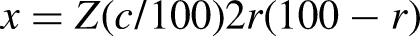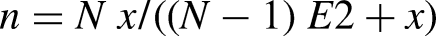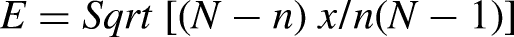Abstract
Introduction
5-Fluorouracil (5-FU) is a chemotherapeutic agent used to treat various types of cancers. Although widely used, it has consistently been attributed to cardiotoxicities after administration. The purpose of this study was to assess the parameters and predictors of cardiotoxicities associated with various 5-FU-based chemotherapeutic protocols in patients with GI/colorectal cancer, as well as the correlation of these cardiotoxic events with age, sex, cumulative dose, and risk factors such as obesity, hypertension, and family history of cardiac diseases.
Methods
A prospective study consisting of 396 patients of both sexes was conducted in the oncology ward of Nishtar Hospital in Multan, Pakistan. Patients were grouped according to the therapeutic protocol they received (5-FU monotherapy or in combination, with different dosing regimens). Electrocardiography and serum troponin levels were used to assess 5-FU-induced cardiotoxicity. In cases where cardiotoxicity was detected, 5-FU treatment was interrupted; nitroglycerin, nitrates, and calcium channel blockers were administered; and cardiac monitoring was initiated. 5-FU was discontinued in all cases of acute myocardial infarction.
Results
Of the 396 patients, 28.5% reported different cardiotoxic symptoms after receiving various 5-FU-containing protocols. 35% had anginal pain, 13% suffered a myocardial infarction, 11% developed hypertension, and 10% presented heart failure. Patients receiving 5-FU combination therapy showed cardiotoxic events that were significantly different from those on 5-FU monotherapy. Based on the ECG results, only the QTc-d interval increased significantly (p < 0.001) after therapy. 68% of the patients had troponin levels > 2 ng/mL at the end of treatment.
Conclusions
Pre-existing cardiac diseases, treatment duration, smoking, and obesity were found to be influential components in the development of cardiotoxicity, and patients with cancer should be closely monitored during 5-FU chemotherapy.
Introduction
A high proportion of cardiac events has been reported in patients receiving chemotherapy in the past few years. 1 Cardiac abnormalities that develop in response to chemotherapy need more attention in comparison with the usual cardiac events caused by drug toxicity due to overdose, for instance, as they could be spontaneous and occur suddenly. Therefore, the cardio-oncology field has recently emerged, which is a new discipline that provides knowledge and information for early screening and clinical monitoring of cardiovascular changes during cancer treatment to reduce oncological comorbidities and mortality. 2
5-Fluorouracil (5-FU) is an antineoplastic chemotherapeutic agent that suppresses the growth of tumour cells and is used clinically for the treatment of various types of cancer, including gastrointestinal cancers such as colorectal carcinoma. 3 Although 5-FU is tolerable in many patients, numerous cases have been reported with a wide spectrum of cardiac events associated with 5-FU administration, including angina, myocardial infarction, and cardiac arrest. 4 In general, previous studies of 5-FU-associated cardiotoxicities demonstrated that between 1.2 and 18% of patients experienced cardiac events, depending on the dosing regimen, treatment duration, risk factors, and use of 5-FU alone or in combination therapy.5–8
5-FU-associated cardiotoxicity was first reported in 1969, 9 and was attributed to myocardial toxicity and coronary artery constriction.10,11 Furthermore, it has been reported that 5-FU treatment caused severe vasoconstriction of the coronary arteries of rabbits during in vivo experiments.12,13 Additionally, it was found that most of the adverse effects associated with 5-FU administration are reversible and can be controlled by reducing the dose or temporary termination of drug administration. 13
In a case series study, it was reported that a combinational chemotherapy regimen (FOLFOX), which consisted of folinic acid, 5-FU, and oxaliplatin, led to various comorbidities that were caused by 5-FU-induced cardiotoxicity, such as myocardial damage and coronary spasm. 14 The prevalence of chemotherapy-induced cardiotoxicity, including that caused by 5-FU, has been extensively addressed in developed countries such as the United States and Europe.15,16
To the best of our knowledge, research studies on chemotherapy-induced cardiotoxicities in developing countries are insufficient and must be performed on large scales to gather more precise and representative results. Moreover, no single study to date has investigated the cardiotoxicity associated with 5-FU used in different combinational chemotherapeutic protocols as well as the impact of demographical data of developing countries’ populations on 5-FU-induced cardiotoxic events. Therefore, the aim of the current study was to evaluate the prevalence, clinical signs, and symptoms of cardiotoxic events associated with 5-FU treatment used in combinational therapeutic protocols in patients with gastrointestinal (GI) and/or colorectal cancers in Pakistan. This study also aimed to determine the influence of cardiovascular risk factors on 5-FU-induced myocardial toxicity. The outcomes of this study could be helpful in designing more elaborate research on various aspects such as pharmacokinetics, pharmacodynamics, and possibly pharmacogenetics of 5-FU, as well as in providing inferential data and conclusions about the clinical guidelines that must be set for cancer treatment schedules in Pakistan.
Methods
Study design
This prospective research study was conducted at the NISHTAR Hospital 25 B clinical oncology ward in Multan, Pakistan, from January 2020 to December 2020. Data collection was completed by accessing the patients’ medical records before and after treatment, who were given chemotherapeutic medications through protocols that predominantly contained 5-FU.
Additionally, a questionnaire (supplementary information), structured to record the demographic and socio-economic characteristics of the patients, was filled in by the hospital. Before enrolment in the study, written informed consent was obtained from all participating patients. Both electrocardiogram (ECG) and serum cardiac troponin levels are used as indicators for the assessment of cardiotoxicity, myocardial damage or reduced cardiac functions. This research study was approved by the ethical committee (Ref No: 366/pharmacypractice,2021).
