Abstract
Ephedrine abuse has spread in many parts of the world and severely threatens human health. The mechanism of ephedrine-induced toxicity still remains unclear. This study was performed to investigate the effects of ephedrine treatment on the liver and explore the underlying mechanisms. Sprague Dawley rats were divided into saline and ephedrine groups. Rats were treated with ephedrine at 20 mg/kg or 40 mg/kg (n = 10) by oral gavage daily for 7 days. Pathological changes were examined by hematoxylin and eosin staining and terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling assay. Enzyme-linked immunosorbent assays were used to measure the liver functional markers, oxidative stress markers, and inflammatory cytokines. Real-time polymerase chain reaction and Western blot were used to measure gene and protein expression, respectively. Our data showed that ephedrine treatment increased hepatocellular cell apoptosis and impaired liver function. Moreover, ephedrine treatment increased oxidative stress and inflammatory responses, which may be due to the increase of transforming growth factor β (TGF-β)/Smad3 expression. Our study demonstrated that short-term treatment of ephedrine caused liver toxicity in rats through regulating TGF-β/Smad pathway.
Introduction
Ephedrine has been used for centuries as one component of various traditional herbal medicines for the treatment of multiple symptoms, and was recently introduced into use in the United States and Europe as a weight loss agent. 1,2 Ephedrine and pseudoephedrine are classified as sympathomimetic alkaloids. These alkaloids are structurally similar to amphetamines and have direct alpha- and beta-agonistic properties and catecholamine-releasing actions. 3 Ephedrine is purported to exert its effects through an increase in sympathetic nervous system activity and thermogenesis. Clinical cases have shown that ephedrine has potential hepatotoxicity. 4 However, the mechanism of ephedrine-induced hepatotoxicity remains to be elucidated.
In liver injury, activated Kupffer cells release a number of cytokines, such as transforming growth factor β (TGF-β) and tumor necrosis factor α (TNFα), 5 and nuclear factor kappa B (NF-κB) translocates to the nucleus, in which it binds to the promoter of target genes of pro-inflammatory cytokines. 6 These factors act on hepatic stellate cells (HSCs), which are activated in response to injury and differentiate into proliferating and migrating myofibroblast-like cells that synthesize extracellular matrix proteins as a wound-closing action. Activated HSCs are the primary collagen-producing cells in the liver. 7 TGF-β is a cytokine that promotes fibrosis and regulates conversion of quiescent HSCs into activated HSCs. 8 TGF-β promotes the process of liver fibrosis mostly by activating its downstream Smads signaling pathway and TGF-β/Smads has been considered to be a major pathway in processes of liver damage. 9 –11
Methamphetamine is reportedly being abused by millions of people worldwide. 12 Chronic use and acute methamphetamine intoxication can cause substantial medical consequences, including kidney, liver, cardiovascular, cerebrovascular, and psychiatric problems. Studies showed that methamphetamine caused liver damage through increasing oxidative stress. 13 –15 As a precursor of methamphetamine, ephedrine has attracted extensive attention in its toxicity. Studies have shown that ephedrine can produce amphetamine-like effects in rat animal models, similar to methamphetamine in some respects. 16,17 Furthermore, herb extracts containing ephedrine (equivalent to 48 mg/kg) caused neurotoxicity in rats through oxidative damage. 18 Instead of using herb extracts, this study investigated the effects of ephedrine on livers in the rat model. Moreover, the mechanism of ephedrine-induced hepatotoxicity was further explored, especially related to the TGF-β/Smad pathway.
Materials and methods
Chemicals and reagents
Caspase-3/9 activity and cytochrome-c assay kits were purchased from R&D systems (Minneapolis, Minnesota, USA). Aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin, alkaline phosphatase (ALP), interleukin 1β (IL-1β), IL-6, TNFα, glutathione peroxidase (GPx), superoxide dismutase (SOD), and malondialdehyde (MDA) assay kits were purchased from Jiancheng Institute of Biological Engineering (Nanjing, China). Terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling (TUNEL) assay kit was purchased from Roche (Mannheim, Germany). Ephedrine was purchased from National Institutes for Food and Drug Control (Beijing, China) with purity more than 99%. Ephedrine was dissolved in saline for treatment.
