Abstract
Background:
Chlorpyrifos (CP) is an organophosphorus pesticide that induces oxidative stress through the production of free radicals and depletes intracellular antioxidant reserves. In this study, the efficacy of three antioxidants (melatonin, coenzyme Q10 (CoQ10), and vinpocetine) on alleviation of toxic effects of CP was evaluated.
Materials and Methods:
Cytotoxicity of CP, in the presence or absence of effective doses of melatonin, CoQ10, and vinpocetine, was determined in human peripheral blood lymphocytes after 72-h exposure. The levels of acetylcholinesterase (AChE) activity along with tumor necrosis factor α (TNF-α), as inflammatory index, were measured. Further, the viability and oxidative stress markers including cellular mitochondrial activity, cell death modes (apoptosis vs. necrosis), total antioxidant power (TAP), total thiol molecules (TTM), lipid peroxidation (LPO), and myeloperoxidase (MPO) activity were measured.
Results:
CoQ10 and also the combination of the three antioxidants were the most notable in opposing toxicity of CP and led to increasing TAP and TTM; improvement of AChE activity; and lowering LPO, MPO, TNF-α, and apoptosis compared to CP alone.
Conclusion:
CP toxicity overwhelms the intracellular antioxidant defense mechanisms. Exogenous supplementation with antioxidants, such as the ones we have investigated, seems to be effective in the prevention of cytotoxicity of CP.
Introduction
Organophosphorus pesticides (OPs), widely used in agriculture and other purposes, count as one of the main factors in environmental contamination.
1
One of the most frequently used OPs is chlorpyrifos [
Recent studies have demonstrated that toxic effects of OPs result from induction of oxidative stress and consequently an increase of reactive oxygen species (ROS), lipid and protein oxidation, and direct cellular DNA damage that lead to inflammation and cellular apoptosis. 6 –8 In addition to that, CP and other OPs may inhibit mitochondrial adenosine triphosphate (ATP) synthesis, which can increase ROS generation. 9 As mentioned, lymphocytes are cells that are affected by OPs and consequent oxidation. 10 Tumor necrosis factor α (TNF-α) as an important endogenous inflammatory mediator excreted from lymphocytes and monocytes, by itself, can induce oxidative stress and cellular apoptosis. 11
To counteract oxidative agents, the primary defense mechanism is an internal cellular antioxidant mechanism, including enzymatic and nonenzymatic antioxidant systems. 2 These systems work by scavenging free radicals and ROS. CP lowers the antioxidant potential of the cells by overcoming intracellular antioxidant reserves. 12 Therefore, it seems that providing cells with exogenous antioxidant resources can have protective properties against toxic effects of CP. For example, previous studies have shown that exogenous antioxidants such as vitamin A, C, and D or zinc can decrease OP-induced lipid peroxidation (LPO), ROS production, and DNA damage. 13 –17
Melatonin (
Coenzyme Q10 (CoQ10), which is a component of the mitochondrial chain, has been known for its antioxidant role since many years. Its reduced form (CoQ10H2) behaves as a potent antioxidant by protecting phospholipids from peroxidation and keeping membrane proteins and DNA resistant to oxidative damage. 19
Vinpocetine (eburnamenine-14-carboxylic acid ethyl ester) is a synthetic vincamine derivative designed for the treatment of central nervous system and cognitive disorders. In these disorders, oxidative stress plays a major role and it is assumed that the effect of vinpocetine is mainly due to its antioxidant properties (ROS scavenging) in addition to nootropic effects. 20,21
Regarding the above explanations, the aim of the present study was to investigate the antioxidant properties of melatonin, CoQ10, and vinpocetine in an in vitro setting on human lymphocytes in the presence of CP by performing biochemical assays including AChE activity, TNF-α release, total antioxidant power (TAP), total thiol molecules (TTM), LPO, and myeloperoxidase (MPO) activity and additionally assay of cells viability, mitochondrial activity, and cell death type (apoptosis and necrosis). Our data indicate that exogenous supplementation with these three compounds protects cells from CP’s toxicity in the human lymphocytes.
