Abstract
Background
Non-alcoholic steatohepatitis (NASH) is a global medical problem and macrophages’ activation is closely related to the pathogenesis of NASH. Curcumin is a polyphenol from turmeric with significant anti-inflammatory activity.
Objective
The objective of present study was to observe the effect of curcumin on macrophages’ activation and secretion of pro-inflammatory cytokines in NASH.
Methods
Hematoxylin and eosin and TUNEL staining were used to observe the hepatic function. RT-PCR was conducted to evaluate the hepatic mRNA expression of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β). Flow cytometry was adopted to detect the M1 polarization of macrophages. The RAW264.7 macrophage was pretreated with different doses of curcumin, and then lipopolysaccharide (LPS) and interferon-γ (IFN-γ) were given to activate the M1 macrophage. The activation ratio of M1 macrophage was observed by flow cytometry, and IL-1β and TNF-α expression was detected by RT-PCR and ELISA.
Results
After treatment with curcumin, the activities of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), the mRNA expression of TNF-α and IL-1β, and M1 polarization of macrophages were significantly decreased. Hematoxylin and eosin and TUNEL staining showed that inflammation and apoptosis in the liver were improved. What is more, curcumin can effectively inhibit M1 macrophage activation induced by lipopolysaccharide and IFN-γ and reduce the secretion of IL-1β and TNF-α.
Conclusion
Curcumin can effectively improve NASH and reduce hepatic cell necrosis by inhibiting the M1 polarization of macrophages and the secretion of inflammatory factors.
Introduction
In recent years, the number of patients suffering from non-alcoholic fatty liver disease (NAFLD) has increased significantly worldwide because of the change in eating habits and weight gain. 1 Non-alcoholic fatty liver disease can cause a series of liver diseases, eventually leading to higher morbidity and mortality. Non-alcoholic steatohepatitis (NASH) is a more severe form of NAFLD, characterized by steatosis, inflammation, and hepatocyte ballooning, which may further lead to fibrosis, cirrhosis, and hepatocellular carcinoma. 2 Recent studies found that the incidence rate of cirrhosis and hepatocellular carcinoma in NASH patients was dramatically higher.3,4 The pathogenesis of NASH was closely related to genetic factors, intestinal bacterial changes, inflammation, and oxidative stress. 5 Miura et al. 6 found that macrophages play an essential role in the pathophysiological process of NASH. Macrophages are roughly divided into classically activated (M1) and alternatively activated (M2) according to the different surface markers and cytokines. 7 The hepatic inflammation and hepatocyte apoptosis are predominantly mediated by M1 macrophages (activated by IL-1β and TNF-α), while the M2 macrophages could secrete IL-4 and IL-10 to inhibit it. 8 A recent study shows that NASH could induce macrophages to activate M1 type. 9 Therefore, the inhibition of M1 polarization of macrophages is an essential target for the treatment of NASH.
Curcumin, a natural polyphenol compound derived from turmeric, exhibits antioxidant, anti-inflammatory, and anticancer bioactivities. 10 Several studies have confirmed that curcumin has a protective effect on the liver by preventing inflammation, 11 oxidative stress, 12 and lipid accumulation. 13 Also, several in vitro and in vivo experiments have verified that curcumin has a therapeutic effect on NASH by reducing lipid accumulation, inflammatory response, and improving antioxidant function through the Nrf2/ARE and NF-κB signaling pathways.14,15 Liu et al. 16 found that curcumin can induce macrophages to M2 polarization and inhibit M1 polarization in vivo and in vitro. Cunningham et al. 17 found that curcumin can treat NASH induced by CCl4 administration in rats. However, the specific mechanism of curcumin on macrophages in NASH is mostly unknown.
Therefore, the aim of this study was to explore the effects of curcumin on the NASH mice and macrophage line RAW264.7. We hypothesized that curcumin may relieve the hepatic injury of the NASH mice and inhibit the activation of M1 macrophages.
Materials and methods
Animal experimental design
All the experimental procedures followed the regulations on laboratory animal management of Jiangsu Provincial People’s Hospital.
