Abstract
Clear vision is dependent on features that protect the anatomical integrity of the eye (cornea and sclera) and those that contribute to internal ocular homeostasis by conferring hemangiogenic (avascular tissues and antiangiogenic factors), lymphangiogenic (lack of draining lymphatics), and immunologic (tight junctions that form blood–ocular barriers, immunosuppressive cells, and modulators) privileges. The later examples are necessary components that enable the eye to maintain an immunosuppressive environment that responds to foreign invaders in a deviated manner, minimizing destructive inflammation that would impair vision. These conditions allowed for the observations made by Medawar, in 1948, of delayed rejection of allogenic tissue grafts in the anterior chamber of mouse eye and permit the sequestration of foreign invaders (eg, Toxoplasma gondii) within the retina of healthy individuals. Yet successful development of intraocular drugs (biologics and delivery devices) has been stymied by adverse ocular pathology, much of which is driven by immune pathways. The eye can be intolerant of foreign protein irrespective of delivery route, and endogenous ocular cells have remarkable plasticity when recruited to preserve visual function. This article provides a review of current understanding of ocular immunology and the potential role of immune mechanisms in pathology observed with intraocular drug delivery.
Immune Privilege
Unlike other sites in the body including skin, demonstrating prolonged and sometimes indefinite survival of allogeneic skin grafts placed in the anterior chamber (AC) of the eye and into the brains of rabbits, forms the basis of the term immune privilege (IP) introduced by Sir Peter Medawar over 70 years ago. 1 The clinical success of corneal allografts observed by ophthalmic surgeons was then linked to IP by Rupert Billingham introducing the concept of immune privileged sites and immune privileged tissues, which tolerates foreign antigens. 2 Cornea, AC, vitreous cavity, and subretinal space are immune privileged sites in the eye where extended survival of foreign grafts is observed; and cornea, lens, pigment epithelium, and retina are immune privileged tissues that survive transplantation to nonimmune privileged conventional sites. 3 The concept of ocular IP encompasses the specialized protection available to the eye against destructive inflammation that can cause the loss of vital nonrenewable ocular cells and compromise visual integrity. Originally, IP was considered possible due to sequestration of antigens due to the presence of anatomical and physiological barriers but animal experiments demonstrate that systemic immune responses are generated against allogeneic lymphocytes injected in the AC. 4 This immune response deviates from the conventional immunity in that antibody responses are generated but cell-mediated immunity that causes graft rejection are downregulated. This distinct immune response is termed AC-associated immune deviation (ACAID). Such immune response is elicited, in humans and nonhuman primates (NHP), by both cell-bound antigens 5 and soluble antigens. 6,7 In NHPs, experimental pharmacologic redirection of a substantial fraction of aqueous humor (AH) outflow into the uveoscleral pathway, rather than trabecular meshwork (TM), results in loss of immune deviation. This finding suggests the TM to be the likely path of escape of intraocular antigens into the systemic circulation under physiological conditions. Subsequent studies in mice have confirmed this path by demonstrating splenic localization of blood-borne cell-associated signal derived from the eye that generates antigen-specific regulatory effectors. 8
The overall protective aspects of ocular IP and related systemic regulatory immune response, although applicable to most antigens, are known to vary in some cases. Cell-associated antigens on lymphoid cells that express major histocompatibility complex class II (MHC class II) and particulate bacterial antigens such as Mycobacterium and Listeria fail to induce systemic immune deviation when injected into the eye. 5,8,9 Similarly, soluble antigens such as ovalbumin, bovine serum albumin, and simian virus 40 large T antigen, once conjugated to latex beads, red blood cells, or expressed on tumor cells do not induce systemic immune deviation like their unconjugated forms. 10 Thus regulatory systemic immune response related to IP is dependent on the physical conformation of proteinaceous antigens introduced in the eye. 11 Particulate and conjugated antigens stimulate increased expression of pro-inflammatory interleukin 12 (IL-12) in ocular antigen-presenting cells (APCs) in contrast to soluble antigens that stimulated anti-inflammatory IL-10. 10 These observations indicate variable effect of antigens on intraocular APCs, which likely impacts the subsequent antigen-specific immune responses. These findings are particularly relevant to the emerging development of intraocular therapeutics that harness ocular IP to prevent adverse responses.
Innate Immunity and IP
Innate immunity is characterized by a nonspecific pro-inflammatory response that develops immediately after engagement of evolutionarily conserved “pattern recognition receptors” (PRRs) on cell surfaces. These receptors bind pathogen-associated molecular patterns (PAMPs) on microbes or danger-associated molecular patterns (DAMPs) represented by endogenous molecules released from damaged or dying cells. Examples of PRRs include Toll-like receptors (TLRs), co-receptor cluster of differentiation 14 (CD14) and scavenger receptor CD36, while PAMPs include bacterial cell wall components such as lipopolysaccharides (LPS) and lipoteichoic acid, and DAMPs include heat shock proteins and proteolytic fragments of extracellular matrix (ECM).
Expression of TLRs is also reported in nonimmune cells of the cornea, uvea, and retina. Human corneal epithelial cells express all TLRs; however, functional receptors were detected for TLR-2-5, -7, and -9. 12,13 Of these, TLR-2 and TLR-4 are detected intracellularly and fail to activate pro-inflammatory nuclear factor-κB (NF-κB) pathway in response to LPS, 14 and TLR-5 is expressed in the corneal basal layer with likely activation only after epithelial breach. It is suggested that such expression pattern of TLRs in the cornea represents a protective adaptation to prevent damaging effects of innate inflammation potentially triggered by normal bacterial flora. 12 Although transcripts for all TLRs were detected in primary cultures of human iris and retina pigment epithelial cells (IPE and RPE), functional TLR-2, -3, and -4 were detected in both IPE and RPE cells. 15 One limitation of these studies is that expression of TLRs is assessed in cultured cells and it is not known if similar pattern of expression is detectable in intact tissue. Also, it is not known if the expression of TLRs in ocular tissues is altered under inflammatory conditions or after injury; however, pro-inflammatory chemokines and cytokines are expressed in animal models (see below).
The ability of TLRs to activate NF-κB triggers release of pro-inflammatory cytokines such as tumor necrosis factor α (TNF-α), IL-1β, and IL-12 and results in activation of APCs such as macrophages and dendritic cells that shape the adaptive immune response. The lack of specificity of innate immune responses poses a risk of indiscriminate and deleterious tissue damage. Several factors have been identified in the AH that can limit such damage. These include inhibitors of complement activity (complement regulatory protein [CD46], complement decay accelerating factor [DAF; CD55], and membrane attack complex inhibitory protein [CD59]), inhibitors of neutrophil activation (α-melanocyte-stimulating hormone [α-MSH], CD95L), and inhibitors of natural killer (NK) cell activity (macrophage migration inhibitory factor [MIF]; transforming growth factor β2 [TGF-β2]). Soluble factors such asTGF-β2, thrombospondin-1 (TSP-1), and calcitonin gene-related peptide (CGRP) inhibit pro-inflammatory activation of macrophages and dendritic cells (factors reviewed by Streilein 3 ).
The existence of multiple mechanisms that preserve ocular IP is demonstrated in mice with experimental intraocular inflammation induced with an intravitreal (IVT) injection of a small concentration of LPS. 16 Despite the profound breakdown of the blood–ocular barrier (BOB) and intense inflammation, the inflamed eye continued supporting IP-related systemic suppression of cell-mediated immunity against antigen injected in the eye and immunosuppressive property of AH. In contrast, systemic LPS injection in susceptible mice causes transient loss of immunosuppressive property of AH that is restored within 48 hours. 17 These findings highlight the persistence of the ocular IP under innate inflammatory responses. However, it should be noted that these studies did not evaluate humoral immunity in animals with intraocular inflammation.
Adaptive Immunity and IP
Immune privilege-associated adaptive immune response includes cell-mediated as well as humoral immune response. Despite the absence of conventional lymphatic connections between the eye and secondary lymphoid organs, experimental studies in animals have demonstrated that antigenic material placed in the AC elicits antigen-specific immune response characterized by cytotoxic CD8+ T cells, CD4+ regulatory T cells and noncomplement fixing antibodies (immunoglobulin G [IgG]1), while excluding inflammatory CD4+ T helper 1 (Th1) and T helper 2 (Th2) subsets and B cells that secrete complement-fixing antibodies (IgG2a, IgG2b, IgG3).
Earlier studies in mice demonstrate that after injection of an antigen in the AC, antigen-carrying eye-derived APCs (F4/80+ cells) are detectable in the blood. 18 These findings suggest that cell-associated antigens exit via normal AH outflow path through TM and canal of Schlemm (“conventional outflow”) that drains into episcleral veins. 3 Later trafficking studies using fluorescence tagged molecules in rats indicate that soluble antigens rapidly reach draining lymph nodes within 30 minutes and other lymphoid organs by 24 hours after injection. 19 Similar studies in mice using cell-associated antigen indicate cellular traffic from the eye to the lymph nodes and spleen by 24 hours. 20 Therefore, in addition to antigen presented by ocular APCs that have been conditioned by TGF-β2, it is suggested that soluble antigens captured by lymph node resident APCs with low MHC class II expression prevents generation of inflammatory cell-mediated immune response. 3,21
In human eyes, human leukocyte antigen class II expressing APCs are not only located in the stroma of iris and ciliary body but within the anterior iridocorneal angle (ICA), which encompasses the area between the clear cornea and pectinate ligaments, also referred to as the internal limbus. 22 Expression of lymphatic vessel markers is detected on cells in both conventional and uveoscleral outflow routes of AH. 23 Pronounced expression of these markers in the conventional outflow tissue suggests this to be a favored route for eye-derived APCs supporting original observations from animal studies regarding their entry into blood circulation en route to spleen. Overall, the availability of lymphatic markers has facilitated identification of previously unknown uveolymphatic outflow pathway for AH drainage. 24,25 Formation of lymphatic vessels is reported in the human cornea under inflammatory conditions 26 and intraocularly as a putative wound healing response after scleral border compromise. 27 In mice, corneal inflammation induces growth of lymphatic vessels from lymphatic marker-expressing innate immune cells such as macrophages and dendritic cells in response to vascular endothelial growth factor C (VEGF-C). 28,29 Similar responsiveness to VEGF-C is demonstrated in endothelial cells of Schlemm’s canal (SC) in mice suggesting their potential to develop lymphatic vessels under pathological conditions involving VEGF-C overexpression. 30 In the mouse, inflammatory cells invading the cornea have been identified as a major source of VEGF-C. 28 Together these studies in mice suggest that cells in the anterior segment (AS) of the human eye that express lymphatic markers are lymphatic precursor cells capable of initiating lymphangiogenesis in response to suitable intraocular stimuli derived from infiltrating inflammatory cells. Thus, the existence of ocular lymphatic drainage implicates the ability of eye-derived APCs to induce systemic adaptive response against ocular antigens and that the anti-inflammatory or pro-inflammatory phenotype of these APCs can determine the nature of this systemic response.
Humoral response against soluble antigen injected in the AC of mice includes predominantly noncomplement fixing IgG1 antibodies against the antigen, unlike the complement-fixing IgG2 antibodies induced by the subcutaneous injection of the same antigen. 31 Being least opsonophagocytic, IgG1 antibodies are unable to clear circulating antigens. 32 In contrast, IgG2 antibodies rapidly clear circulating antigen. Murine studies have established that γ interferon (IFN-γ) derived from antigen-specific inflammatory CD4+ Th1 lymphocyte subset regulates IgG2a secretion while IL-4 derived from CD4+ Th2 lymphocyte subset regulates IgG1 antibody isotype secretion. 33,34 These studies have identified these antibody isotypes as indicators of underlying Th1 or Th2 response in mice. Immune privilege-related suppression of Th1 is consistent with the suppression of IgG2-secreting B cells in mice receiving intraocular antigen. Both mouse IgG1 and human IgG4 are reported to block complement activation and prevent IgG-induced inflammation. 35 Therefore, predominance of IgG1 response elicited by intraocular antigens is consistent with the protective function of ocular IP. It is not known if intraocular antigens in humans induce predominantly antigen-specific IgG4 antibodies. Induction of complement binding IgG1 or IgG3 antibodies against an intraocular therapeutic may serve as an indicator of potential inflammatory responses. Specific cytokines like IL-21 identified as switch factor for the production of IgG1 and IgG3 isotype during human B cell activation 36 may provide additional markers to shed light on potential humoral response elicited by any intraocular therapeutic.