Sample size
In this study, 396 patients of both sexes, with ages ranging from 18 to 70 years, following 5-FU treatment regimens were entered the study. The sample size was calculated using the Rao- Soft equation below, at a 95% confidence level. The error boundary in this study was approximately 5%.
where, n is the required sample size, Z is the confidence level at 95%, and E is the margin of error, which was 5% (standard value is 0.05).
Physical examination and electrocardiogram (ECG) recordings of about 30 min were performed prior to the administration of 5-FU as well as 1- and 3-months post-treatment with 5-FU regimen. Blood samples for troponin measurements were collected at the same time points for each treatment.
Inclusion and exclusion criteria
The study included patients who were diagnosed with solid tumours and were receiving 5-FU treatment. A history of cardiac diseases was also an inclusion criterion for the study. Patients who chose a proactive termination or change in the chemotherapeutic treatment regime against medical advice, were unwilling to participate in the study, were unconscious, or had unstable medical condition were excluded. If pre-existing cardiac diseases were detected during the initial phase of cardiac evaluation, these patients were excluded from the study.
Biomarker's measurement
In the present study, plasma concentrations of the biomarker troponin, a cardiac enzyme indicative of cardiac toxicity or stress, were measured. For biomarker measurement, blood samples were collected before and after the administration of 5-FU in each combinational therapeutic protocol. Blood samples (5 mL) were collected for measurement of cardiac troponin I (cTnI), a subtype that denotes cardiac damage. Blood samples were centrifuged immediately after collection, plasma was separated, transferred into EDTA glass tubes (purple-capped), and stored frozen at − 40 °C until analysis. The serum cTnI concentration was determined using a commercially available cTnI human assay kit (SMC® Human cTNi High Sensitivity Kit (03-0154-00), MERCK). The assay detection limit was 0.2 ng/mL and cTnI plasma concentration was considered abnormal if it exceeds 0.2 ng/mL.
Response to cardiotoxicity symptoms
Patients who experienced 5-FU-induced cardiotoxic events were treated with drugs used for cardiovascular diseases such as nitroglycerin, nitrates, and calcium channel blockers (CCB). Myocardial ischemia, arrhythmias, and anginal pain were monitored after drug administration.
Statistical analysis
Logistic regression analysis was performed to analyze the effects of various variables such as age, sex, pre-existing cardiac diseases, and history of chemotherapy and radiation on the development of cardiotoxicity after 5-FU administration. The results were considered statistically significant if the P value was < 0.05. Chi-square statistical analysis (α = 5%) was used to analyze the differences in ECG parameters in all groups. SPSS version 26 software was used to statistically analyze all variables.
