Abstract
We conducted a double-blinded, placebo-controlled, crossover study to investigate the occurrence of adverse effects such as headache as well as pain and mechanical sensitivity in pericranial muscles after oral administration of monosodium glutamate (MSG). In three sessions, 14 healthy men drank sugar-free soda that contained either MSG (75 or 150 mg/kg) or NaCl (24 mg/kg, placebo). Plasma glutamate level, pain, pressure pain thresholds and tolerance levels, blood pressure (BP), heart rate and reported adverse effects were assessed for 2 h. No muscle pain or robust changes in mechanical sensitivity were detected, but there was a significant increase in reports of headache and subjectively reported pericranial muscle tenderness after MSG. Systolic BP was elevated in the high MSG session compared with low MSG and placebo. These findings add new information to the concept of MSG headache and craniofacial pain sensitivity.
Introduction
Monosodium glutamate (MSG) is the sodium salt of the excitatory amino acid, glutamic acid. MSG is a food additive (flavour enhancer), which is widely used in the preparation of certain ethnic foods as well as in a large percentage of canned, frozen and prepared foods, especially soups, sauces, broths and dressings. MSG has long been suggested to cause headache in susceptible individuals and there is some discussion in the literature about the existence of a so-called Chinese restaurant syndrome, which may be brought on by ingestion of MSG (1–5). Chinese restaurant syndrome includes symptoms of headache, thoracic, cervical and facial tightness, palpitations, asthma, and gastrointestinal upset, and it has been suggested to more appropriately name this group of symptoms ‘the MSG symptom complex’ (6). However, the use of MSG is currently considered to be without safety concerns (7).
There is some evidence to suggest that elevated tissue concentrations of glutamate contribute to pain and sensitivity in various musculoskeletal pain conditions (8–11). In patients with ‘jumper's knee’ and ‘tennis elbow’, tissue glutamate levels are elevated compared with healthy controls (8,9), but mixed findings have been observed in patients with chronic or experimental pain in the trapezius muscle (11–13). However, local injections of high doses of glutamate (1 M, 0.2 ml) into the masseter muscle of healthy humans have been found to induce intense but short-lasting pain and increased mechanical sensitivity (14,15). Although more than one-third of an oral dose of MSG is taken up by skeletal muscle (16), interstitial glutamate concentrations obtained after systemic administration are substantially lower than those achieved by intramuscular injection of glutamate (17), and thus it is currently not known to what extent the level of glutamate needs to be increased in order to cause changes in mechanical sensitivity of skeletal muscle in humans (10).
Recently it was shown that a two- to threefold elevation of interstitial glutamate levels in the rat masseter muscle, caused by intravenously administered MSG, was sufficient to excite and induce mechanical sensitization through activation of N-methyl-D-aspartate (NMDA) receptors (10). NMDA receptors are expressed by Aδ and C-fibres, which innervate the skin, muscle and viscera (18,19). The pain and mechanical sensitivity in humans and afferent fibre discharge in rats caused by such activation may be attenuated by local administration of the NMDA receptor antagonist, ketamine (14,19,20).
The effect of systemically administered MSG on muscle pain sensitivity has not yet been examined in detail in humans. Therefore, in this three-session, double-blinded, placebo-controlled, crossover study we aimed to investigate the influence of oral administration of MSG to healthy young male volunteers on: the occurrence of headache, sensitivity to pressure pain in masseter and temporalis muscles, blood pressure, heart rate and the occurrence of side-effects. Male volunteers were chosen in order to avoid the potential confound of menstrual cycle-related variations in pain sensitivity that have been reported in some studies in women (21).
Methods
Subjects
Fourteen healthy pain-free young men [>18 years, mean age
(±
The study was performed in three sessions at least 7 days apart to avoid possible crossover effects. Subjects received financial compensation.
