Abstract
Doxorubicin (DOX) is an anticancer drug which is used for treatment of several types of cancers. But the clinical use of doxorubicin is limited because of its cardiotoxicity and cardiomyopathy. Mitochondrial-dependent oxidative stress and cardiac inflammation appear to be involved in doxorubicin-induced cardiotoxicity. Betanin as a bioactive compound in Beetroot (Beta vulgaris L.) displays anti-radical, antioxidant gene regulatory and cardioprotective activities. In this current study, we investigated the protective effect of betanin on doxorubicin-induced cytotoxicity and mitochondrial-dependent oxidative stress in isolated cardiomyocytes and mitochondria. Isolated cardiomyocytes and mitochondria were treated with three concentrations of betanin (1, 5 and 10 µM) and doxorubicin (3.5 µM) for 6 h. The parameters of cellular and mitochondrial toxicity were analyzed using biochemical and flow cytometric methods. Our results showed a significant toxicity in isolated cardiomyocytes and mitochondria in presence of doxorubicin which was related to reactive oxygen species (ROS) formation, increase in malondialdehyde (MDA), increase in oxidation of GSH to GSSG, lysosomal/mitochondrial damages and mitochondrial swelling. While betanin pretreatment reverted doxorubicin-induced cytotoxicity and oxidative stress in isolated cardiomyocytes and mitochondria. These results suggest that betanin elicited a typical protective effect on doxorubicin-induced cytotoxicity and oxidative stress. It is possible that betanin could be used as a useful adjuvant in combination with doxorubicin chemotherapy for reduction of cardiotoxicity and cardiomyopathy.
Introduction
Doxorubicin (DOX) as an anticancer antibiotic belonging to the anthracycline family was discovered in the late 1960s. 1 Doxorubicin is widely used in clinic for treatment of several types of tumors, such as solid tumors, soft-tissue sarcomas, different forms of lymphomas and leukemia. 2 Despite the beneficial effectiveness of this drug in the treatment of cancers, a few years later after its discovery, severe cardiotoxic side effects such as cardiomyopathy and cardiac toxicity have been reported in patients. 3 In addition, the use of doxorubicin is another important reason of dilated cardiomyopathy that creates congestive heart failure. 4 Therefore, Doxorubicin-induced cardiotoxicity leads to limiting its clinical application. Despite above major side effects induced by doxorubicin, now this drug is widely used in clinic and is still one of the strongest anti-tumor drug available for clinical use. 5
The mechanism of action of doxorubicin-induced cardiotoxicity is controversial, and there are several hypotheses. 6 At first, it was accepted that doxorubicin-induced cardiotoxicity is entirely independent from its anti-tumor effect. 7 Therefore, the majority of studies focused on doxorubicin -induced mitochondrial dysfunction and cardiotoxicity. 8 Doxorubicin accumulates in mitochondria and can bind to mitochondrial DNA and other biomolecules such as cardiolipin and the mitochondrial abundant phospholipids. 6 Accumulation of doxorubicin in mitochondria can create intramitochondrial ROS and reactive nitrogen species (RNS) formation mainly through nonenzymatic mechanisms, leading to activation of PARP-dependent cell death pathways. 6 Doxorubicin-induced mitochondrial DNA oxidation is a major contributor of heart failure development and cardiomyopathy. 9 Other major mechanisms of doxorubicin-induced ROS production are the redox cycling-dependent formation of superoxide anion, alteration in iron metabolism and formation of hydroxyl radicals through Haber-Weiss reaction. 10 Subsequently enhanced ROS formation causes activation of proinflammatory transcription factor nuclear factor kappa B (NF-κB), inducible nitric oxide synthase and finally cell death in cardiomyocytes. 11 Therefore, mitochondrial dysfunction, a decrease in the activity of endogenous antioxidants and an increase of free radicals in the cardiomyocytes play important roles in doxorubicin-induced cardiotoxicity. 12 Therefore, a number of compounds with potential cardioprotective activity, including natural compounds with ROS scavenger properties and mitochondrial protective effect can probably play a promising role in reducing the cardiotoxicity caused by doxorubicin.
