Abstract
The aim of this study was to compare the cytotoxic effects of a newly synthesized thialo benzene derivative 2,4-dithiophenoxy-1-iodo-4-bromobenzene (C18H12S2IBr) and a well-known antifungal agent, fluconazole, in L929 cells. L929 cells were treated with 250, 500, or 1000 µg/mL of C18H12S2IBr and with the same doses of fluconazole. Cytotoxicity tests including 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), lactate dehydrogenase (LDH) leakage, and protein content were compared. Glucose and lactate concentrations were measured to determine alterations in metabolic activity. Apoptosis was investigated by TUNEL test and results were supported with survivin enzyme-linked immunosorbent assay. Treatment with C18H12S2IBr resulted in a concentration-dependent cytotoxicity as indicated by MTT, LDH leakage assay, and decreased protein concentration. The loss of cell viability and the increased LDH leakage in 500 µg/mL and 1000 µg/mL C18H12S2IBr and fluconazole groups indicated cell membrane damage and necrotic cell death. In all groups, metabolic activities were altered but apoptosis was not induced. We have previously investigated lower doses of C18H12S2IBr; there was no cytotoxicity in L929 cells. In this study, higher doses caused cytotoxicity and alterations in metabolic activity . When we consider the similar results obtained from fluconazole and especially the lowest dose of C18H12S2IBr, this newly synthesized compound may be a good alternative antifungal agent.
Introduction
Recently, resistance to antifungal agents increased significantly worldwide. 1 Hence, new compounds have been developing for the treatment of fungal infections. Major antifungal agent groups were based on the inhibition of ergosterol synthesis or interaction with ergosterol which is a part of the fungi cell membrane. 2 Azole groups belong to major antifungal agents whose mechanisms of action are based on inhibiting fungal ergosterol synthesis, including ketoconazole, fluconazole, and intraconazole. Efficiency of these drugs are proven in overcoming of human antifungal infections.
Fluconazole is a very well-known antifungal agent and has a widespread usage in the clinic for treatment and prevention of Candida infections. 3 It has a common usage in treatment of vaginal candidiasis, esophageal infection, urinary tract infection, and peritonitis. 4 Additionally, fungal infections are also common and prominent in immunosuppressed or immunodeficient patients. 5 However, widespread usage of antifungal agents may be accompanied with resistance against them. 6 For this reason, researchers are in quest of exploring new drugs that are active against organisms. Loğoğlu et al synthesized a novel antifungal agent (2,4-dithiophenoxy-1-iodo-4-bromobenzene [C18H12S2IBr]) which is a thialo benzene derivative and they evaluated the biological activities of this compound. 7 In that study, antimicrobial activities of agents containing different configurations of halogens and thio groups against fungi, gram-negative, and gram-positive bacteria were evaluated and it was determined that the newly synthesized agent had 4 times lower minimum inhibition concentration than fluconazole. But there was no report on the effect of this new compound on mammalian cells.
The effects of potential drugs and their mechanism can be tested quickly in different cell lines under in vitro conditions. 8 The results obtained from these studies contribute to determination of further analyses, dose selection in vivo, and so on.
One of the easiest methods for evaluating the destructive effects of the chemicals are cytotoxicity tests. Several markers exist for screening cytotoxicity of chemicals including plasma membrane integrity characterized by a cytosolic lactate dehydrogenase (LDH) enzyme release and mitochondrial dehydrogenase activity based on the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) reduction of viable cells. 9 Additionally, the protein assay measures the protein content of viable cells which is an indirect measurement of cell viability and evaluates cell toxicity. 10,11 Although it not a specific end point, protein content data contribute to other cytotoxicity assays.
The induced type of cell death after the administration of a chemical or a drug is important in order to elucidate the mechanisms of action of that compound. In order to identify necrotic cell death due to the cell membrane damage, extracellular level of LDH, which is one of cytosolic enzymes, can be investigated. Examining the presence or absence of apoptotic proteins and/or inhibitor of apoptosis protein family (IAP) is another aspect of defining the cell death type. Survivin (baculoviral inhibitor of apoptosis repeat-containing 5, BIRC5) is a member of IAP which regulates apoptosis and cell cycle. Although increased survivin expression has been evaluated in cancer cells, current studies have defined the role of survivin in regulating function in several proliferating normal adult cell types. 12,13
This study was designed to compare a newly synthesized drug, C18H12S2IBr, with the well-known antifungal agent fluconazole in terms of cytotoxicity in the mammalian L929 cell line. Cytotoxicity assays included MTT, LDH release, and protein concentration assays. In order to identify the apoptotic cell death, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay was performed and the findings were supported with survivin enzyme-linked immunosorbent assay (ELISA).