The study protocol was approved by the local ethics committee (Aarhus County, Denmark) and informed consent was obtained from all participants. The study was conducted in accordance with the Declaration of Helsinki in a randomized, double-blinded, placebo-controlled, crossover manner. Randomization was performed by a computer and the single examiner and all subjects were blinded with regard to the sequence of the sessions until after finishing data collection.
Baseline measurements
In each session at baseline, a blood sample was taken and the following parameters were evaluated: spontaneous pain on a 0–10 visual analogue scale (VAS), pressure pain threshold (PPT) and pressure pain tolerance (PPTol) with a pressure algometer (Somedic, Hörby, Sweden) at four sites; right masseter muscle (MAR), left masseter muscle (MAL), right anterior temporalis muscle (TAR), and left anterior temporalis muscle (TAL), systolic (SBP) and diastolic (DBP) blood pressure, and heart rate (HR) with the use of a digital blood pressure monitor (UA-767plus; A&D Medical, Abingdon, UK). PPTs and PPTols were measured at the most prominent part of the masseter muscles and at the anterior part of the temporalis with a 1 cm diameter probe and a rate of increase in pressure of 30 kPa/s (22–24). During measurements, the subjects sat comfortably in a dental chair in a quiet room and the head of the subjects was gently supported by the opposite hand of the examiner. Subjects were instructed to keep their teeth slightly apart to avoid contraction of the jaw-closing muscles during pressure stimulation (22).
Administration of MSG or placebo
All subjects came for each session after an overnight fast. In each session a drink was prepared of 400 ml sugar-free lemon soda (Sprite Zero®; The Coca-Cola Company) by a research assistant in a separate room. The beverage contained carbon dioxide, citric acid, sodium citrate, aromas, artificial sweeteners (aspartame, acesulfame potassium) and sodium benzoate. MSG [150 mg MSG per kg bodyweight (high MSG session) (25), 75 mg MSG per kg (low MSG session)] or placebo (24 mg/kg NaCl) was added to the soda. A comparable taste of ‘saltiness’ was targeted with this dose of NaCl because it has previously been reported that subjects otherwise will have no difficulty in recognizing the MSG taste (1) and thereby the blinding would be compromised. The drink was ingested over a few minutes and the subjects were asked to spontaneously report if they had any sort of side-effects. Any spontaneous remarks about the taste were noted by the investigator. No food or drinks other than water were ingested for 2 h after each drink.
At time points 15, 30, 45, 60, 90 and 120 min after MSG or placebo ingestion, spontaneous pain, PPT measured once at each of the four sites mentioned above, SBP, DBP and HR were measured. PPTol was measured only at time 30 and 60 min after ingestion to avoid unnecessary sensitization. If the subject reported pain, he was asked about the location of pain and all subjects were repeatedly reminded to report any positive or negative side-effects. All these side-effects and their duration were noted by the blinded female examiner. Thirty minutes after ingestion, a second blood sample was drawn (16). Hence, a total of six blood samples were taken from each subject, two from each session. The concentration of glutamate in the plasma from these blood samples was analysed by the use of an enzyme-based assay that measures L-glutamic acid activity with fluorescence (Amplex Red Glutamic Acid/Glutamate Oxidase Assay Kit; Molecular probes, Inc., Eugene, OR, USA). The plasma glutamate concentrations at 30 min after ingestion in the two MSG sessions are given as a percentage of the plasma glutamate concentration at 30 min in the placebo session.
As a control experiment to rule out sodium loading as an explanation for any change in blood pressure, five healthy male subjects were asked to ingest the same drink, containing an equimolar dose to high MSG of NaCl, 52 mg/kg bodyweight (equivalent to 0.89 mmol Na/kg) and the SBP, DBP and HR were followed over 2 h as the study sessions.