Betanin (betanidin-5-O-b-glucoside) as a major phytochemical agent found in Beetroot (Beta vulgaris L.) displays the antioxidant potential and anti-inflammatory effects. 13 Because of the resistance to gastrointestinal digestion, Betanin is absorbed by the epithelial cells and reaches the plasma. 14 This betanin shows free-radical scavenger potential through electron or hydrogen donation. 15 Also, betanin suppresses the pro-inflammatory NF-κB pathways and simultaneously induces the transcription of antioxidant genes through the nuclear factor erythroid-2-related factor 2. 16 It has been reported that betanin can reduce low-density lipoprotein (LDL) and lipid membranes peroxidation, modulate gene expression and ROS formation in order to decrease inflammatory cytokines and enhancing antioxidant enzyme activities. 15 As a supportive therapy, betanin shows potential to attenuate the pathophysiological effects caused by the inflammation and oxidative stress that lead to cardiovascular disease (CVD). 17 Due to the promising effect of betanin on ROS scavenger, antioxidant, anti-inflammatory and cardioprotection, it deserves to be more studied. Therefore, here we aim to evaluate the effect of betanin against doxorubicin-induced toxicity in isolated cardiomyocytes and mitochondria obtained from rat heart.
Materials and methods
Chemicals and reagents
Collagenase (Type II), Penicillin and Streptomycin Solution, Fetal Bovine Serum (FBS), 199 Medium, Creatine, 2′,7′-Dichlorofuorescin Diacetate (DCFH-DA), Carnitine, Potassium chloride, Taurine, N-(2-hydroxyethyl) piperazine-N′-(2-ethanesulfonic acid) (HEPES), Hank’s Trypan blue, Rhodamine123, Balanced Salt Solution (HBSS), Bovine Serum Albumin (BSA), Dimethyl sulfoxide (DMSO), -Amino-2-hydroxymethyl-propane-1,3-diol (TRIS), D-mannitol, Sucrose, 4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 2 Monopotassium phosphate, 3-morpholinopropane-1-sulfonic acid (MOPS), Sodium succinate, Rotenone, Magnesium chloride and Acridine Orange were purchased from Sigma (St. Louis, MO USA). Xylazine and Ketamine were obtained from CEVA Santé Animale (Naaldwijk, the Netherlands). Doxorubicin with CAS number 25316-40-9 and a purity of about 99%, was gifted from Dr. Abidi Pharmaceuticals Co. Iran. It was freshly prepared before use and dissolved in 0.05% DMSO. Also, betanin with CAS number 0007659952 and a purity of about 99%, were purchased from Sigma (St. Louis, MO USA) and dissolved in 0.05% DMSO.
Animals
Adult male Wistar rats (8- to 9-week-old) weighting 180–220 g were obtained from the experimental animal center at Baqiyatallah University of Medical Sciences, Tehran, Iran. The Committee of Experimental Animals of Shahid Beheshti University of Medical Sciences approved this study. All animals were cared and experimented on in accordance with the recommendations in the Guide for the Use and Care of Laboratory Animals approved by the Ethics Committee at the Shahid Beheshti University of Medical Sciences, Tehran, Iran. The approval code is IR.SBMU.RETECH.REC.1399.725. In the current study, isolated cardiomyocytes (106 cells/ml) and mitochondria (1000 µg/ml) obtained from five animals were used.