Materials and methods
Chemicals
All of chemicals were purchased from Sigma-Aldrich Chemie (Steinheim, Germany) unless otherwise stated. Human specific TNF-α ELISA kit from Bender MedSystems® (Vienna, Austria) and ApoFlowEx® FITC Kit from Exbio (Vestec, Czech Republic) were used. CoQ10 and vinpocetine were purchased from the Osveh Pharmaceutical Company (Tehran, Iran) and Advance Physician Formulas (Irvine, California, USA), respectively.
Cell culture
This part of the study was approved by applying all ethical aspects according to Institute Review Board’s approval with code number 90-04-151-16052. The venous blood samples were collected from healthy volunteers into Vacutainer™ tubes containing sodium heparin as an anticoagulant. Then human lymphocytes were isolated using Ficoll-Paque density gradients and cultured as described previously.
22
Briefly, blood was mixed with Ficoll-Paque and centrifuged at 400
Treatment conditions
According to previous studies, 23,24 we used peripheral blood lymphocytes exposed to 12 μg/ml of CP to induce oxidative stress. In this manner, cell culture was incubated with 12 μg/ml CP for 72 hs at 37°C and 5% carbon dioxide (CO2) humidified atmosphere. For protective treatment, optimization of dose was done by pretreating CP-induced cells with various concentrations of melatonin (0, 50, 100, 200, 400, and 800 μM), CoQ10 (0, 25, 50, 100, 200, and 400 μM), and vinpocetine (0, 0.005, 0.05, 0.5, 5, and 50 μM) for 72 h to reach the median effective doses (ED50). The 3-4,5-dimethyl thiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT) assay was used to calculate the concentration of the mentioned drugs that could provide 50% increase in cell viability. After determining their ED50, lymphocytes were divided into six, nontreated control and experimental groups that are shown in Table 1. Then the lymphocytes were incubated at 37°C and 5% CO2 humidified atmosphere. After a 72-h period, the cell suspension in all groups was centrifuged. The supernatant was collected for the biochemical assays and the deposited cells were used in viability and cell-death (apoptosis vs. necrosis) assays.
Treatment conditions of experimental groups.
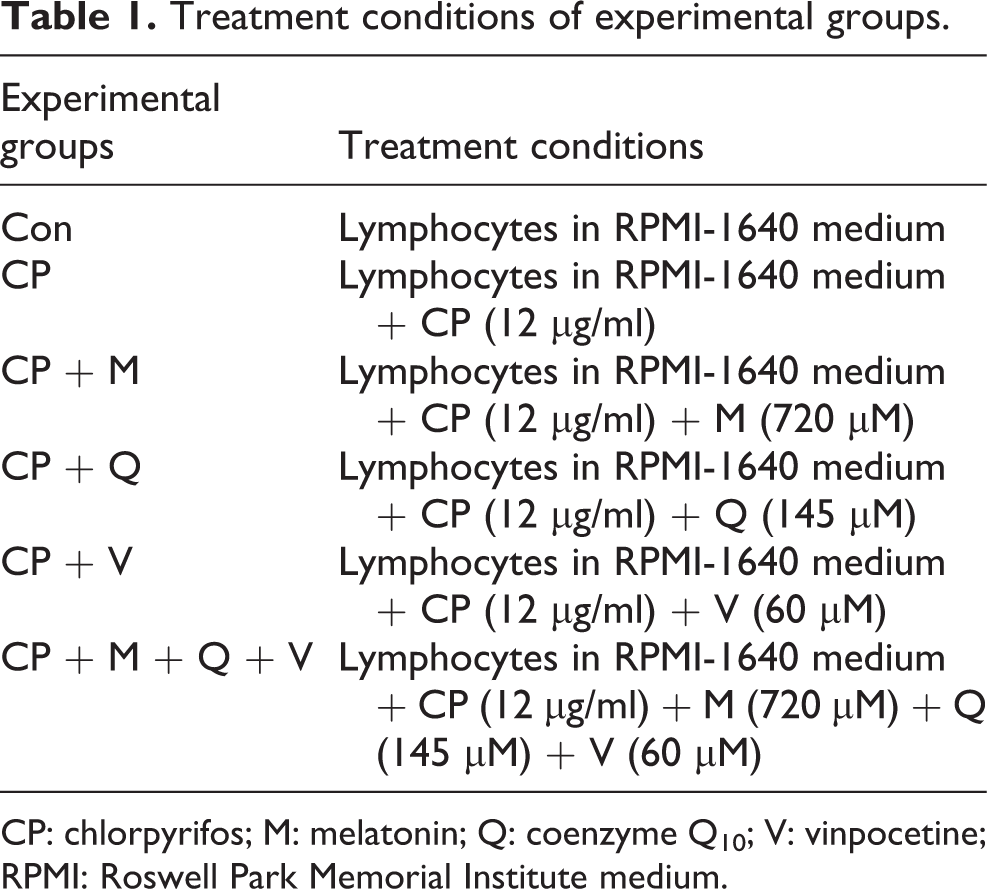
CP: chlorpyrifos; M: melatonin; Q: coenzyme Q10; V: vinpocetine; RPMI: Roswell Park Memorial Institute medium.