Statistics
The number of subjects was based on a paired-design sample size calculation. We
wished to be able to detect a 25% reduction in PPT, and the intraindividual
coefficient of variance of the psychophysical measures was estimated to 20%,
giving a minimum of 10 healthy subjects. Results are presented as means
± standard error of the mean (
The frequency of each specific side-effect was compared between sessions with McNemar's tests. Values of P < 0.05 were considered statistically significant.
Results
Plasma glutamate level
The mean baseline glutamate level (glu-level) in the three sessions was 4.2 ± 0.7 µM. At 30 min after ingestion, the glu-level increased by 556% in the high MSG session and by 395% in the low MSG session compared with baseline. In the placebo session, the glu-level at 30 min was 97.5% of the baseline level. There was a significant effect of session (P < 0.001) and time (P < 0.001) as well as a significant interaction between the two factors (P < 0.001). Post hoc tests revealed that the glu-level in the high and low MSG session was significantly increased compared with the placebo session (P < 0.001), and that the glu-level in the high MSG session was significantly higher than in the low MSG session at 30 min (P = 0.010).
Headache
None of the subjects had spontaneous pain at baseline on the days of the study. Four subjects experienced headache during the low MSG session. They scored 4, 4, 1 and 1 on the 0–10 VAS, respectively. One of these subjects also experienced headache (VAS 3.5) in the high MSG session. No spontaneous pain was experienced during the placebo session.