Anti-Inflammatory Regulatory Mechanisms and IP
Conceptually, ocular IP affords protection to ocular tissues from the collateral damage of an inflammatory immune response. Such protection is achieved through several mechanisms that include anti-inflammatory cytokines, soluble as well as cell-bound receptors, neuropeptides (Table 1) and regulatory T cells induced as part of the systemic immune response. A high degree of redundancy exists among these mechanisms with respect to their ability to contribute to IP status of the eye. Although several inflammatory factors have been identified to counter some of these mechanisms, so far a only few factors have been experimentally demonstrated to result in a complete loss of ocular IP (Table 2).
Immunomodulatory Factors That Contribute to Immune Privilege.

Abbreviations: APC, antigen-presenting cell; CGRP, calcitonin gene-related peptide; CTLA-2α, cytotoxic T lymphocyte-associated protein 2 α; FasL, Fas ligand; IL, interleukin; LPS, lipopolysaccharide; MIF, macrophage migration inhibitory factor; MSH, melanocyte-stimulating hormone; NK, natural killer; NPY, neuropeptide; PEDF, pigment epithelium-derived factor; RPE, retinal pigment epithelial; SOM, somatostatin; TGFβ2, transforming growth factor β 2; TSP-1, thrombospondin-1; α-melanocyte stimulating factor; VIP, vasoactive intestinal peptide.
Select Factors Associated With Ocular Inflammation.

Abbreviations: IL, interleukin; IP, immune privilege; MCP-1, monocyte chemoattractant protein-1; TNFα, tumor necrosis factor α; TSP-1, thrombospondin-1; VEGF-α, vascular endothelial growth factor α.
a Factors experimentally demonstrated to result in complete loss of ocular IP.
Therefore, while some interacting components of IP are dispensable, the loss of certain critical factors, as indicated in Table 2, will destabilize the balanced mechanisms that maintain IP resulting in complete loss of IP.
Innate response inhibitors
In addition to TGF-β, macrophage MIF suppresses NK cell activity 37 while soluble Fas ligand (sFasL) suppresses neutrophil recruitment and activation. 38,52 Complement regulatory proteins (CRPs) such as membrane cofactor protein (MCP, CD46), DAF (CD55), and membrane attack complex inhibiting protein (CD59) are expressed by many intraocular cells and are also detected in aqueous and vitreous humor. 50,65,66 These CRPs prevent damage to ocular tissues due to activated complement system during an inflammatory response.
Cell surface receptors
The pigmented epithelia of retina and iris/ciliary body express cell surface receptors that contribute to an immunosuppressive ocular environment. Expression of CD95L/FasL on RPE cells promotes T effector cell death, 53 while the expression of galectin-1, 55 PD-L1 and CTLA-2α, and B7-2 on iris pigment epithelia promotes generation of regulatory T cells. 56,67 Soluble pigment epithelium-derived factor (PEDF) secreted by RPE cells inhibits LPS-driven macrophage activation to contribute to innate IP. 39 Pigment epithelial cells of both retina and iris/ciliary body express matricellular protein TSP-1 that facilitates activation of their latent TGF-β and generation of regulatory T cells. 64,68
TGF-β and regulatory T cells
Among soluble factors, immunosuppressive activity of TGF-β was identified the earliest in rabbit as well as human AH and 80% to 90% of the biologic activity was attributed to the TGF-β2 isoform. 40 This TGF-β2 is produced locally by ocular cells as a latent inactive form that requires proteolytic cleavage of latency-associated peptide (LAP) to release a biologically active form capable of binding TGF-β receptors on target cells to drive signal transduction. Active TGF-β2 regulates intraocular inflammation by suppressing activation of T cells, NK cells, and macrophages and confers an immunologic tolerance-inducing function on local APCs. 3,40 Antigen carrying APCs exposed to local TGF-β are able to secrete their own TGF-β2 69 and in the spleen through complex cellular interactions that include NKT cells, marginal zone B, and γδ T cells, eye-derived APCs activate regulatory T cells. 3,70 These regulatory cells have the capacity to suppress delayed type hypersensitivity (DTH) responses including Th1- and Th2-mediated pathologies. 71
Thrombospondin-1
Expression of the glycoprotein, TSP-1 in ocular parenchymal cells and ocular APCs facilitates the activation of latent TGF-β2, making its biologically active form available for immunosuppressive activities. 64,72 The significant contribution of TSP-1 to ocular IP is evident from its loss in TSP-1 deficient mice and resultant irreversible damage to the retina from uveal inflammation in these mice. 64 Dependence of ocular tissues on TSP-1 for their activation of latent TGF-β2 can be explained by the fact that while TGF-β1 and TGF- β3 isoforms are activated by integrins induced during inflammation, the LAP associated with the abundantly available TGF-β2 lacks integrin-binding site. 42 Activation of latent TGF-β is orchestrated by TSP-1 at the cell surface via its CD36 receptor thus allowing localized activation while avoiding deleterious fibrotic effects on ocular tissues. In addition to activating latent TGF-β, TSP-1 maintains the anti-inflammatory phenotype of ocular APCs by regulating their expression of inflammatory cytokines IL-12 and IL-23. 42 As an early response gene, expression of TSP-1 in mouse corneal cells is upregulated within 30 minutes of injury. 73 Therefore, it is possible that the technical procedure of intraocular delivery of antigens causes increased expression of TSP-1 at the site of injection facilitating localized activation of latent TGF-β in the eye to exert immunomodulatory effects. This is also relevant for intracameral (IC) and IVT injections as expression of TSP-1 is also reported in the sclera, choroid and Bruch’s membrane in addition to aqueous and vitreous humor. 74 –76
Neural regulation
Majority of TGF-β in the AH exists in its latent form which cannot exert its immunosuppressive effect without being activated. Neuropeptides, typically released from nerve termini, are immunomodulatory in AH. Vasoactive intestinal peptide (VIP) in AH inhibits T cell activation, while somatostatin and α-MSH inhibit secretion of IFN-γ from activated T cells that otherwise can abolish IP. 46,48,43,77 Calcitonin gene-related peptide inhibits secretion of pro-inflammatory nitric oxide by macrophages activated by endotoxin and IFN-γ. 47 Although the cornea is rich in afferent sensory nerves with termini releasing various neuropeptides, the proximities of the iris and ciliary body to AH makes their sympathetic (from the superior cervical ganglion) and parasympathetic (from the ciliary ganglion) nerves the likely source of neuropeptides in the AH. The contribution of sympathetic afferent nerves to IP is confirmed by the loss of IP in mice after surgical removal of the superior cervical ganglion that also results in reduced TGF-β concentration in AH. 78 In contrast to immunoregulatory neuropeptides, neuroinflammatory substance P abolishes IP. The increase in substance P and reduced VIP in the nerve termini of iris and ciliary body of mice reared in the dark (or dark adapted) results in loss of IP. 62 Similarly, release of substance P after retinal injury and severing of the corneal nerves causes loss of IP. 63,79 These findings suggest that tonic stimulations derived from the afferent nerves in the cornea and iris and ciliary body support maintenance of the local immunosuppressive microenvironment and that the nervous system makes critical contribution to ocular IP. 77
Blood Ocular Barrier
The BOB provides physical protection supporting IP in the eye. Several unique features of the ocular tissues and microvasculature of the anterior and posterior segments of the eye (Figure 1) limit flux of inflammatory cells and some solutes between blood and most parts of the eye. These anatomical features present restrictive barriers that tightly regulate ocular homeostasis and protect vision from toxins, pathogens, inflammation, injury, and disease. Two major components of BOB are blood–aqueous barrier (BAB) and blood–retinal barrier (BRB; Figure 2). In the anterior segment (AS) of the eye (Figure 3), tight junctions of the nonpigmented epithelial cells of the ciliary body and continuous nonfenestrated capillaries of the iris allow BAB to limit passage of small and large molecules in the AH. 11,55,56 In the posterior segment (PS; Figure 4), BRB is formed at 2 levels; while tight junctions of the RPE cells form the outer BRB, the nonfenestrated retinal vessels form the inner BRB to regulate fluid and solute permeability. 80 The restrictive nature of BOB also prevents diffusion of the drugs to intraocular structures, requiring efforts to modulate or bypass BOB for delivery of therapeutics. Although BOB is permissive to specific molecular size, it allows the passage of immune cells like T cells and bone marrow–derived myeloid cells. In animal studies, specific observations are made with regard to the ability of activated, but not naive (resting), T cells to cross BRB, which is facilitated by vascular adhesion molecules. In contrast, activated macrophages fail to cross BRB. 80
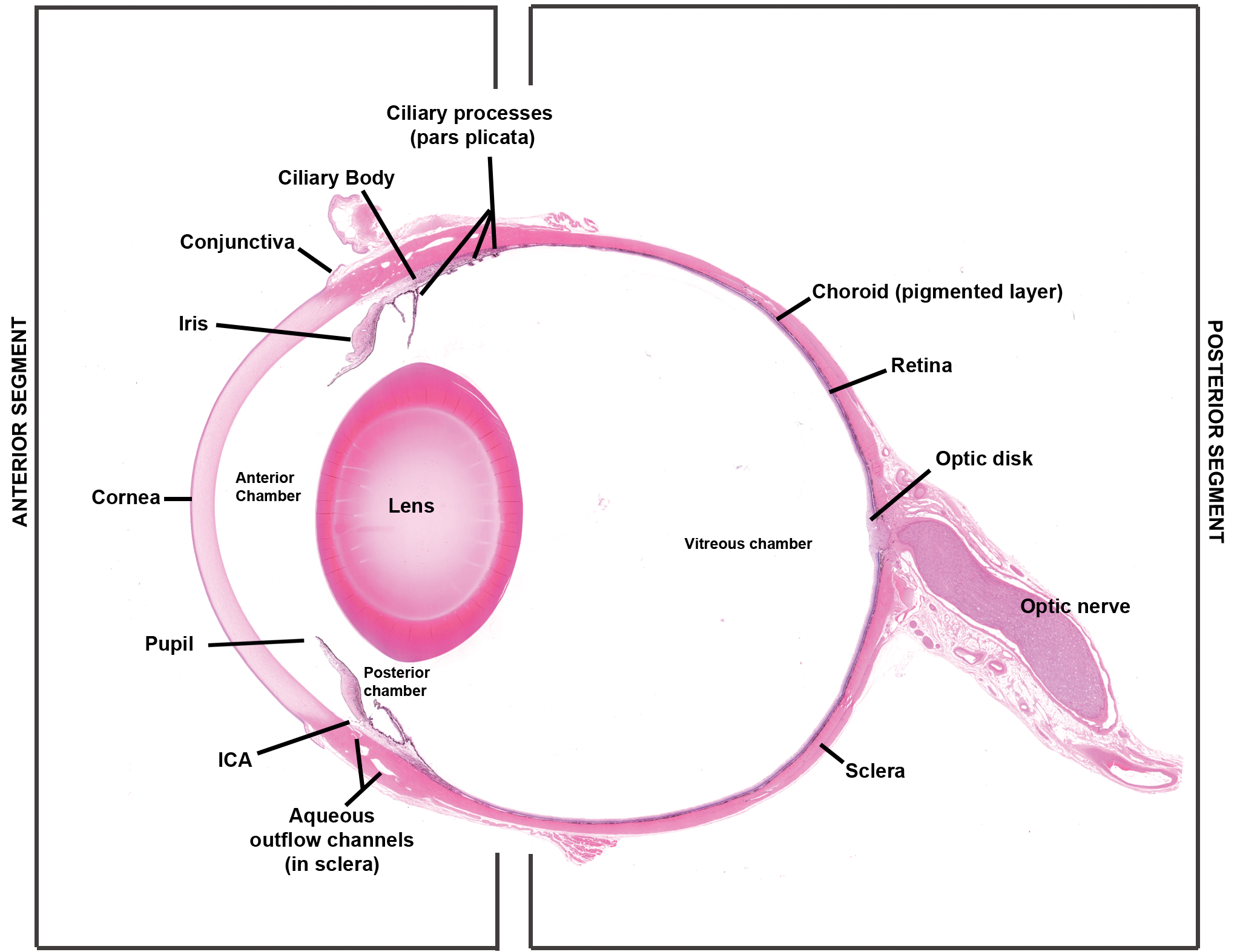
Structures and compartments of the eye. The eye can be divided into 2 segments: anterior and posterior. The anterior segment consists of the lens and all structures anterior to it and is divided into 2 chambers: anterior chamber and posterior chamber. The anterior chamber is delimited by the anterior surface of the iris and the posterior surface of the cornea. The posterior chamber is delimited by the posterior surfaces of the iris and ciliary body, the ciliary processes and zonules, and the anterior capsule of the lens. Aqueous humor is secreted by the ciliary epithelium lining the ciliary processes and enters the posterior chamber. Aqueous is formed by the ciliary body and released into the posterior chamber. It circulates through the pupil to the anterior chamber and exits the anterior chamber by passive flow through the iridocorneal angle. The posterior segment is bound anteriorly by the posterior lens capsule and peripherally by the sclera. H&E stained section of a dog eye.