Experimental procedures and treatment
Male Sprague Dawley (SD) rats weighing 180–220 g were purchased from Charles River Ltd (Beijing, China). Rats were maintained in humidity (40–70%) and temperature-controlled (23 ± 2°C) facility on 12:12 h light:dark cycles under specific pathogen-free condition. Rats had free access to food and water during the experimental period. All animal procedures were approved by the Ethical Committee on Animal Care and Use of Shangluo Central Hospital. Rats were treated with ephedrine at 20 mg/kg or 40 mg/kg (n = 10) by oral gavage daily for 7 days, rats treated with saline (vehicle, n = 10) as negative control. The dosage of ephedrine was selected based on our preliminary study and literatures. 18 –20 The plasma levels (0.3–1.2 µM) after treatment with ephedrine at 4 × 25 mg/kg intraperitoneally in rats 19 were comparable with the plasma levels (0.08–0.7 µM) observed in humans with neurotoxicity resulting from ephedrine exposure. 20 Body weight was measured every day. Blood was collected from rats before euthanasia, and plasma was separated by centrifugation at 5000 r/min for 10 min.
Hematoxylin and eosin staining
After euthanasia, livers were harvested and fixed with 4% formalin for 24 h, and embedded in paraffin. Serial 5-µm sections of prefrontal cortex tissue were stained with hematoxylin and eosin (H&E) for microscopic examination. Histological analysis was performed in a blinded manner by an experienced pathologist using an Olympus BX60 microscope (Olympus, Tokyo, Japan).
TUNEL staining
TUNEL staining was performed with an in situ cell death detection kit (Roche). In brief, sections were incubated with proteinase K for 30 min at room temperature and then cleaned with phosphate-buffered saline (PBS), followed by incubating with TUNEL reaction mixture for 60 min at 37°C. After rinsing with PBS, sections were sealed with VECTASHIELD mounting medium containing 4′,6-diamidino-2-phenylindole, and photographs were taken by an Olympus BX60 microscope.
Cytochrome-c assessment
After euthanasia, livers were removed and homogenated to harvest tissue lysate. Cytochrome-c was measured by a microplate reader at 450 nm according to the manufacturer’s instructions.
Caspase activity measurement
The caspase activities were measured according to the manufacturer’s instructions. Briefly, liver tissue lysate containing 25 mg protein were mixed with the reaction buffer. The reactions were initiated after adding substrates (50 mM at the final concentration): Ac-DEVD-Amc for caspase-3, Ac-LEDH-Afc for caspase-9, followed by incubation for 2 h at 37°C. The cleavage of substrates was measured (Amc: 390/475 nm; Afc: 400/505 nm) by a microplate reader.
Enzyme-linked immunosorbent assay
AST, ALT, total bilirubin and ALP in plasma, IL-1β, IL-6, TNFα, GPx, SOD, and MDA in liver tissue lysate were measured by enzyme-linked immunosorbent assay kits according to the manufacturers’ instructions.
Real-time polymerase chain reaction
Total RNA was extracted from liver tissues using Qiagen (Qiagen, Hilden, Germany) RNeasy reagents and quantified by spectrophotometry; 0.5 µg of total RNA was reverse-transcribed into cDNA with SuperScript master mix (Bio-Rad, Hercules, CA, USA). Quantitative polymerase chain reaction was carried out in triplicate for each sample using SYBR green Supermix (Thermo Fisher, Waltham, MA, USA) with comparative C t value method to quantify the expression of target genes in different samples. β-actin was served as the internal control.
Western blot analysis
Protein was extracted from liver tissues and quantified using the bicinchoninic acid (BCA) protein assay kit (Beyotime, China). Samples with equal amounts of protein (50 μg) in 4× loading buffer were boiled for 5 min, and then subjected to electrophoresis on a 4–12% (v/v) sodium dodecyl sulfate (SDS)-polyacrylamide gel. After protein was electroblotted to a polyvinylidene difluoride (PVDF) membrane, the membrane was blocked with phosphate-buffered saline with Tween 20 (PBST) containing 1% bovine serum albumin (BSA) at room temperature for 1 h, and incubated with primary antibodies at 4°C overnight, followed by incubating with the horseradish peroxidase-conjugated secondary antibody for 1 h. After washing three times, the antigen–antibody complexes were visualized by the enhanced chemiluminescence system (PerkinElmer, Waltham, MA, USA).