Pressure pain threshold and pressure pain tolerance
There were no differences between sides in baseline values of PPTs [MAL 193 ± 14 kPa, MAR 181 ± 122 kPa, TAL 225 ± 25 kPa, TAR 217 ± 21 kPa (P > 0.300)] or PPTols [MAL 461 ± 45 kPa, MAR 415 ± 36 kPa, TAL 470 ± 43 kPa, TAR 445 ± 42 kPa (P > 0.05)].
The normalized PPTs at the four muscle sites MAL, MAR, TAL and TAR were not
significantly influenced by MSG ingestion
(P > 0.089). The normalized
PPT was not changed over time at MAL or MAR, but at TAL and TAR there was a
significant main effect of time
(P < 0.004). Post hoc tests
revealed that the PPT increased over time and that it was significantly
different from baseline at 45, 60 and 90 min after MSG ingestion
(P < 0.043). No
interaction between session and time was detected
(P > 0.179) (Fig. 1). Mean (± 
Overall, the normalized PPTols at the four muscle sites were not significantly
different between sessions
(P > 0.073) (Fig. 2). The PPTol was not significantly changed over time
(P > 0.260), except at
MAL, where the PPTol was decreased at 30 and 60 min compared with
baseline ( Mean (± 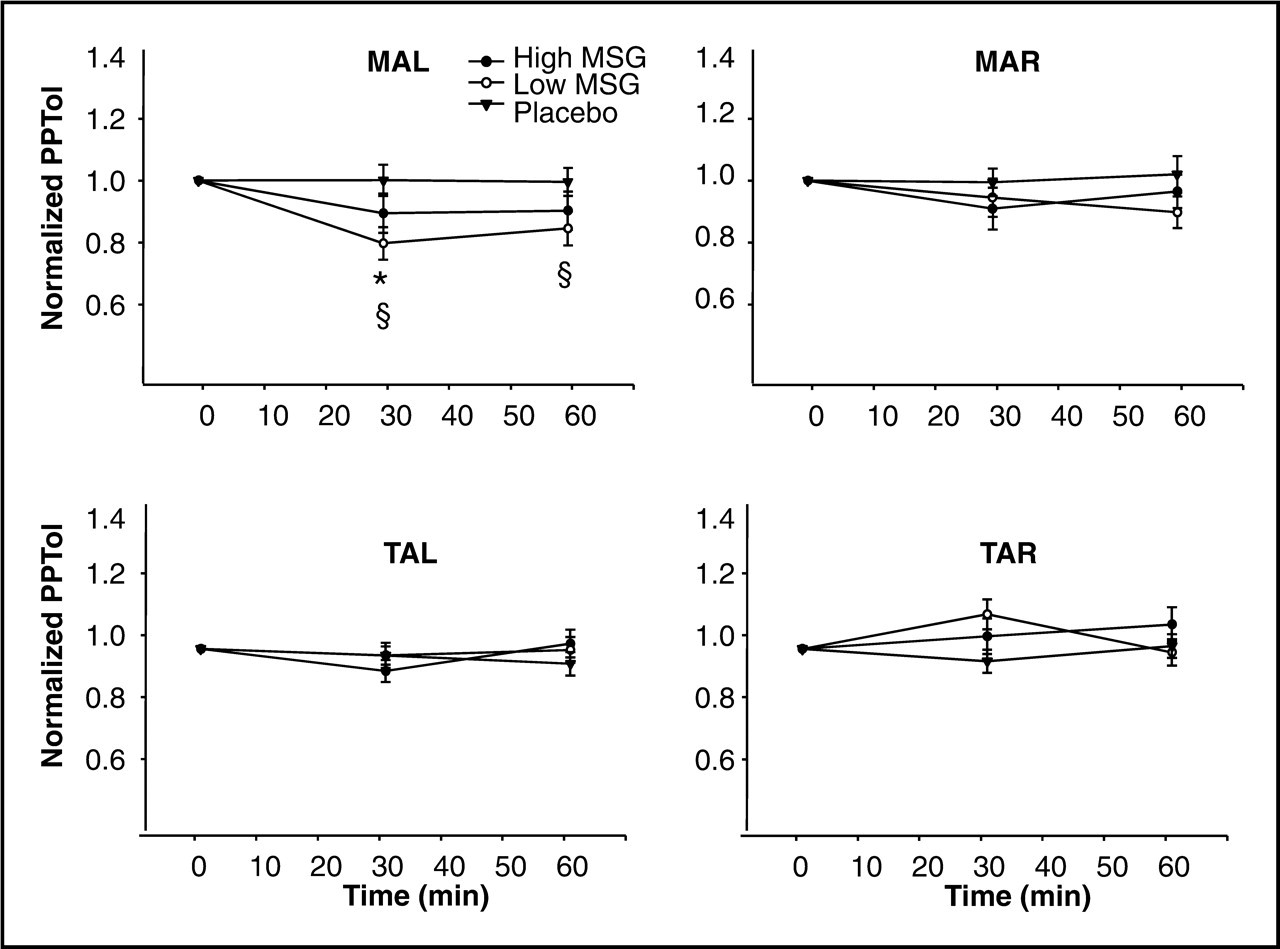
Autonomic parameters
The mean baseline SBP in the three sessions was
120 ± 3 mmHg and did not differ
between the three sessions. The normalized systolic blood pressure (SBPn) was
significantly different between sessions
(P = 0.003) and time points
(P = 0.032).