Glucose consumption is a good biomarker both for alterations in cell metabolism and for cell viability. As a result of elevated or decreased glycolysis, lactate production is also affected related to metabolism. Additionally, alterations in glucose consumption and lactate production after 24 hours exposure either to C18H12S2IBr or fluconazole were investigated.
Materials and Methods
Cell Line and Culture Conditions
The L929 cell line (mouse fibroblast) was obtained from HUKUK, Foot and Mouth Disease Institute (Ankara, Turkey). The cells were routinely maintained in Dulbecco’s modified eagle medium (HyClone Laboratories, Inc, Logan, Utah) supplemented with 10% fetal bovine serum (PAA Laboratories, Linz, Austria) at 37°C and 5% CO2 in a humidified incubator.
Chemicals and Treatment
2,4-Dithiophenoxy-1-iodo-4-bromobenzene is one of the thiohalo benzene derivatives synthesized from 1,2,3,5-tetrahalogene benzene. 7 Fluconazole is one of the 1,2,4-triazole-based azole antifungal agents which is used commonly in the clinic. 14 In this study, fluzole tablet (pure fluconazole) 150 mg which was purchased from Biofarma (İstanbul, Turkey) and pure C18H12S2IBr (as powder) were used. Final concentrations for both the compounds (250 µg/mL, 500 µg/mL, and 1000 µg/mL) were determined according to preliminary experiments, and test compounds were dissolved in phosphate-buffered saline (PBS). Cells were cultured for 24 hours and were incubated either with fluconazole or with C18H12S2IBr for 24 hours and then the analyses were performed.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) Assay
Cells were suspended in a complete medium (HyClone Laboratories, Inc), seeded into 96-well microculture plates and incubated for 24 hours. After C18H12S2IBr and fluconazole treatment, MTT (Sigma, St Louis, Missouri) was added. After incubation for 4 hours, isopropanol was added and optical density at 570 nm was recorded with a microplate reader (BIO-TEK µQuant, BIO-TEK Instruments, Inc, USA). Experiments were performed in triplicate at concentrations of 250 µg/mL, 500 µg/mL, and 1000 µg/mL and percentage of surviving cells was defined as the treatment group/control group (control group assumed as 100% survival).
Lactate Dehydrogenase (LDH) Cytotoxicity Assay
Cytotoxicity of antifungal agents was evaluated by quantification of the plasma membrane damage measuring LDH enzyme in cell culture media. Lactate dehydrogenase cytotoxicity was measured using LDH cytotoxicity assay kit (Biovision, USA). Briefly, L929 cells were seeded at a density of 2 × 104/well in 96-well plates for control, fluconazole, and C18H12S2IBr treatment for 24 hours at concentrations of 250 µg/mL, 500 µg/mL, and 1000 µg/mL. At the end of the incubation time, culture media were collected and centrifuged at 600g for 10 minutes. Supernatants (10 µL/well) were transferred into 96-well plates. Lactate dehydrogenase reaction mix (100 µL) was added and incubated for 30 minutes. Absorbance of all groups was measured with microplate spectrophotometer (BIO-TEK µQuant, BIO-TEK Instruments, Inc, USA) at 450 nm with 650 nm reference wavelength. The results were expressed as cytotoxicity percentage.
Determination of Protein Concentration
L929 cells were seeded in 25-cm2 culture flasks for control, fluconazole, and C18H12S2IBr treatments (concentrations of 250 µg/mL, 500 µg/mL, and 1000 µg/mL) for 24 hours. Cells were then washed with ice-cold PBS, scraped off, and solubilized in ice-cold radio-immunoprecipitation assay lysis buffer containing 25 mmol/L Tris-HCl pH 7.6, 150 nmol/L NaCl, 1% NP-40, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate (Pierce Biotechnology 89901, Illinois, USA) with protease inhibitor cocktail (Roche 11873580001, Mannheim, Germany). Then lysates were centrifuged at 13 000 rpm for 30 minutes and supernatants were stored at −80°C. Protein concentration of lysates was measured at 562 nm with microplate spectrophotometer (BIO-TEK µQuant, BIO-TEK Instruments, Inc, USA) using BCA protein analysis kit (Pierce Biotechnology 23227, Illinois, USA).