AChE activity assay
AChE activity in lymphocytes was measured according to the modified Ellman method using acetylthiocholine iodide as the substrate and 5-5-bis dithionitrobenzoic acid as a coloring agent. 25 Enzyme activity was expressed as unit per milligram protein.
Determination of TNF-α
A human-specific ELISA kit (BenderMed System, Austria) was used to quantify TNF-α in the supernatant of lymphocyte culture. To assess the amount of TNF-α, the absorbance of the sample was measured at 450 nm as the primary wavelength and 620 nm as the reference wavelength by the ELISA reader as described in the kit brochure. Data were shown as milligram per milliliter.
Determination of TAP
The method is based on the reduction of ferric (Fe3+) tripyridyltriazine (TPTZ) complex (colorless) to Fe2+ TPTZ (blue colored) formed by the action of the electron-donating antioxidants at low pH. The ferric reducing antioxidant power reagent was prepared by mixing 300 mM acetate buffer, 10 ml TPTZ in 40 mM hydrochloric acid, and 20 mM ferric chloride in the proportion of 10:1:1 at 37°C. Ten microliters of supernatant of sample was then added to 300 μl freshly prepared reagent warmed at 37°C. An intense blue color complex was formed when Fe3+ TPTZ complex was reduced to Fe2+ form, and the absorbance at 593 nm was recorded against a reagent blank after 30 min incubation at 37°C. Data were shown as millimole per microgram protein. 26
Determination of TTMs
To determine TTM in the control and test groups, 0.6 ml Tris-ethylenediaminetetraacetic acid (EDTA) buffer (Tris base: 0.25 M; EDTA: 20 mM; pH 8.2) was added to 0.2 ml of supernatant, and after quick vortex mixing, 40 μl 5,5ʹ-dithiobis-2-nitrobenzoic acid (10 mM in pure methanol) was added. The final volume of this mixture was made up to 4.0 ml by an extra addition of pure methanol. After 15 min incubation at room temperature, the samples were centrifuged at 3000
Determination of LPO
To measure LPO, thiobarbituric acid-reactive substances were measured. Thiobarbituric acid reacts with lipid peroxides in the samples producing a measurable pink color that has an absorbance at 532 nm, as described in our previous work. 28 The activity was shown as millimolar.
Determination of MPO activity
To assay MPO activity, we measured it spectrophotometrically as follows: 0.1 ml of supernatant was added to 2.9 ml of 50 mM phosphate buffer containing 0.167 mg/ml
Determination of cell death (apoptosis vs. necrosis)
To find out the mode of lymphocyte cell death induced by CP in the presence and absence of melatonin, CoQ10, vinpocetine, and their combination, the Annexin V-FITC/propidium iodide (PI) staining was carried out. The staining of Annexin V-FITC and PI indicates the type of death induced by the test compound, that is, apoptosis or necrosis. Annexin V binding was used as an indicator of phosphatidyl serine surface exposure in early apoptotic cells and PI staining as the necrosis indicator. Presently, the most widely used analytical assesses are based on checking the translocation of phosphatidylserine from inner phospholipid layer to the cell surface by use of a fluorochrome-labelled Annexin V in combination with appropriate vital dyes. ApoFlowEx® FITC kit is based on standard setup that employs Annexin V-FITC conjugate and PI. The flow cytometry test can discriminate intact cells (Annexin V−/PI−), early apoptotic cells (Annexin V+/PI−), late apoptosis cells (Annexin V+/PI+), and necrotic cells (Annexin V−/PI+). Human lymphocytes (1 × 106/200 µl) were incubated with indicated concentrations of CP and antioxidants for 72 h, washed, and stained with Annexin V-FITC/PI as per the instructions given by the manufacturer. The cells were scanned for fluorescence intensity in FL-1 (FITC) and FL-2 (PI) channels. The fraction of cell populations in different quadrants was analyzed using quadrant statistics. The values shown in the lower left, lower right, upper left, and upper right quadrants of each panel represent the percentage of viable, early apoptotic, necrotic, and late apoptotic (post-apoptotic necrotic) cells, respectively. 30 Flow cytometry analysis was performed with a flow cytometer (Apogee, UK).