Furthermore, there was a significant two-way
session × time interaction
(P = 0.001). Post hoc
analyses revealed that SBPn was elevated in the high MSG session compared with
low MSG and placebo
(P < 0.036). In all
sessions, SBPn was lower at 60 min than at 15 min after
ingestion (P < 0.05). In
the high MSG session, SBPn was elevated compared with the low MSG session at 30,
45, 60 and 90 min after MSG ingestion
(P < 0.003) and compared
with the placebo session at 15 and 30 min after ingestion
(P < 0.001) (Fig. 3). Mean (± 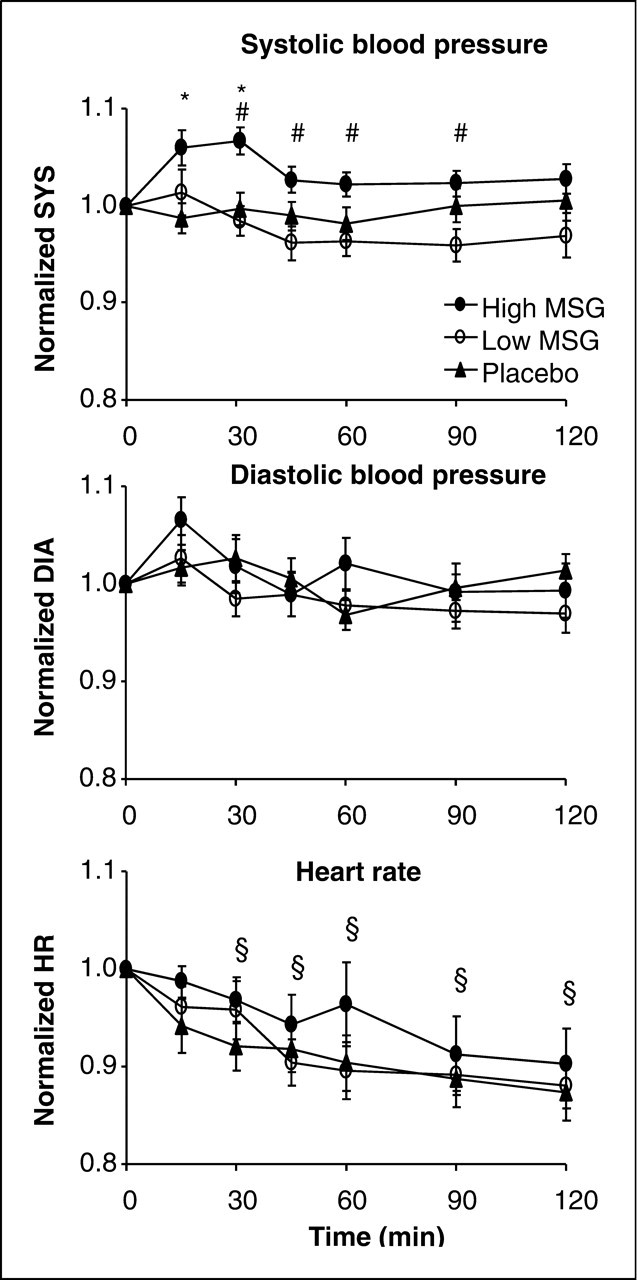
The mean baseline DBP in the three sessions was 74 ± 2 mmHg and did not differ between the three sessions. The normalized diastolic blood pressure (DBPn) was not different between sessions (P = 0.355) but varied over time (P = 0.004). Post hoc analysis showed that DBPn was significantly higher 15 min after MSG ingestion compared with the time points 45, 60, 90 and 120 min after ingestion (P < 0.035). No interactions were detected between session and time (P = 0.220) (Fig. 3).
The mean baseline HR in the three sessions was 64 ± 3 bpm. The normalized heart rate (HRn) was not significantly affected by MSG ingestion (P = 0.321), but decreased over time in all sessions (P < 0.001). At 30, 45, 60, 90 and 120 min after MSG ingestion, HRn was lower than at baseline (P < 0.017) (Fig. 3).
