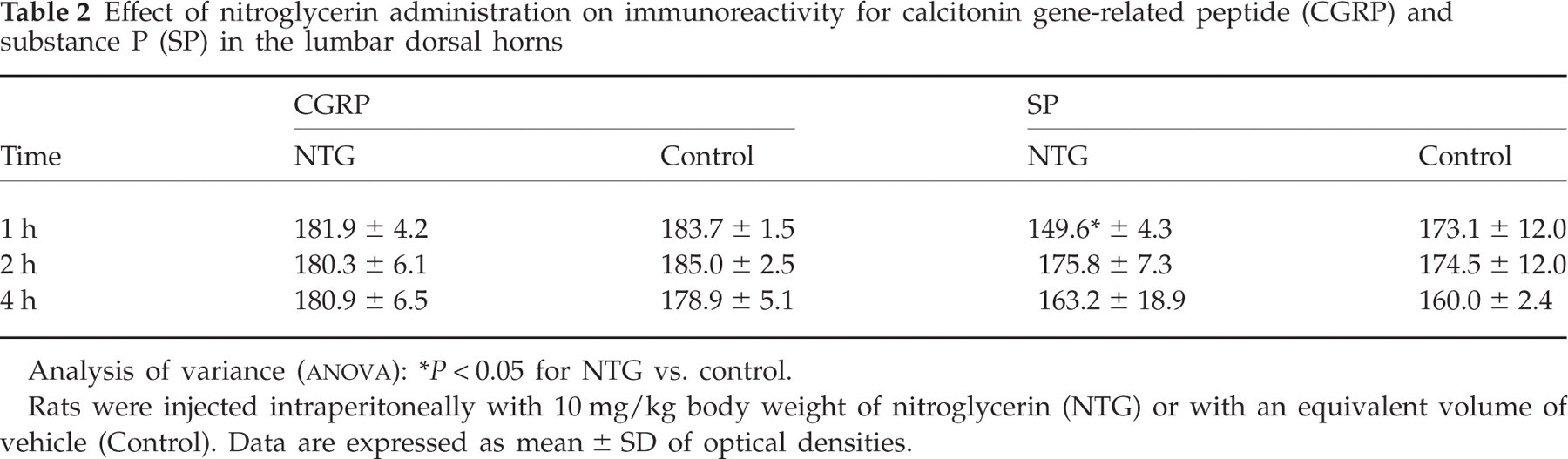
Analysis of variance (
P < 0.05 for NTG vs. control.
Rats were injected intraperitoneally with 10 mg/kg body weight of nitroglycerin (NTG) or with an equivalent volume of vehicle (Control). Data are expressed as mean ± SD of optical densities.
Quantitative analysis of the changes in the number of varicosities immunoreactive for calcitonin gene-related peptide (CGRP) (A) and substance P (SP) (B) within the lumbar dorsal horns of animals treated with vehicle (Cont) or nitroglycerin (NTG) 1, 2 and 4 h before. Data are expressed as mean ± SD. ∗P < 0.05 vs. controls.
Formalin test
Nucleus trigeminalis caudalis
The injection of 1% formalin into the hindpaw induced the characteristic nociceptive behavioural response, characterized by two subsequent phases of flinches and shakes: phase I lasted from 1 to 5 min, and phase II from 10 to 60 min (data not shown) (19).
CGRP-ir was significantly reduced in the ipsilateral NTC 1 h after formalin administration, compared with both the contralateral side and with both sides of the control group (Table 3, upper panel and Fig. 4A,B). Accordingly, the number of CGRP-positive varicosities was decreased on the injected side 1 h after formalin injection (Fig. 4C,D).