Male C57BL/6 mice (18∼20 g) were purchased from GemPharmatech Co., Ltd. (Jiangsu, China) and reared in a restricted pathogen-free condition (23 ± 2°C, 50% relative humidity) for 1 week; the mice were divided into three groups: Con group (control group given a basal diet), model group (model group given an MCD diet), and Cur group (curcumin group given a MCD diet and 100 mg/kg curcumin by gavage) with eight rats in each group. Moreover, the experiment lasted for 8 weeks. Curcumin was resuspended with 0.5% sodium carboxymethyl cellulose (CMC-Na) solution. Mice in the model group and Con group were given 0.5% CMC-Na solution of the same dose as the Cur group every day.
Sample collection
Twenty-four hours after the last administration, blood was obtained by tail vein puncture and centrifuged (3000 × g, 15, at 4°C). The mice were killed after anesthesia, and the liver samples were collected quickly. The right lobe of the liver was fixed in 4% paraformaldehyde. The left lobe of the liver was preserved in liquid nitrogen for further analysis.
Cell culture and treatment
The RAW264.7 cells were purchased from the Cell Resource Center of the Chinese Academy of Medical Sciences (Shanghai, China). They were cultured in Dulbecco’s modified Eagle’s medium (DMEM, 89% DMEM medium + 10% fetal bovine serum + 1% double-antibody) with 37°C and 5% CO2. Then, the RAW264.7 cells were resuspended in DMEM complete medium to a 2×105 cells/mL concentration and then added into a 6-well plate with 4 mL per well. They were treated in different dose of curcumin (0 μM, 2.5 μM, 5 μM, 10 μM) for 3 h, according to the previous cytotoxic experiment of curcumin in supplementary materials. We added 2.5 ng/mL interferon-γ (IFN-γ) and 400 ng/mL lipopolysaccharide (LPS). The cells and culture supernatant were collected for further study after 12 h.
Hematoxylin and eosin staining
The fixed right liver tissue was taken out from formalin liquid and embedded in paraffin, and then the samples were sectioned at six μm. Sections were stained with hematoxylin and eosin (HE). A section of each mouse was observed and photographed at ×100 magnification with an inverted optical microscope. We invited three experienced histologists to evaluate the images, and they were blinded to the experimental information.
TUNEL staining
After dewaxing and hydrating, the sections were stained in a 3% H2O2, 50% methanol, and 50% dH2O solution for 15 min and then incubated with proteinase K solution. Next, they were washed in PBS twice and stained with TUNEL reaction mixture at 37°C for 60 min. After another PBS wash, we used diaminobenzidine as the chromogen. Mounting tablets were used to observe it under the light microscope.
Serum aminotransferase assay
The activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) in the serum were measured by the kits purchased from the Nanjing Jiancheng Institute of Bioengineering (Nanjing, Jiangsu, China).
Cell cytokines assay
The cell contents of tumor necrosis factor α (TNF-α) and interleukin 1β (IL-1β) were measured by commercial ELISA kits purchased from the Nanjing Jiancheng Institute of Bioengineering (Nanjing, Jiangsu, China).
Quantitative real-time PCR (qRT-PCR)
The liver and cell RNA was extracted by the TRIzol reagent obtained from TaKaRa Biotechnology (Dalian, Liaoning, China). The cDNA was obtained using a reverse transcription kit from TaKaRa Biotechnology. The expression of the target gene was quantified by qRT-PCR. The sequence of related primers was as follows: IL-1β forward 5′-GATGTCTCGAACCTGCTGCC-3′, reverse 5′-GTTTCA-TGTTCGTGGTTGAA-3′. TNF-α forward 5′-AATTC-GAGGTCCGGTCGATCGC-3′; reverse 5′-CCGTGGACAGACGTTCTAA-3′. β-actin forward 5′-AGCGGTTGTATTCCTATCCA-3′; reverse 5′-CCTGGTTGATATGTGATA-3′. The PCR products’ predicted sizes were 165 bp, 180 bp, and 178 bp for IL-1β, TNF-α, and β-actin.