Statistical analysis
Data were presented as mean ± standard deviation. All comparisons were made by one-way analysis of variance using the SigmaStat statistical software (SPSS Inc., Chicago, Illinois, USA). A value of p < 0.05 was considered significant.
Results
Ephedrine treatment caused liver damage
During the experimental period, none of the rats exhibited distinct behavioral signs of intoxication or stress. Ephedrine treatment slightly delayed the increase of rat body weight, compared to the saline group, but there was no significant difference between control and treatment groups. At the end of treatment, livers were harvested for histopathological analysis. No pathological changes were observed in the saline group. However, liver damage was observed from rats in ephedrine groups. The number of apoptotic cells was significantly higher in livers of rats treated with ephedrine, shown by TUNEL staining. Moreover, ephedrine treatment increased the release of cytochrome-c and caspase activities. Pro-apoptotic protein Bax expression increased, but anti-apoptotic protein Bcl-2 decreased after ephedrine treatment (Figure 1).
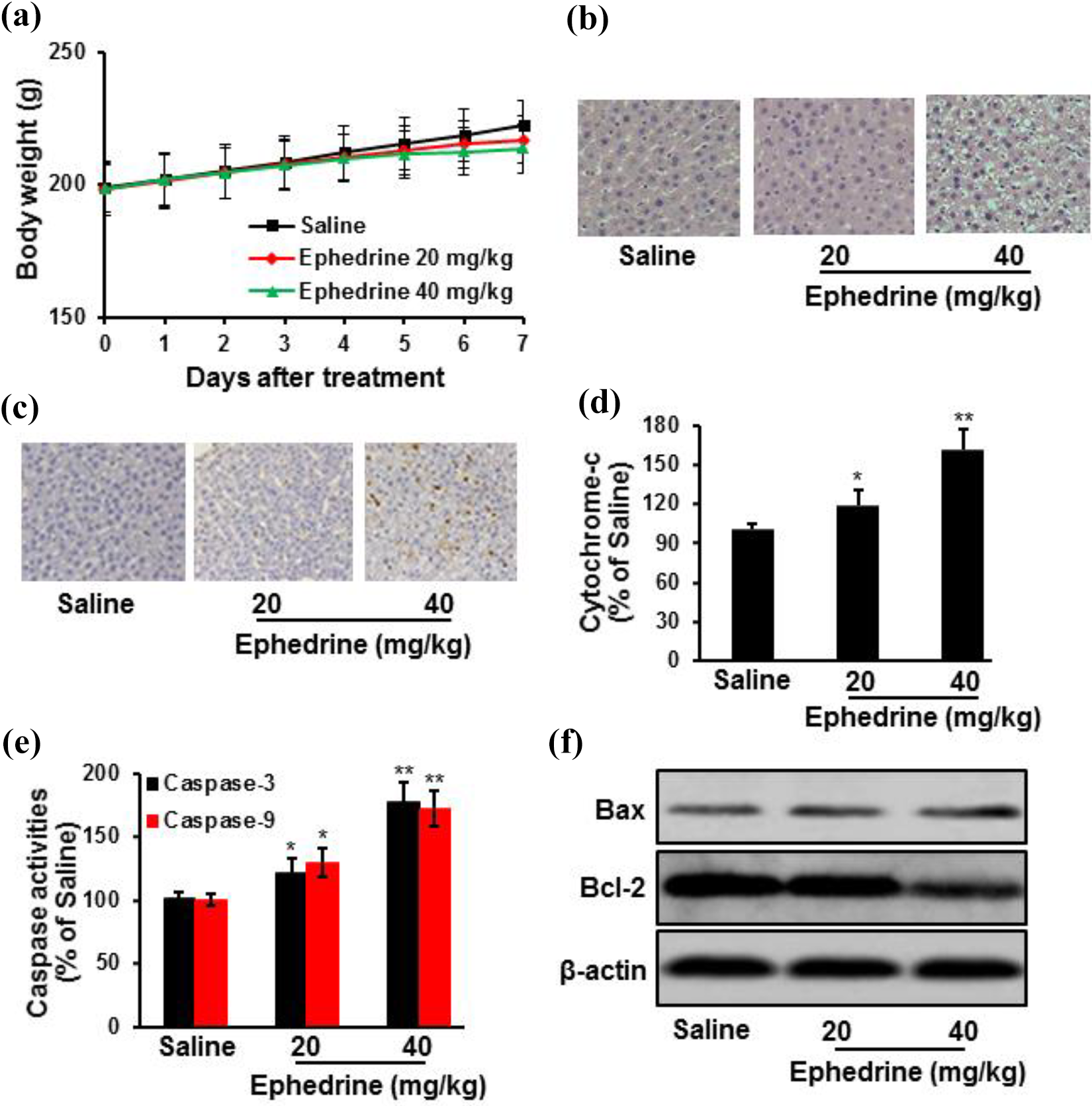
Ephedrine treatment caused liver damage in SD rats. Rats were treated with ephedrine (n = 10) or saline (n = 10) by oral gavage for 7 days. Ephedrine treatment slightly delayed the increase of rat body weight, compared to the saline group (a). Representative images of liver sections with H&E staining from rats were presented (b). Ephedrine treatment significantly increased DNA damage, shown by TUNEL staining (c), cytochrome-c release (d), and caspase activities (e) in liver tissues. Ephedrine treatment also increased pro-apoptotic protein Bax expression, while decreasing anti-apoptotic protein Bcl-2 expression (f). Data were expressed as mean ± standard deviation (n = 10). *p < 0.05; **p < 0.01 versus saline group. SD: Sprague Dawley; H&E: hematoxylin and eosin; TUNEL: terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling.
Ephedrine treatment impaired liver function
Biochemical measurements were performed using plasma obtained from rats. Results showed that AST, ALT, ALP, and total bilirubin in plasma significantly increased, indicating ephedrine treatment impaired liver function of rats (Figure 2).