Control experiment
The SBPn, DBPn and HRn were not significantly influenced by ingestion of
52 mg/kg bodyweight of NaCl (dose of sodium equimolar to the high
MSG session) (P > 0.327)
(Fig. 4). Mean (± 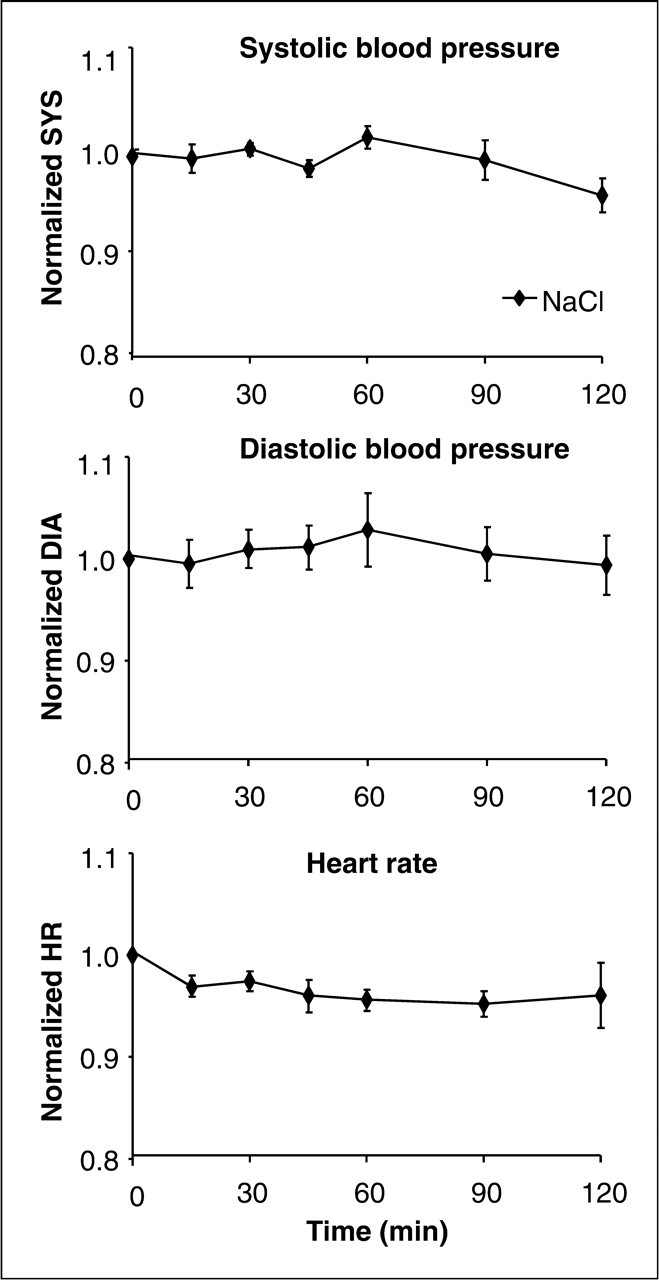
Side-effects
The mean number of side-effects reported by each subject in the three sessions
were 2.4 ± 0.4 (high MSG),
1.8 ± 0.3 (low MSG) and
0.4 ± 0.2 (placebo). Stomach ache
(P = 0.014), the feeling of
pressure/tenderness in the zygomatic region
(P = 0.008) and tender
masseter muscles
(P = 0.045) were reported
more frequently in the high MSG session than in the other two sessions. On the
other hand, headache occurred significantly more in the low MSG session,
compared with high MSG and placebo
(P = 0.045) (Fig. 5). Other side-effects such as burning skin or facial
flushing were not reported significantly more often in any of the MSG sessions
compared with placebo
(P > 0.157) (Fig. 5). Frequency of side-effects experienced in the three sessions with high
monosodium glutamate (MSG), low MSG, and placebo.
*Higher occurrence in the high-MSG session compared with
low MSG and placebo
(P < 0.045).
#Higher frequency in the low-MSG session compared with high MSG and
placebo
(P = 0.045).
Blinding procedure
The success of the blinding procedure was not tested systematically, but all spontaneous remarks about the taste of the beverage were noted by the examiner. In the high MSG, low MSG and placebo sessions, a salty taste was reported by two, three and two subjects, respectively. A bad taste was reported by six, seven and five subjects in the three sessions.
