Abstract
Endometrial cancer (EC) is the fourth most common malignancy in women in developed countries. The prognosis of EC is extremely poor, and it is an important factor that contributes to the death of patients. Therefore, studying EC pathogenesis and therapeutic targets, and exploring effective drugs are the primary tasks to improve the prognosis of EC. In the present study, we aimed to explore the function of ginkgolic acid (GA) in EC cell apoptosis and autophagy through PI3K/Akt/mTOR signal pathway in vitro and in vivo. Firstly, MTT assay and clone formation assay were employed to analyze the Ishikawa and HEC-1-B cell viabilities and proliferation after treatment with GA. The results showed that GA inhibited endometrial cancer cell survival. Flow cytometry assay and western blot assay were applied to examine the apoptosis and apoptosis related protein Bcl-2, Bax, Cleaved caspase-3 expression levels of Ishikawa and HEC-1-B cells after treatment with GA. Next, we applied western blot assay to analyze the autophagy associated proteins LC3I, LC3II, p62 and Beclin-1 in GA treated Ishikawa and HEC-1-B cells. We found that GA promoted apoptosis and induced autophagy of endometrial cancer cells. Meanwhile, western blot assay was also used to determine the expression levels of the PI3K/Akt/mTOR signal pathway related protein and the results revealed that GA inhibited the activity of PI3K/Akt/mTOR pathway. Finally, we found that GA inhibited tumor growth in vivo through immunohistochemistry assay. In conclusion, GA induces apoptosis and autophagy of EC cells via inhibiting PI3K/Akt/mTOR pathway in vivo and vitro.
Introduction
Endometrial cancer (EC) accounts for 20–30% of endometrial tumors in women’s reproductive system, and it is the fourth most common malignancy in women in developed countries.1,2 In recent years, clinical studies have shown that EC occurrence has a trend toward younger age. Although the survival rate of early-stage EC has improved after standardized treatment, the prognosis is extremely poor and it is an important factor that contributes to the death of patients. 3 Therefore, the primary task to improve the prognosis of EC is to study its pathogenesis and therapeutic targets, and to explore effective therapeutic drugs.
Autophagy is a dynamic process that involves the rearrangement of subcellular membranes to isolate the cytoplasm and organelles, and transport to lysosomes or vacuoles, where the isolated cargo is degraded and recycled. 4 Autophagy is involved in the pathogenesis of cancer and is often regarded as a “double-edged sword” due to its role in tumor progression and tumor inhibition. 5 Previous studies have shown that several cytokines and hormones regulate autophagy in different cancers. The phosphatidylinositol 3-kinase (PI3K)/AKT/mTOR pathway is an important regulator of transcription, translation, migration, metabolism, proliferation and survival.6,7 More and more studies have shown that the PI3K/AKT/ mTOR pathway regulates autophagy. 8 Generally, activation of PI3K class I reduces autophagy. On the other hand, PIK3C3/VPS34, the catalytic subunit of Class III PtdIns3K, generates phosphatidylinositol 3-phosphate, which is crucial for the occurrence and development of autophagy. 9
Ginkgolic acid (GA) is a natural compound isolated from ginkgo seed. 10 GA has a wide range of biological activities, including anti-bacterial, anti-HIV and anti-mollusc activities. 11 It has been reported that GA has an effective inhibitory effect on tumor cells by regulating autophagy and apoptosis. For example, GA induces the interaction between ROS production and regulation of apoptosis and autophagy in colon cancer, inhibiting tumor cell survival, 12 induces HepG2 cell death by a combination of apoptosis, autophagy and mitochondrial pathways. 13 GA can inhibit the PI3K/AKT/ mTOR pathway in lung cancer. 14 However, whether GA has any effect on EC has not been reported.
In this study, we aimed to explore the function of GA in EC cells apoptosis and autophagy through PI3K/Akt/mTOR signal pathway in vitro and in vivo.