Flow cytometry analysis
The fresh liver samples were cut into 3–4 mm3 pieces and put into the disposable decomposer immediately, and then 1 mL PBS was added and treated for 1 min. The treated cell suspension was aspirated with a 2-mL syringe and filtered through a 70 μM cell filter to obtain a single-cell suspension, stained with different combinations of fluorochrome-coupled antibodies (CD45, F4/80, and CD86). Incubated at room temperature in the dark for 25 min, and then washed with PBS. Finally, it was detected by flow cytometry.
RAW264.7 cells were collected, washed, and resuspended with PBS. Flow cytometric antibodies (anti-mouse CD45, anti-mouse F4/80, and anti-mouse CD86) were added and incubated for 25 min in the dark at room temperature and then washed with PBS. Finally, it was detected by flow cytometry.
Statistical analysis
All data analyses in this study were done using SPSS 20.0 statistical software. One-way analysis of variance was used for statistical differences between different groups, followed by multiple pairwise comparisons (Duncan’s multiple range test, used for multigroup comparisons), and p-values ≤ 0.05 were considered statistically significant. Data were presented as mean ± standard deviation.
Results
Curcumin alleviates liver inflammation induced by MCD diet
As shown in Figure 1(a), the model group showed many vacuolated hepatocytes, severe cystic steatosis, and inflammatory cell infiltration, which suggested that the NASH model was successfully established. After treatment with curcumin, the number of vacuolated hepatocytes, severe cystic steatosis, and inflammatory cell infiltration was reduced. Further, compared with the Con group, ALT and AST levels were significantly increased (p < 0.01) in the model group. As expected, in the Cur group, ALT and AST levels were significantly