Lymphocyte’s viability assay
The assay is based on the conversion of yellow, water-soluble MTT to the purple, water insoluble end product, formazan, by mitochondrial dehydrogenase. The amount of formazan formed is proportional to the number of metabolically active cells. 31 After 72-h incubation and centrifugation, the precipitated lymphocytes were washed twice by phosphate buffer. Then, 50 µl of MTT solution was added. After 4 h incubation at 37°C and 5% CO2 humidified atmosphere, colored crystals of formazan were dissolved with 150 µl of dimethyl sulfoxide solution. The plates were kept on a orbital shaker for 10 min and optical density was read on a multiwell scanning spectrophotometer (ELISA reader) at 570 nm. The viability of the treatment groups was expressed as the percentage of control which put on 100%.
Protein assay
To determine total protein concentration of cells, Bradford reagent was added to dilute samples and the absorbance was measured by the spectrophotometer at 595 nm after 5 min. The bovine serum albumin was used as the standard. 32
Statistical analysis
At least four independent experiments in repetition were carried out. Data are presented as mean ± SEM. One-way analysis of variance and Tukey’s multi-comparison tests were carried out by Stats-Direct 3.0.139 to determine the statistical differences while the level of significance had been set at
Results
ED50 of melatonin, CoQ10, and vinpocetine
As shown in Figure 1, by increasing the concentration of melatonin and vinpocetine, the cell viability increased and ED50 for these two compounds was estimated as 720 and 60 µM, respectively. In case of CoQ10, by increasing the dose up to 200 µM, the viability of the cells increased, but further after 200 µM, by increasing the dose, viability of cells is significantly decreased relative to the 200 µM dose. In our experiment, we found out that ED50 for CoQ10 is 145 µM.

Effects of various concentrations of melatonin (a), CoQ10 (b), and vinpocetine (c) on CP-induced cytotoxicity in human lymphocytes in vitro after 72-h incubation. Data are expressed as mean ± SEM. *
AChE activity
Regarding our data (Figure 2), the activity of AChE is substantially decreased in the CP group compared to control group (

Effects of ED50 of melatonin (M), CoQ10 (Q), vinpocetine (V), and their combination (M + Q + V) in AChE activity of isolated human lymphocytes for 72-h incubation, in the presence of CP. Data are expressed as mean ± SEM. aa
TNF-α release
As illustrated in Figure 3, a rise in TNF-α release is observed in the CP group compared with control group (
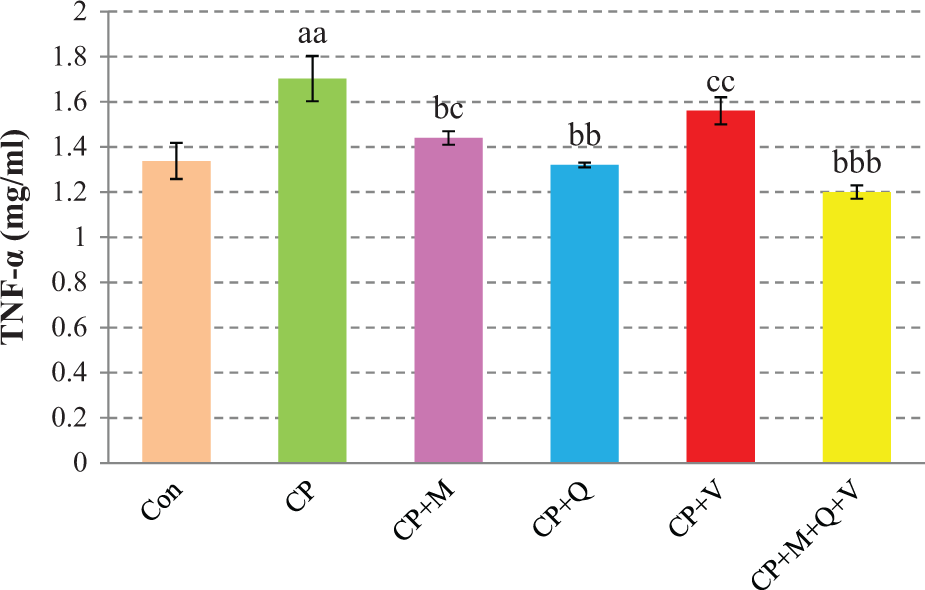
Effects of ED50 of melatonin (M), CoQ10 (Q), vinpocetine (V), and their combination (M + Q + V) in TNF-α release of isolated human lymphocytes for 72-h incubation, in the presence of CP. Data are expressed as mean ± SEM. aa
Total antioxidant power
Results in Figure 4(a) show that the activity rate of TAP in CP group is decreased in comparison to control group (

Effects of ED50 of melatonin (M), CoQ10 (Q), vinpocetine (V), and their combination (M + Q + V) in TAP values (a), TTM levels (b), MPO activity (c), and LPO levels (d) of isolated human lymphocytes for 72-h incubation, in the presence of CP. Data are expressed as mean ± SEM. a