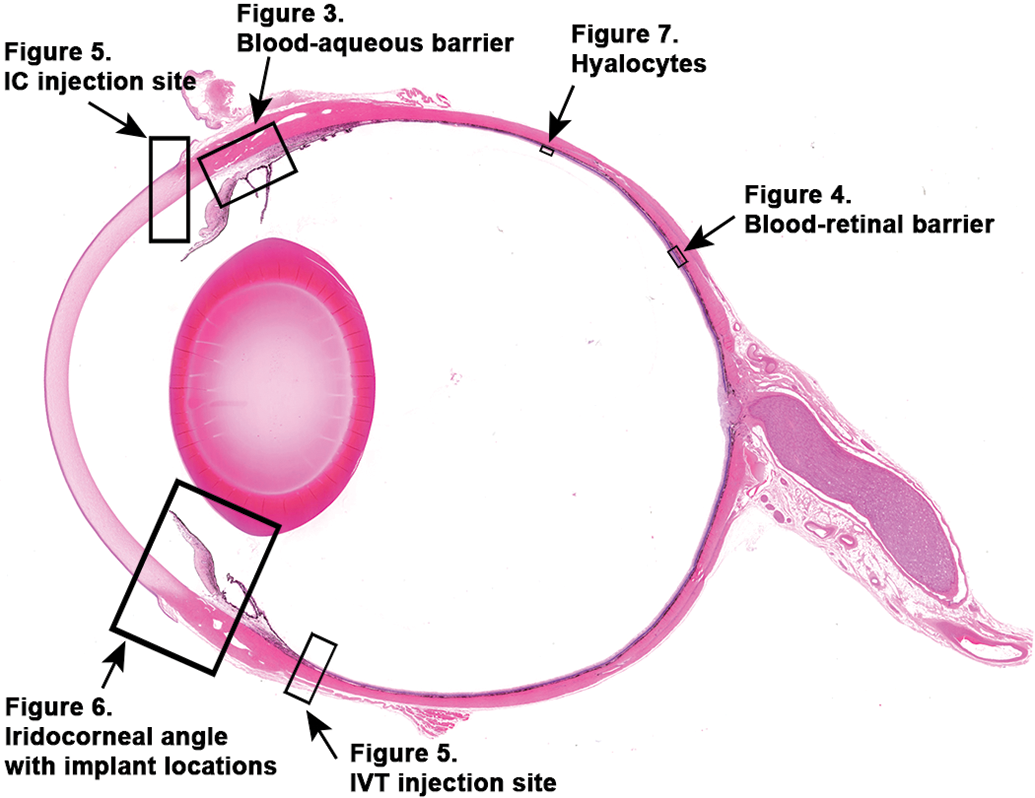
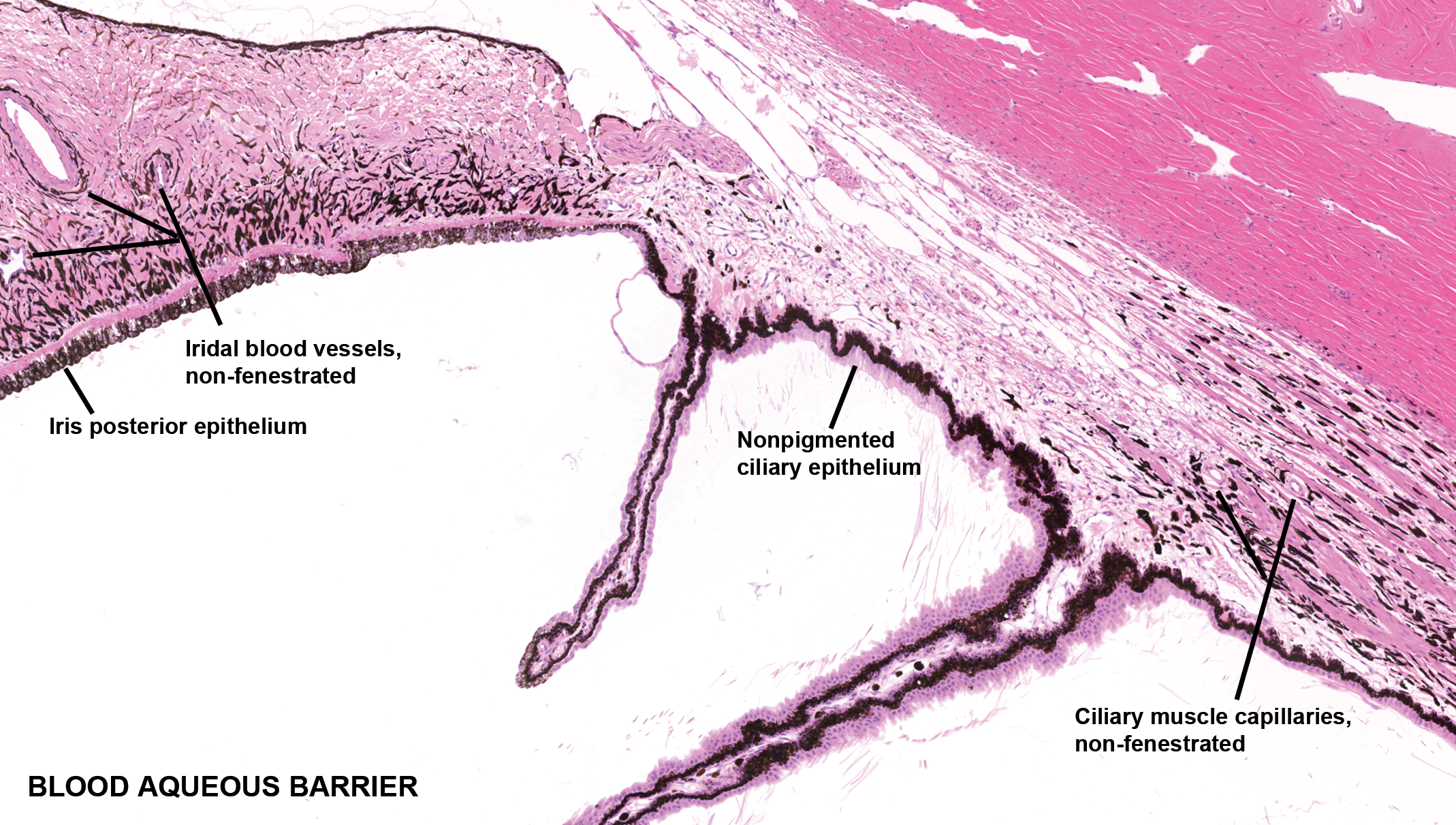
Blood–aqueous barrier (BAB) of the uvea (iris and ciliary body) in the anterior segment. Components of the BAB in the anterior segment are composed of tight junctions between pigmented epithelial cells of the iris and nonpigmented epithelial cells of the ciliary body and vascular endothelial cells of nonfenestrated blood vessels in the iris. H&E stained section of a dog eye.
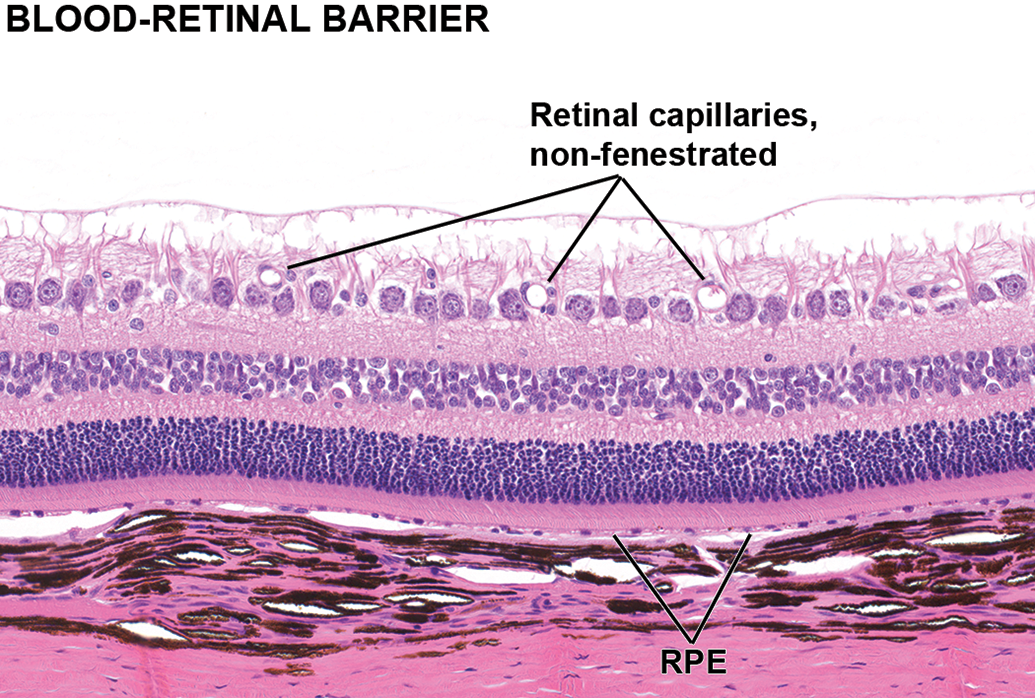
Blood–retinal barrier (BRB) of the posterior segment. The BRB is composed of tight junctions between endothelial cells of nonfenestrated retinal blood vessels and between retinal pigment epithelial (RPE). The capillary bed immediately subjacent to Bruch’s membrane, the choriocapillaris, is fenestrated, while the other choroid vessels are nonfenestrated. H&E stained section of a dog eye.