Methods
Cell culture
The EC cell lines Ishikawa and HEC-1-B cells were purchased from American Type Culture Collection (ATCC, Manassas, VA). Ishikawa and HEC-1-B cells were cultured in culture medium (DMEM; Biological Industries, Kibbutz Beit Haemek, Israel) supplemented with 10% fetal bovine serum (FBS; Biological Industries, Kibbutz Beit Haemek, Israel), 1% penicillin/streptomycin (Gibco), and 0.025 µg/ml amphotericin B (Sigma-Aldrich, St. Louis, MO, USA). The cells were cultured in a humidified 5% CO2 incubator at 37°C. GA was initially dissolved in pure methanol at the stock concentration of 20 μM, 50 μM and 100 μM. Working dilutions for GA were made in culture medium immediately and 0 μM was used as control in all experiments.
MTT assay
To assess the extent of cell viability, MTT method was used. Briefly, the cells (2.5 × 10 3 cells/ml) were plated into 96-well plates in triplicates and treated in different conditions as indicated in each experiment. Following treatment, a final concentration of 0.5 mg/mL MTT solution (Beyotime) was added to each well, and the cells were incubated for another 4 h at 37°C. Subsequently, the culture medium was discarded and 100 µL Dimethyl Sulfoxide (DMSO, Sigma) was added to visualize. The optical density (OD) value of each samples was detected at 490 nm through a microplate reader (BioTek, Winooski, VT, USA).
Clone formation assay
To assess the extent of cell proliferation, crystal violet staining method was used. Briefly, 4.5 × 10 3 Ishikawa and HEC-1-B cells were plated into 6-well plates in triplicates and treated in different conditions as indicated in each experiment. After culture for 3 weeks, the supernatant was removed, and 4% formaldehyde was added for 15 min. Following treatment, cells were stained with 0.25% crystal violet solution for 25 min. The number of cells was counted after drying the culture plate.
Cell apoptosis
Annexin V/FITC and PI apoptosis detection kit (Sigma-Aldrich, St. Louis, Carlsbad, CA, USA) was applied to analyze apoptosis. Flow cytometry results were obtained from BD Accuri™ C6 (CA, USA). Briefly, the cells were digested, washed, and resuspended in Annexin V incubation solution. Then the cells were kept in dark for at least 30 min, 37°C and quantified through flow cytometry.
Western blot
Briefly, cells were washed in pre-cooled PBS buffer three times, and the total protein was extracted by RIPA buffer (Beyotime, Shanghai, China). For autophagy experiment, cells were treated with chloroquine first before protein extraction. Protein concentration was determined by using BCA protein assay kits (CoWin Biotechnology). An equal amount of total proteins was electrophoresed using SDS-PAGE. Then, they were transferred to the polyvinylidene difluoride membranes (PVDF; Millipore) and blocked by 5% non-fat milk at room temperature for 1 h. The protein was identified by incubating with specific primary antibodies Bcl-2 (Rabbit Anti-Bcl-2 antibody, ab182858, 1:2000; Abcam, Cambridge, MA, USA), Bax (Rabbit Anti-Bax antibody, ab32053, 1:5000; Abcam, Cambridge, MA, USA), Cleaved Caspase-3 (Rabbit Anti- Cleaved Caspase-3 antibody, ab32042, 1:500; Abcam, Cambridge, MA, USA), LC3 (Rabbit Anti-LC3 antibody, ab192890, 1:2000; Abcam, Cambridge, MA, USA), P62 (Rabbit Anti-P62 antibody, ab109012, 1:30,000; Abcam, Cambridge, MA, USA), Beclin-1 (Rabbit Anti-Beclin-1 antibody, ab210498, 1:1000; Abcam, Cambridge, MA, USA), p-PI3K (Rabbit Anti-PI 3 Kinase p85 alpha (phospho Y607) antibody, ab182651, 1:3000; Abcam), PI3K (Rabbit Anti-PI 3 Kinase p85 alpha antibody, ab86714, 1:3000; Abcam), p-AKT (Rabbit Anti-AKT (phospho T308) antibody, ab38449, 1:3,000; Abcam), AKT (Rabbit Anti-pan-AKT antibody, ab8805, 1:3000; Abcam), p-mTOR (Rabbit Anti-p-mTOR antibody, ab109268, 1:5000; Abcam), mTOR (Rabbit Anti-mTOR antibody, ab134903, 1:10,000; Abcam), GAPDH (Rabbit anti-GAPDH antibody, ab8245, 1:5000; Abcam, Cambridge, MA, USA), overnight at 4°C. Then, the membranes were further incubated with HRP-conjugated goat anti-rabbit immunoglobulin G secondary antibody (ab205718, 1:1,500; Abcam) and the bands on the membranes were visualized by the ECL chemiluminescence reagent (Beyotime). GAPDH was used to normalize the amount of the analyzed samples and protein bands were quantified by gray scale analysis through ImageJ software (National Institutes of Health).