Ephedrine treatment impaired liver function of SD rats. Rats were treated with ephedrine (n = 10) or saline (n = 10) by oral gavage for 7 days. Biochemical measurements were performed using plasma obtained from rats. Ephedrine treatment significantly increased (a) AST, (b) ALT, (c) ALP, and (d) total bilirubin in plasma. Data were expressed as mean ± standard deviation (n = 10). *p < 0.05; **p < 0.01 versus saline group. SD: Sprague Dawley; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase.
Ephedrine treatment increased oxidative stress
Compared to saline control, oxidative stress significantly increased after ephedrine treatment, evidenced by increased MDA level and decreased SOD as well as GPx activities, together with the increase of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 1 protein expression (Figure 3).

Ephedrine treatment increased oxidative stress in livers of SD rats. Rats were treated with ephedrine (n = 10) or saline (n = 10) by oral gavage for 7 days. Livers were harvested to measure the oxidative stress: (a) MDA content, (b) SOD and (c) GPx activities, and (d) oxidative stress-related protein expression. Data were expressed as mean ± standard deviation (n = 10). *p < 0.05; **p < 0.01 versus saline group. SD: Sprague Dawley; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathione peroxidase.
Ephedrine treatment increased inflammatory responses
Compared to saline control, IL-1β, IL-6, and TNFα levels in liver tissues significantly increased after ephedrine treatment. Ephedrine treatment also increased the protein expression of NF-κB (Figure 4).

Ephedrine treatment increased inflammatory responses in livers of SD rats. Rats were treated with ephedrine (n = 10) or saline (n = 10) by oral gavage for 7 days. Livers were harvested to measure the inflammatory factors: (a) IL-1β, (b) IL-6, and (c) TNFα levels, and (d) protein expression of NF-κB. Data were expressed as mean ± standard deviation (n = 10). *p < 0.05; **p < 0.01 versus saline group. SD: Sprague Dawley; IL: interleukin; TNFα: tumor necrosis factor α; NF-κB: nuclear factor kappa B.
Ephedrine treatment increased α-SMA, TGF-β, and Smad3 expression
Effects of ephedrine treatment on TGF-β and Smad expression in liver tissues were investigated. The mRNA expression of TGF-β and Smad3 significantly increased after ephedrine treatment, compared to saline group. Moreover, ephedrine treatment increased the protein expression of α-SMA, TGF-β, and Smad3, while decreasing Smad7 protein expression (Figure 5).