Results
The chemotherapeutic treatment regimens used in the study are described in Table 1 below:
Patient groups based on chemotherapeutic treatment protocols.

IDFA/5-FU: Intermediate-dose folinic acid and 5-FU.
IDFA/5-FU/DP: Intermediate-dose folinic acid and 5-FU plus dipyridamole.
IDFA/5-FU after EAP relapse: Patients re-treated with 5-FU following a relapse from etoposide, doxorubicin (Adriamycin), and cisplatin (EAP) combination therapy.
ELF: Etoposide, Leucovorin and 5-FU.
In the current study, the ECG parameters of patients were recorded before and after therapy with the 5-FU regimen, as shown in Figure 1. Evaluation of the basal (before therapy) and post-treatment ECG parameters of the patients exhibited statistically significant differences only in the QTc-d interval (p < 0.001). The ejection Fraction decreased from 88.6% to 72.2%, indicating a dramatic reduction in cardiac function due to 5-FU therapy. Troponin plasma levels were determined before and after therapy, and the results demonstrated that 35% of male patients and 33% of female patients had troponin plasma levels greater than 1 ng/mL before treatment with 5-FU. After 5-FU treatment, approximately 43% of male patients and 23% of female patients had troponin plasma levels greater than 2 ng/mL.
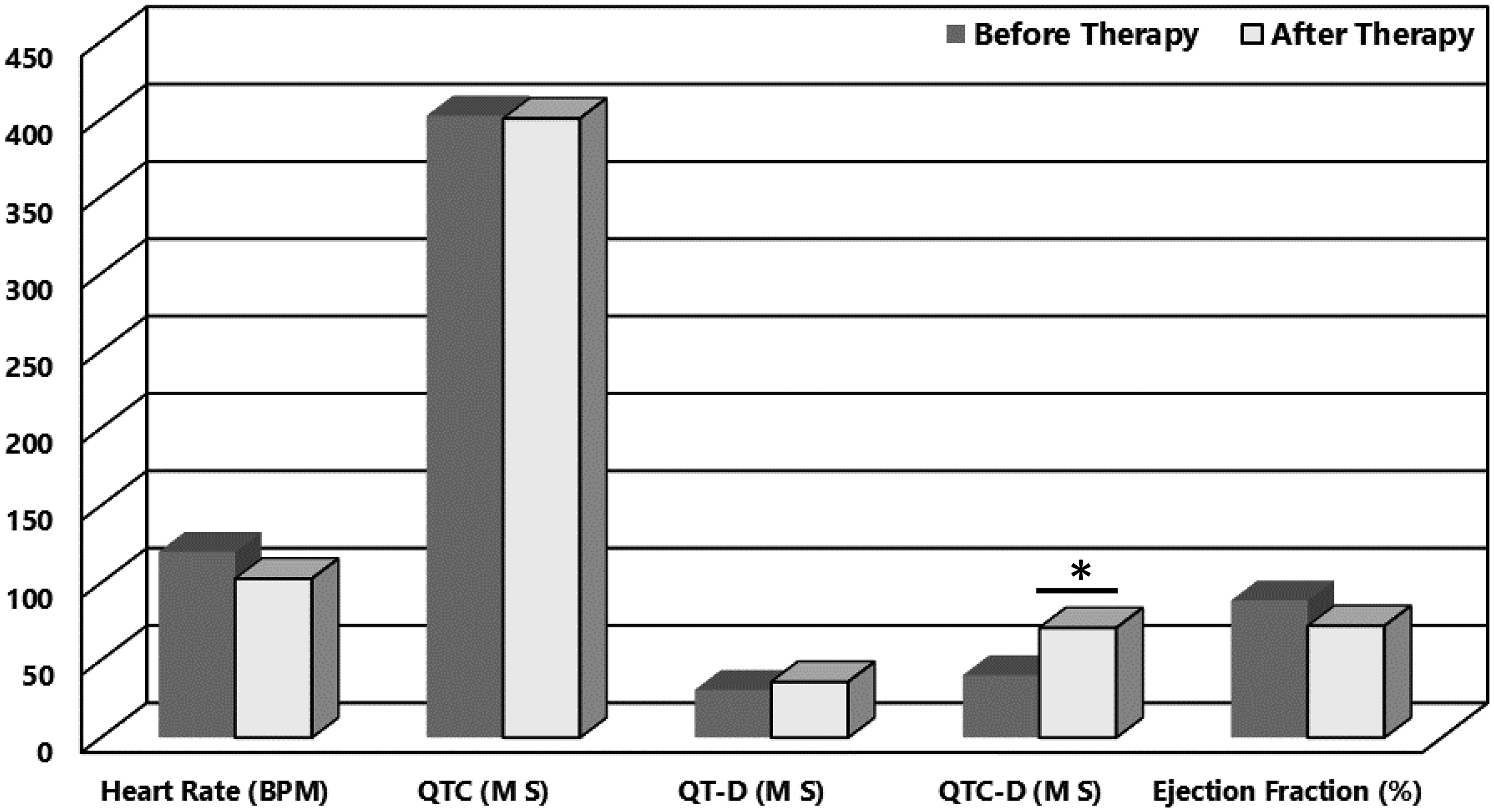
Electrocardiogram (ECG) parameters before and after treatment with the different 5-FU containing combinational therapies. Heart Rate is the estimated heart rate indicative for heart rhythm. *Denotes p > 0.05.
Cardiotoxic events such as myocardial ischemia, arrhythmias and angina that occurred to patients as a result of receiving the different treatment protocols were monitored and recorded. Table 2 displays the cardiac toxicities manifested in patients after treatment with intermediate-dose folinic acid & 5-fluorouracil (IDFA/5-FU) for 1 day and for 3 days, the same treatment protocol due to disease recurrence following a treatment with etoposide, doxorubicin (Adriamycin) and cisplatin (EAP), intermediate-dose folinic acid & 5-fluorouracil in addition to dipyridamole (IDFA/5-FU/DP), and combination of etoposide, leucovorin and 5-fluorouracil (ELF).
Reported cardiotoxic events associated with 5-FU treatment used in the different therapeutic protocols.
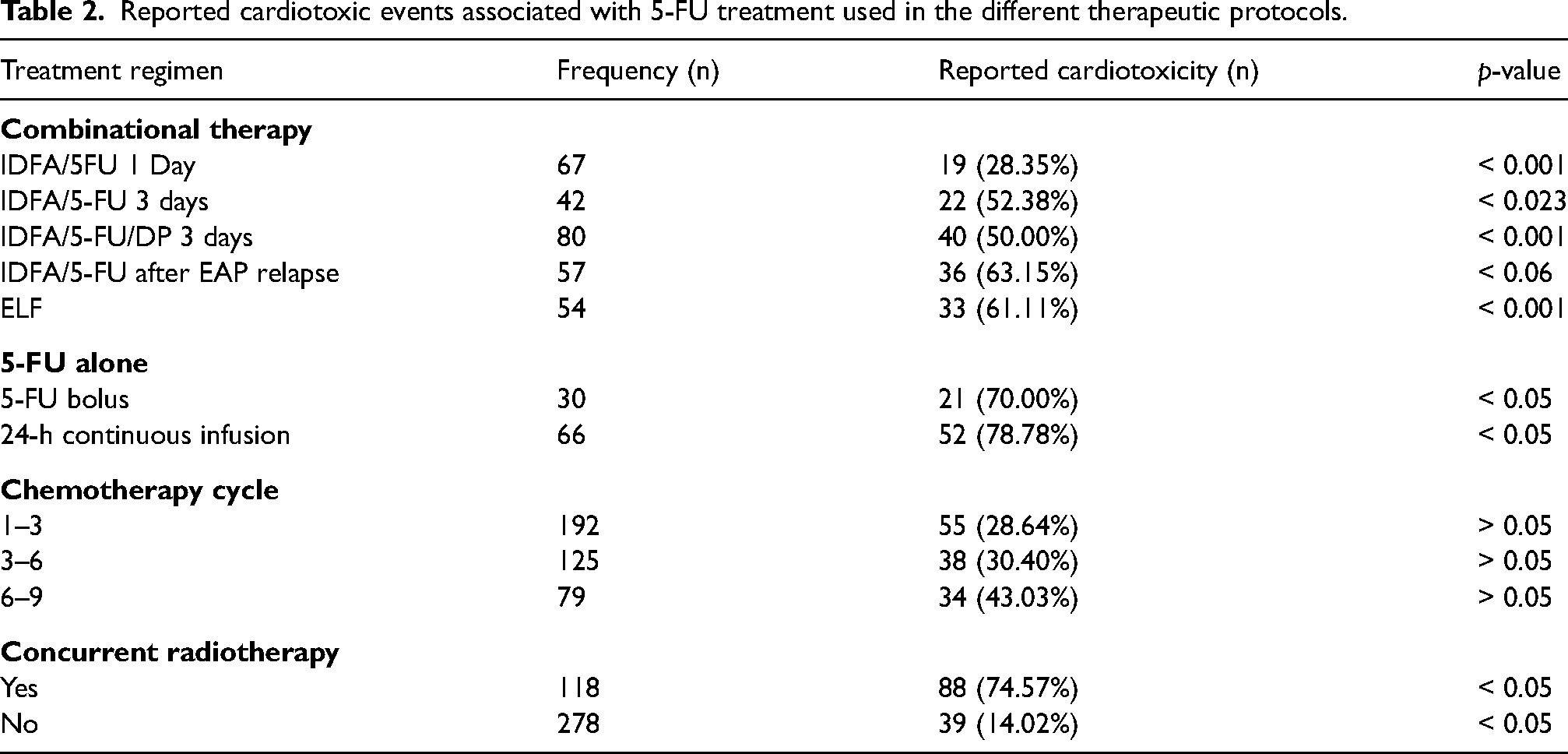
About 19 out of 67 patients developed minor yet significant (p < 0.001) changes in ECG after 1 day of treatment with IDFA/5-FU, while 22 out of 42 patients reported cardiotoxic events such as myocardial ischemia, arrhythmias, and anginal chest pain after 3 days of treatment. Moreover, 50% of patients receiving the IDFA/5-FU/DP therapeutic protocol reported ST elevation and ST depression. In contrast, about 63.1% (36 patients) of the group that received IDFA/5-FU after disease relapse from EAP treatment reported supraventricular arrhythmias. In the patient group of the ELF therapeutic protocol, approximately 61.1% (33 patients) of the total cohort reported cardiotoxicity.
