Abstract
People in the fast-food era rely on pre-packaged foods and engage in limited physical activity, which leads to a shift in eating patterns. Monosodium glutamate (MSG), a dietary ingredient used in this sort of cuisine, has been found to be hazardous to both experimental animals and humans. The objective of this study was to explore at the unnecessary changes caused by consuming MSG in secret and exceeding the recommended dosage. Hence, we decided to evaluate the impact of MSG by using three different doses (200, 400, and 600 mg/kg body weight orally) for 28 days in rats. We uncovered that all three MSG dosages result in a rise in body weight, dyslipidemia, inflammatory response, and hepato-cardiac marker enzymes, all of which imply hepatic and cardiac toxicity. Furthermore, changes in redox status suggest oxidative stress, which was higher in all three MSG dosages although not as much as in the MSG-600 group when compared to control. Such effects eventually manifested themselves in tissue architecture of the liver and heart, resulting in severe hepato-cardiac derangement, but the degree of tissue damage was greater in the MSG-600 group. As a result, it is possible that MSG has a negative influence on the liver and heart. However, the MSG-600 group showed a substantial effect, indicating that MSG should not be used in food preparation. Therefore, the findings of the study may aid in the formulation of health-care strategies and serve as a warning to the general public regarding the use of MSG in daily diet.
Keywords
Introduction
Unnecessary changes in eating habits and tasty food are directly linked to people’s health problems all around the world. People nowadays are preoccupied with their technology-based hectic work schedules in a variety of sectors, industries, and businesses. They have very little time to prepare their meals and engage in physical exercises since they live in such a fast-paced environment. As a result, the majority of them rely on fast food purchased from the neighboring eateries having flavor-enhancing chemicals like monosodium glutamate (MSG). Such practices eventually lead to a shift in their eating habits and cause the body to produce reactive oxygen species (ROS), which ultimately leads to systemic anomalies.1,2 MSG is a common food ingredient that improves the palatability of food, especially in ultra-processed foods. 2 However, food processors utilize MSG extensively, either directly or indirectly, to increase the selling of fast food in the market because they have very little information regarding the safe limit of use (i.e., 16 mg/kg body weight) and the adverse effect of it on human health. 2 Ultimately, regular consumption of such kind of food disrupts our natural physiological homeostasis.2–4 MSG alters various signaling pathways in experimental animals and humans, causing significant systemic damage. 2 However, there is no standard dosage kinetic research to determine the effects of the human equivalent dosage of MSG on the liver and heart as an alternative to a human trial.
In light of this, the current research aims to determine the impact of three different doses of MSG, as well as the most hazardous dose, on hepato-cardiac toxicity. As a result, we are interested in filling in the gaps in the study by conducting a preliminary evaluation of these three different doses of MSG on male albino rats, elucidating the probable mechanisms of these effects, and revealing the toxic dose.
Materials and methods
Reagents and chemicals
Reagents and chemicals were acquired from Sigma–Aldrich Chemicals Company (St. Louis, MO, USA). MSG (L-Glutamic Acid Monosodium Salt extrapure) was purchased from Sisco Research Laboratories Pvt. Ltd., India. All the cell culture media, buffer, reagents, and collagenase type II were purchased from Gibco (Waltham, MA, US) and all other reagents used for this study were of highest quality grade.
Method of preparation of MSG
Monosodium glutamate was dissolved in distilled water by previously published method of Banerjee et al. (2020).3,4
Experimental design
Adult male albino Wistar rats (120–125 g) were taken randomly and divided into four equal groups (n=8) and treated with or without MSG (200, 400, and 600 mg/kg body weight, orally) for 28 days shown in Figure 1. The control group of rats were treated with normal saline (10 mL/kg body weight/day, orally). Serampore College Institutional Animal Ethics Committee, West Bengal, India (Approval No. 07/P/S/IAEC/2017, 24/P/S/SC/IAEC/2019), registered under Committee for the Purpose of Control And Supervision of Experiments on Animals (CPCSEA), Government of India (Reg. No. 1946/PO/Re/S/17/CPCSEA) suggested the experimental conditions and procedure. Animals were kept in a temperature-controlled animal housing (24°C) with a 12-h light/dark cycle and free access to water. The rats in all groups were fed a control meal consisting of 71% carbohydrate, 18% protein, 7% fat, and 4% salt combination, as well as water ad libitum.3–5 Experimental design. The plan of the experiment was represented as a schematic diagram. Duration of the force feeding of monosodium glutamate was 28 days with a control group.