decreased (p < 0.05) than those in the model group (Figure 1(b)). Effect of curcumin on the hepatic function of the NASH mice. (a) HE staining of the liver tissues (magnification × 100). (b) The levels of AST and ALT in the serum of NASH mice. Notes: Con, the control group, was given a basal diet; model, the model group, was given an MCD diet; and Cur, the curcumin group, was given an MCD diet and 100 mg/kg curcumin by gavage. AST: aspartate aminotransferase; ALT: alanine aminotransferase. Values are mean ± SEM *p < 0.05, **p < 0.01.
Curcumin inhibits hepatocyte apoptosis in NASH model mice
As shown in Figure 2, the number of TUNEL-positive cells in the model group was significantly increased (p < 0.01). After treating with curcumin, in the Cur group, the number of TUNEL-positive cells was significantly decreased (p < 0.05) than those in the model group. Effect of curcumin on the hepatocyte apoptosis of the NASH mice. (a) TUNEL staining of the liver tissues (magnification × 100). (b) The number of TUNEL-positive cells in the liver of NASH mice. Notes: Con, the control group, was given a basal diet; model, the model group, was given an MCD diet; and Cur, the curcumin group, was given an MCD diet and 100 mg/kg curcumin by gavage. Values are mean ± SEM *p < 0.05, **p < 0.01.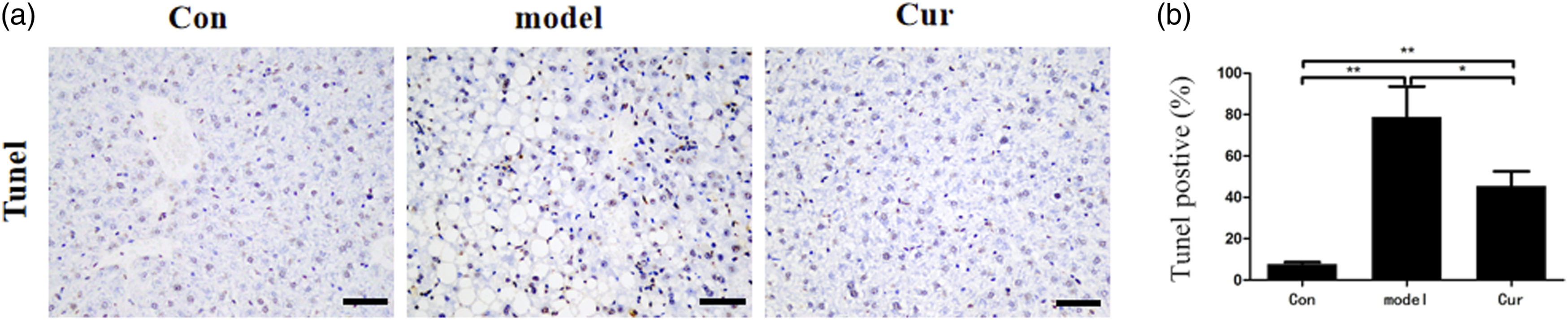
Curcumin inhibits the secretion of pro-inflammatory cytokines in NASH model mice
As shown in Figure 3, in the model group, the hepatic mRNA expressions of IL-1 β and TNF-α were significantly increased (p < 0.01) compared with the Con group. In the Cur group, the hepatic mRNA expressions of IL-1β and TNF-α were significantly decreased (p < 0.01) than those in the model group. Effect of curcumin on the mRNA expressions of IL-1β and TNF-α in the living NASH mice. Notes: Con, the control group, was given a basal diet; model, the model group, was given an MCD diet; and Cur, the curcumin group, was given an MCD diet and 100 mg/kg curcumin by gavage. IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α. Values are mean ± SEM *p < 0.05, **p < 0.01.