Effect of the formalin test on immunoreactivity for calcitonin gene-related peptide (CGRP) (upper panel) and substance P (SP) (lower panel) in the nucleus trigeminalis caudalis
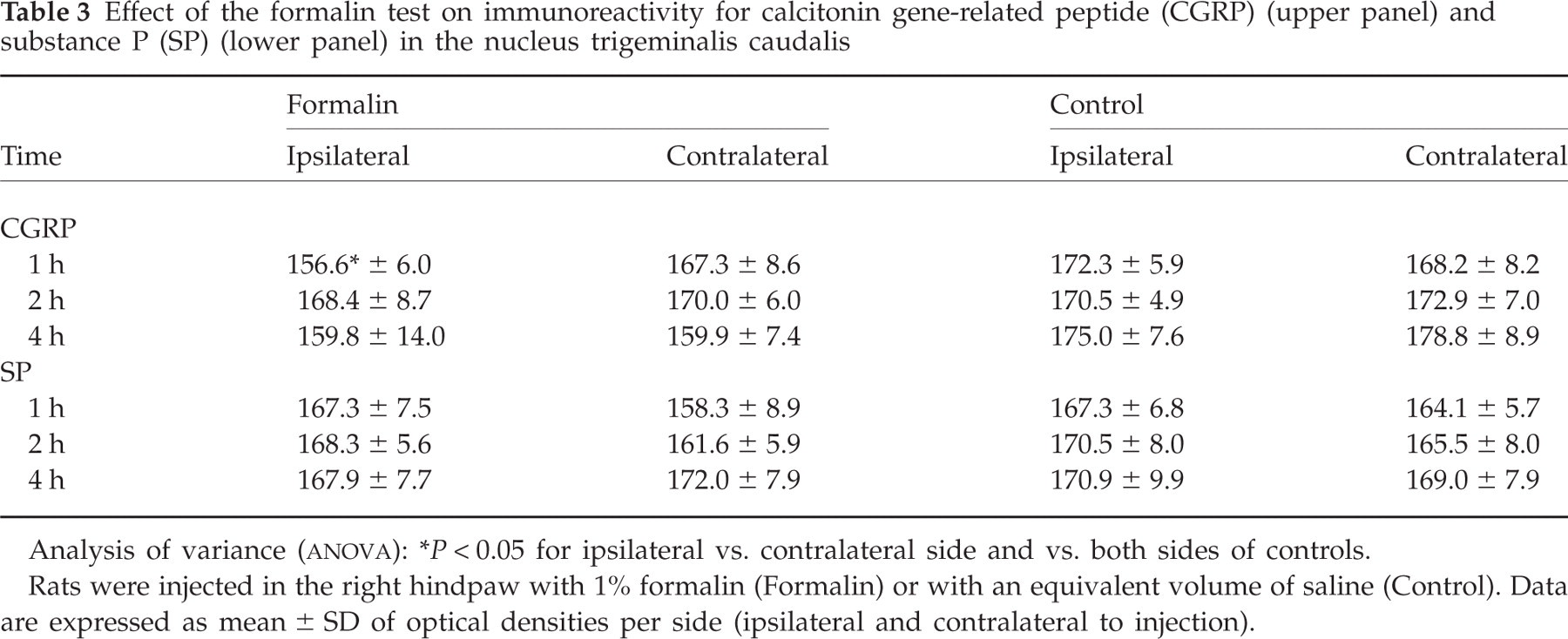
Analysis of variance (
P < 0.05 for ipsilateral vs. contralateral side and vs. both sides of controls.
Rats were injected in the right hindpaw with 1% formalin (Formalin) or with an equivalent volume of saline (Control). Data are expressed as mean ± SD of optical densities per side (ipsilateral and contralateral to injection).

Experssion of CGRP in representative sections of the nucleus trigeminalis caudalis of rats injected with formalin in the right hindpaw 1 hour before: A) injected side, B) contralateral side. Scale bar, 10μm. Histograms show quantitative analysis of the changes in the number of CGRP-(A) and SP-immunoreactive (B) varicosities in the nucleus trigeminalis caudalis of animals injected with formalin in the right hindpaw. Data are expressed as means number of varicosities ± SD on the side ipsilateral (ipsi) and contralateral (contra) to the stimulated side. ∗P < 0.05 vs. contra 1h.
Following formalin injection, no changes were observed in SP-ir (Table 3, lower panel) or in the number of SP-positive varicosities (data not shown).
Lumbar dorsal horns
Formalin injection caused a significant increase (P < 0.05) both in CGRP-ir (Fig. 5) and in SP-ir (Fig. 6) at 1 and 2 h, ipsilaterally to the injection site. This increase was paralleled by an increased number of varicosities in the ipsilateral dorsal horn at 1 and 2 h (Table 4 and Fig. 7).
Effect of the formalin test on immunoreactivity for calcitonin gene-related peptide (CGRP) (upper panel) and substance P (SP) (lower panel) in the lumbar dorsal horn

Analysis of variance (
P < 0.05 for ipsilateral vs. contralateral side and vs. both sides of controls.
Rats were injected in the right hindpaw with 1% formalin (Formalin) or with an equivalent volume of saline (Control). Data are expressed as mean ± SD of optical densities per side (ipsilateral and contralateral to injection).