Blood collection and serum preparation
After the experiment was completed (on the 29th day), overnight starved rat from various groups were sedated with an intraperitoneal injection (ip) of ketamine (87 mg/kg body weight), as recommended by IAEC. A heart puncture was used to collect blood, and serum was prepared to measure several biochemical parameters.3–5
Biochemical assay
Biochemical parameters such as alanine amino transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase (ALP), triglycerides (TG), total cholesterol (TC), low density lipoprotein (LDL), high density lipoprotein (HDL), and very low-density lipoprotein (VLDL) were assessed by ACCUREX Biomedical Pvt. Ltd., Mumbai; then, the level of LDL and VLDL were calculated. 6 Atherogenic index (AI) was calculated by previously published formula. 3 Serum lactate dehydrogenase (LDH) was measured by using commercially available standard test packs (Stanbio Laboratory, USA); at 450 nm, the readings were recorded by UV double beam spectrophotometer (Shimadzu Model 1800). Creatine kinase-MB (CK-MB) was measured by using an inexpensive enzyme-linked immunosorbent assay kit at 450 nm in a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA).
Determination of cytokine level
The levels of serum TNF-α and IL-6 were determined at 450 nm by using a rat ELISA kit (Raybiotech, USA) according to the manufacturer’s instructions.
Preparation of tissue extract and isolation of hepatocytes and cardiomyocytes
With protease inhibitor cocktail, liver and heart tissue homogenates were prepared from all groups of animals for assessment of enzymatic and nonenzymatic antioxidant related to oxidative stress.3–5 Hepatocytes and cardiomyocytes were also isolated using a two-step collagenase type II digestion perfusion technique.7,8
Estimation of lipid peroxidation and nitric oxide (NO)
TBA test was used to assess the formation of thiobarbituric acid reactive substance (TBARS) as a result of lipid peroxidation, and the absorbance was measured at 530 nm using an ELISA Reader (Thermo Scientific). 9 Using the molar extinction coefficient (1.56×105 cm2/mM), the results were represented as nmoles of TBARS per milligram of protein. In aerated fluids, NO decomposes quickly to generate stable nitrite/nitrate compounds. Previously reported method used the Griess reaction to estimate nitrite buildup, and the absorbance was measured at 550 nm using an ELISA Reader (Thermo Scientific). 10 NO was measured as µmole/mg of protein in the sample.
Estimation of SOD and CAT activity
The nitroblue tetrazolium technique was used to estimate SOD activity, which is dependent on NBT hindrance being reduced by SOD. 10 The relative absorbance at 560 nm was measured and then translated to a unit of SOD activity/mg of protein.
The breakdown of H2O2 at 240 nm was used to calculate CAT using a well-established technique developed by Bhattacharjee et al. (2018). 10 The difference in absorbance per unit time was used as a measure of CAT activity, and the results were reported as U/mg of protein.
Estimation of glutathione (GSH) level
The GSH level was estimated using DTNB. At 412 nm, the absorbance of the reduced chromogen was measured spectrophotometrically. The level of GSH was calculated using a standard curve and reported as nmoles/mg of protein. 11
Estimation of protein
The protein content of the liver and heart was estimated by the Lowry method by using BSA as standard. 12
Determination of hepatocytes and cardiomyocytes viability by MTT assay
As directed by the MTT test kit’s instruction manual, the MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) test was performed to determine the viability of cells (Thermo Fisher Scientific, Waltham, MA, USA). Percentage of viable cells were calculated by the formula: (Absorbance of the treated cells/Absorbance of control cells)×100.
Histopathological analysis by hematoxylin and eosin (H & E) staining
Banerjee et al. (2020) method was adopted for H & E staining of the liver and heart.3–5 A compound microscope with camera attachment (Carl Zeiss, Primostar model) was used to examine the microscopic slides, and pictures of the various sections were taken.