This study also investigated the influence of 5-FU monotherapy (not in combination with any other chemotherapeutic agent) on the induction of cardiotoxic events. Seventy percent of patients receiving 5-FU alone as an IV bolus injection and 78.8% of patients receiving 5-FU alone as an IV infusion experienced cardiotoxicity.
As patients received treatment in different cycles, the influence of the number of chemotherapy cycles during the treatment on 5-FU induced cardiotoxicity was also investigated. The number of patients who experienced 5-FU cardiotoxic incidences was 55 out of 192 (28.6%), 38 out of 125 (30.4%), and 34 out of 79 (43%) after 1–3, 3–6 and 6–9 cycles, respectively. However, these differences were not statistically significant (p > 0.05). In addition, the correlation between concurrent radiotherapy treatment and 5-FU induced cardiotoxicity was investigated. The findings showed that 46.6% (55 patients out of 118) of patients who were receiving radiotherapy had cardiotoxic side effects, while only 14% (39 out of 278) of patients who were not receiving radiotherapy experienced 5-FU cardiotoxicity.
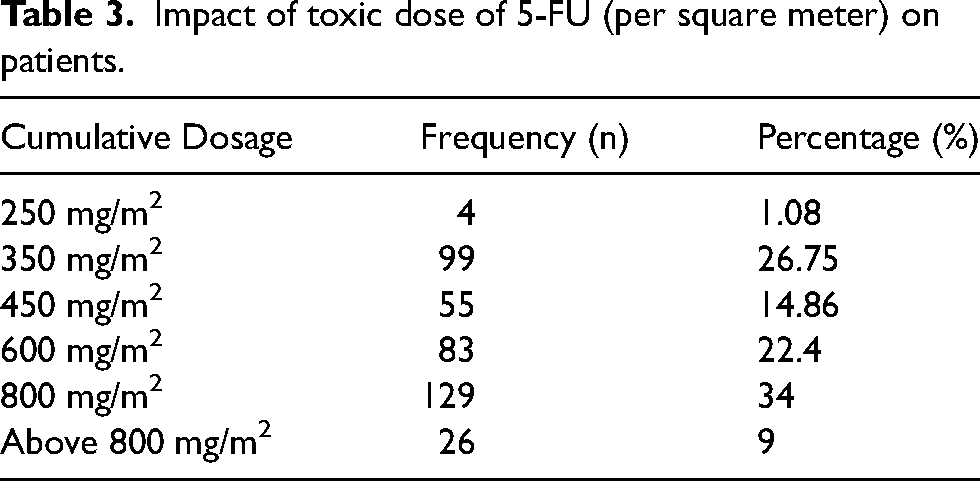
In addition, we investigated and analyzed the impact of the 5-FU dose per m2 (cumulative dose) on induced cardiotoxic events (Table 3). Our findings demonstrated that the incidence of 5-FU induced cardiotoxicity increases significantly at higher doses (≥ 350 mg/m2). At 250 mg/m2 5-FU dose, 1.08% of the patients had hypertension along with changes in ECG, whereas only minor abnormalities in ECG were observed in 26.75% of the patients (15 patients) treated with a 5-FU dose of 350 mg/m2. At a dose of 450 mg/m2 5-FU, 55 patients (14.86%) complained of chest pain and myocardial infarction, whereas cardiotoxic events were reported in 83 (22.4%) and 180 (34%) patients treated with 5-FU doses of 600 mg/m2 and 800 mg/m2 respectively. In the group of patients treated with a 5-FU dose greater than 800 mg/m2, 9% experienced sudden death due to congestive heart failure (CHF).
Impact of toxic dose of 5-FU (per square meter) on patients.
After the introduction of the different therapeutic protocols, patients who presented 5-FU-induced cardiotoxic symptoms were administered nitroglycerin, nitrates, and calcium channel blockers (CCB) at standard doses. The results showed that the administration of nitroglycerin, nitrates, and calcium channel blockers significantly decreased anginal pain by 80%, 70%, and 50%, respectively, reduced arrhythmias by 60%, 40%, and 45%, respectively, and reduced myocardial ischemia by 40%, 45%, and 60%, respectively, as shown in Figure 2.