Cardiomyocyte isolation
Cardiomyocytes were isolated from adult male Wistar rats. Rats were intraperitoneally anesthetized with combination of ketamine (50 mg/kg) and xylazine (10 mg/kg). After rats appeared calm, hearts were quickly excised, and directly perfused on a modified Langendorff perfusion apparatus with Powell medium (containing: 110 mM NaCl, 1.2 MgSO4 7H2O, 2.5 mM KCl, 1.2 mM KH2PO4, 25 mM HEPES and 10 mM D (+)-Glucose monohydrate in ultra-pure sterile water with pH = 74 adjusted with 2 M NaOH) for 5 min. Powell medium were replaced by enzyme mixtures containing 30 µM CaCl2 and 5 mg/ml collagenase, in warm Powell medium for 25 min. Then, hearts were dissected mechanically to small pieces and shaken in flasks with enzyme mixture for another 10 min. The collagenase digested ventricle suspensions were then filtered through a sieve (180 µm) to remove remnants of connective tissues. After rinsing with Powell medium, cell suspensions were placed on top of BSA solution where non-cardiomyocytes were removed by a density gradient. Isolated cardiomyocytes were suspended in CCT medium containing taurine, creatine and carnitine in medium 199 supplemented with antibiotics (100 µg/ml penicillin and 100 µg/ml streptomycin) at 37°C under a 5% CO2−95% air atmosphere. One hour after plating, the CCT medium was changed to remove unattached dead. 18 The isolated cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and 10 µM betanin group for 6 h.
Measurement of cell viability
After the cardiomyocytes were seeded and allowed to attach, the MTT assay was used to evaluate the effects of doxorubicin and betanin on cell viability. Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 96 well plate (104 cells per well) at 37°C for 6 h. 25 µl of 0.5 mg/mL MTT was added to each well. After 2 h of incubation the formazan crystals were dissolved in 100 µL DMSO and the absorbance was measured at 570 nm. 19 The viability was represented as the percentage absorbance compared with untreated control group.
Intracellular ROS measurement
Intracellular ROS accumulation was examined using the fluorescence probe 2′-7′dichlorofluorescin diacetate (DCFH-DA). Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 24 well plate (105 cells per well) at 37°C for 6 h. After incubation, the cells were washed twice with PBS and incubated with 5 µM DCFH-DA dissolved in CCT medium for 15 min in a dark chamber. Then the fluorescence intensity of DCF was detected by a Cyflow Space-Partec flowcytometry. 20
Measurement of mitochondrial damages
Mitochondrial membrane potential collapse was examined using the fluorescence probe rhodamine 123. Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 24 well plate (105 cells per well) at 37°C for 6 h. After incubation, the cells were washed twice with PBS and incubated with 1 µM rhodamine 123 dissolved in CCT medium for 15 min in a dark chamber. Then the fluorescence intensity of rhodamine 123 was detected by a Cyflow Space-Partec flowcytometry. 21
Measurement of lysosomal damages
Lysosomal membrane integrity was measured using the fluorescence probe acridine orange (AO). Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 24 well plate (105 cells per well) at 37°C for 6 h. After incubation, the cells were washed twice with PBS and incubated with 5 µM acridine orange dissolved in CCT medium for 15 min in a dark chamber. Then the fluorescence intensity of acridine orange was detected by a Cyflow Space-Partec flowcytometry. 19
Measurement of the reduced (GSH) and oxidized (GSSG) glutathione levels
The levels of glutathione (GSH) and glutathione disulfide (GSSG) were measured by Hissin and Hilf method. 22 Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 24 well plate (105 cells per well) at 37°C for 6 h. After incubation, the cells were washed twice with PBS and mechanically lysed using a glass homogenizer in phosphate buffer (0.1 M with pH 7.4), then the cells were centrifugated at 8,000 ×g at 4°C for 10 min. Then 100 µl supernatant was blended with 3 ml reaction solution (500 mM TRIS-HCl and 10 mM DTNB with pH = 8.0) for measurement of GSH. The optical density was measured at 412 nm, after 15 min of incubation at 25°C. Similarly, 100 µl of supernatant was blended to 3 ml of reaction solution (150 μM NADPH, 500 mM TRIS–HCl buffer, 1 mM EDTA, glutathione reductase, 10 mM DTNB and3 mM MgCl2) for measurement of GSSG. The optical density was measured at 412 nm, after 15 min of incubation at 25°C.