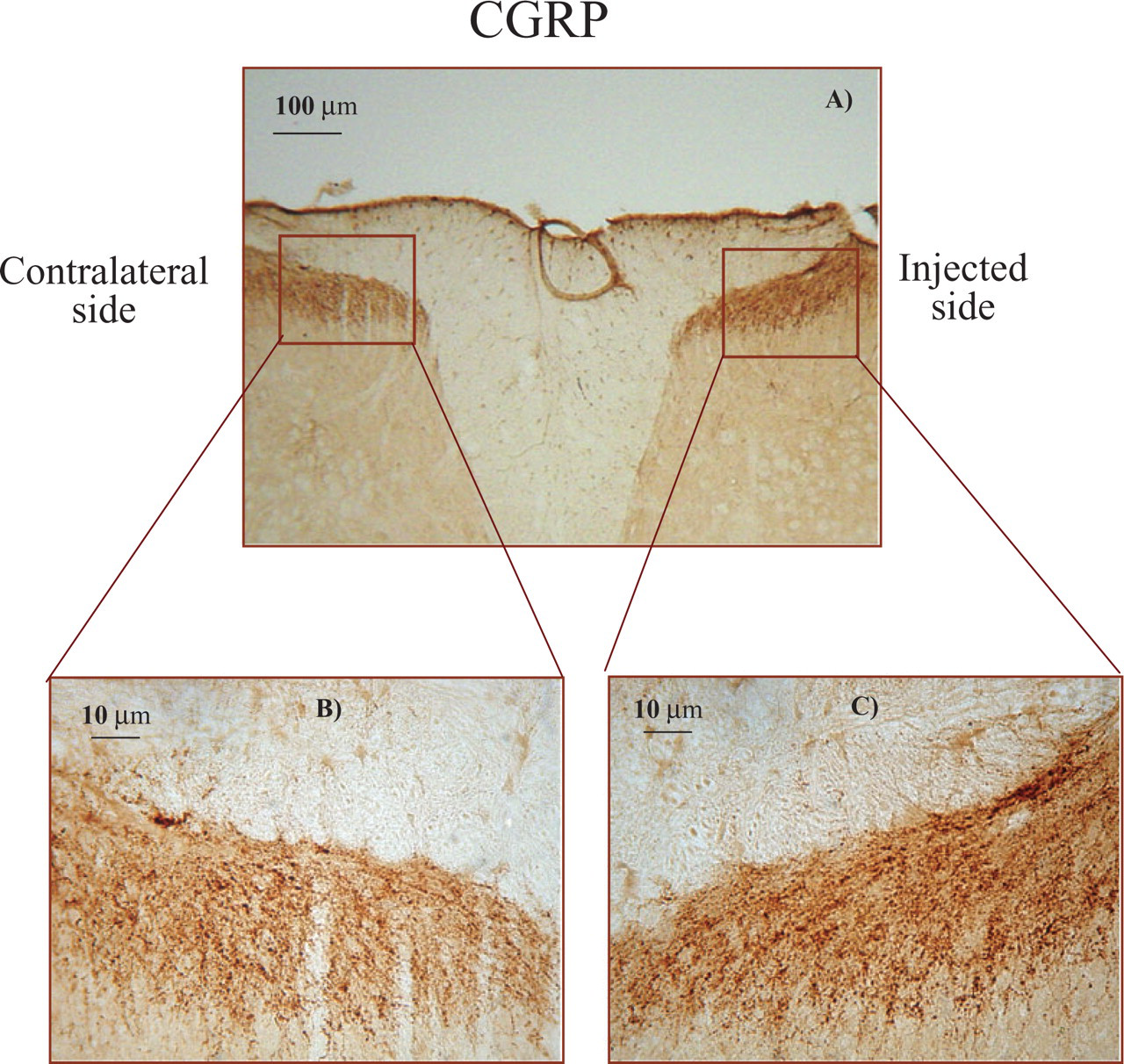
Photomicrographs of representative sections of the lumbar dorsal horns stained immunohistochemically for calcitonin gene-related peptide (CGRP) in the contralateral and ipsilateral sides relative to the site of injection of formalin 1 h before. (A) Low-power photomicrograph of the lumbar dorsal horn. Scale bar, 100 μm. (B,C) High-power photomicrographs of the two sides of the section illustrated in (A), scale bar, 10 μm.

Photomicrograph of representative section of the lumbar dorsal horns stained immunohistochemically for substance P (SP) in contralateral and ipsilateral sides relative to the site of injection of formalin 1 h before. Scale bar, 100 μm.