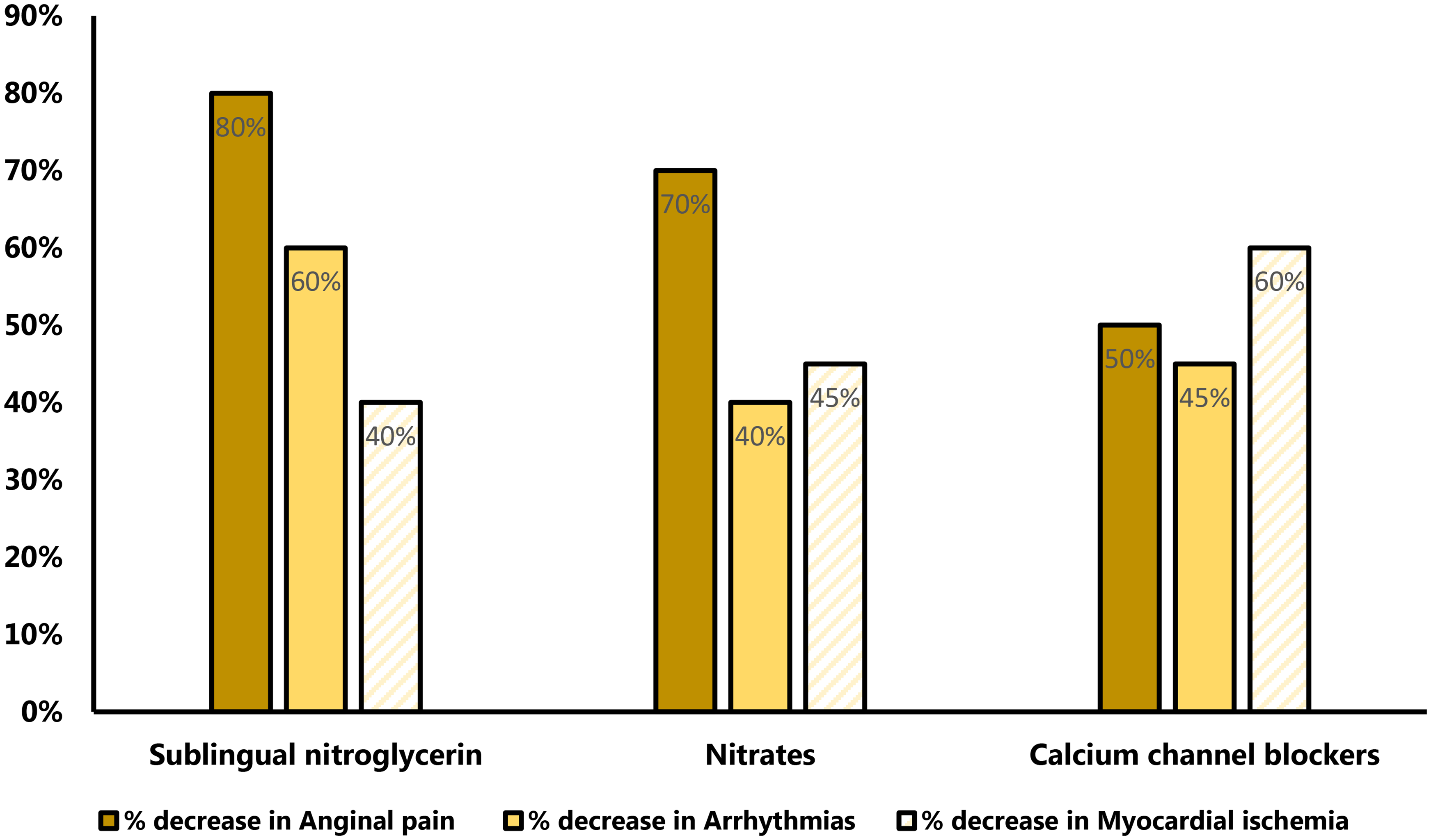
Percent reduction in the 5-FU-induced cardiotoxic events (anginal pain, arrhythmias and myocardial ischemia) after administration of various drugs used for the treatment of cardiovascular diseases such as sublingual nitroglycerin, nitrates and calcium channel blockers.
As patient's age can be a factor determining susceptibility to toxicity induced by drugs, the correlation between the 5-FU induced cardiotoxicity and patients’ age was also analysed. As in Figure 3, a direct correlation (statistically significant, p < 0.05) between patients’ age and development of 5-FU-induced cardiotoxicity was found, in such way that elderly patients show more toxicity than their younger counterparts.