Measurement of malondialdehyde
As an indicator of lipid peroxidation, malondialdehyde (MDA) levels was measured. Briefly, cardiomyocytes were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin (IC50 6 h) group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 24 well plate (105 cells per well) at 37°C for 6 h. After incubation, the cells were washed twice with PBS and mechanically lysed in 1 ml 0.1% (w/v) trichloroacetic acid (TCA) by glassy homogenizer and centrifugated at 10,000 ×g for 10 min. The obtained supernatant was combined with 4 ml of 20% TCA and 0.5% TBA. The mixture was boiled for 15 min at 90°C. After cooling on ice and centrifugation at 1000 ×g for 10 min, the absorbance was measured at 532 nm. 23
Mitochondria isolation
Mitochondria were isolated from the rat heart as previously described by Gostimskaya and Galkin. 24 Briefly, after deep anesthesia the animals with combination of ketamine and xylazine, the heart was cleared from blood vessels, chopped and homogenized with a glass homogenizer in isolation buffer (10 mM Tris-HCl (pH 7.4), 75 mM sucrose, 1 mM EDTA, 225 mM mannitol at 4°C). After centrifugation of the homogenate at 1000 ×g for 10 min, the pellet was removed and supernatant was collected. The mitochondria in the supernatant were sedimented at 10000 ×g for 10 min at 4°C. The protein content in mitochondria was measured using the Bradford assay. Bovine serum albumin (BSA) is used as a protein concentration standard.
Measurement of mitochondrial function
Mitochondrial Function was measured by the activity of succinate dehydrogenase (complex II). Briefly, isolated mitochondria were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 96 well plate (100 µg/ml) at 37°C for 1 h. Then 25 µl of 0.4% MTT was added to each well and incubated for 30 min at 37°C. The formazan crystals were dissolved in DMSO and the optical density was detected at 570 nm. 25 The succinate dehydrogenase was represented as the percentage absorbance compared with untreated control group.
Measurement of mitochondrial swelling
Mitochondrial swelling was estimated by examining changes in light scattering as monitored spectrophotometrically at 540 nm. 26 Briefly, isolated mitochondria were randomized into six experimental groups: 0.05% DMSO as the control group, 3.5 µM doxorubicin group, 3.5 µM doxorubicin + 1 µM betanin group, 3.5 µM doxorubicin + 5 µM betanin group, 3.5 µM doxorubicin + 10 µM betanin group and betanin group in a 96 well plate (100 µg/ml) at 37°C for 1 h. After 1 h, the absorbance at 540 nm was monitored during 60 min using a spectrophotometer.
Statistics
The results are presented as mean ± SD from three repeated experiments. Statistical analysis was performed using GraphPad Prism 5 (GraphPad Software, La Jolla, CA). Data were subjected to one- and two-way ANOVA followed by post hoc Tukey and Bonferroni test respectively. A value of <0.05 was considered to indicate a significant difference.
Results
Cytoprotective effect of betanin on doxorubicin-induced cytotoxicity in isolated cardiomyocytes
Addition of doxorubicin (3.5 µM) to isolated cardiomyocytes resulted in a decrease in the number of cells as shown by MTT assay at 6 h (Figure 1(A)). As seen in Figure 1(B), pretreatment of isolated cardiomyocytes with 10 µM betanin, 30 min before doxorubicin addition prevented cytotoxicity caused by the drug. The obtained results showed that 10 µM betanin significantly (p < 0.001) prevented of doxorubicin-induced cytotoxicity.

Effects of betanin treatment on doxorubicin-induced cytotoxicity (A). DOX, doxorubicin; Data presented are the mean ± SD (n = 3). ***p < 0.001 compared with the group with no pretreatment; ###p < 0.001 compared with the group with doxorubicin. (B) Effects of betanin treatment on doxorubicin-induced lipid peroxidation. Data presented are the mean ± SD (n = 3). ***p < 0.001 compared with the group with no treatment; ###p < 0.001 compared with the group with doxorubicin.