Intraocular Therapeutics and Related Pathology
Anatomic barriers and physiologic clearance mechanisms pose hurdles in delivering drugs to intraocular targets. Topical administration of drugs for intraocular delivery is relatively safe; it may be associated with contact irritation leading to keratitis and conjunctivitis but rarely leads to toxic injury of intraocular structures. However, due to the complicated anatomical structure and a small absorptive surface of the eye, it is difficult to achieve and maintain effective concentrations of drugs targeted to intraocular target sites when administered topically. Intraocular delivery of drugs allows for more direct administration of an ocular drug to its target tissues in both the anterior and posterior segments of the eye. At present, compared to topical and systemic drug delivery routes, the intraocular drug delivery route is the most direct approach to overcome BOBs and to achieve highest intraocular bioavailability of the drugs. Effective drug concentrations can be maintained in these tissues with frequent dosing or through the use of sustained release drug delivery systems (DDS). However, intraocular drug administration is the most invasive form of drug delivery and involves direct penetration by injection across ocular barriers. There are real and potential risks to intraocular drug delivery that are associated with administration and with long-term exposure of intraocular structures to the drug or drug delivery device. Immunological and pathological complications associated with these potential risks will be presented and discussed here.
Anterior Segment
The AS is the front third of the eye and includes all ocular structures anterior to the vitreous chamber: cornea, iris, ciliary body, internal limbus, TM, and lens (Figure 1). The AS is divided into two AH-filled spaces, the anterior and posterior chambers. The anterior chamber (AC) is bordered anteriorly by the corneal endothelial layer and posteriorly by the iris anterior limiting layer. The posterior chamber (PC) is bordered anteriorly by the iris posterior epithelium and ciliary posterior epithelium, and posteriorly by the anterior lens capsule. The AH is secreted by the ciliary epithelium into the PC and flows across the posterior epithelial surface of the iris and the anterior surface of the lens capsule and through the pupil into the AC. Within the AC, AH flows across the corneal endothelial surface and anterior iris limiting membrane before exiting peripherally through the ICA, an angular recess formed by the junction of the internal limbus, TM, iris root, and ciliary body 81 (Figure 6). The AH passes through the nonfiltering area of the ICA consisting of the internal limbus and pectinate ligaments and follows either the trabecular outflow path or uveoscleral outflow path. Via the trabecular outflow path, AH passes through the porous filtering region, the TM. The TM surface cells (TMC) are phagocytic and function as prefilters that remove cellular debris from AH as it flows into the less porous juxtacanalicular connective tissue (JCT). The TM creates most of the AH outflow resistance at the level of the JCT, 82 and this resistance generates the intraocular pressure (IOP). Intraocular pressure stabilizes the shape of the eye and maintains constant distances between the retina and refractive surfaces of the cornea and lens. From the JCT, AH enters SC in primates or the aqueous angle plexus in other mammals and drains through the aqueous veins into general circulation. 83 –86 Aqueous humor also flows via the unconventional or uveoscleral outflow path, where it passes through intercellular spaces of the ciliary muscle and drains either through the sclera into the orbital veins or through the choroid into the vortex veins. The contribution of the uveoscleral path to the total AH outflow varies significantly between species and appears to diminish with age. 85 Aqueous humor contains many soluble factors that modulate immunity and inflammation including complement regulating proteins, leukocyte inhibitors, anti-inflammatory cytokines, and noncomplement-fixing IgG1. 31,40,50,65,66 Thus, AH factors promote self-regulation by ocular immune cells and suppression of inflammation, which is critical to maintaining IP. 3 Two important AH factors are the immunomodulators TSP-1 and TGF-β2. 40,75 Many ocular cells produce TSP-1, including the iris epithelium, ciliary epithelium, corneal endothelium, and TMCs. 42,68,73,87 –89 As a matricellular protein, TSP-1 mediates cell-to-cell adhesion, cell to matrix adhesion, migration-proliferation, ECM remodeling, and TGF-β2 activation. It is also known to play contrasting pivotal roles in the AS based on the cell type and cell receptors involved. For example, TSP-1 promotes corneal endothelial cells (CECs) migration in wound healing but suppresses vascular endothelial cell migration and proliferation and promotes apoptosis. 42
The predominant cytokine in AH and AC, TGF-β2, is synthesized and released in a latent form by ciliary epithelium and lens anterior capsule epithelium. It is also secreted by lens capsule epithelium, corneal epithelium, keratocytes, CECs, anterior lens epithelium, Schwalbe’s line cells (SLCs) of the internal limbus, and TM cells into their respective basement membranes or ECM. 75,40 Together, TSP-1 and TGF-β2 regulate intraocular inflammation by suppressing pro-inflammatory activation of macrophages and dendritic cells, and promote wound healing through transdifferentiation of various cell types into fibroblasts and myofibroblasts in the cornea, lens, and ICA. 28,40,89,90
Anterior segment therapeutics
Intracameral injection (injection into the AC) is an increasingly utilized technique for administration of drugs or therapeutic devices to the AS. The most common drug indications are glaucoma and postsurgical antibiotic and anti-inflammatory prophylaxis. Intracameral injection (Figure 5) of drugs in solution or suspension allows for administration of an ocular drug directly onto target tissues within the AC rather than indirectly by topical application; however, effective drug exposure is still limited by the rate of AH exchange, which ranges from 1% to 1.5% of the AC volume (ACV) per minute and results in the replacement of the entire volume of AH within 2 hours. 82 Intracameral sustained release DDS are designed to provide prolonged localized drug exposure to target tissues while limiting systemic exposure and potential side effects. 91,92 Intracameral DDS in the form of biodegradable implants have recently been approved for delivery of anti-inflammatory and antiglaucoma therapeutics to circumvent the limitations associated with topical and IC solution and suspension formulations, which include poor transcorneal or transscleral tissue penetration into the AC and short half-life within the AC. 93 –97
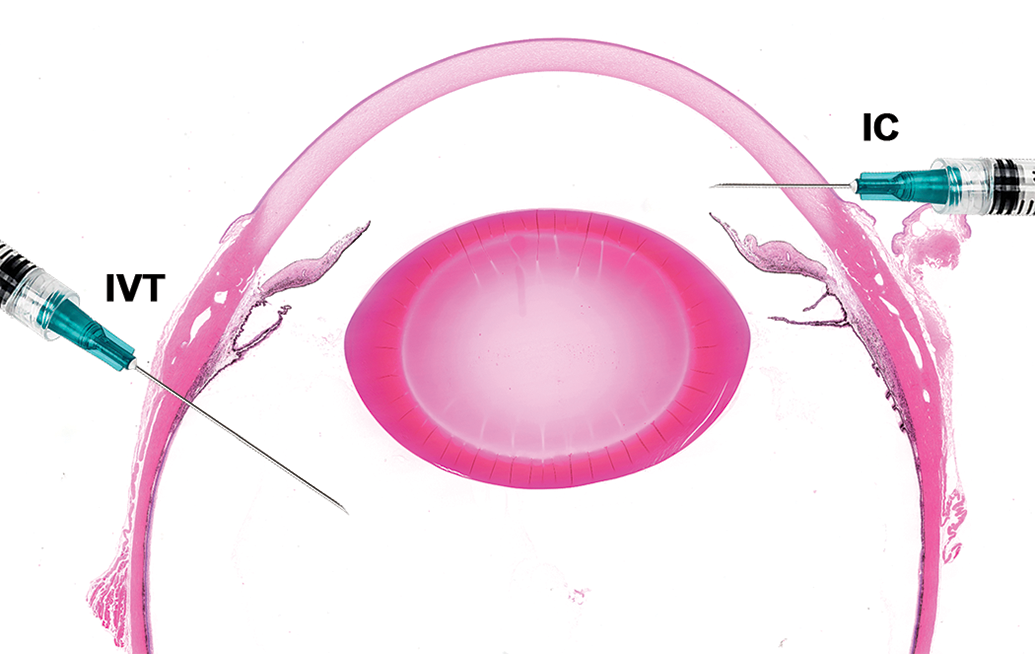
Injection sites for intracameral (IC) and intravitreal (IVT) drug delivery. Intracameral injection: needle entry into the anterior chamber through clear cornea adjacent to the limbus (corneoscleral junction) in the superotemporal quadrant with the needle positioned parallel to the iris plane and needle bevel facing the cornea to avoid injecting the drug solution or implant toward the anterior surface of the iris, the needle tip does not extend to the pupil to avoid contact with the anterior lens capsule. Intravitreal injection: needle entry into sclera posterior to the superotemporal or inferotemporal limbus initially at 15° to 30° angle directed toward the posterior pole of the globe, then needle angle is increased to 45° to 60° as it passes through the sclera and into the mid-vitreous. H&E stained section of a dog eye.
Species used for preclinical evaluation of IC drugs
Anterior chamber dimensions of some species such as mice and rats preclude the IC administration of a certain volume and may not accommodate a solid injectable implant because the ACV are small and the AC depths (ACD) are relatively shallow. 98 In rabbits, cats, dogs, pigs, and NHP, the ACV and ACD are great enough to accommodate larger volumes of drug solutions, 98,99 but their ICA dimensions may not be large enough to accommodate a solid drug implant designed to fit within the internal limbus at the opening of the human ICA. The relevant ICA size parameters for determining whether an angle can accommodate an implant are AC angle, angle opening distance, and angle recess area. Anterior segment optical coherence tomography (OCT) technology has been used to measure ICA parameters in multiple species, including the New Zealand White rabbit, domestic cat, Beagle dog, cynomolgus macaque, and rhesus macaque. 100 –102 The dog and rhesus macaque ICA dimensions are the closest to humans and can be used in safety studies for large IC implants or multiple IC implants. 103 Figure 6, a sagittal section of the ICA in the dog, shows the typical location of the IC implant(s) deep within the angle.
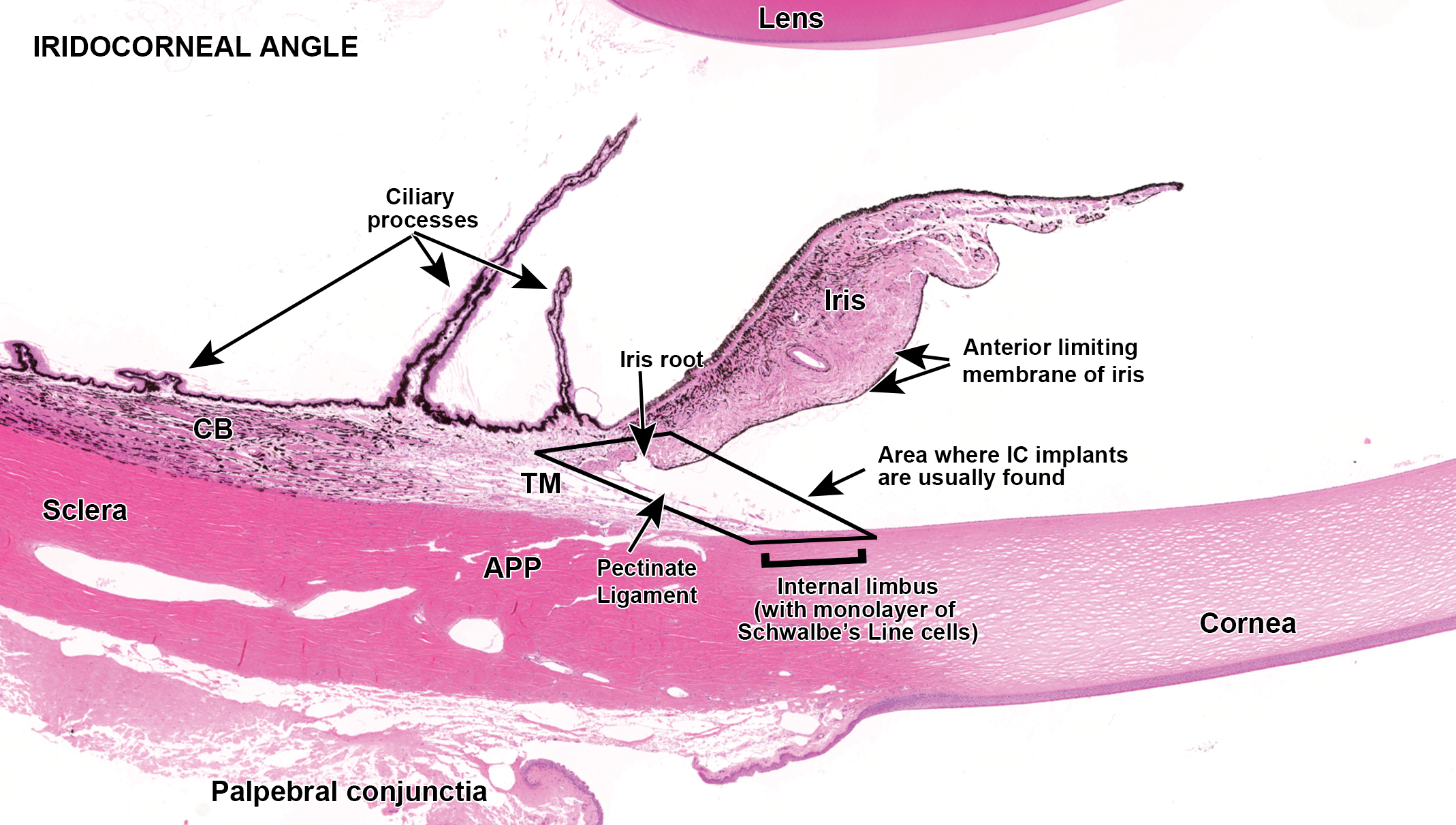
Intracameral implant placement at the entrance of the iridocorneal angle (ICA) and location where intracameral implant(s) settle (rhomboid box). The implants adhere to the ICA surface in an area delineated by the rhomboid box. Implants injected into the anterior chamber are swept by aqueous flow into the entrance of the ICA and in time adhere to the Schwalbe’s line cells lining the inner limbal surface, the trabecular meshwork (TM) cells lining the pectinate ligaments at the opening of the TM, the iris anterior limiting membrane at the iris root, or all 3 if the implant is large or there are multiple implants adhered to each other. APP indicates angle plexus. H&E stained section of a dog eye.