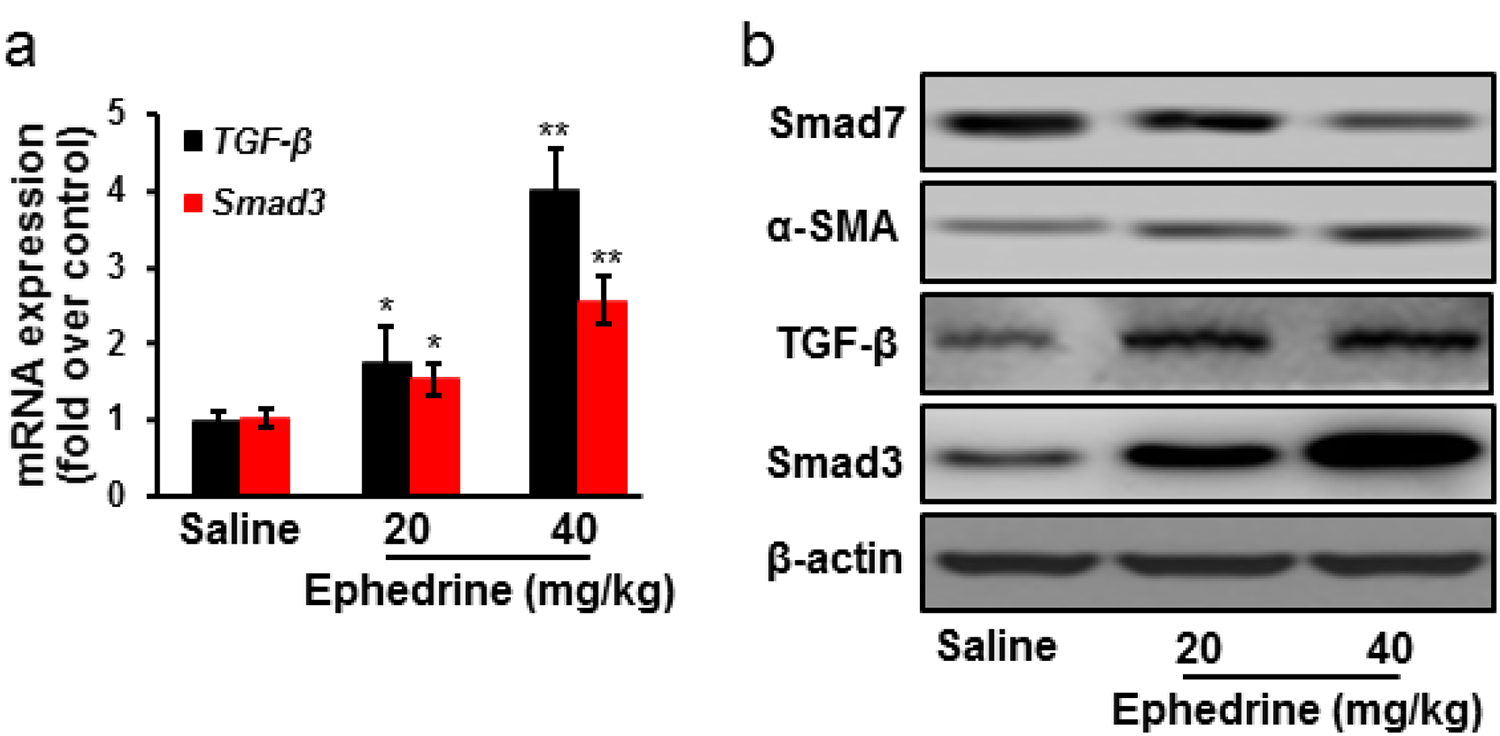
Effects of ephedrine treatment on TGF-β/Smad signaling pathway. Rats were treated with ephedrine (n = 10) or saline (n = 10) by oral gavage for 7 days. Ephedrine treatment significantly increased TGF-β and Smad3 mRNA expression (a), and increased α-SMA, TGF-β, and Smad3 protein expression, while decreasing Smad7 protein expression (b). Data were expressed as mean ± standard deviation (n = 10). *p < 0.05; **p < 0.01 versus saline group. TGF-β: transforming growth factor β.