Total thiol molecules
As seen in Figure 4(b), the level of TTM in CP group in comparison with control group decreased significantly (
MPO activity
Figure 4(c) demonstrates that the activity of MPO enzyme is boosted in the CP group compared to control group (
Lipid peroxidation
According to the results (Figure 4(d)), LPO level shows a significant elevation in the CP group compared to control group (
Lymphocyte death (apoptosis vs. necrosis)
As demonstrated in Figure 5(a) and (b), CP group showed a 21% decrease (

(a) Flow cytometric analyses of apoptosis and necrosis in human lymphocytes induced by CP, alone, or in combination with melatonin (M), CoQ10 (Q), vinpocetine (V), and their combination (M + Q + V), using Annexin V-FITC and PI double staining. Quadrant analysis of fluorescence intensity of non-gated cells in FL1 (Annexin V) vs. FL2 (PI) channels was from 5000 events. (b) Changes in the percentage of live, early, and late apoptotic cells. aa
Mitochondrial activity
The result of MTT assay after 72-h incubation time is shown in Figure 5(c). The viability of the cells in the CP group shows a significant decrease compared with control group (
Discussion
One of the most used substances with potential hazard in agriculture is pesticides specially OP compounds. CP is a broad spectrum OP that is in use for agriculture and other purposes. Both in vivo and in vitro studies have reported that CP causes neurotoxicity and immune toxicity in animals and human. 2,33,34 The major toxicity mechanism of CP is inhibition of AChE activity. Results of previous studies suggest that inhibition of AChE activity by CP may occur as a consequence of accumulation of ROS and other oxidative agents. 35 Since AChE has some effects on structural and functional modifications of various immune cell populations including lymphocytes, inhibition of that could interrupt the immune system. 36 Additionally, OPs by means of induction of oxidative stress and production of ROS could induce apoptosis in lymphocytes. 24 Results of the present study demonstrated that exposure of human lymphocytes to CP increases the cell death and the levels of oxidative stress and inflammation-related factors. However, it has been observed that pretreatment of lymphocytes with melatonin, CoQ10, and vinpocetine has a positive effect on relieving CP’s toxicity especially in the case of CoQ10 and melatonin. Additive effect was also seen in the combination of melatonin, CoQ10, and vinpocetine.
Some previous studies have shown that both in vitro and in vivo exposure of human cells to CP induces LPO 37 and increases all other markers of oxidative stress. 38 In another study on lung, liver, and brain homogenates of rat, the results indicated that malonedialdehyde is markedly increased and in the meantime, some endogenous antioxidant enzymes are inhibited. 2 On the other hand, together with LPO, the activity of the lysosomal enzyme MPO increases. MPO, as a heme protein, is an oxidant enzyme that produces inflammatory mediators that can aggravate LPO. 39 The present study shows that by use of melatonin, CoQ10, and their combination, the increment of LPO and MPO is reduced possibly through amplification of TAP and TTM.