Statistical analysis by Statsdirect 3.0 software
To see if the scores of different groups differed substantially, the Kruskal–Wallis nonparametric analysis of variance (ANOVA) test was used. Mann–Whitney U multiple comparison tests were used to determine the correlation between the research variables in order to assess inter-group significant differences. For statistical analysis, StatsDirect 3.0 (United Kingdom) was used. p < 0.05 was used to determine if differences were significant.3–5
Results
Monosodium glutamate induced changes in body weight and serum lipid level
In the present study, we have observed a significantly increased body weight (Figure 2(a)) in both MSG-200 and MSG-400 (p < 0.05) groups but not up to the level of MSG-600 (p < 0.01) as compared to the control. In addition, body weight was 1.35-fold higher in MSG-600 as compared to the control group and 1.16-fold and 1.1-fold higher in the MSG-600 group as compared to the MSG-200 and MSG-400 groups, respectively. Hence, it can be inferred that the MSG-600 group induces overweight in male rats as compared to the rest of the groups. Monosodium glutamate (200, 400, and 600 mg/kg body weight/day for 28 days) induced changes in body weight (a), serum lipid profile (b), and AI (c). Significance level based on the Kruskal–Wallis test [###p < 0.001. (a): Control versus MSG (200 mg/kg body weight, orally); (b): control versus MSG (400 mg/kg body weight, orally); (c): control versus MSG (600 mg/kg body weight, orally): **p < 0.01, *p < 0.05. MSG: monosodium glutamate.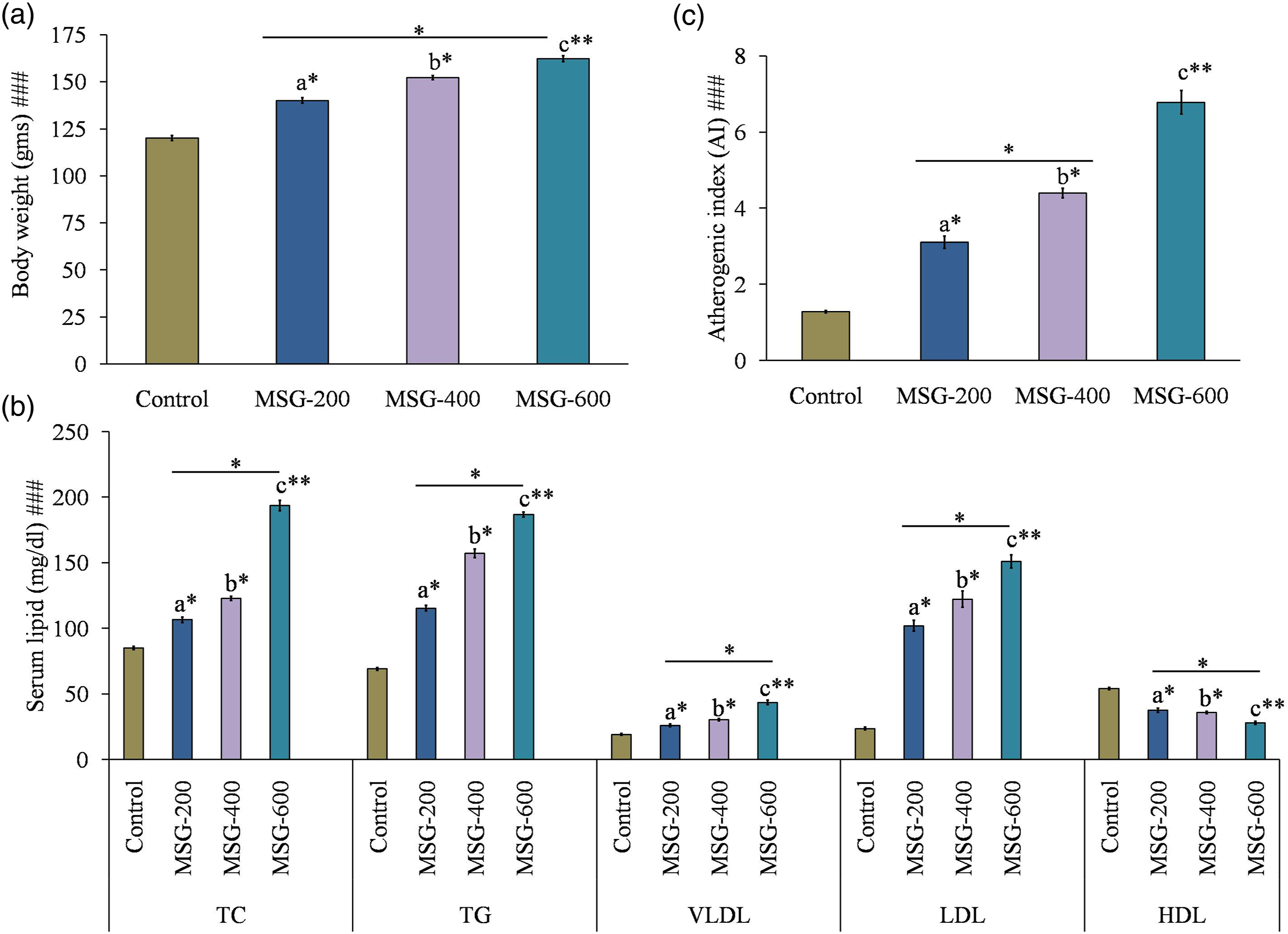
Moreover, TC, TG, VLDL, and LDL levels were 2.28-fold, 2.71-fold, 2.27-fold, and 6.43-fold higher in MSG-600 as compared to the control group; TC, TG, VLDL, and LDL levels were 1.82-fold, 1.62-fold, 1.67-fold, and 1.48-fold higher in the MSG-600 group as compared to the MSG-200 and 1.58-fold, 1.19-fold, 1.44-fold, and 1.24-fold higher than the MSG-400 group, respectively, with a significant decrease (MSG-600: p < 0.01; MSG-200, MSG-400: p < 0.05) in the HDL level (Figure 2(b)). HDL was 1.94-fold lower