Discussion
The toxic effects of methamphetamine on the brain are well-known and are linked to oxidative stress. However, little information is available about the oxidative stress induced by methamphetamine in other organs. As a substituted amphetamine and a structural methamphetamine analogue, we hypothesize that ephedrine also caused toxicity through oxidative stress. Lee et al. have evaluated mitochondrial toxicity/mitophagy as the proximate cause of hepatotoxicity following ephedrine treatment in vitro. 21 The exact mechanism of liver injury with ephedrine exposure is still to be examined. Therefore, in this study, a rat model was developed with ephedrine treatment over 7 days. The extent of alterations in hepatocellular morphology was examined by H&E staining and TUNEL assay. To confirm if these changes in morphology were indicative of damage, AST, ALT, bilirubin, and ALP in plasma were measured. AST and ALT are hepatocellular enzymes which are released into the blood during cell damage. 22,23 Both AST and ALT significantly increased after ephedrine treatment, consistent with the changes in cellular morphology. Comparing to the relatively small change of AST and ALT, there was more striking increase in bilirubin, which could be due to the principally apoptotic rather than necrotic responses of the hepatocytes, or due to previous depletion of the cytosolic transaminases in the earlier phase of ephedrine treatment.
Ephedrine causes a massive release of dopamine by damaging dopaminergic neurons. 19 Dopamine then reacts with molecular oxygen to form reactive oxygen species (ROS), which can easily initiate the peroxidation of membrane lipids, leading to the accumulation of lipid peroxides. Under pathological conditions, ROS are overproduced and result in oxidative stress. Imbalance between ROS and antioxidant defense mechanisms leads to oxidative modification in cellular membrane or intracellular molecules. 24,25 It has been suggested that the hepatic catalase level is negatively associated with the severity of liver injury 26 and that SOD scavenges hydroxyl peroxides generated in the cytosol and mitochondria, thereby terminating autoxidation. In this study, ephedrine imbalanced the hepatocellular antioxidant system, as evidenced by the decrease of GPx as well as SOD activities and the increased level of MDA in hepatic tissues. Moreover, ephedrine treatment increased NADPH oxidase 1 protein expression.
Hepatocyte apoptosis increases recruitment of inflammatory cells to damaged liver and release of pro-fibrogenic cytokines (TGF-β, IL-1β, IL-6, TNFα). 27 Inflammation is the main characteristic in liver fibrosis. Among pro-inflammatory mediators, IL-1β plays a key role, by increasing the synthesis of short-time pro-inflammatory mediators such as TNFα and IL-6. 28,29 TNFα is a multifunctional cytokine involved in inflammation, immunity, and a variety of diseases. TNFα also induces apoptosis in a variety of cell types. In the liver, TNFα-induced apoptosis is thought to contribute to injury from hepatotoxins. 30 As a major regulator of the acute phase response in the liver, IL-6 is also essential for liver regeneration after injury or partial hepatectomy. 31 Overproduction of the pro-inflammatory factors resulted in intracellular toxic events to cause cell death. 32 In this study, ephedrine treatment significantly increased the levels of IL-1β, IL-6, and TNFα in liver tissues. Moreover, ephedrine treatment increased NF-κB protein expression.
Bone marrow-derived and liver-resident macrophages produce TGF-β in the fibrotic liver. 27 The increasing expression of TGF-β is involved in the pathogenesis of liver fibrosis and injury, which promotes the process of liver fibrosis mostly by activating its downstream Smads signaling pathways. 10,11 As the downstream mediator of TGF-β signaling, Smad3 acts as a transcription factor to regulate the expression of collagen genes in pathological conditions. Smad7 is an inhibitory factor induced by TGF-β, which negatively regulates the fibrogenic signaling. The low expression of Smad7 in liver fibrosis may be the promotion factor to the progress of liver damage. 33,34 Therefore, we speculated that TGF-β/Smads pathway played a key role in the progress of liver injury caused by ephedrine. Our data showed TGF-β/Smad pathway participated in liver injury, evidenced by increased expression of α-SMA, TGF-β, and Smad3 in liver tissues, and decreased expression of Smad7.
In conclusions, severe liver damage was observed in SD rats after ephedrine treatment, with increased oxidative stress and inflammatory responses through TGF-β/Smads pathway, providing a scientific rationale for mechanistic studies for ephedrine-caused liver injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