Discussion
In this first study of the effect of systemically administered MSG on sensitivity of pericranial muscles in healthy humans, no spontaneous muscle pain or decreases in PPT were detected. This could be speculated to be due to the use of single PPT measurements instead of, for example, triplicate measurement performed at each site. Single measurements are almost always used for PPTol measurements and have also been used for evaluations of the PPT (23,24), and in this study single measurements were used in order to include four muscle sites in a limited amount of time. The present study would have been strengthened by the use of triplicate measurements at each site. However, instead of lowered PPT values, in fact, an increase consistent with an adaptation over time to the pressure stimulus was observed. Such adaptation has been reported in several other studies (22,26,27). However, there was a (minor) indication of mechanical sensitization of the pericranial muscles because a post hoc test revealed a decrease in PPTol of the left masseter muscle 30 min after ingestion of the low dose of MSG. This change, however, could not be detected in the other muscles in either of the MSG sessions. Conflicting results on intramuscular glutamate levels in clinically painful muscles do exist, indicating the involvement of complex mechanisms, such as release of other neuroactive substances (28). It has been proposed that a brief elevation in glutamate levels may trigger a cascade of events, involving different peripheral glutamate receptors and ion channels, which may alter the response properties of muscle afferent fibres (29).
Orally ingested MSG undergoes a significant first-pass effect, minimizing the amount of glutamate reaching the systemic circulation unless relatively high doses (75–150 mg/kg) are administered (16,30). Peak plasma concentration occurs 30–45 min after administration of an oral dose of 150 mg/kg glutamate, after which plasma concentration declines rapidly, returning to near pre-administration levels within 90 min post ingestion (16,30). The apparent terminal half-life is ∼30 min (16,30). Skeletal muscle content (mol/g) of glutamate parallels changes in plasma glutamate concentration, although the interstitial concentration of glutamate in skeletal muscle has not been measured after oral administration of MSG (16). In this study, plasma glutamate levels were somewhat lower than previously reported levels (16). This could probably be due to the use of different methods of analysis. Such methodological differences can result in significant differences in the estimated concentrations of glutamate. The relative increase in plasma glutamate levels after ingestion of MSG, on the other hand, is similar to earlier reports (16). In rats (male and female), intravenous administration of MSG causing a two- to threefold elevation in interstitial glutamate concentration is sufficient to induce mechanical sensitization of the masseter muscle afferent fibres through activation of peripheral NMDA receptors (10). Activation of peripheral NMDA receptors has also been demonstrated to contribute to muscle pain and mechanical sensitization produced by local injection of a high concentration of glutamate (1 M, 0.2 ml) into the human masseter muscle (14,31). Our finding of a lack of overall mechanical sensitization suggests that the 150-mg/kg dose of MSG may not have raised skeletal muscle interstitial glutamate concentrations sufficiently to cause mechanical sensitization in all our healthy male subjects. Furthermore, it may be that some individuals are more sensitive to the effects of elevated glutamate concentrations than others, as witnessed by the variability in adverse effects such as headache and craniofacial sensitivity.
Since the present study group consisted of healthy young men alone, the results may not be reflective of the response to MSG in the general population. A study group more sensitive to muscle pain, e.g. women patients with chronic musculoskeletal pain conditions such as fibromyalgia or temporomandibular disorders, may be more sensitive to MSG than our healthy male subjects, since it has been proposed that muscle afferent fibres in women are more sensitive to glutamate that those in men (29). However, in this first study using quantitative sensory testing in the form of PPTs and PPTols after MSG ingestion, we wanted to avoid the possible extra variation in sensitivity caused by fluctuations of female sex hormones (21). Data from animal studies have indicated that the temporalis and masseter muscle afferent fibres show different sensitivity to peripheral NMDA receptor activation (32). Therefore, we analysed the temporalis and masseter PPTs and PPTols separately in this study. However, pooling the PPTs from all four muscles in the interest of reducing variation (analysis not reported) did not produce a different result. No overall reductions in PPTs or PPTols after MSG or placebo ingestion could be detected in such an analysis.