Xenograft experiments
All animal experiments in this study were in agreement with the Guide for the Care and Use of Laboratory Animals 15 and approved by the Ethics Committee of Henan University of Chinese Medicine(Approval No.DWLL201904110). We purchased 6-week-old male BALB/c nude mice (n = 6) from Experiment Animal Center (Shanghai, China). These mice were subcutaneously injected with equal numbers of Ishikawa cells (2 × 106). After 1 week, the mice were randomly divided into two groups and received an intraperitoneal injection of GA (25 mg/kg) or vehicle. Tumor volume was measured and noted every 3 days. Finally, the mice were sacrificed after injection for 18 days. The tumor tissues were isolated, and their weight was measured.
Immunohistochemistry
For assessment of tumor cells proliferation, the tumor tissues of mice were fixed in 4% (v/v) paraformaldehyde, embedded in paraffin, cut into sections about 4 µm and stained with antibodies. The rabbit anti-human Ki-67 antigen monoclonal antibodies (1:400, Cell Signaling Technology, Boston, USA) were employed to detect nuclear Ki-67 expression. Lastly, the optical microscope (Olympus, Japan) was used to observe the tumor tissues sections of mice.
Statistical analysis
All data are presented as mean ± standard deviation from three independent assays. Student’s t-test was employed to calculate the comparisons between two groups. We employed GraphPad Prism 5 (GraphPad Software, Inc., San Diego, CA, USA) for analysis. P values of <0.05 was considered statistically significant.
Results
Ginkgolic acid inhibited endometrial cancer cell survival
MTT assay was carried out to examine the effect of GA on the viability of EC cell lines, Ishikawa and HEC-1-B. The results showed that compared with control group, high concentration of GA significantly inhibited Ishikawa and HEC-1-B cells viability (Figure 1(A)). Further, clone formation assay was used to evaluate the proliferation of Ishikawa and HEC-1-B cells, the results revealed that high concentration of GA could decrease the number of cell clones in two cell lines (Figure 1(B)). From these results, a dose dependent down-regulation in cell viability was observed, suggesting that GA inhibited endometrial cancer cell survival.

Ginkgolic acid inhibited endometrial cancer cell survival. (A) The effect of GA at concentration of 0, 20, 50, 100 μM on the viabilities of Ishikawa and HEC-1-B cells were examined by MTT assay, *p < 0.05, **p < 0.01. (B) Clone formation assay was used to measure the proliferation of Ishikawa and HEC-1-B cell lines after GA treatment at concentration of 0, 20, 50, 100 μM, **p < 0.01.
Ginkgolic acid promoted apoptosis of endometrial cancer cells
The modulatory effect of GA on endometrial carcinoma cell apoptosis was analyzed by flow cytometry assay. The results revealed that after GA induction, Ishikawa and HEC-1-B cells apoptosis were increased (Figure 2(A)). Western bolt results showed that GA decreased the expression level of anti-apoptotic protein Bcl-2, while increased the expression levels of Bax and Cleaved Caspase-3 in Ishikawa and HEC-1-B cells (Figure 2(B)). These results revealed that GA promoted apoptosis of endometrial cancer cells.