Protective effect of betanin on doxorubicin-induced oxidative stress in isolated cardiomyocytes
The MDA amounts in isolated cardiomyocytes after the treatment with doxorubicin and betanin was shown in Figure 1(B). The MDA amounts increased rapidly 6 h after the treatment with 3.5 µM doxorubicin when compared with untreated control group (p < 0.001). While these data revealed that pretreatment of isolated cardiomyocytes with 10 µM betanin, 30 min before doxorubicin addition, significantly (p < 0.001) decreased the MDA level in the culture medium. That means betanin could inhibit the lipid peroxidation and subsequent cardiomyocytes membrane damage under doxorubicin-induced oxidative stress.
Protective effect of betanin on doxorubicin-induced GSG depletion in isolated cardiomyocytes
Altered intracellular redox status (GSH/GSSG) has a pivotal role in doxorubicin-induced cell death in the cardiomyocytes. To determine whether the intracellular redox status (GSH/GSSG) contributes to the protective effect of betanin on doxorubicin-induced cytotoxicity, we measured GSH and GSSG levels in isolated cardiomyocytes. As shown in Table 1, doxorubicin exposure at 3.5 µM resulted in increased oxidation of GSH to GSSG, and the GSH/GSSG ratio fell significantly (p < 0.01) after doxorubicin addition. Pretreatment with 10 µM betanin 30 min before doxorubicin addition, prevented GSH reduction and GSSG elevation, leading to almost complete recovery of GSH/GSSG ratio.
Effects of betanin pretreatment on doxorubicin-induced GSH depletion in cardiomyocytes.
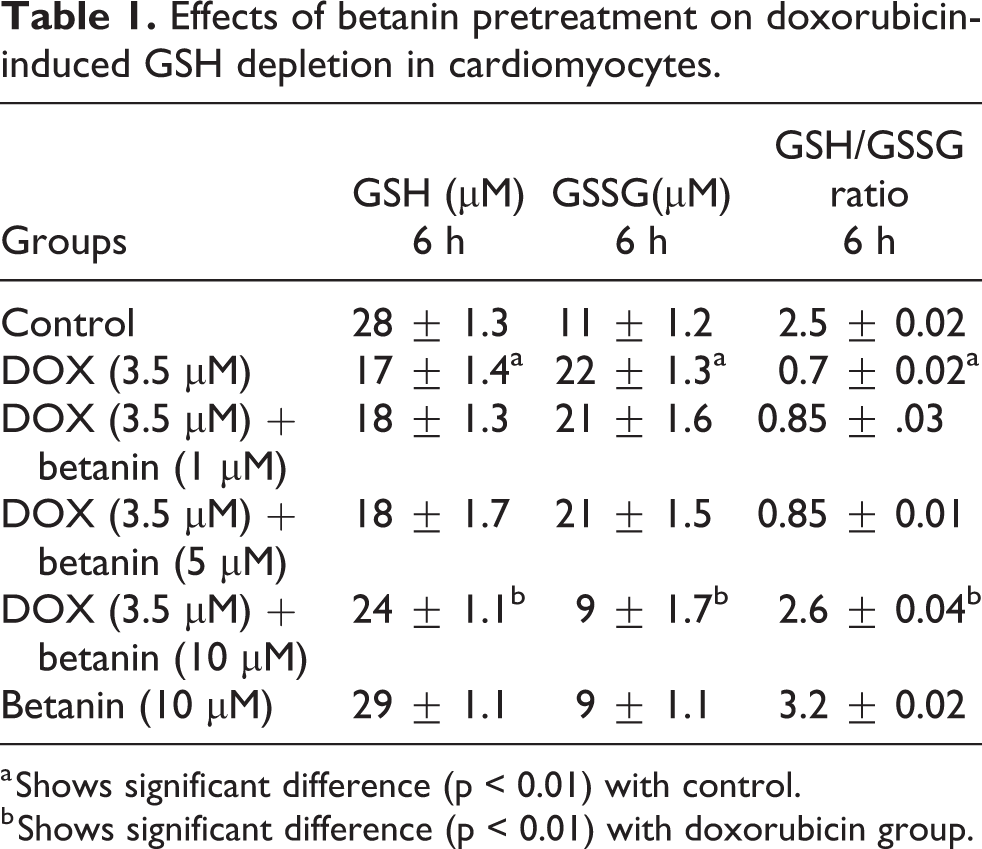
a Shows significant difference (p < 0.01) with control.
b Shows significant difference (p < 0.01) with doxorubicin group.