Quantitative analysis of the changes in the number of calcitonin gene-related peptide (CGRP)- (A) and substance P (SP)-immunoreactive (B) varicosities in the lumbar dorsal horns of animals injected with formalin in the right hindpaw. Histograms show the means number of varicosities ± SD on the side ipsilateral (ipsi) and contralateral (contra) to the stimulated side. ∗P < 0.05 vs. controls.
Discussion
Different animal models are used extensively in basic pain research to shed light on the complex mechanisms of pain transmission in physiological and pathological conditions and to predict the potency and efficacy of the pharmacological actions of different drugs (18). Although numerous studies have been conducted in the attempt to advance our understanding of the neurochemical changes in the spinal cord that are involved in the generation and maintenance of the hyperalgesic state, these mechanisms are still not fully understood. Increased release of CGRP and SP in the spinal cord may contribute to the generation and maintenance of hyperalgesia, which is attributable to sensitization of peripheral receptors and of spinal neurons. In this study we have reported specific changes in the immunohistochemically detectable levels of these neuropeptides in an animal model (NTG administration) that is specific for primary headaches, migraine in particular (17, 18), and in an animal model of spinal hyperalgesia (formalin test).
Nitroglycerin model
A decrease was found in the density of CGRP-immunoreactive fibres in laminae I and II of the NTC (lower brainstem and cervical spinal cord) associated with a decrease in the number of CGRP-positive varicosities following NTG administration. This decrease in CGRP-ir lasted for the entire observation period (4 h) and is possibly related to the release of the neuropeptide from the storage vesicles, as suggested by the observed decrease in the number of CGRP-positive varicosities. These data support both the findings of Pardutz et al. (31), who showed a decrease in the size of the area occupied by CGRP-immunoreactive fibres in the NTC 4 h after the administration of NTG, and previous reports demonstrating that NTG evokes the release of CGRP from vascular tissue and causes sensory fibres to release the neuropeptide (32, 33). More recently, Bellamy et al. (34) have demonstrated that NO can directly stimulate release of CGRP from trigeminal ganglia neurons. Furthermore, a clinical study has revealed a strong correlation between plasma CGRP concentrations and severity of migraine headaches induced by NTG administration (35). Taken together, these findings strongly suggest that NTG administration may cause significant and sustained release of CGRP from trigeminal afferents. We speculate that this phenomenon may be related to the activation of trigeminovascular fibres (17, 35, 36). A possible alternative mechanism could be NTG-induced activation of endogenous NO synthesis, as suggested by the previously reported increase in nitric oxide synthase activity in the cervical spinal cord following NTG administration (31, 37) and by the demonstration that NTG accumulates in the rat brain following systemic administration (38).
NTG induces a condition of hyperalgesia, which is detectable in the formalin test and in the tail flick test (13, 14), 4 h after its administration. The time course of the evolution of CGRP-ir suggests that the neuropeptide is involved in the development of NTG-induced hyperalgesia.
NTG causes a moderate-to-marked decrease in blood pressure that is concomitant with an increase in plasma concentrations of CGRP (39). We have previously reported that NTG causes an increase in cGMP immunoreactivity in the NTC and in the superficial cortical arterioles 2, 3 and 4 h after drug administration (40). Garry et al. (4) have reported a positive association between increasing levels of cGMP in the dorsal horn and increased release of CGRP from slices of the dorsal horn after stimulation with sodium nitroprusside. Therefore, theoretically, the same mediators (CGRP and cGMP) may be involved in NTG-induced hypotension and hyperalgesia. At present, however, evidence is lacking as regards the possible influence of NTG-induced hypotension per se on the development of hyperalgesia.
At variance with the changes observed in CGRP staining, SP staining showed a short-lasting increase in the NTC following NTG administration. Previous reports have suggested that CGRP and SP are co-released in nociception (41) and during neurogenic inflammation (42, 43). Furthermore, previous reports have shown that stimulation of the trigeminal ganglion in the rat causes a reduction in SP-ir and CGRP-ir in the NTC, ipsilateral to the stimulated side (44, 45). Therefore, the differential direction of the changes in immunoreactive densities of the two neuropeptides here reported is somewhat surprising. Previous studies have reported an accumulation of SP in the neuronal terminal following nociceptive stimulation. For example, Kantner et al. (46) have shown an increase in the level of SP in the dorsal horn in response to formalin injection, probably as a consequence of increased electrical activity due to activation of free nerve endings or rapid axoplasmic transport. Also, Zhang et al. (47) have reported an increase in SP immunoreactivity in the lumbar dorsal horn in response to formalin injection and interpreted their findings as the result of an increased biosynthesis, transport and release of these peptides in primary afferents and spinal intrinsic neurons in response to long-lasting inflow of noxious messages.