Interestingly, we could demonstrate a change in SBP and DBP due to MSG ingestion. In a control experiment with an equimolar concentration of NaCl, no such elevations were present, suggesting that the blood pressure elevations were not caused by elevated sodium levels. Such blood pressure elevation caused by MSG ingestion may have implications for chronic pain patients, since functional interactions exist between the pain regulatory and cardiovascular systems (33). In healthy humans, increases in resting arterial blood pressure are related to low pain sensitivity, whereas in chronic pain patients this relationship seems to be reversed (34). To our knowledge, elevations in blood pressure have not been reported in early MSG safety evaluation studies. However, in animal studies, both intramuscular and intravenous injection of NMDA has been shown to elevate mean blood pressure significantly for 5–15 min post injection, which suggests that activation of peripheral NMDA receptors may mediate the elevated blood pressure observed in the present study (32,34). Glutamate has also been shown to vasodilate blood vessels in skeletal muscle and in the cerebral cortex through activation of peripheral NMDA receptors (31,35). Plasma glutamate concentration is higher in migraine sufferers that in healthy controls (36). Taken together, these findings suggest that the combination of increased SBP and cerebral vasodilation may, in part, explain the increased incidence of headache that occurred after ingestion of MSG.
Adverse side-effects were experienced by a large proportion of the subjects and to a significantly higher extent in the MSG sessions compared with placebo. Before inclusion in the study, none of the subjects reported any experience with reactions to MSG. A high proportion of the side-effects in this study were located in the craniofacial region, for example in the form of a sense of pressure or tenderness in the zygomatic region as spontaneously reported by half of the subjects in the high MSG session. Interestingly, this symptom is listed as being frequently associated with MSG-induced headache in the International Headache Society (IHS) Classification (5). In contrast, other symptoms described in relation to MSG-induced headache in the IHS Classification, such as facial flushing and burning sensations, did not occur significantly more frequent in the MSG sessions than in the placebo session. It is important to note that the studies cited in the IHS diagnostic criteria for MSG-induced headache mainly consist of case reports, questionnaire studies, and only very few randomized controlled trials (RCTs) (5,6,37–39). In these RCTs, MSG is not dosed according to body weight and the maximum dose of MSG is 5 g, which is fairly low compared with this study (mean body weight 80.9 kg × 150 mg/kg = 12 g). In addition, a large multicentre study with 130 self-reported MSG-sensitive persons has shown that MSG given without food may elicit more symptoms than placebo, but that these symptoms are neither persistent nor serious (1). Again, MSG was not individually dosed and the maximum dose (5 g) was low. Furthermore, the self-reported MSG-sensitive subjects were asked to report the occurrence of 10 specific predefined symptoms (feeling of general weakness, feeling of muscle tightness, feeling of muscle twitching, feeling of flushing, sweating sensation, burning sensation, headache-migraine, chest pain, palpitations, feeling of numbness-tingling), which may have biased their report of side-effects (1). In contrast, the subjects in the present study were asked to describe symptoms without ‘guidance’.
Although no systematic evaluation of the blinding procedure was performed, we believe it was successful, based on the spontaneous remarks about the taste of the drinks. Lack of successful blinding has been a problem in earlier studies, especially those performed in patients who believed they were ‘MSG sensitive’ (37,40). In addition, this study was strengthened by the fact that the MSG dose was based on the body weight of the individual subject and not a ‘standard dose’ as used in several earlier studies of side-effects of MSG ingestion (1,2,4).
Conclusion
Although no spontaneous pericranial muscle pain or statistically significant reductions in PPT levels due to MSG ingestion were detected, there was an increased occurrence of headache and side-effects such as subjectively reported muscle tenderness located in the craniofacial region. We suggest that additional studies on the effect of systemic MSG on pericranial muscle sensitivity should be performed in women and in patients with chronic musculoskeletal pain, e.g. temporomandibular disorder or fibromyalgia.
Footnotes
Acknowledgements
This study was financially supported by the Danish Dental Association. The authors wish to thank dental nurse Bente Haugsted for skilful assistance in the laboratory and laboratory technician Karin Trollsås for analyses of glutamate levels.