Protective effect of betanin on doxorubicin-induced ROS formation in isolated cardiomyocytes
A significant increase of ROS was seen when isolated cardiomyocytes were treated with 3.5 µM doxorubicin (Figure 2), which could suggest the occurrence of oxidative stress after doxorubicin addition to cardiomyocytes. Pretreatment with 5 and 10 µM betanin 30 min before doxorubicin addition reduced the level of ROS formation caused by the drug. Betanin (5 and 10 µM) effectively reduced the levels of ROS formed after doxorubicin. Butylated hydroxytoluene (BHT) as a standard antioxidant was able to reduce ROS levels in isolated cardiomyocytes after doxorubicin addition.

Effects of betanin treatment on doxorubicin-induced ROS Formation in cardiomyocytes. Representative fluorescence intensity of ROS staining with DCF-DA. Cardiomyocytes with DCF-DA fluorescence indicate elevated intracellular ROS. As shown the fluorescence intensity is increased after exposure with doxorubicin. The peak is translocated to the right compared to control. While treatment of betanin (5 and 10 µM) with doxorubicin showed that the fluorescence intensity is decreased and the peaks are translocated to the left.
Protective effect of betanin on doxorubicin-induced mitochondrial damages in isolated cardiomyocytes
A significant increase of fluorescence intensity of rhodamine 123 as indicator of the mitochondrial permeability transition and mitochondrial damages was observed when isolated cardiomyocytes were treated with 3.5 µM doxorubicin (Figure 3), which could show the mitochondria damages after doxorubicin addition to cardiomyocytes. Pretreatment with 5 and 10 µM betanin 30 min before doxorubicin addition reduced the fluorescence intensity of rhodamine 123 as indicator of the mitochondrial damages caused by the drug. Betanin (5 and 10 µM) effectively reduced the mitochondrial damages after doxorubicin. Cyclosporine A as a mitochondrial permeability transition (MPT) pore sealing agents was able to reduce mitochondrial damages in isolated cardiomyocytes after doxorubicin addition.

Effects of betanin treatment on doxorubicin-induced loss of mitochondrial ΔΨm in cardiomyocytes. (A) Representative fluorescence intensity of rhodamine 123 staining. (B) Quantitative analysis of rhodamine 123 staining. DOX, doxorubicin. Data presented are the mean ± SD (n = 3). ***p < 0.001 compared with the group with no treatment; ###p < 0.001 compared with the group with doxorubicin.
Protective effect of betanin on doxorubicin-induced lysosomal damages in isolated cardiomyocytes
A significant increase of fluorescence intensity of acridine orange as indicator of the lysosomal membrane permeabilization was observed when isolated cardiomyocytes were treated with 3.5 µM doxorubicin (Figure 4), which could show the lysosomal damages after doxorubicin addition to cardiomyocytes. Pretreatment with 5 and 10 µM betanin 30 min before doxorubicin addition reduced the fluorescence intensity of acridine orange as indicator of lysosomal membrane disintegrity caused by the drug. Betanin (5 and 10 µM) effectively reduced the lysosomal membrane disintegrity after doxorubicin. Chloroquine as a lysosomotropic agent was able to reduce the lysosomal membrane disintegrity in isolated cardiomyocytes after doxorubicin addition.

Effects of betanin treatment on doxorubicin-induced loss of lysosomal membrane destabilization in cardiomyocytes. (A) Representative fluorescence intensity of acridine orange staining. (B) Quantitative analysis of acridine orange staining. DOX, doxorubicin. Data presented are the mean ± SD (n = 3). ***p < 0.001 compared with the group with no treatment; ##p < 0.01, ###p < 0.001 compared with the group with doxorubicin.