in MSG-600 as compared to the control group; 1.35-fold and 1.29-fold lower in MSG-600 group as compared to the MSG-200 and MSG-400 groups, respectively (Figure 2(b)). Furthermore, higher AI (Figure 2(c)) also indicates serious cardiac anomaly which was well corroborated with altered lipid profile. These data proposed that MSG may cause dyslipidemia in the rat model. The most noticeable effect (p < 0.01) was found in the MSG-600 group of animals as compared to the control group.
Monosodium glutamate induced changes in hepatic and cardiac marker enzymes
Hepatic marker enzymes ALT, AST, and ALP (Figure 3(a)) were found to be increased in each of the three experimental groups when compared with those of the control group (ALT, AST, and ALP levels were increased by 3.03-fold, 2.75-fold, and 1.47-fold in the MSG-600 group as compared to the control) where the most increased (ALT, AST, and ALP levels were increased by 1.32-fold, 1.25-fold, and 1.17-fold in MSG-600 as compared to the MSG-200 and 1.1-fold higher ALT, AST, and ALP levels in the MSG-600 group as compared to the MSG-400 group) outcome were seen in the MSG-600 group (p < 0.01). Cardiac marker enzymes LDH and CK-MB (Figure 3(b) and (c)) were similarly increased (LDH and CK-MB levels were 2.99-fold and 2.01-fold higher in MSG-600 as compared to the control) in all three concentrations of MSG-fed groups (MSG-400, MSG-600: p < 0.01; MSG-200: p < 0.05) but not up to the degree of MSG-600-fed animals (LDH and CK-MB levels were increased by 1.56-fold and 1.31-fold, and 1.24-fold and 1.16-fold in the MSG-600 group as compared to the MSG-200 and MSG-400 groups, respectively). Monosodium glutamate (200, 400, and 600 mg/kg body weight/day for 28 days) induced changes in hepatic marker enzymes (a), cardiac marker enzymes (b, c). Significance level based on the Kruskal–Wallis test [###p < 0.001]. (a): Control versus MSG (200 mg/kg body weight, orally); (b): control versus MSG (400 mg/kg body weight, orally); (c): control versus MSG (600 mg/kg body weight, orally): **p < 0.01, *p < 0.05. MSG: monosodium glutamate.