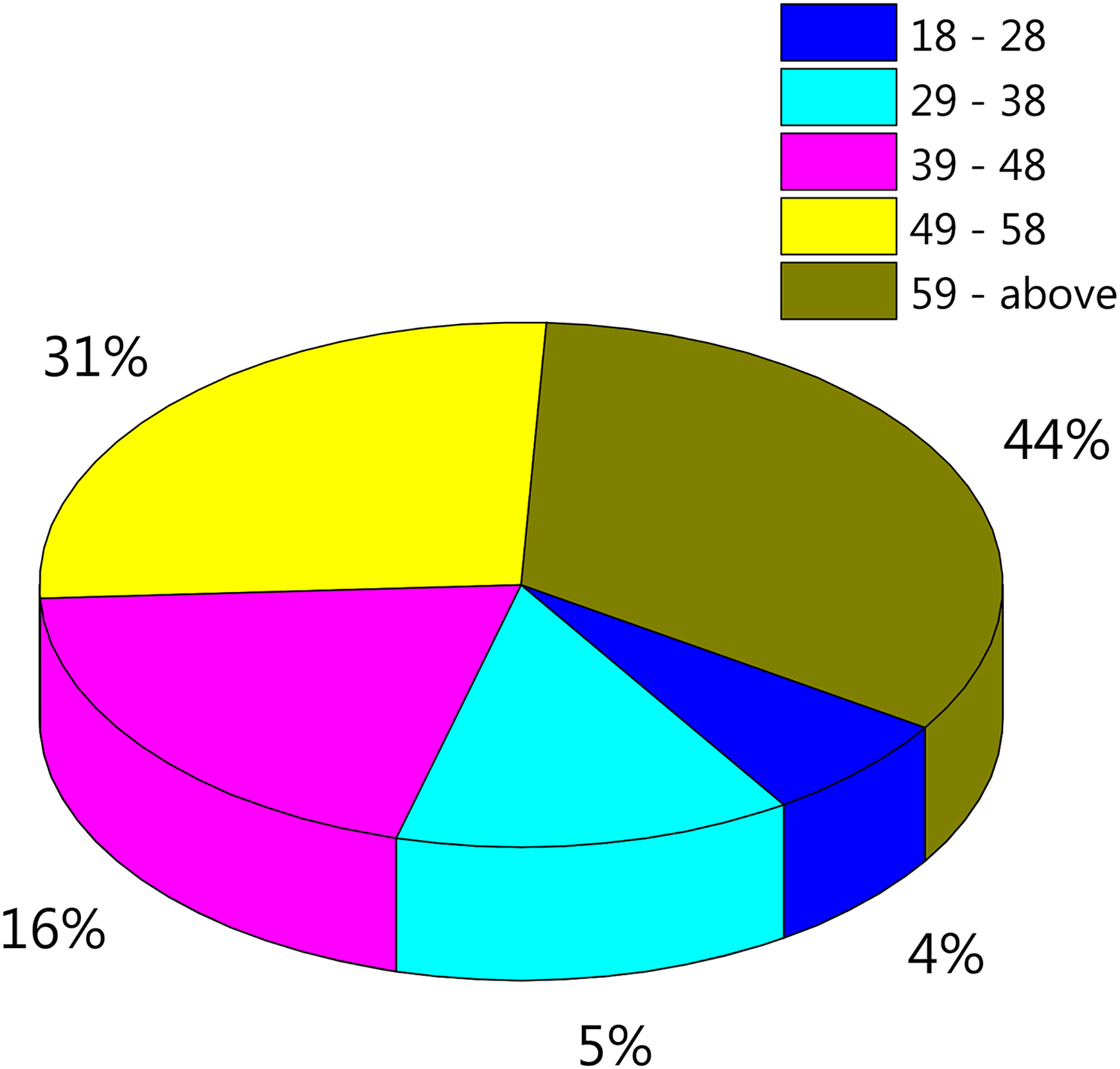
Percent cardiotoxic events occurrence in the different patients’ age groups. Patients were divided into 5 age groups, 18–29, 29–39, 39–49, 49–59 and above 59 years old.
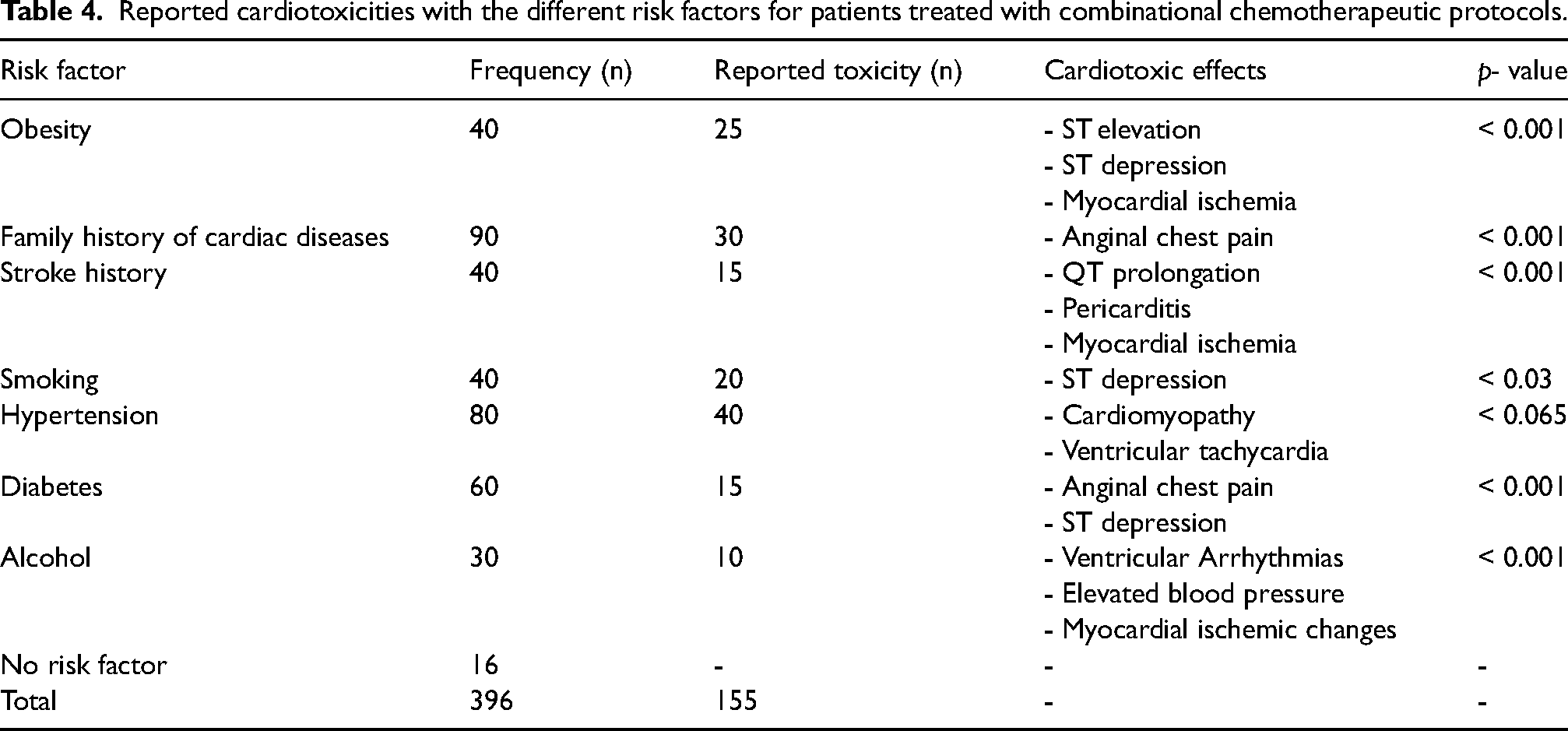
Risk factors such as family history of certain diseases can contribute to development of drug-induced toxicities. The correlation between different risk factors such a family history of cardiac diseases, history of stroke, comorbid diseases such as diabetes and hypertension as well as smoking, and alcohol consumption and the 5-FU induced cardiotoxicity was also studied as demonstrated in Table 4. The findings reported that about 33.3% of patients with family history of cardiac diseases had cardiotoxic events such as elevated blood pressure, anginal chest pain, and QT prolongation. Patients with history of cardiac diseases such as stroke had greater chances (37.5%) of developing cardiotoxicity. Examples are QT prolongation, pericarditis and myocardial ischemia caused by the administration of 5-FU. It was also reported that 62.5% of patients who were obese and received the 5-FU containing therapy had developed cardiotoxic events including ST elevation, ST depression, and myocardial ischemia. Moreover, it was noticed that about 25% of diabetic patients had cardiotoxicity, whereas 50% of hypertensive patients were reported to have cardiotoxic effects such as cardiomyopathy and ventricular tachycardia. On the other hand, 33.3% of alcoholic patients were reported to experience cardiotoxic effects such as elevated blood pressure, myocardial ischemic changes, and ventricular arrhythmias, and 50% of patients who were smokers had cardiotoxic side effects such as ST depression as displayed in Table 4.
Reported cardiotoxicities with the different risk factors for patients treated with combinational chemotherapeutic protocols.
Discussion
Although 5-flourouracil (5-FU) is one of the main chemotherapeutic agents used to treat gastrointestinal malignancies, it has consistently been attributed to cardiotoxicities. In this prospective study, the impact of cardiotoxicity induced by different 5-FU-containing treatment regimens in cancer patients with gastric and/or colorectal carcinomas was evaluated. Our results showed that patients who received an intermediate-dose folinic acid and 5-FU combination regimen (IDFA/5-FU) for one day exhibited minor changes in ECG parameters and a lower percentage of cardiotoxic events (28%). In contrast, patients who received the same regiment for 3 days continuously developed cardiotoxic events such as anginal chest pain, tachyarrhythmia, and myocardial ischemia. Also, administration of the same chemotherapeutic protocol (i.e., IDFA/5-FU) to patients who experienced relapse following treatment with the ELF regimen resulted in more severe cardiotoxic events such congestive heart failure (CHF) and cardiac cell death; however, p-value was more than 0.05. Combinational therapy with intermediate-dose folinic acid and 5-FU plus dipyridamole also resulted in cardiotoxicity. These cardiotoxic events lead to a substantial limitation of treatment options for patients receiving a 5-FU-containing chemotherapeutic regimen.17,18
Studies on 5-FU cardiotoxicities have reached a degree of consensus that treatment schedules have a direct effect on 5-FU-related toxicities. 19 Our present study showed that the 5-FU chemotherapy schedule had a clear influence on the development of cardiotoxicity, showing that intravenous 5-FU infusion increases the risk of cardiotoxicity in comparison with bolus treatment (Table 2). These results could be correlated to the short half-life of 5-FU (approximately 15–20 min) which means that it is rapidly cleared from the blood when administered as a bolus, while it accumulates when administered as a continuous infusion and hence poses an increased propensity for cardiotoxicity.20–22