More recent data show that SP rapidly desensitizes NK1 receptors, which are quickly internalized in second order trigeminal neurons (28, 48); therefore, we can speculate that SP synthesized in first order trigeminal neurons, ultimately may accumulate in the terminals, before recycling takes place (49). Further studies, aimed at evaluating earlier time points, are necessary in order to verify this hypothesis. Furthermore, the differential behaviour of CGRP and SP in this animal model of migraine may have a neuroanatomical basis in the partial segregation of the two neuropeptides in the trigeminal ganglion, where more than half of the CGRP-positive neurons are not SP-positive (50, 51).
Considering the timing of the changes observed in this and in previous studies (13–17), it seems likely that SP is released as a result of the activation of the trigeminovascular system in response to the rapid vasodilatory changes induced by NTG at the meningeal level. This peptide is therefore not likely to play a direct role in the NTG-induced hyperalgesia observed at later time points (4 h). Transposed to the clinical field, these findings allow us to speculate that SP may be involved in the immediate, non-specific headache response to NTG, but this neuropeptide plays only a minor role in the specific (migraine-like), delayed response (11, 52). This hypothesis seems consistent with the failure of NK1 receptor antagonists to abort migraine (5, 53) and with the failure to detect any changes in SP plasma levels following NTG-induced headache (54).
Conversely, the long-lasting involvement of CGRP fits in with the recently demonstrated efficacy of a CGRP antagonist in the symptomatic treatment of migraine attacks (55) and with the reported increase in plasma levels of CGRP associated with NTG-induced headache (56).
In the lumbar spinal cord, NTG administration did not induce any significant change in CGRP staining, thus confirming the specificity of this model for evaluating phenomena related to cephalic pain and, in particular, the selectivity of CGRP in the modulation of cephalic pain (57). In contrast, SP staining in the lumbar spinal cord was significantly reduced 1 h after the administration of NTG. This may ensue from the local formation of NO from NTG and its interaction with glutamate (58), which might ultimately cause a release of SP (59, 60).
Formalin test
The formalin test was used in this study for the comparative analysis of CGRP and SP behaviour in a model of extracephalic pain. It is well known that injection of formalin into the hindpaw causes peripheral and central sensitization of dorsal horn neurons and evokes Fos expression in the spinal cord (19). Previous reports, based on different nociceptive models, have demonstrated that CGRP and SP are released in the lumbar spinal cord following noxious stimulation (61, 62). Our findings are very much in agreement with the data from the literature, as they show a significant, simultaneous and short-lasting increase in both SP-ir and CGRP-ir, paralleled by a similar increase in SP+ and CGRP+ varicosities in the dorsal horn of the lumbar spinal cord, ipsilateral to the injection site of formalin (63–65). This increase has been interpreted as a consequence of an increased axonal transport and, in the case of SP, may be associated with phenomena of receptor desensitization (66, 67).
The formalin test did not influence SP-ir in the NTC, where we instead detected a decrease in CGRP-ir. This decrease may reflect a transient release of the peptide at the medullary/cervical level of the spinal cord in response to heterosegmental stimuli, thus raising the interesting possibility of an effect of the descending inhibitory control system on CGRP release (29). In a previous report, the coeruleospinal modulation system has actually been shown to play a role in suppressing hyperexcitability of nociceptive dorsal horn neurons extended through the propriospinal pathways during inflammation (68).
Unfortunately, available literature data on this aspect are currently so limited that it is not possible to advance an evidence-based hypothesis.
Conclusions
The data obtained show specific neurochemical changes at different levels of the neuraxis in different models of pain. NTG administration selectively induces significant and sustained changes in CGRP-ir in areas of the brain involved in the transmission and modulation of cephalic pain, whereas SP-ir changes are shorter lasting and less anatomically specific. In the cephalic/cervical segment, CGRP emerges as a likely candidate for a role in NTG-induced hyperalgesia, whereas SP may play a role in the early phase of the response to pain, but not in the maintenance of central sensitization, as other authors have already suggested (42). The effect of the formalin test on the dynamics of CGRP and SP suggests a parallel and synchronous involvement of peptides at lumbar levels.
Footnotes
Acknowledgements
This study has been funded by Eurohead project (LSHM-CT-2004-504837).