Monosodium glutamate induced inflammatory response and changes in cell viability
Monosodium glutamate plays a key role in the development of inflammation by increasing both TNF-α (Figure 4(a)) and IL-6 (Figure 4(b)) as a pro-inflammatory marker (TNF-α and IL-6 were increased by 1.55-fold and 1.36-fold in MSG-600-fed animals as compared to the control group of rats). Both TNF-α and IL-6 were significantly increased in MSG-fed rats (MSG-600: p < 0.01; MSG-200, MSG-400: p < 0.05) as compared to the control group of rats. Further, this degree of change in MSG induced inflammation by increasing the pro-inflammatory cytokines were maximum in the MSG-600 group (pro-inflammatory TNF-α and IL-6 were significantly higher in MSG-600 group by 1.38-fold and 1.27-fold as compared to the MSG-200, and 1.15-fold and 1.18-fold as compared to the MSG-400 groups, respectively). Monosodium glutamate (200, 400, and 600 mg/kg body weight/day for 28 days) induced changes in pro-inflammatory cytokines level (a, b) and cell viability (c). Significance level based on the Kruskal–Wallis test [###p < 0.001]. (a): Control versus MSG (200 mg/kg body weight, orally); (b): control versus MSG (400 mg/kg body weight, orally); (c): control versus MSG (600 mg/kg body weight, orally): **p < 0.01, *p < 0.05. MSG: monosodium glutamate.
Monosodium glutamate induced changes in hepatocytes and cardiomyocytes viability were observed by the MTT assay. The results of the present experimental setup revealed that three distinctive concentration of MSG could intensely reduce the viability of both hepatocytes and cardiomyocytes (Figure 4(c)). Hepatocellular viability were decreased from 100% (control) to 68.75% (MSG-200), 62.88% (MSG-400), and 51.13% (MSG-600) in hepatocytes, respectively; viability of cardiomyocytes were reduced from 100% (control) to 56.25% (MSG-200), 50.88% (MSG-400), and 38.88% (MSG-600). Hence, it can be suggested that cell morphology of the liver and heart was diminished with the increased concentration of MSG. The average numbers of viable cells were decreased over incubation time and with increased concentration of MSG (i.e., MSG-600).
Monosodium glutamate induced oxidative stress and histopathology of the liver and heart
Monosodium glutamate induced changes in oxidative stress-related parameters.

TBARS: thiobarbituric acid reactive substance; MSG: monosodium glutamate.
MSG (200, 400, and 600 mg/kg body weight/day for 28 days) induced changes in oxidative stress parameter of liver and heart tissue homogenates, hepatocytes, and cardiomyocytes, respectively. Significance level based on the Kruskal–Wallis test [###p < 0.001]. a: Control versus MSG (200 mg/kg body weight, orally); b: control versus MSG (400 mg/kg body weight, orally); c: control versus MSG (600 mg/kg body weight, orally): **p < 0.01, *p < 0.05.
We also looked at the histology of the liver and heart, using H & E staining to see whether there was any tissue damage. The normal architecture of the liver in the control group of animals showed no pathological alterations in the current experimental setup (Figure 5(a)). Furthermore, in MSG-fed rats, a changed radiating pattern of cell plates surrounding the central vein, central vein damage, hepatocytes with inflammatory cell infiltration, necrotic alterations, hepatocytes lysis, and hepatic tissue injury were detected and highlighted by an arrow (Figure 5(a)). When comparing the MSG-600 group to the other groups, the amount of hepatic tissue damage was greater in MSG-600. In the control group, however, there were no pathological alterations with normal cardiac architecture (Figure 5(b)). However, occasional muscle fiber loss with significant aggravation, substantial necrotic alterations of karyolysis, hyalinization of muscle filaments, necrosis of muscle fibers, mild to moderate inflammation, and cardiac tissue damage were detected and highlighted by the arrow in MSG-fed rats (Figure 5(b)). When compared to the other groups, the MSG-600 group had a greater amount of heart tissue damage. Representative photomicrograph of hematoxylin and eosin–stained section (×20) of monosodium glutamate (200, 400, and 600 mg/kg body weight/day for 28 days) induced changes in hepatic (a) and cardiac (b) tissue architecture.
Discussion
Monosodium glutamate is widely utilized as a food ingredient in a variety of ready-to-eat meals. The body tries to control the balance between endogenous antioxidants and free radicals in normal homeostasis; however, MSG increases intracellular reactive free radicals and electrophiles, which disrupts the body’s normal physiological state and leads to oxidative stress-mediated cellular damage. Pro-inflammatory responses have been related to ROS production and therefore oxidative stress-mediated systemic abnormalities, according to mounting evidence.3,4 The production of ROS increased lipid peroxidation, disrupting the intracellular antioxidant/FR balance. 13
In the present experiment, we found that MSG causes adverse effect on the liver and heart by (i) altering serum lipid profile, (ii) increasing level of hepato-cardiac marker enzyme and pro-inflammatory cytokines, and (iii) causing oxidative stress via disturbing the redox status to reduce cellular viability and tissue damage in dose-dependent manner. Moreover, the injurious impact of MSG was higher in 600 mg/kg body weight dose as compared to the rest of the groups.