Intracameral injections
The IC injection involves needle entry into the AC through clear cornea at the corneoscleral junction with the needle positioned parallel to the iris plane (Figure 5). The needle bevel faces the cornea to avoid injecting the drug solution or implant toward the anterior surface of the iris and the needle tip does not extend to the pupil to avoid contact with the anterior lens capsule. Common consequences of IC injections include physical tissue injury from the needle injection and implant placement, hemorrhage (hyphema), and infectious endophthalmitis. 98 The immunopathology of wound healing subsequent to IC injection-related injury is discussed below. The immunopathology of endotoxin introduction and infectious endophthalmitis is addressed in the sections for innate immunity and the posterior segment.
Common procedure-related consequences
Transcorneal needle puncture wound
Wounding of the cornea upregulates the synthesis of soluble TSP-1 and expression of TGF-β receptors types I and II (TGFβRI, TGFβRII), and αv integrins in the epithelium, keratocytes, and endothelium. 73,87,104,105 Latent TGF-β2 is introduced to the wound via the tear film, AH, and upregulated expression in injured corneal epithelial cells and endothelial cells. It is activated through binding to TSP-1 and CD36. 28,42,73,88,106 –114 Active TGF-β2 stimulates exposed corneal epithelial basal cells at the wound edge to cover the epithelial defect through integrin β1-mediated migration and to restore the basement membrane through fibronectin secretion. 115
Within the exposed corneal stroma, TGF-β2 stimulates transdifferentiation of keratocytes into fibroblasts and myofibroblasts at the wound edge. 116 –118 Platelet-derived growth factor (PDGF) from tears and secreted by injured corneal epithelial cells binds to PDGF receptors on keratocytes to modulate the TGF-β2 effect. 112,119,120 Stromal wound repair consists of myofibroblasts migrating into the wound and filling the stromal void by creating an interwoven and interconnected network of cells that simultaneously deposit ECM. Stromal wound closure is complete when the epithelial basement membrane and Descemet’s membrane are fully restored and TGF-β2 and PDGF access to the stroma is blocked. With the absence of TGF-β2, the remaining myofibroblasts undergo apoptosis and the stroma is repopulated by keratocytes that gradually remove and reorganize the disordered ECM and restore corneal stromal transparency. 73,111,115,116,121 –123
Wound healing in the corneal endothelial layer is accomplished primarily through CEC migration, spreading, and occasionally transient transdifferentiation; CEC proliferation plays a secondary role. 115 TGF-β2 promotes CEC migration and modulates CEC proliferation. 115 Expression of TSP-1 is necessary for CEC migration and there is upregulated expression of TSP-1 in CECs at the leading edge until the defect is covered. 108,124 The fibronectin receptor α5β1 is upregulated in wounded corneal endothelium, 125 suggesting that fibronectin interactions also mediate cell migration. When the corneal endothelial layer is intact, TSP-1 returns to a more diffuse expression pattern within the CECs. 88 Binucleated or multinucleated endothelial cells occasionally are formed in the wound healing process. These are observed most often in rats and rabbits but can be observed in cats, NHPs, and humans when there is significant wounding. 126 It has been hypothesized that the formation of multinucleated cells in the normally amitotic corneal endothelial layer represents a compensatory response in the absence of mitosis. 126 –128
Complications arising from IC injections
Hyphema
When the injection point of entry is posterior to the corneoscleral junction, the needle may pass through blood vessels in the limbal sclera and result in minor hemorrhage into the anterior chamber. Limbal hemorrhage is usually self-limiting due to IOP and the small number of intact erythrocytes introduced into the anterior chamber are readily removed by aqueous outflow. Significant complications may arise when large amounts of erythrocytes or fibrin clots are introduced into the anterior chamber through vascular injury in the iris stroma from needle-related tears or implant-induced blunt trauma. Large numbers of erythrocytes can partially occlude the TM at the level of the JCT, impede aqueous flow, and result in elevated IOP. With more severe vascular injury, there is introduction of fibrin into the anterior chamber that may lead to the development of fibrovascular adhesions between posterior iris and the anterior lens surface (posterior synechiae), fibrovascular adhesions of the peripheral iris to the structures in the ICA (peripheral anterior synechiae), TM fibrosis, and removal of corneal endothelium through erythrocyte-fibrin clot contraction. Synechiae and TM fibrosis can result in obstruction of the ICA and lead to glaucoma. If uncontrolled or unresolved, glaucoma leads to irreparable injury to the retina and optic nerve in the posterior segment. 129 –133
The AC mechanisms for the prevention and resolution of fibrin clot formation and maintenance of aqueous flow include: Tissue plasminogen activator: Tissue plasminogen activator catalyzes the conversion of plasminogen to plasmin, which is the major enzyme responsible for fibrin solubilization.
134
Tissue plasminogen activator is present in AH
135
and is expressed in a constitutive and upregulated manner by CECs, TMCs, and uveal microvasculature.
114,136
–138
Trabecular meshwork cell phagocytosis: TMCs utilize integrin receptors to capture fibrin (mediated by RGD binding)
139,140
and erythrocytes (mediated by CD47 binding).
84,141
–143
TMCs maintain aqueous flow through the TM via removal of fibrin blood clots and remodeling if TM fibrosis develops.
Anterior subcapsular fibrous plaque
Anterior lens capsule punctures may occur if the injection needle is inserted too deep. With wounding of the lens capsule, latent TGF-β2 from AH and secreted by anterior lens epithelial cells (LEC) binds to exposed TβR-IIs and αvβ3 expressed on LECs and stroma and is activated by TSP-1 from the AH and LECs. Active TGF-β2 promotes LEC migration from the wound edge into the defect, LEC proliferation to form an epithelial plaque, and epithelial to mesenchymal transdifferentiation (EMT) of the superficial and midlayer LECs to form a fibrous plaque consisting of collagens and other adhesive proteins. 88,90,144 –148 The LECs in the deepest layer of the epithelial plaque do not undergo EMT. They ultimately line the posterior edge of the fibrous plaque and secrete basement membrane to form the new lens capsule. The entrapment of the fibrous layer between the original lens capsule and the newly formed lens capsule results in an anterior subcapsular defect that has the appearance of a focal lens opacity when observed clinically. Often, secondary globular (Morgagnian) degeneration of lens fibers develops posterior to the subcapsular fibrous-plaque. 149
Complications arising from injection of an IC DDS implant
When an implant is injected into the AC periphery, it is swept by aqueous flow into the limbal region of the ICA. Typically, the implant stays within the ICA due to AH outflow pressure; however, the implant is initially mobile and may be temporarily displaced from the ICA through head movement and come into contact with the corneal endothelial layer. If the implant contact is brief (“touch” contact), there is focal CEC loss and exposure of Descemet’s membrane, which results in localized corneal stromal edema at the site of injury. The adjacent surviving CECs upregulate TSP-1 expression along the leading edge as they migrate from the periphery to cover the exposed Descemet’s membrane. Latent TGF-β2 in Descemet’s membrane and from AH is activated through binding with TSP-1 and modulates CEC migration and proliferation and promotes basement membrane secretion. Endothelial changes following touch contact are subtle and may not be observed by light microscope. However, if the endothelial cell response is limited to spreading without endothelial cell proliferation, increased individual cell surface area and decreased endothelial cell density are detectable via specular microscopy. 150
Prolonged implant contact with the corneal endothelial surface occurs if the implant is too large to fit within the ICA. This results in chronic mechanical injury with continual removal of the CECs and Descemet’s membrane until the implant is removed. With the ongoing trauma, repair of Descemet’s membrane and subsequent wound closure are delayed. This leads to chronic exposure of the corneal stroma to TSP-1 and TGF-β2, the transdifferentiation of large numbers of keratocytes to stromal myofibroblasts, and the production of abundant disordered ECM. Immune and angiogenic privileges are lost under these conditions, leading to corneal neovascularization, fibrosis, and corneal opacification. 42,89,90,106,108,111,112,115,116,118,121,122,124,151 –157
Immunopathologic changes associated With IC implants
Biodegradable copolymer (polylactic acid and a polyglycolic acid) or a polylactic co-glycolic acid implants have been used in both the AC and the vitreous. In the AC, proteins from the AH adsorb to the implant surface immediately upon entry. 158 –161 Aqueous humor proteins detected on polymer surfaces include the TSPs, TGF-βs, fibrinogen, and fibronectin. 40,88,161 –163
Protein adsorption is influenced by physiochemical characteristics of the proteins and the biomaterial surface, and the biological surroundings including pH, salinity, and temperature. 164 –167
Protein adsorption to the implant surface is followed by the migration and adherence of phagocytic cells, presumably from the TM and iris, 22,168 to the implant surface. Phagocyte attachment to the implants is likely mediated through the binding of MAC-1 (αMβ2) and other integrin receptors to adhesive proteins adsorbed to the implant surface. 158,161,163,169 Gonioscopically, the initially mobile implant gradually becomes fixed to the inner limbal surface in the inferior aspect of the AC. Microscopically, the phagocytes covering the cast appear to be attached to the SLCs lining the internal limbus between the termination of Descemet’s membrane and the pectinate ligaments. Implant phagocytes can also appear attached to the anterior limiting layer of the iris. It is possible that TSP-1 and TGF-β2 in the AH and the iris stroma are involved in the formation of cellular adhesions that lead to implant immobilization. For example, TGF-β2 has been shown to promote SLC transdifferentiation and β-catenin upregulation, which enhances cadherin-mediated cell–cell adhesion. 170 These cell–cell adhesions could also form between activated SLCs and implant phagocytes. A similar TGF-β2-mediated mechanism may also promote cell-to-cell adhesion between implant phagocytes and iris fibrocytes and/or iris resident dendritic cells, or these interactions may be facilitated by TSP-1 through its interactions with various other ECM proteins and receptors. 42,171
Over time (3-6 months post implant immobilization) hypertrophy, hyperplasia and spindle cell transdifferentiation of SLCs in contact with the implant occurs, suggestive of EMT, which has been reported in SCs and TMCs in vitro. 172,173 Schwalbe’s line cells have been identified as adult stem cells for TMCs, and implant-related changes in the SLCs are similar to those described in activated TMCs 81,143,174 –176 The transdifferentiated SLCs form layers 2 to 3 cells thick over a basement membrane thickened due to accumulation of a fibrillar matrix.