The cardiotoxic events associated with 5-FU could be attributed to the direct lethal effect of 5-FU on cardiac muscles, where 5-FU is initially metabolized into α-fluoro-β-alanine (FBAL), followed by fluoro-acetate metabolites. These metabolites are associated with cardiotoxic events such as supraventricular arrhythmias. 23 These toxic effects can be prevented by the co-administration of di-hydro-pyrimidine dehydrogenase (DPD) enzyme inhibitors during 5-FU treatment, which inhibits the breakdown of 5-FU to FBAL, thus reducing the concentration of FBAL and its metabolites, consequently preventing the occurrence of detrimental adverse effects in patients experiencing 5-FU-associated cardiotoxicity. 24 Moreover, the proposed mechanism of 5-FU associated cardiovascular toxic events is thought to be due to a direct toxic effect on the vascular endothelium, including the inhibition of endothelial nitric oxide synthase, and consequently leading to coronary spasms and endothelium-independent vasoconstriction through protein kinase C.24,25
Our results showed that patients who were administered a 5-FU dose of 600 mg/m2 to 800 mg/m2 in different treatment regimens had a higher risk of developing cardiotoxicity than those who received low 5-FU doses of 200–400 mg/m2 (initial dose). This suggests that the cardiotoxic events associated with 5-FU administration could be dose dependent. Although some of the literature reports indicated that the dose dependence of 5-FU-related cardiotoxicity is unclear,17,26 similar findings to our results were reported in another study, where researchers concluded that patients treated with 5-FU dose greater than 600 mg/m2 had a high possibility of developing cardiac toxicity. 18
Patient monitoring and management of cardiotoxic events during 5-FU administration are crucial steps in the course of treatment to avoid further complications and lower mortality rates. In our study, patients were closely monitored, and nitroglycerin, nitrates, and calcium channel blockers were introduced to those who experienced anginal pain, arrhythmias, and myocardial ischemia. Our results showed that sublingual nitroglycerin and nitrates have a greater impact on reducing anginal pain than calcium channel blockers do. Moreover, sublingual nitroglycerin is more effective in reducing cardiac arrhythmias than the other drugs, whereas calcium channel blockers are more effective in decreasing myocardial ischemia. The results of a COHART study concluded that ECG monitoring, dose reduction, and the use of nitrates or calcium antagonists were effective in the early detection and control of adverse events of 5-FU administration. Also, some previous studies have supported that the use of vasodilator agents such as sublingual nitrates and calcium channel blockers is very beneficial in the management of cardiotoxicity.27,28
The incidence of 5-FU associated cardiotoxicity among different age groups was also assessed, and approximately 150 of 300 patients (50%) experienced cardiotoxic effects, as demonstrated in Figure 3. Our findings showed that the incidence of cardiotoxicity increased with age (Figure 3). This could be attributed to the fact that elderly people have declined renal functions, which reduces the renal clearance of 5-FU, thus leading to accumulation of the drug. In a previous study conducted by Cerny et al., most patients who developed 5-FU-related cardiotoxicity were aged greater than 55 years. 29
In the current study, we also investigated the influence of various risk factors and comorbidities on cardiotoxic events induced by 5-FU treatment (Table 4). Our data revealed that patients with a smoking history had a higher probability of developing ECG abnormalities than did non-smokers. This is ascribed to the fact that smoking has detrimental effects on cardiac functions, where it increases oxidative stress and inflammation. Smoking also causes cardiac systolic and diastolic dysfunction, ECG abnormalities, increased heart rate, risk of heart failure, diabetes, and cardiotoxicity. 30 Additionally, patients with a history of cardiovascular disorders, such as coronary artery disease, were more vulnerable to 5-FU related cardiotoxicity. Although previous cardiovascular disorders are not predictive of 5-FU associated cardiotoxicity, they may worsen this effect. 31 Moreover, patients who had a previous stroke were more vulnerable to 5-FU induced cardiotoxicity. This could be attributed to the fact that stroke deteriorates some parts of the central nervous system that regulate the heart, such as the impairment of the right hemisphere, which causes irregular cardiac rhythm complications. 32