We showed in the present work that MSG causes hepatic and cardiac tissue damage by causing LDH to leak as a cytoxicity marker, which is followed by a rise in ALT, AST, ALP, and CK-MB to trigger tissue damage. Furthermore, lipid peroxidation induces oxidative stress-mediated liver and cardiac damage by lowering endogenous antioxidant levels, resulting in the production of TBARS and nitric oxide. Furthermore, changes in LDL and HDL levels in MSG-fed rats reveal hyperlipidemia linked to lipid metabolic aggravation, resulting in enhanced lipogenesis and reduced lipolysis, which is consistent with previous research. 14
Monosodium glutamate promotes dyslipidemia by changing the lipid profile, oxidative stress generates inflammatory response by raising the amount of pro-inflammatory cytokines, and hepato-cardiac derangement stimulates cellular toxicity, according to our findings. MSG in all three concentrations has a negative effect on the liver and heart but not as much as MSG-600.
Increased levels of liver functional enzymes such as ALT, AST, and ALP in MSG-fed rat indicate a change in hepatocellular metabolism, which leads to hepatic damage, as previously reported. 15 Increased levels of AST, ALT, and ALP as indicators of liver functional status 16 and LDH and CK-MB as cardiac markers 17 were linked to lipid peroxidation-induced disturbance of cell membrane structure and integrity in the current study. Both hepatic and cardiac functional indicators were shown to be disrupted in rats treated with MSG-600 in this investigation. As a result, we postulated that MSG may affect hepatic and cardiac function by disrupting these marker enzymes. MSG in three different concentrations (200, 400, and 600 mg/kg body weight, orally) has been found to disrupt liver metabolism and cause liver damage. Furthermore, MSG has been directly associated with oxidative stress.3,4,13 MSG causes oxidative stress in this study by lowering SOD, CAT, and glutathione levels while increasing TBARS and NO levels in hepatic and cardiac tissue. All of these events have resulted in enhanced hepatic and cardiac damage, as seen by the other two elevated cardiac marker enzymes 17 LDH and CK-MB. Inadequate oxygen supply to the heart or MSG-induced heart damage might increase penetrability or even burst the pericardium and myocardium, causing spilling of cytosolic enzymes such as myocardial infarction (MI) markers CK-MB, AST, LDH, and ALT, resulting in increased serum concentrations. Because of the substantial rise in CK-MB in the myocardium and its virtual absence in most other tissues, the CK-MB is a major and accurate diagnostic indicator for MI. 18 Furthermore, higher AI is a good predictor for cardiovascular disorders 3 which was well supported with the increased level of cardiac marker enzymes and altered lipid profile.
Furthermore, changes in lipid concentration as well as qualitative changes in lipoprotein fractions in obesity are linked to many types of obesity-related deleterious effects.19,20 In both human and animal models of obesity, it has been hypothesized that oxidative stress and decreased endogenous antioxidant defense systems play a key role.21,22 Obesity-induced pathophysiology is thought to include lipid peroxidation. 23 The current study also found that MSG-induced obesity is triggered by increased NO production in the liver and heart, which leads to an increase in TBARS formation and, as a result, an increase in the rate of lipid peroxidation. Decrease levels of SOD and CAT and more GSH will be utilized as an acyl radical and other ROS scavenger. However, increasing TBARS levels resulted in a decrease in GSH, a free radical scavenger and membrane structure stabilizer that removes acyl peroxides produced in response to lipid peroxidation. Depletion of glutathione is a sign of tissue degradation, and the degree of depletion correlates with the severity of the damage.24–26 Obesity can also induce increased lipid peroxidation as a result of gradual and cumulative cell damage caused by the big body mass’s pressure. When cells are injured, cytokines are released, particularly IL-6 and TNF-α, which induce oxidative stress, which in turn cause lipid peroxidation. 27 Hypertriglyceridemia in obese rats may have a role in the oxidant–antioxidant balance, implying that an increase in free fatty acid bioavailability might promote lipid peroxidation. 23 As a result, oxidative stress and lipid peroxidation products stimulate stellate cells, resulting in fibrosis.28,29 Lipid peroxidation in the heart causes oxidative alteration of lipids and proteins, which can lead to arrhythmias, poor contractility, infarction, cardiac failure, or sudden death. 30 Increased lipid peroxidation in cardiac tissue may be attributed to an increase in lipid substrate inside the heart, which can serve as a bigger target for free radical oxidation.30,31