Ginkgolic acid promoted apoptosis of endometrial cancer cells. (A) The effect of GA at concentration of 0, 20, 50, 100 μM on the apoptosis of Ishikawa and HEC-1-B cells were analyzed by flow cytometry assay, **p < 0.01. (B) Western blot assay was used to measure the expression levels of apoptosis related genes Bcl-2, Bax and Cleaved Caspase-3 in Ishikawa and HEC-1-B cells after GA treatment at concentration of 0, 20, 50, 100 μM, **p < 0.01. GAPDH was used as an internal control.
Ginkgolic acid induced autophagy of endometrial cancer cells
Then we further explored the function of GA in endometrial cancer. Western blot results showed the protein expression levels of LC3I, LC3II, p62 and Beclin-1. Then we calculated the gray value of LC3II/LC3I, p62 and Beclin-1 bands. The results showed that LC3II and Beclin-1 were highly induced after treatment of GA at concentration of 0, 20, 50, 100 μM, while p62 was downregulated markedly, supporting that GA induced autophagy of endometrial cancer cells (Figure 3).

Ginkgolic acid induced autophagy of endometrial cancer cells. Western blot analysis of LC3I, LC3II, p62 and Beclin-1 in Ishikawa and HEC-1-B cells after treatment of GA at concentration of 0, 20, 50, 100μM, **p < 0.01. GAPDH was used as an internal control.
Ginkgolic acid inhibited the activity of PI3K/Akt/mTOR pathway
To investigate the underlying mechanisms through which GA induces the apoptosis and autophagy of endometrial cancer cells, we employed the western blot assays to examine the PI3K/Akt/mTOR pathway protein levels in Ishikawa and HEC-1-B cells after treatment of GA at concentration of 0, 20, 50, 100 μM. The western blot results showed that GA suppressed p-PI3K, p-Akt and p-mTOR expression, suggesting that GA served as a strong inhibitor of PI3K/Akt/mTOR pathway activity (Figure 4).

Ginkgolic acid inhibited the activity of PI3K/Akt/mTOR pathway. Western blot analysis of p-PI3K, PI3K, p-Akt, Akt, p-mTOR and mTOR in Ishikawa and HEC-1-B cells after treatment of GA at concentration of 0, 20, 50, 100 μM, **p < 0.01. GAPDH was used as an internal control.
Ginkgolic acid inhibited tumor growth in vivo
The effects of GA on endometrial tumor was further assessed in BALB/c nude mice models with subcutaneous injection of Ishikawa cells. One week after the injection of Ishikawa cells, the mice were randomly divided into two groups and received an intraperitoneal injection of GA or vehicle. The subcutaneous tumor formation results showed that the tumor volume was remarkable decreased in GA injection group compared to control group (n = 3) (Figure 5(A)). The tumor volume and tumor weight were significantly lower in group treated with GA in comparison with control group (Figure 5(B)). Consistent with the above in vivo results, immunohistochemical results revealed that GA treatment weakened the expression of Ki67 (Figure 5(C)). Taken these results together, GA inhibited the xenograft tumors growth in vivo.