Transdifferentiated SLCs cover or encapsulate the implant over time, presumably through a combination of cell-to-cell adhesions with the adherent phagocytes and migration over newly formed basement membrane. Ultimately, 1 to 3 layers of spindle cells cover the implant and are continuous with the SLC layer. Nonpigmented fibroblasts and pigmented stellate cells from the anterior iris surface can encapsulate the implant in a similar fashion. Very rarely, SLCs and iris mesenchymal cells attach to the implant simultaneously, creating a transient bridging structure between the iris anterior limiting membrane and the limbal SLC layer that has the appearance of a synechia by gonioscopy. However, when examined microscopically, the bridging structure lacks a fibrovascular component that is a consistent feature of synechiae 177 and are termed instead as implant-associated adhesions (IAA). The absence of vascular tissue involvement in IAA formation may be due to TSP-1- and TGF-β2-mediated angiogenic inhibition. The antiangiogenic effect of TSP-1 is mediated directly via CD36 and CD47 receptors expressed on vascular endothelial cells, regulating their migration, proliferation, and apoptosis and subsequently inhibiting growth of new blood vessels. 75,178 –180 TSP-1 also binds with latent TGF-β2, CD36, and CD47 on peripheral (limbal or scleral) vascular endothelium (directly) and dendritic cells/tissue macrophages (indirectly) to inhibit of angiogenesis and restrict of leukocyte migration. 42
Once the implant material completely degrades, as evidenced in-life by gonioscopic examination and microscopically by the absence of the cellular cast, the inner limbal surface gradually remodels to a smooth surface of a monolayer of SLCs. Localized minimally thickened and irregular basement membrane is the final vestige of a previous implant adhesion site at 18 months postimplant injection. 103
It should be noted that the typical foreign body response to biomaterials as described and reviewed by Anderson in 2001 and 2008, respectively, 158,160 is not seen with IC implants presumably due to the immunoregulatory environment of the AC. 143,181,182 Moreover, any capture of implant-derived antigens by resident APCs within the internal limbus and TM will result in induction of the antigen-specific systemic immunoregulatory response (ACAID) that prevents an inflammatory reaction against the implant.
Posterior Segment
The posterior segment (Figure 1) is composed of the vitreous body (occupying the posterior compartment), the pars plana, retina, optic nerve head, the portion of the optic nerve that passes through the lamina cribosa of the posterior sclera, the choroid, and surrounding sclera.
The vitreous is currently the most common site of administration for intraocular retinal therapeutics. Insertion site and technique varies slightly with species. 183 For NHPs, the needle should be inserted into the sclera at a 15° to 30° angle, 2 to 3 mm posterior to the temporal limbus (either inferior or superior), and then angled 45° to 60°, extending needle insertion 4 to 6 mm into the vitreous 183 (Figure 5). Understanding the physical nature of the vitreous and its role in maintaining ocular health is important to successful development of therapies intended for this space. An important physical role of the vitreous is to maintain the interior shape of the eye by ensuring correct placement of the ocular tissues that it contacts: the lens, retina, and optic nerve head. This is aided by the surrounding external sclera, a fibrous tunic with limited expansibility. Together with slow fluid dynamics, the eye is relatively fixed in shape and volume. These properties collectively protect the interior ocular tissues and maintain the shape of the eye and IOP. These physical characteristics also contribute to the challenges of intraocular drug development including volume of delivery, successful delivery of therapeutic doses to the intended site, and any resulting pathology.
Vitreous Composition and Structure
The vitreous is an extracellular viscoelastic gel-like matrix that is ≥95% water by weight 184 ; it generally does not survive typical methods of tissue processing, limiting postmortem assessment in otherwise healthy eyes. However, the vitreous is a complex tissue consisting of collagen fibrils arranged in a lattice-like scaffold made 3-dimensional through the attraction of water molecules by hyaluronan, a hydrophilic glycoprotein interspersed between the collagen fibrils. Additional molecules of fibronectin, fibrillin, and opticin are spatially arranged within the vitreous in a manner that permits transparency. 184,185 Differential distribution of collagen with the other molecules results in zonal differences between the medullary core and the cortex, particularly in human eyes, where the medulla contains less collagen fibrils and exhibits a higher state of liquidity. As a consequence, in human eyes the medulla becomes progressively liquified with age, developing pockets of fluid; this can result in collapse of the cortical vitreous, placing traction on the adjacent retina, and can manifest in spontaneous disease such as retinal tears and/or vitreous detachment (see below). Aging changes such as these are generally not observed in animals used in preclinical studies. Structural differences also vary across species with respect to percentage differences in both hyaluronan and collagen. Compared to humans, most preclinical species have a predominantly gel-like vitreous with a higher concentration of collagen overall relative to hyaluronan. 186 Drug delivery may result in structural alterations to the vitreous that may or may not be clinically relevant.
Vitreous Cortex
The cortex is relatively more condensed and fibrillar than the medulla, with the highest density of collagen located within the anterior base of the vitreous, the region spanning the inner space of the PS immediately posterior to the lens that is circumferentially rimmed by the ora serrata. Fibers of the vitreous base are arranged perpendicular to the surface of the retina and form tight adhesions through interdigitations with cells of the pars plana and glial cells of the peripheral retina. 187 Fibers in the posterior vitreous course parallel to the retina and form tight adhesions with the internal limiting membrane (eg, basement membrane of Müller cells) and glia surrounding the optic disc (and macula of primates). Between these 2 regions, adherence of the vitreous to the retina is currently thought to occur through biomechanical properties of molecular components (chondroitin sulfate, glycosaminoglycans, fibronectin, opticin, and laminin) of the ECM residing in the vitreoretinal interface. 187 Because of these adhesions (both physical and biochemical), disturbances in the vitreous–retinal space place tension on the adhering cells, causing mechanical stress that can lead to pathology in this region (described below). Nonetheless, the interface between the vitreous and the retina represents a “path of least resistance” for delivery of drugs and devices and thus is often where pathological changes are observed.
Hyalocytes, cells of monocyte/macrophage lineage, are an important constituent of the vitreous cortex. Hyalocytes reside near the retina in the peripheral margins of the vitreous, generally in the vicinity of the optic nerve head, in Cloquet’s canal, or anteriorly in the vitreous base (Figure 7). Hyalocytes express CD45 (leukocyte common antigen), CD11a (leukocyte-function antigen-1), CD64 (Fc receptor I), MHC II antigen, and (most hyalocytes) ED2 antibody, an antigen expressed by tissue macrophages. 188 Hyalocytes are resident phagocytes with roles in modulating ocular immune conditions. However, hyalocytes are responsive to PAMPs, increase in number and activity when stimulated, recruit leukocytes under inflammatory conditions, and secrete ECM and VEGF, contributing to formation of membranes physically and through recruitment of other cell types (see Cellular Transdifferentiation, below) within the vitreous and surface of the retina. 189 –191
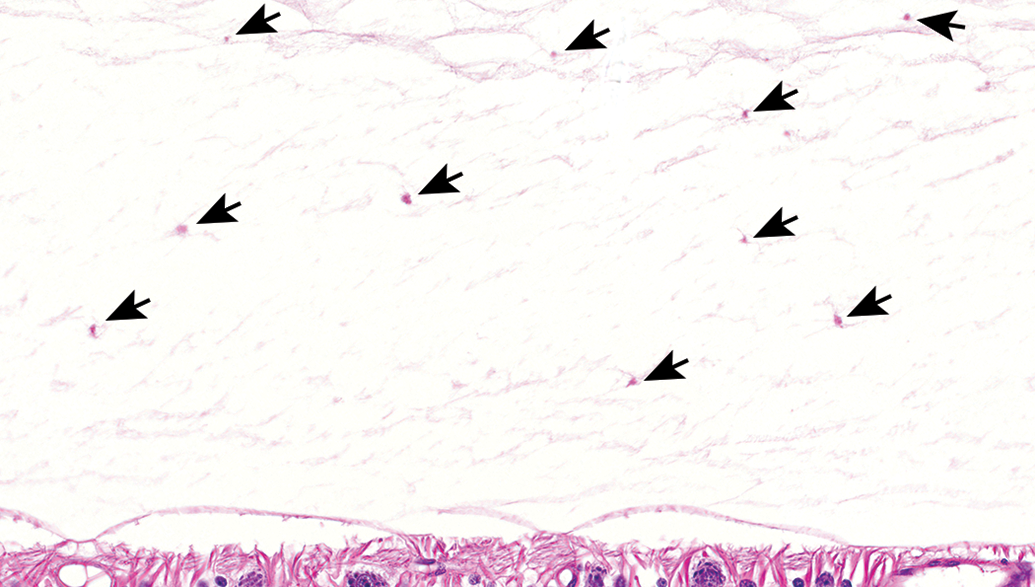
Hyalocytes located near the inner surface of the retina. Arrows point to individual mononuclear cells dispersed within the peripheral margins of the vitreous. Hyalocytes tend to have indistinct cellular features and can be easily overlooked. The numbers of cells in this field indicate that a robust population of hyalocytes are present even in healthy eyes. H&E stained section of a dog eye.
Intravitreal therapeutics
Structural aspects of the vitreous, and the relationship between the vitreous and the retina have important roles in pathology that occurs spontaneously and in preclinical development of ocular therapeutics. Physically, the vitreous residing within the enclosed space of the posterior segment represents a potential barrier to successful drug delivery, and inflammatory responses and subsequent reparative processes commonly occur at the vitreous–retinal interface.
Responses to changes in IOP
The physical dynamics of the eye limits the volume of intravitreous (IVT) administration to 50 µL in large animal species, 192 and to 150 µL in human eyes. 193 Even these low volumes can be associated with transient elevations in IOP. Intraocular pressure coupled with the physical attributes of the vitreous creates a certain amount of passive resistance to IVT administration. Injections are administered through the densest region of the vitreous under positive pressure and thus reflux of fluid through the injection site upon needle withdrawal is not uncommon and likely represents some amount of formulation rather than that of the highly viscous vitreous. Observations of perivascular cuffs of lymphocytes in the sclera at the injection site following IVT administration of a biologic is both suggestive of leak of the administered biologic exterior to the eye and of subsequent immune response. Additionally, inflammatory reactions inside the eye following IVT injection tend to occur at the periphery of the vitreous, near the injection site (Figure 8), pars plana and adjacent peripheral retina reflecting chemokine gradients. More severe inflammation may extend posteriorly to the optic nerve head and into the vitreous proper. Similarly, IVT delivery of devices, degradable biomaterials or gel formulations can result in reactions near the site of insertion, where they appear relatively immobilized by reactive, membranous bands of tissue (described further below).