Oxidative stress and pro-inflammatory factors, on the other hand, play a major role in regulating the development of steatosis to non-alcoholic steatohepatitis, fibrosis, and cirrhosis, which leads to cardiovascular complications such as coronary heart disease.4,32 MSG-induced oxidative stress is recognized to have a significant role in the development and progression of hepatic fibrosis. MSG induces a large amount of intracellular ROS related to cellular lipid and DNA molecules in the in vivo experiment, causing lipid peroxidation by generating TBARS and a single strand to double strand break in DNA, respectively. When ROS attack polyunsaturated fatty acids in cells, aldehyde by-products such as malondialdehyde (MDA) are produced. These by-products might potentially spread from their origins to reach far-flung intracellular and extracellular targets, amplifying the effect of oxidative stress. 33 According to a recent study, α-ketoglutarate dehydrogenase, glutamate receptors, and cysteine-glutamate anti-porters all up-regulate the production of ROS in MSG-induced abnormalities and therefore oxidative stress. 34 It is possible that hepatocytes and cardiomyocytes are the main sources of oxidative stress production in response to MSG intake, as evidenced by elevated pro-inflammatory cytokines IL-6 and TNF-α.
Increased levels of IL-6 and TNF-α, which are connected to oxidative stress caused by MSG, are linked to cellular damage. The vicious cycle promotes tissue damage by increasing the levels of pro-inflammatory cytokines such as IL-6 and TNF-α. MSG promotes oxidative stress by activating intracellular transcription factors such as NF-kB, which causes inflammation. 35 On interaction of a particular sequence of the target gene’s promoter region with NF-kB, the expression level of various pro-inflammatory mediators TNF-α and IL-6 is raised.36,37 Furthermore, the viability of hepatocytes and cardiomyocytes were reduced in the current research due to elevated pro-inflammatory cytokines IL6 and TNF-α, which was well supported by an earlier report. 38 The rising dosage of MSG, particularly in the MSG-600 group, decreased cellular morphology. This finding revealed MSG’s inhibitory effect on hepatocyte and cardiomyocyte growth. Hepatocytes and cardiomyocytes had the inhibitoriest and cytotoxic effects in the MSG-600 group of rat.
The investigation suggests that MSG damages hepatic and cardiac tissue by changing lipid profile, resulting in dyslipidemia, changed cell toxicity indicators, and FR/EA, resulting in oxidative stress with inflammatory response. Our findings echo of some earlier published study, where MSG induced cellular damage by oxidative stress and inflammation via loss of mitochondrial membrane potential and activated the mitochondria-mediated apoptotic pathway to cause systemic damage.4,39–41 Furthermore, the current experimental setup revealed that all three MSG doses produce hepato-cardiac abnormalities but not to the same degree as MSG-600. In male rats, Figure 6 depicts the overall negative impact of MSG on the liver and heart. Negative impact of monosodium glutamate on the liver and heart in male rats (figure is made with biorender: https://biorender.com).
Conclusions
In this era of metabolic disorders, dietary risk factors should be examined to raise public awareness about eating habits. This endeavor is critical in combating the threat of metabolic disorders like hepatic and cardiovascular problems. The MSG content with far crossing the safer limit (i.e., MSG in 600 mg/kg body weight) was shown to be highly relevant in this investigation, and it is the primary source of systemic harm. Further studies are needed to uncover its impact on humans.
Footnotes
Acknowledgments
Authors are grateful to Prof. Dhrubajyoti Chattopadhyay (Vice Chancellor, Sister Nivedita University) for his continuous encouragement and valuable suggestions. Authors are indebted to Dr. Vansanglura (Principal, Serampore College) for his active administrative support and encouragement during the experiment. Authors are thankful to Ms. Debasmita Das, Mr. Rajarshi Paul (Department of Physiology, Serampore College), and Mr. Rathin Maji for their technical help during this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding received from University Grants Commission (UGC), Government of India, for funding this study [Memo No.F.PSW-125/15-16 (ERO)] as Minor Research Project.