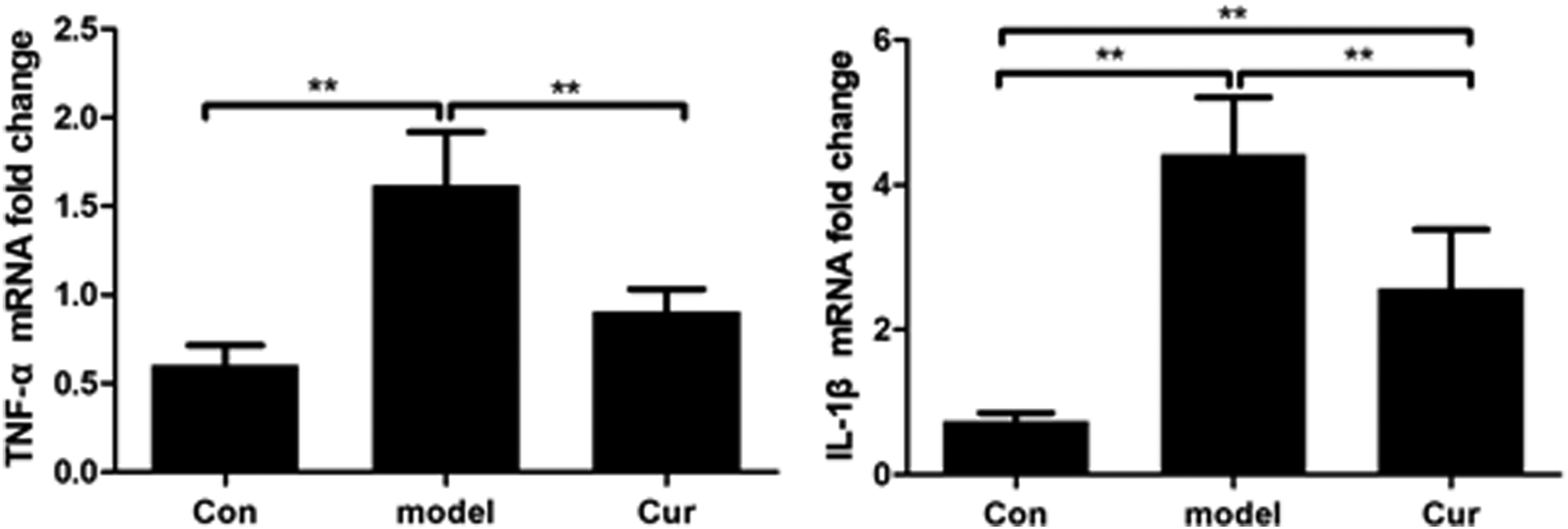
Curcumin inhibits M1 polarization of macrophages in NASH model mice
As shown in Figure 4, in the model group, the hepatic number of M1 macrophages was significantly higher (p < 0.01) than that in the Con group. After treatment with curcumin, the number of M1 macrophages in the liver of Cur group was significantly decreased (p < 0.05) than the model group. Effect of curcumin on the M1 polarization of macrophages in the liver of NASH mice. (a) Gating strategy of activated M1 macrophages. (b) Proportions of M1 macrophages in total macrophages. Notes: Con, the control group, was given a basal diet; model, the model group, was given an MCD diet; and Cur, the curcumin group, was given an MCD diet and 100 mg/kg curcumin by gavage. Values are mean ± SEM *p < 0.05, **p < 0.01.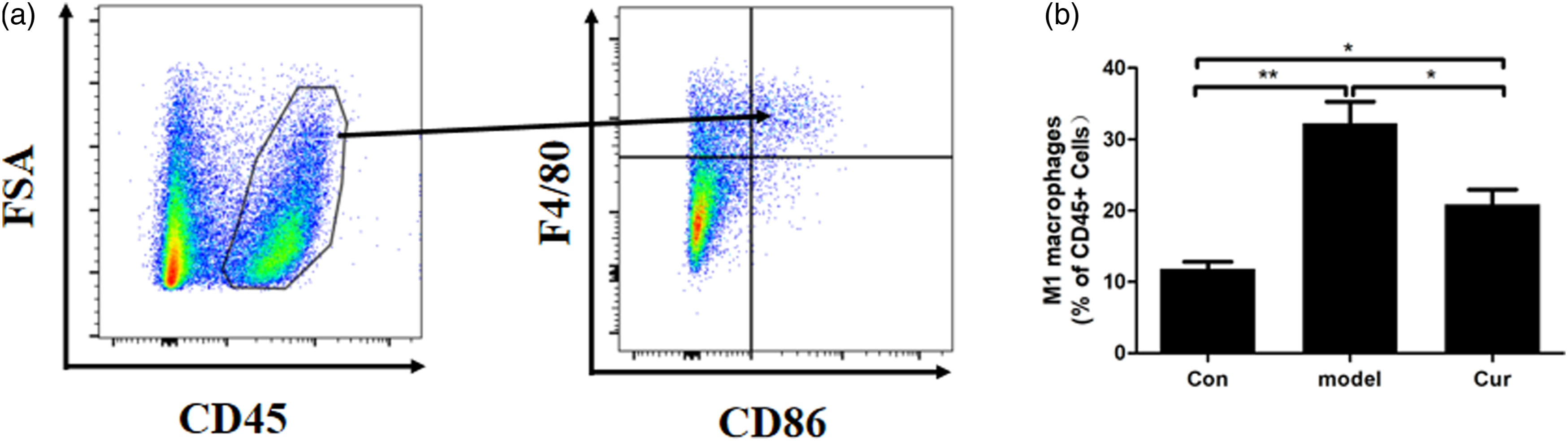
Curcumin inhibits M1 polarization of macrophages induced by LPS+IFN-γ
As shown in Figure 5, M1 polarization of macrophages in the model group was significantly increased (p < 0.01) in comparison with the Con group. After treatment with curcumin, M1 polarization of macrophages was significantly reduced (p < 0.01) in comparison with the model group. Moreover, curcumin inhibited the activation of RAW264.7 cells to M1 type in a dose-dependent manner. Effect of curcumin on the M1 polarization of macrophages induced by LPS+IFN-γ. Notes: Gating strategy of activated RAW 264.7 (CD45+F4/80+CD86+ cells), with the schematic diagram and statistical results of fluorescence values of different groups. Values are mean ± SEM *p < 0.05, **p < 0.01.