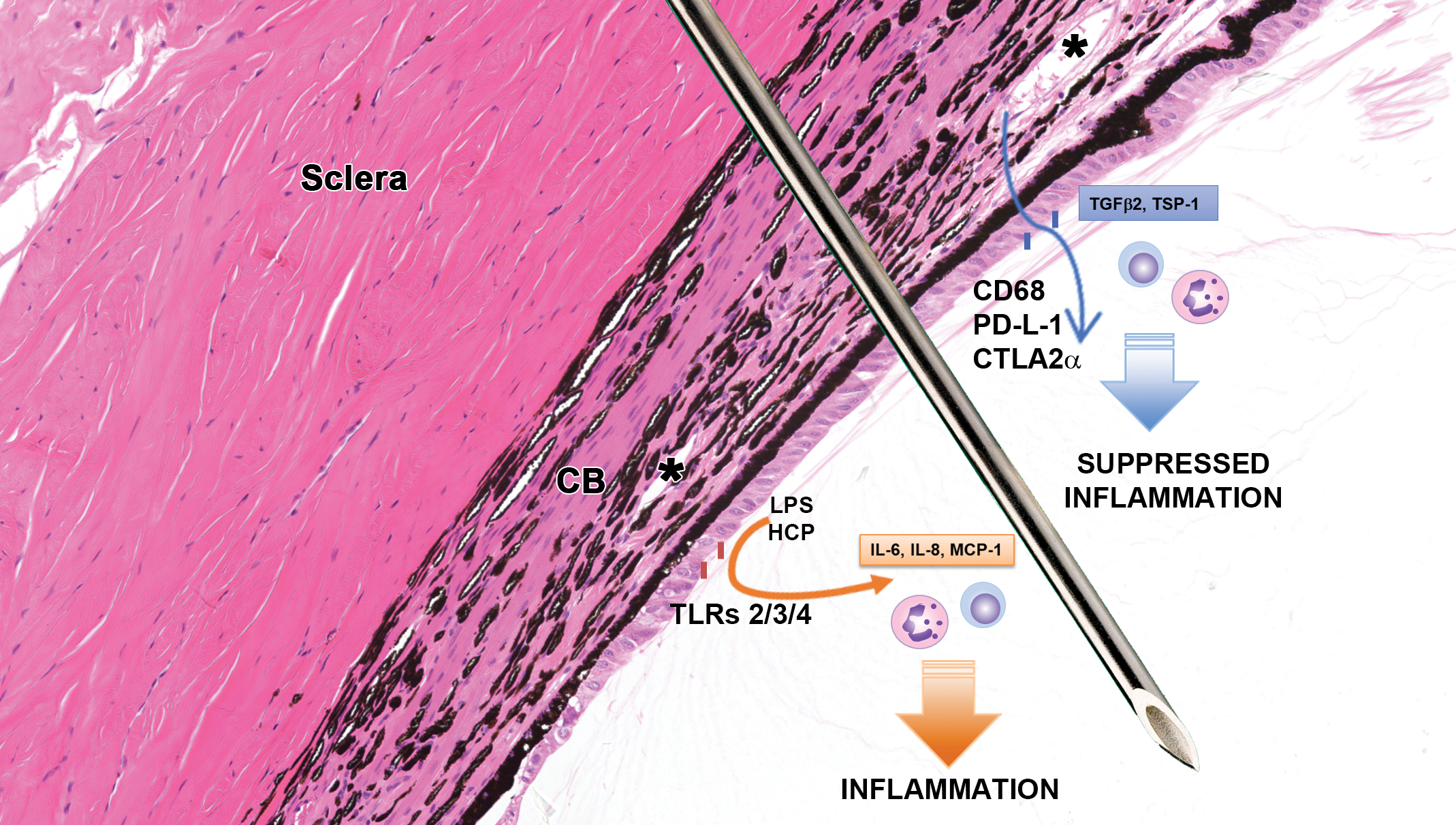
Immunoregulatory mechanisms associated with intravitreal (IVT) injections. After IVT injection through the pars plana, there is a transient breakdown of the ocular barrier (tight junctions between pigmented and nonpigmented epithelia), which may allow limited entry of leukocytes. immune privilege has the ability to suppress development of inflammatory responses through expression of cell surface molecules expression (CD86, PD-L1, CTLA2α) and release of soluble factors (TGFβ2, TSP-1) expressed by epithelial cells of the pars plana, ciliary processes, and iris. In the presence of toll-like receptor (TLR) ligands such as endotoxin and host cell proteins in formulations, epithelial cells may express inflammatory molecules (IL-6, IL-8, MCP-1). Unregulated persistence of these pro-inflammatory molecules may result in intraocular tissue damage. CB indicates ciliary body; IL, interleukin; MCP-1, monocyte chemoattractant protein-1; TSP-1, thrombospondin-1; “*” denote blood vessels. H&E stained section of a dog eye.
Given the limited exterior expansibility and interior volume, the comparatively delicate retina is susceptible to injury from sustained increase pressure. In order to avoid pressure increases, extraction of vitreous fluid of comparable volume prior to intraocular injection has been advocated to enable larger volumes to be administered but is not easily accomplished given the viscosity of the vitreous even with low gauge needles. Additionally, vitreous extraction can be associated with trauma at the insertion site with possible fibrotic reactions, a reparative response of the sclera, extending into the vitreous, or occurring de novo within the vitreous, thus limiting this method as standard practice.
Innate immune response to ocular drug delivery
Knowledge of the immune pathways that occur in the eye in response to bacterial infection provides a framework for understanding the subsequent pathology that occurs following IVT administration of a biologic. Biological drugs manufactured in bacterial or yeast cultures can contain contaminants of the host cells, including endotoxin, wall components, and nucleic acids that can activate ocular TLRs and incite acute inflammatory responses similar to those directly caused by the pathogens. Regulatory limits to endotoxin for solutions intended for the posterior compartment of the eye are currently not specified although the FDA recommends ≤0.2 EU/mL for medical devices intended for intraocular implantation in humans. 194 However, inflammation in the rabbit eye has been elicited by 0.02 EU endotoxin in an ocular viscosurgical device, 195 and a study conducted by Bantseev et al 196 established a no-observed-effect level of IVT administration of endotoxin to rabbits at 0.01 EU/eye, suggesting ocular sensitivity may exceed current device recommendations. Immune responses to formulation excipients, including endotoxin and host cell proteins, are subject to both species differences and individual variation, with some animals apparently more sensitive and exhibiting inflammation of increased severity. Rabbits may be particularly sensitive to endotoxin. Because any inflammation in the eye can be confounding, care should be taken to minimize formulation components known to be pyrogenic in preclinical studies.
In the intact eye, leukocytes enter the posterior compartment through the uveal tissues at the ciliary body and processes, or through vessels located in and around the optic nerve head following chemokine gradients. On histological evaluation, a trail of leukocytes can sometimes be observed extending from the head of the optic nerve towards the lens, corresponding to a remnant of Cloquet’s canal. Acute inflammatory infiltrates following IVT drug administration are commonly observed near the injection site and at the adjacent, peripheral margins of the vitreous (primarily posterior to the ciliary processes, at the retinal surface, and behind the lens; see Figure 3, for IVT injection site). Collection and microscopic examination of injections sites is critical to safety assessment as findings may be missed through examination of standard sections alone (see considerations for ocular tissue assessment in this issue [Short B. Selected aspects of ocular toxicity studies with a focus on high-quality pathology reports: a pathology/toxicology consultant’s perspective. Toxicol Pathol. 2021.]). Leukocytes are recruited and migrate along chemokine gradients, and thus cellular infiltrates are anatomic indicators of activated cells elaborating inflammatory mediators; although speculative, it may be of value to consider that the location of inflammation is an indirect reflection of drug distribution within the eye. Activated ocular cells include the hyalocytes, but retinal cells that express TLRs can also contribute to the inflammatory milieu when activated, secreting chemokines that attract leukocytes (Table 2; Factors associated with ocular inflammation). Activated neutrophils can elaborate reactive oxygen species and their proximity to the delicate retinal tissues can cause injury that further amplify the inflammatory response through the release of DAMPs. The physical attributes and slow fluid dynamics of the vitreous may have roles in restricting acute inflammatory responses to the peripheral margins of the vitreous. This may be particularly apparent in laboratory species that have a predominantly gel vitreous (eg, rodent, rabbit, dog). However, exudative effusions tend to be associated with more extensive inflammation of the vitreous body. Such inflammatory responses can resolve with changes to the vitreous matrix, altering the molecular components and resulting in opacities that impair vision.
At times, an acute influx of nonreactive leukocytes into the vitreous–retina interface may occur without apparent impact to the retina or visual function. Possibly these represent regulatory lymphocytes and regulatory neutrophils, a recently described subtype of neutrophils shown to have immunomodulating characteristics in several disease models (Perobelli et al for review. 197 ) Upon crossing the BOB, neutrophils may be subject to the same epithelial and cytokine modulations that induce regulatory T and B cells under conditions that invoke vitreous cavity-associated immune deviation (VCAID). Resolution, however, may be prolonged due to slow turnover of vitreous fluid resulting in the persistence of leukocytes within the vitreous–retinal interface. While innocuous appearing, the presence of any leukocytes within the eye tend to have a negative interpretation, and the ability to differentiate regulatory from pro-inflammatory leukocytes could be impactful in ocular drug development.
Adaptive immune reponses to ocular drug delivery
Conditions of VCAID do not preclude an adaptive immune response to antigens that cross BOBs, although the generation of effector CD4-T cells that traffic back to the eye, compliment-fixing antibodies, and DTH are inhibited. 57,198 –201 Lymphocyte infiltrates and/or antidrug antibodies (ADA) can be observed in the posterior segment following the administration of a biologic in preclinical species for which histological evidence for tissue injury may be lacking (personal observation). Such occurrences may be suggestive of VCAID induction, but supportive literature is lacking.
However, we often do see associated ocular tissue injury, and the conditions that promote inflammation are not clearly understood. Significant informational gaps exist in understanding if inflammation observed following delivery of a biologic antigen is caused primarily by innate responses with loss of IP, or by adaptive responses generating complement-fixing antibodies, or DTH associated with autoimmunity to retinal antigenic targets now vulnerable to immune surveillance. Innate inflammation enables adaptive responses to occur that can amplify inflammation and further tissue injury, and this may occur in the eye when IP is lost.
Chemokine gradients created by the innate immune response are antigen nonspecific but result in the migration and activation of macrophages that play a key role in initiating adaptive immune responses through antigen presentation. Recruited macrophages secrete several cytokines including proteases that increase vascular permeability and amplify inflammation through additional leukocyte recruitment. Cytokines in turn activate hyalocytes, retinal microglia, and RPE. While they do not normally express MHCII, they can be stimulated to do so under disease or inflammatory conditions and assume characteristics of APCs, shaping the adaptive response. 57,202,203 Toll-like receptors on activated ocular cells may elaborate additional inflammatory mediators such as IL-6, IL-8, IFN-β, monocyte chemoattractant protein-1 (MCP-1), and intracellular adhesion molecule 58 (Table 2; Factors associated with ocular inflammation). Interleukin-6 has roles in inciting acute-phase responses, TH17 cell differentiation, and CD4-T cell stimulation resulting in antibody production. 59 Activated retinal microglial have also been shown to produce high levels of TNF-α, MCP-1, regulated upon activation-normal T cell expressed and secreted nitric oxide synthase 2, and matrix metalloproteinases in disease models of age-related macular degeneration (AMD), glaucoma, retinitis pigmentosa, and diabetic retinopathy. 60 The unintentional activation of ocular TLRs following drug delivery may have a similar pathogenesis.
Perivascular cuffs of lymphocytes are often observed in the retina and/or optic nerve in preclinical species following the ocular administration of a biologic intended for humans and are assumed to be an adaptive immune response. The optic nerve can serve as a route of clearance for drugs injected into the vitreous, 204 and thus inflammatory cells may migrate to this area. In rabbits, which have a merangiotic vascular supply, inflammation may be largely confined to vessels around the optic nerve head. Perivascular inflammation may resolve with minimal impact to the adjacent tissues or to vision. However, perivascular inflammation that is more extensive and includes activated neutrophils can result in necrosis of the adjacent retina (personal observation). Evidence of immune-complex disease (vascular fibrinoid necrosis, plasma cells, antigen–antibody/compliment complexes) can be helpful in study interpretation and risk assessment. However, the role for immune-complex disease is speculative in spontaneous ocular diseases 57 and should not be our default interpretation for vascular injury following administration of a biologic without supporting evidence. Careful consideration should be given to downstream effects of on-target pharmacology as part of our safety assessment. For example, biologics that target vascular disease (eg, anti-VEGF, anti-PDGF) could potentially also target normal vessels; vascular occlusion, ischemic necrosis, hemorrhage, and subsequent inflammation of adjacent tissues could be difficult to differentiate from an exuberant ADA immune-mediated inflammatory response. In-vivo fundic imaging, fluorescent angiography, and OCT over the course of a study can significantly aid in monitoring potential vascular changes, subsequent retinal injury, and inform study interpretation.