Succinate dehydrogenase activity in isolated mitochondria
Addition of doxorubicin (3.5 µM) to isolated rat heart mitochondria resulted in a decrease in the succinate dehydrogenase activity as shown by MTT assay at 60 min. As seen in Figure 5(A), pretreatment of isolated rat heart mitochondria with 5 and 10 µM betanin, 30 min before doxorubicin addition prevented mitochondrial dysfunction caused by the drug. This observation confirmed that the used concentration (5 and 10 µM) of betanin significantly (p < 0.001) prevented of doxorubicin-induced mitochondrial dysfunction.

Effect of doxorubicin on succinate dehydrogenase activity and protective effect of betanin in isolated mitochondria (A). Mitochondrial SDH activity was evaluated by MTT assay. Presented data showed doxorubicin (3.5 µM) significantly decreased SDH activity compared to control group. Also, data showed betanin significantly increased SDH activity compared to treated groups with doxorubicin (3.5 µM). Effect of doxorubicin on mitochondrial swelling and protective effect of betanin in isolated mitochondria (B). Mitochondrial swelling was evaluated by monitoring absorbance at 540 nm. Presented data showed doxorubicin (3.5 µM) significantly induced mitochondrial swelling compared to control group. Also, data showed betanin significantly inhibited mitochondrial swelling compared to treated groups with doxorubicin (3.5 µM). SDH, Succinate Dehydrogenase Activity; DOX, doxorubicin. Data presented are the mean ± SD (n = 3). ***p < 0.001 compared with the group with no pretreatment; ###p < 0.001 compared with the group with doxorubicin.
Mitochondrial swelling in isolated mitochondria
The decreased absorbance at 540 nm, as a criteria of mitochondrial membrane permeability and the mitochondrial swelling in isolated rat heart mitochondria was evaluated after addition of doxorubicin and betanin. Doxorubicin addition (3.5 µM) significantly caused mitochondrial swelling in treated isolated rat heart mitochondria as compared with untreated control group (p < 0.001). While pretreatment with 5 and 10 μM betanin significantly (p < 0.001) decreased mitochondrial swelling as compared with doxorubicin treated group (p < 0.001) (Figure 5(B)).
Discussion
Doxorubicin as a potent anticancer and the first-choice drug is used for many cancers such as the leukemia, lymphoma, breast, colon and liver cancer. 27 However, its use is decreased due to toxicity in several organs such the heart, skeleton, kidney, brain, muscle, liver and lung. 27 Among the above toxicities, the cardiotoxicity and cardiomyopathy are the most feared and serious side effect of this anticancer. 28 Despite the apparent toxicity of this drug due to its good effectiveness it is still widely used in the clinic. Since the main mechanism of action of doxorubicin is based on the inhibition of the enzyme topoisomerase II, causing DNA damage and induction of apoptosis and this mechanism is independent of the mechanism of its toxicity and oxidative stress. 29 Therefore, finding a solution to reduce the toxicity of doxorubicin during treatment can help to increase the effectiveness of the drug and reduce its toxic effects, especially cardiac toxicity. Therefore, this study aimed to provide a solution to reduce the toxicity of doxorubicin based on its mechanism of action through the mitochondrial-dependent oxidative stress at the cellular and mitochondrial level.