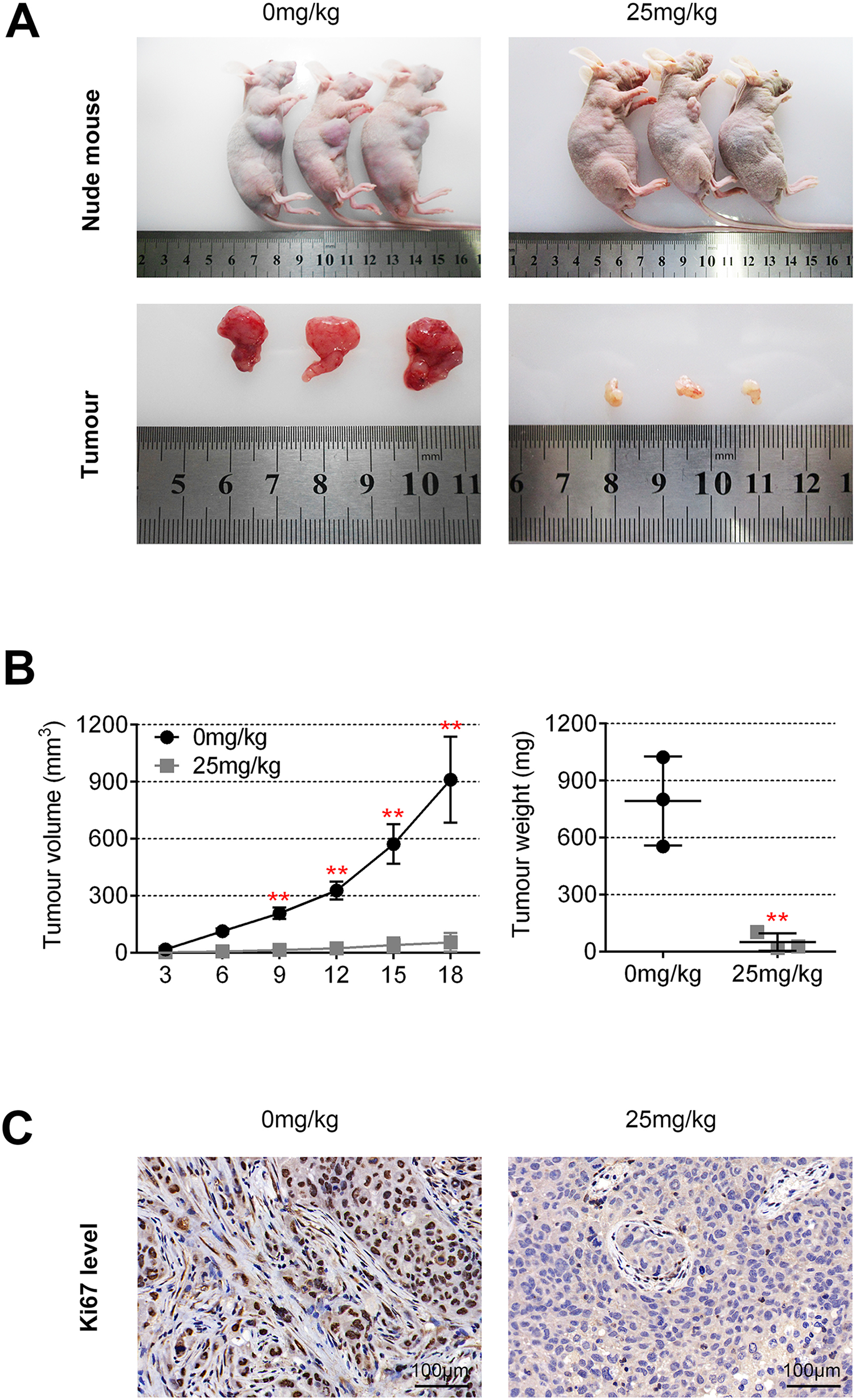
Ginkgolic acid suppressed tumor growth in vivo. (A) Photograph of subcutaneous xenograft tumors isolated from BALB/c nude mice in GA group and control group, n = 3. (B) The average tumor volume was calculated every 3 days. The tumor weights of GA treatment group and control group were measured at the end of the experiment, n = 3, **p < 0.01. (C) Image of immunohistochemical staining for Ki67 in GA group and control group, Scale bar = 100 μm.
Discussion
Endometrial cancer causes approximately 6000 deaths each year in the United States. 16 Our understanding of endometrial cancer has evolved through improvements in molecular biology, allowing improved target-specific therapies. 17 Many drugs have been validated that have effects in clinical therapies of endometrial cancer. 18 One study analyzed effects of Lenvatinib plus pembrolizumab in patients with advanced EC. 19 Another study reported effect of taxane plus platinum regimens compare to doxorubicin plus cisplatin as adjuvant chemotherapy for EC. 20 A third study reported a phase II study of frontline paclitaxel/carboplatin/bevacizumab, paclitaxel/carboplatin/temsirolimus, or ixabepilone/carboplatin/bevacizumab in advanced/recurrent EC. 21 Aspirin can improve the survival of women whom suffer from endometrial adenocarcinoma. 22 Ornithine decarboxylase was found as a therapeutic target for EC. 23 However, there are no research on GA’s action on EC occurrence and growth. In this study, we found that GA restrained endometrial cancer cells proliferation and viability. This is the first time GA treatment on EC development was explored.