Cellular transdifferentiation
Both retinal glial cells and hyalocytes are capable of myofibroblastic transdifferentiation, and formation of contractile cellular membranes. Although abundantly available cytokine TGF-β isoforms are known to maintain an anti-inflammatory environment within the eye at homeostasis, it is also known to play a significant role in fibrotic processes observed in resolution of wound healing. These processes are recapitulated in inflamed eyes. Soluble TGF-β2 together with other growth factors (Table 3; Factors associated with cellular transdifferentiation) promote this process that entails the cellular transdifferentiation, proliferation, and migration of epithelial barrier cells and other endogenous non-neural ocular cells such that they assume a membranous, and/or fibroblastic-like characteristic that can be reminiscent of fibroplasia. 188,206 –208
Select Factors Associated With Cellular Transdifferentiation.

Abbreviations: bFGF, basic fibroblastic growth factor; CTGF, connective tissue growth factor; ECM, extracellular matrix; EGF, epidermal growth factor; GDNF, glial cell-line derived neurotrophic factor; IGF-1, insulin-like growth factor 1; NGF, nerve growth factor; PDGF, platelet-derived growth factor; TGFβ, transforming growth factor β.
Given its pleiotropic nature, TGF-β2 activation is a tightly regulated process. TGF-β protein is synthesized as an inactive latent complex that must be biologically activated. In the eye, TSP-1 plays a key role in the activation of membrane-bound TGF-β2 on ocular APCs and parenchymal cells that aids the conversion of activated lymphocytes to regulatory T-cells both systemically and within the ocular environment. 57,199,201 Several studies report that physiological activation of latent TGF-β requires combined activities of proteases and integrins, which in the eye can be altered under inflammatory conditions. 209 In vivo, integrins are known to activate latent TGF-β1 and TGF-β3 but not TGF-β2. 210 Müller glia can also express TGF-β3 211 and TGF-β3 derived from Müller cells could be activated by integrins on retinal glial cells that in a progressive loop amplify the fibrotic process. In addition, hyalocytes express TGF-β1, TGF-β2, and TGF-β3 under inflammatory conditions. 211 TGF-β1 reportedly enhances transdifferentiation of the monocyte/macrophage lineage by increasing smooth muscle cell-specific marker expression 212 and thus it’s likely that hyalocytes activated by TGF-β1 have a role in membrane formation at the retinal surface and within the vitreous.
Immunohistochemistry studies conducted on surgical or experimentally derived ocular membranes show that the cell populations are derived from endogenous ocular cells, demonstrating their plasticity under promoting conditions. Cellular constituents can variably include glial cells, hyalocytes, RPE, fibroblasts, and myofibroblasts. 213
Membranes in spontaneous disease and ocular drug development
Spontaneous ocular diseases resulting in membranous tissue include subretinal membranes (eg, AMD), retinal surface membranes (epiretinal membranes), and vitreous membranes. Membrane development consists of linear proliferations of stromal cells that may be accompanied by varying degrees of ECM components; remodeling of ocular tissues can be significant and impair vision. Similar findings may also occur following intraocular delivery of biologics or devices and likely are derived from similar pathogeneses, specifically, perturbations of the vitreous–retinal, or retinal–RPE interfaces.
Subretinal membranes
Neovascularization in the subretinal space is a hallmark feature of AMD. Exudate and inflammatory mediators result in RPE detachment and dissociation, and growth factors promote RPE proliferation and transdifferentiation. 206 Retinal pigment epithelial detachment and dissociation are crucial steps in subretinal membrane formation that also can occur with drug or device delivery into this space. Animal models and immunohistochemistry indicate that membranes are primarily sourced from RPE although other cells recruited include cell populations found in spontaneous disease. 206
Retinal membranes
Retinal membranes develop in response to retinal surface perturbations and as such are often seen spontaneously in humans with age-related changes to the vitreous, such as vitreous collapse following liquefaction or other causes of vitreous separation from the retina. Retinal membranes are also thought to occur as a form of protection for the retinal surface and thus may be observed following the administration of a device that comes to reside at the ventral retinal surface (as may occur over time with gravity). Disruptions to adhesions between the vitreous and retina result in tractional forces that are transferred to Müller cells, resulting in their hypertrophy and proliferation. 61 Proliferation is induced through stretch-activated calcium channels resulting in calcium influx, activation of potassium channels, and the release of growth factors such as basic fibroblastic growth factor (bFGF) and ATP. 214 In spontaneous disease, subsequent breaks in the retina allow for contributions by migrating RPE. 208 Retinal membranes can contain variable amounts of ECM and can be contractile, resulting in retinal buckling. Retinal membrane formation in patients with proliferative vitreoretinopathy is associated with intraocular inflammation. 208 Inflammatory events that activate TLRs located on retinal cells result in release of growth factors may additionally promote membrane formation. Expression of growth factors found in retinal membranes include TGF-β1, bFGF, nerve growth factor, and glial cell-line derived neurotrophic factor 213,215 suggesting that they are locally derived.
Retinal membranes observed in preclinical studies can vary in extent of ECM and retinal impact. Devices that contact the retina may occasionally result in membranes composed primarily of Müller cells, as demonstrated through predominant glial fibrillary acidic protein expression (Booler H, Larsen T, Shelton A, et al. Foreign body reaction, retinal degeneration and epiretinal membranes associated with intravitreal administration of PLGA rods. Toxicol Pathol. 2021.). However, these can be finely fibrillar and lacking in ECM; as focal findings they may be minimal and inconsequential. Adversity determination should consider both impact to the underlying or adjacent retina and to visual function. Differences in outcome between patients with ocular disease and those observed in preclinical studies may in part be attributable to inherent differences in vitreous structure, and/or the health of the vitreous.
Vitreous membranes
Vitreous membranes are composed of fusiform cells and variable amount of ECM oriented in linear arrays at the peripheral margins of, or occasionally within, the vitreous. Vitreous membranes may develop following posterior vitreous detachment, retinal tears, injury, or penetrating foreign bodies. Cell origins potentially include hyalocytes, glial cells, local fibroblasts, and RPE, which migrate through breaks in the retina. 188,208 Hyalocytes may enable the process by secreting components of ECM (fibronectin, collagens, and glycosaminoglycans) under the influence of TGF-β, PDGF, and CTGF 188,208,216 providing a substrate for membranes to develop. Vitreous membranes may also incorporate collagen components of the vitreous which act as a scaffold. Vitreous membranes that develop following ocular trauma or foreign body penetration likely have roles in confining and localizing the injury, preserving adjacent tissues. Vitreous membranes that develop in the vicinity of an injection site, typically located at the pars plana, ciliary processes, or the peripheral margins of the anterior face of the vitreous resemble those that occur in ocular injury. Fibroblasts derived from fibrous plugs that form within the sclera at injection sites may extend into the posterior compartment and comingle with cells of the zonule fibers suspending the lens, with both populations contributing to the membranous process. Similarly, devices or matrices intended for sustained delivery to the back of the eye may become immobilized by surrounding membrane formation at the injection site or within the vitreous–retinal interface of the peripheral retina, due to displacement by the vitreous. In inflamed eyes, vitreous membranes are facilitated by the formation of a “provisional” matrix, an exudate of serum proteins that leak into the eye, covering the device or depot, and creating a substrate for recruitment of additional leukocytes and compliment activation. 217,218 This matrix provides a substrate for vitreous membrane formation that ultimately isolate the device within the ocular space and resembles that which occurs following penetration by a foreign body.
Although inflammatory cytokines appear to have an essential role in retinal and vitreous membrane formation, the presence of leukocytes is not imperative for membranes to form in the eye. 208 Membranes can occur with comparatively minimal inflammatory cell infiltrates present. Membranes may result from mechanical stress and the unintended activation of ocular cell TLRs, causing local shifts in cytokines with roles in membrane formation (TGF-β, PDGF, bFGF, and insulin-like growth factor-1). 61,188,205,208,219 Mechanical stress, in combination with active TGF-β1, has been shown to promote the transdifferentiation of fibroblasts into myofibroblasts. 220,221 Formulations, gels, or devices that come to reside in the vitreous–retinal interface may cause retinal membranes to form despite the lack of inflammation through unintentional mechanical stress to Müller cells. Rabbits, which have a highly viscous gel vitreous, are possibly more susceptible than other species to formation of vitreous or retinal membranes.
Optical clarity is dependent on the precise arrangement of cellular constituents and molecules within the multiple matrices of the eye, which can be disrupted by membrane formation. Membranes that contain abundant ECM are opaque, and their location may impede or scatter light transmission and impair vision. In addition, an important characteristic of the stromal cells within membranes is the tendency to express α-smooth muscle actin and myosin light chain, displaying a myofibroplastic phenotype following transdifferentiation. 188,205,206 As with fibrous tissue reactions elsewhere, when these membranes mature, they tend to contract, exerting tension on adjacent structures. Contraction of retinal or subretinal membranes can result in irregularities to the retina that can manifest as perturbations in vision, and when more significant, result in loss of retinal cells. Inflammatory conditions could potentially exacerbate contraction. 207,208,222 Vitreous membranes tend to tether to, and extend between, fixed ocular tissues such as the retina, optic nerve head, lens, and ciliary body and processes. In rabbits, membranes appear to develop in the region of Cloquet’s canal, extending between the optic nerve head and the lens. Upon contraction, significant damage can occur including retinal detachments, displacement of the optic nerve head or lens, and functional impairment to the drainage angle or accommodation by the ciliary body. Chronic hypotony and ciliary body muscle atrophy have been reported following uveitis and the development of cyclitic membranes. 223 In spontaneous clinical disease, surgical intervention may be required to preserve vision. 224
Conclusion
The physical attributes, physiology, and immunology of the interior space of the eye pose unique challenges to ocular drug development. Significant gaps exist in our understanding of the immunological pathways that maintain IP and those activated after administration of a biologic or device placed in the eye. With regard to IC devices, there are still gaps in our understanding and appreciation of the cells that reside in the ICA, their response to drug delivery devices, and the role that IP plays in the local tolerance or intolerance to such devices. Such knowledge would surely enhance our understanding of the varied pathogenic pathways that lead to glaucoma. Additionally, the pathology that occurs in both spontaneous disease and disease models, that is recapitulated in nonclinical studies, suggest pathogenic pathways that mitigation strategies could potentially target. Although this represents an active area of research, it seems that ocular TLR activation is a potential consequence of intraocular drug delivery, particularly in the posterior segment. Disturbances at the vitreous–retinal interface appear to underlie the pathology in this region, leading to challenges with formulations and devices designed for sustained delivery. Yet devices for sustained delivery of anti-inflammatories to the back of the eye has been used successfully in treatment of retinal disease since 2009 225 suggesting the importance of suppression of endogenous pro-inflammatory pathways in this mode of delivery. Future directions should focus on better understanding of the inherent properties of biologics in effort to promote regulatory immune responses associated with IP and to minimize antigenicity. Co-delivery of immunomodulators may provide a path forward for development of intraocular therapeutics or devices and formulations intended to provide sustained delivery.
Footnotes
Acknowledgment
The authors acknowledge the valuable expertise of Ronnie Nie of Allergan, an AbbVie Company, for her contribution of the scanned image of the dog eye used in producing the images for this manuscript.
Declaration of Conflicting InterestsThe author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Meg Ferrell Ramos is an employee of AbbVie, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