Curcumin inhibits the levels of IL-1β and TNF-α secreted by M1 macrophages
As shown in Figure 6, in the model group, the TNF-α and IL-1β levels were significantly increased (p < 0.01) compared with the Con group. Curcumin significantly reduced (p < 0.01) the TNF-α and IL-1β levels compared with the model group. Moreover, curcumin inhibited the secretion of inflammatory factors in RAW264.7 cells stimulated by IFN-γ + LPS in a dose-dependent manner. Effect of curcumin on the levels of IL-1β and TNF-α in the RAW264.7 cells induced by LPS+IFN-γ. Notes: IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α. Values are mean ± SEM *p < 0.05, **p < 0.01.
Discussion
Due to changes in dietary habits, the incidence of NAFLD is increasing worldwide. It is now considered one of the leading causes of end-stage liver failure and is expected to become the leading cause of liver transplantation by 2025. 18 Compared with NAFLD, the liver of NASH patients showed many hepatocyte steatosis and apoptosis, which is one reason for the poor prognosis of NASH patients. 19 Therefore, the inhibition of the hepatic inflammation and hepatocyte necrosis during NASH is also an important research direction. This study found that curcumin can significantly improve the hepatic inflammatory response, inhibit cell apoptosis, and significantly inhibit macrophages’ conversion to M1 type in vivo and in vitro. It means that curcumin may be an effective drug for the treatment of NASH.
The activities of AST and ALT are essential indicators of hepatic function. When the hepatic function is seriously damaged, the ALT and AST levels in the blood circulation will increase significantly. 20 In the present study, we found the ALT and AST activities in serum of the NASH mice were significantly increased, which means the hepatic function was seriously damaged due to NASH. Also, we found a large number of vacuolated hepatocytes, severe cystic steatosis, and inflammatory cell infiltration in liver sections of NASH rats through the HE staining, which further confirmed that the hepatic function of the NASH mice was destroyed. After the treatment of curcumin, the hepatic function has been significantly improved. These results were consistent with previous studies.13,21 Besides, we also found that curcumin can alleviate the apoptosis of hepatocytes induced by NASH. Therefore, we demonstrated that supplementation of curcumin could effectively improve hepatic function.
Interleukin-1β and TNF-α are closely related to the pathological process of NASH. Among many inflammatory factors, TNF-α plays a vital role in intermediary metabolism, including inducing the production of cytokines like IL-1β.22,23 Macrophages mainly secrete IL-1β and TNF-α in acute or chronic liver injury, resulting in the death of hepatocyte through chemotactic neutrophils and inducing other pro-inflammatory production cytokines. 24 Curcumin has been demonstrated to alleviate hepatic injury by reducing the secretion of IL-1β and TNF-α in the liver induced by CCl4. 25 In our study, after administration of curcumin, the hepatic mRNA expression of IL-1β and TNF-α in the NASH model mice was decreased significantly. Additionally, we also found that the number of M1 macrophages in the liver of NASH mice decreased significantly after curcumin administration. Macrophages play an essential role in the pathophysiological process of NASH. 26 Functionally, macrophages can be divided into M1 (pro-inflammatory or “classically activated”) to M2 (tissue-repairing or “non-classically activated”) subtypes. 27 M1 type macrophage can secrete many inflammatory factors, such as IL-1β and TNF-α, leading to severe inflammatory reactions. 27 In in vitro experiments, we found that the proportion of M1 macrophages is significantly increased under the stimulation of IFN-γ and LPS, while curcumin treatment significantly reduced the proportion of M1 macrophages. At the same time, the levels of IL-1β and TNF-α were also significantly decreased after the administration of curcumin. Xu et al. 28 also found that curcumin can inhibit macrophages’ activation from alleviating the lung’s inflammatory reaction, similar to our result. Li et al. 29 found that curcumin can inhibit the secretion of B cell-activating factor by macrophages to treat nephritis. These results confirm that curcumin plays a vital role in inhibiting macrophage activation.
In summary, the results in our study suggested that curcumin may inhibit the activation of macrophages to M1 macrophages and then reduce the expression of IL-1 β and TNF-α to alleviate hepatic dysfunction and inflammation during NASH. It means that curcumin may be a potential candidate for the treatment of NASH. However, further clinical studies are needed to explore the therapeutic effect of curcumin.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