Ginkgolic acid, an antibacterial compound, has been found to be involved in the development of many cancers. Ginkgolic acid suppressed the proliferation, migration and invasion of colon cancer cells. 24 GA 17:1 was found to restrain STAT3 signaling pathway and thus exert its anti-tumor effects against multiple myeloma cells. 25 GA suppresses migration and invasion of lung cancer cells by PI3K/Akt/mTOR inactivation. 14 GA inhibits the development of pancreatic cancer through restraining lipogenesis pathways. 26 However, there are only a few researches on GA’s function in cancer cells apoptosis and autophagy. GA restrains gastric cancer development by inducing apoptosis and inhibiting STAT3/JAK2 signal pathway. 27 Increased apoptosis of Hep-2 cancer cells was observed after GA treatment. 28 Based on those researches on GA’s apoptosis inducing effect, our present study revealed that GA promoted apoptosis and induced autophagy of EC cells. Suppressing the activation of PI3K/Akt/mTOR signaling and activating the critical genes involved in apoptosis and autophagy may be the underlying mechanisms.
Apoptosis and autophagy are closely related to the occurrence and development of EC cancer. Autophagy suppression enhances resveratrol-induced apoptosis in EC cells. 29 Isoliquiritigenin promotes apoptosis and autophagy thus inhibits EC cells growth in vivo. 30 SI113, the SGK1 inhibitor, induces autophagy, apoptosis, and endoplasmic reticulum stress in EC cells. 31 The anti-tumor activities against EC cancer and cervical cancer involved apoptosis, autophagy and transferrin receptor of dihydroartemisinin. 32 FAM83B knockdown silenced the PI3K/AKT/mTOR pathway and promoted autophagy in EC. 33 MiR-205 inhibits cell growth by targeting AKT-mTOR signaling in progesterone-resistant Ishikawa cells. 34 Nifedipine induced autophagy through Beclin1 and mTOR pathway in EC cells. 35 We found that GA inhibited the activity of PI3K/Akt/mTOR signal pathway, thus induced EC cells apoptosis and autophagy directly. There are complex interactions between apoptosis and autophagy, hence future study is necessary to fully reveal the two different mechanisms. Knocking down FAM83B inhibits endometrial cancer cell proliferation and metastasis by silencing the PI3K/AKT/mTOR pathway. 33 AKT-mTOR signal pathway restrains cell growth by targeting to miR-205 in progesterone-resistant Ishikawa cells of EC. 34 PI3K/Akt/mTOR reduces cell proliferation and invasion and enhances apoptosis in EC via targeting to miR-101. 36 Whether PI3K/AKT/mTOR signaling pathway has functions other than proliferation, apoptosis, metasis in EC cells after GA treatment remains further exploration.
In conclusion, in this study, we found that GA inhibited endometrial cancer cells proliferation and viability. In addition, we found that GA promoted apoptosis and induced autophagy of EC cells. Furthermore, we showed that GA restrained the activity of PI3K/Akt/mTOR pathway. Finally, in vivo experiment revealed that GA inhibited tumor growth. All those results suggested that GA induces apoptosis and autophagy of EC cells via inhibiting PI3K/Akt/mTOR pathway in vivo and vitro.
Footnotes
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors’ contributions
Li Zhou designed the study, supervised the data collection, Shurong Li analyzed the data, interpreted the data, Jianhua Sun prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval was obtained from the Ethics Committee of Henan University of Chinese Medicine.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Youth fund of National Natural Science Foundation of China (Grant No. 82004412).