Using Annexin V/PI staining results, it has been found that CP induces apoptosis in lymphocytes. In this respect, increased LPO and cell apoptosis in mice retina cells following CP-induced oxidative stress have been reported. 35 Meanwhile, it was shown that apoptotic effect of CP in human Jukart T-cells and U937 cell line is mediated by activation of intracellular caspase-3. 40,41 Our finding that using these three compounds (melatonin, CoQ10, and vinpocetine) in combination could increase the number of viable cells in the treated groups was a sign of their cell-protective effects.
Among the presently tested compounds, CoQ10 showed the numerically highest, significant effect on the amelioration of CP’s toxic effects. CoQ10 in humans as a lipid-soluble compound is synthesized endogenously. For many years, it was known for its important role in mitochondrial bioenergetics while recent studies demonstrated the existence of CoQ10 in other subcellular fractions and plasma. 42 The reduced form of CoQ10, ubiquinol (CoQ10H2), was identified as a potent antioxidant that protects phospholipids from peroxidation. 19,42 Studies on the association between susceptibility of LDL to peroxidation and the ubiquinol content clearly show that ubiquinol is the most active antioxidant especially in the early stages of peroxidation. Interestingly, supplementation with exogenous CoQ10 raises ubiquinol content. 42 In addition, in another experiment, in vitro supplementation of human lymphocytes with CoQ10 improved DNA resistance against H2O2-induced oxidation. 19
After CoQ10, melatonin showed a considerable effect in neutralizing the negative effect of CP in human lymphocytes. Melatonin, considering its major use as a free radical scavenger, exerts potential protection against free radical-induced DNA, protein, and membrane damage. As a result, melatonin can play an important role in treatment of naturally occurring free radical-related diseases such as diabetes, which with decreasing LPO prevents diabetes-induced nephropathy and neuropathy. 43 In a study on modulating effects of melatonin on apoptosis, it was observed that melatonin is able to reduce endogenous ROS generation induced by calcium signaling pathway in human leukocytes. Also melatonin (as an indolamine compound) demonstrated a protective effect against apoptosis-induced calcium signaling by means of remarkable reduction of caspase-9, caspase-3, and DNA fragmentation. 44 The immune-modulatory effect of melatonin is due to existence of specific binding site on lymphoid cells. In vivo studies show that human lymphoid cells are major physiologic resources of melatonin that endorses immune-modulatory effect of melatonin. 45,46
Vinpocetine is a synthetic alkaloid and a vincamine derivative, which is extracted from the periwinkle (Vinca minor) plant. Vinpocetine is known as a natural phosphodiesterase type 1 inhibitor that enhances cyclic adenosine monophosphate and current good manufacturing practice levels with potential to improve the memory and treatment of cognitive disorders in animals and human. 47 In vitro study shows that vinpocetine prohibits the upregulation of nuclear factor κB by TNF-α in human. 47 In confirmation of antiinflammatory effects of vinpocetine in an in vivo preparation, a significant decrease in polymorphonuclear neutrophil infiltration in the lung tissue was shown. 48 Due to ROS scavenging ability of vinpocetine, it can probably be used in the treatment of disorders with underlying oxidative stress such as Alzheimer’s and other neurodegenerative diseases. 21,49
In conclusion, efficacy of CoQ10 in lessening the oxidative and inflammatory factors associated with CP toxicity is more notable than those of melatonin and vinpocetine (Figure 6). Combination of these compounds also demonstrated additive effects in control of the inspected oxidative stress factors. The results of the present study support the rationale for the use of specific antioxidants in the prevention and treatment of OP toxicity. Of course this remains to be further examined in vivo and in the clinic.

Graphical picture of positive effects of CoQ10 and melatonin against CP-induced cytotoxicity in human lymphocytes. Changes induced by CoQ10 and melatonin are relative to those induced by CP alone. CoQ10: coenzyme Q10; CP: chlorpyrifos.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by a grant from Tehran University of Medical Sciences (90-04-151-16052).