Regarding obesity as a risk factor for 5-FU associated cardiotoxicity, obesity was found to increase the probability of myocardial toxicity in patients receiving 5-FU-containing treatment. This could be attributed to the effect of obesity in increasing cholesterol levels, which can lead to elevated blood pressure, and consequently, ECG abnormalities, indicative of elevated blood pressure, myocardial infarction, cardiomyopathy, and heart failure.33,34
Conclusions
This study was a benchmark for finding facts related to cardiac events associated with 5-Fluorouracil therapy. It highlighted the patient care scenario in Pakistan, especially in the case of GI and colorectal cancer patients. It was also concluded that patients treated with 5-FU in combinational chemotherapeutic protocols had increased chances of cardiotoxic adverse reactions which is suggested to be primarily related to 5-FU, rather than to the other chemotherapeutic agents. Therefore 5-FU administration should be closely monitored, especially when administered in combination with other drugs.
The study also concluded that patients receiving more intense chemotherapy i.e., a high dose for a short time, had an increased risk of cardiovascular events. Furthermore, patients who already suffer from cardiovascular diseases are at a higher risk of stroke and cardiomyopathy than their cardio-normal counterparts. Accordingly, practitioners should make patients aware of their cardiac check-ups during and even after chemotherapy.
The study also revealed poor clinical outcomes of chemotherapy because of the associated risks that were not taken into consideration. The fact that the presence of various comorbidities plays a significant role in triggering cardiac issues after chemotherapeutic treatment needs to be addressed and investigated at large scales, and researchers need to collect more scientific data to justify this in a more precise manner in the cancer patient population.
Our study also showed that risk factors, such as obesity and smoking, can aggravates the incidence and complications of 5-FU-associated cardiotoxicities. Hence, patients who receive 5-FU should be advised to stop smoking and maintain a healthy weight, as they are at a higher risk of developing cardiotoxicity after taking 5-FU drug if they are overweight or obese and smokers.
In summary, well-defined risk factors and their possible mechanisms should be identified and evaluated on a clinical basis and put together into a model for risk stratification, which could be used by clinicians to aid their decisions during treatment schedules for patients undergoing 5-FU based chemotherapy. Furthermore, a wide range of clinical trials should be conducted to assess and evaluate the various risk factors indicated in the present study, as wel as previous studies, and their clinical utility should be demonstrated.
Footnotes
Acknowledgment
All authors are thankful to all doctors, staff and patients at Nishtar Hospital Multan, Pakistan who voluntarily participated in the study
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Before enrolment in the study, an oral consent was taken from all subjects. The institutional ethical committee approved the research work (Ref No.366/pharmacypractice,2021).
Ethical approval
The ethics committee of Faculty of Pharmacy, Bahauddin Zakariya University approved this study (Ref No.366/pharmacypractice,2021).
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Author contributions
HR, MJ, and WR researched literature and conceived the study. SR, ZR, RS were involved in protocol development, gaining ethical approval and patient recruitment. HR wrote the first draft of the manuscript. AJ and MDY were involved in data analysis, review and editing of the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.