Doxorubicin-related cytotoxicity is often irreversible. 3 Doxorubicin accumulates in mitochondria and binds to the phospholipid cardiolipin in the inner mitochondrial membrane. This binding leads to inhibition of complex I and complex II and disruption of the electron transport chain and inducing ROS generation in mitochondria. 30 Also, free radicals might be generated by other doxorubicin-mediated mechanisms, for example a quinone segment in the structure of doxorubicin is reduced by mitochondrial complex I into a reactive semiquinone which transfers an electron to a molecule oxygen and produces the superoxide anion free radical. Then, the produced semiquinone free radical is oxidized and rebounds to the quinone form in a sequence of reactions which is known as the redox cycling of doxorubicin. 31 Other mechanisms such as the alteration in Iron homeostasis, 32 induction of mtDNA damage through binding to endothelial nitric oxide synthase (eNOS) and disruption of Ca2+ homeostasis is involved in doxorubicin-related cytotoxicity. 33 The above mechanisms directly and indirectly lead to ROS and peroxynitrite (ONOO−) formation, mitochondrial membrane permeability, mitochondria dysfunction, cytochrome c and leading to apoptosis or necrosis. 34 The increasing levels of TNFα and NADPH oxidase mediated by doxorubicin can also generate the excessive oxidative stress and lead to redox modifications of macromolecules. 35 Previous studies have been reported that the antioxidative defense of cells reduces after exposure to doxorubicin.12,34 Our studies on isolated cardiomyocytes and mitochondria showed that doxorubicin-induced cytotoxicity is exerted by oxidative stress, depletion of antioxidant defenses, production of reactive oxygen radical, and organelle damage such as lysosome, and especially mitochondria. Our data in the current study, are consistent with previously published studies. As shown in our results and other studies, mitochondria play a main role in doxorubicin-induced cytotoxicity and cardiotoxicity, 34 Therefore, protection of mitochondria and reduction of harmful factors to them can be an important strategy in reducing cytotoxicity and cardiac toxicity induced by doxorubicin.
Scientific documents have demonstrated the protective effect of the plant-derived antioxidant natural products on mitochondrial damage, oxidative stress and cardiovascular disease (CVD). 36 Several mechanisms such as the maintaining a redox state in the cells due to their scavenging ROS abilities, modulating the expression of genes involved in intracellular defense, binding to receptor sites as antagonists and competing for active sites in enzymes in various subcellular structures are suggested. 37 One of the plant-derived antioxidant natural products is betanin which has been proven that it’s antioxidant and anti-inflammatory activities can reduce the cardiovascular disorders. 17 Previous studies have reported the free-radical scavenger activity of betanin. 17 In the current study we showed that betanin significantly decreased ROS formation and oxidative stress induced by doxorubicin. The high level of ROS inside and outside mitochondria after exposure with doxorubicin leads to damaged mitochondrial components, opening of MPT pores, mitochondrial depolarization, high amplitude mitochondrial swelling and outer membrane rupture. 38 In the current study, increased mitochondrial depolarization and mitochondrial swelling were found in the doxorubicin group compared to control group, demonstrating mitochondrial toxicity of doxorubicin in isolated cardiomyocytes and mitochondria. These toxicity parameters were reversed in betanin groups, consistent with a protective effect of betanin on mitochondria.39,40 This has demonstrated that mitochondrial dysfunction damages lysosomal structure in a ROS-dependent manner 41 and that loss of Transient Receptor Potential Mucolipin (TRPML1) as a pore channel in lysosome induces the accumulation of ROS, which in turn cause loss of mitochondrial membrane potential and fragmentation of mitochondria. 42 It has been reported that doxorubicin accumulates in lysosomes as a hydrophobic weak base anticancer drug and may be associated with lysosomal damage. 43 Our findings in this study showed that betanin reduced doxorubicin-induced lysosomal damage in isolated cardiomyocytes.
In summary, we showed that betanin has protective effect against doxorubicin-induced cytotoxicity, oxidative stress and mitochondrial/lysosomal damages in isolated cardiomyocytes and mitochondria. The obtained results of the current study are consistent with previous studies for inhibition and reduction of cardiotoxicity and cytotoxicity induced by doxorubicin though antioxidant action.44–47 We also showed betanin’s antioxidant activities, protection of mitochondria and lysosome, are likely the mechanism responsible for its protection against doxorubicin. In the light of these findings, we concluded that betanin exerted protective role in DOX-induced toxicity in isolated cardiomyocytes and mitochondria, and it would be valuable to examine in vivo effects and its potential in clinical uses.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shahid Beheshti University of Medical Sciences, Deputy of Research with ethics code IR.SBMU.RETECH.REC.1399.725.