TUNEL Assay of L929 Cells
L929 cells were seeded at a density of 15 × 104/mL into chamber slides. Cells were then treated with fluconazole or C18H12S2IBr (at concentrations of 250 µg/mL, 500 µg/mL, and 1000 µg/mL) for 24 hours. Cells were washed with PBS, fixed in a 1% paraformaldehyde, and postfixed in precooled ethanol–acetic acid 2:1 for 5 minutes at −20°C. The endogenous peroxide was quenched in 3% hydrogen peroxide in PBS for 5 minutes at room temperature. Apoptotic nuclei of L929 cells were identified by TUNEL test with commercial ApoptagPlus kit (Millipore) according to the manufacturer’s instructions. Cells were counterstained with 1% methyl green. The TUNEL-positive cells were counted randomly in all groups and scored as percentages of total cells by 3 observers.
Total Survivin ELISA Assay
Total survivin protein levels in the cell lysates were quantified using sandwich ELISA kit according to the manufacturer’s guidelines (Cell Signaling Technology, Massachusetts, USA). Briefly, 100 µL of diluted cell lysates were added to survivin antibody-coated wells, and the plate was incubated overnight at 4°C. After washing steps, 100 µL of survivin antibody was added and plate was incubated for 1 hour at 37°C. Wells were washed 4 times, then 100 µL of horseradish peroxidase-linked secondary antibody was added, and the plate was incubated for 30 minutes at 37°C. After repeated wash steps, 100 µL of 3,3′,5,5′-tetramethylbenzidine (TMB) substrate was added to each well and plate was incubated for 10 minutes at 37°C. At the end of the time, reaction was stopped with 100 µL of stop solution. Absorbance was read at 450 nm with microplate spectrophotometer (BIO-TEK µQuant, BIO-TEK Instruments, Inc, USA). Data were expressed as percentage of survivin based on survivin concentration in the control group.
Metabolic Activity Assay
Cells were seeded at 5 × 104 and treated with fluconazole or C18H12S2IBr at concentrations of 250 µg/mL, 500 µg/mL, and 1000 µg/mL for 24 hours. After C18H12S2IBr and fluconazole treatment, culture media were aspirated, centrifuged at 2000 rpm for 5 minutes, and stored at −20°C. Glucose and lactate concentrations were measured using commercially available glucose (Spinreact Glucose Trinder, GOD-POD, Sant Esteve De Bas, Spain) and lactate (Spinreact, LO-POD Sant Esteve De Bas) kits in microplate spectrophotometer (BIO-TEK µQuant, BioTek Instruments, Inc, USA) in triplicate. Both of the analysis were performed according to the manufacturer’s instructions.
Statistical Analysis
A statistical analysis was performed using a STATISTICA program for Windows. Data were expressed as mean ± standard error and statistical significance was assigned at P ≤ 0.05 level. The homogeneity of variance and normal distribution between groups were evaluated by General Linear Model procedure and Kolmogorov-Smirnov nonparametric test. Data were analyzed using one way analysis of variance (ANOVA) with Tukey post hoc test. Factorial ANOVA was used to compare fluconazole and C18H12S2IBr treatment.
Results
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) Assay
According to our preliminary experiments, concentrations of 10, 25, 50, 100, and 150 µg/mL did not cause cytotoxicity in either C18H12S2IBr or fluconazole treatment groups, compared to control groups (data not shown). In order to determine the lowest observed cytotoxic levels, we used 250, 500, and 1000 µg/mL concentrations for both of the compounds. The loss of cell viability was notable in 500 µg/mL and 1000 µg/mL C18H12S2IBr exposure, but at the lower concentration (250 µg/mL), no statistical difference was observed compared to the control group. At high doses of fluconazole, the cytotoxicity seems to be negligible (Table 1).
Comparison of different cytotoxicity assays after C18H12S2IBr and fluconazole treatment in L929 Cells.a
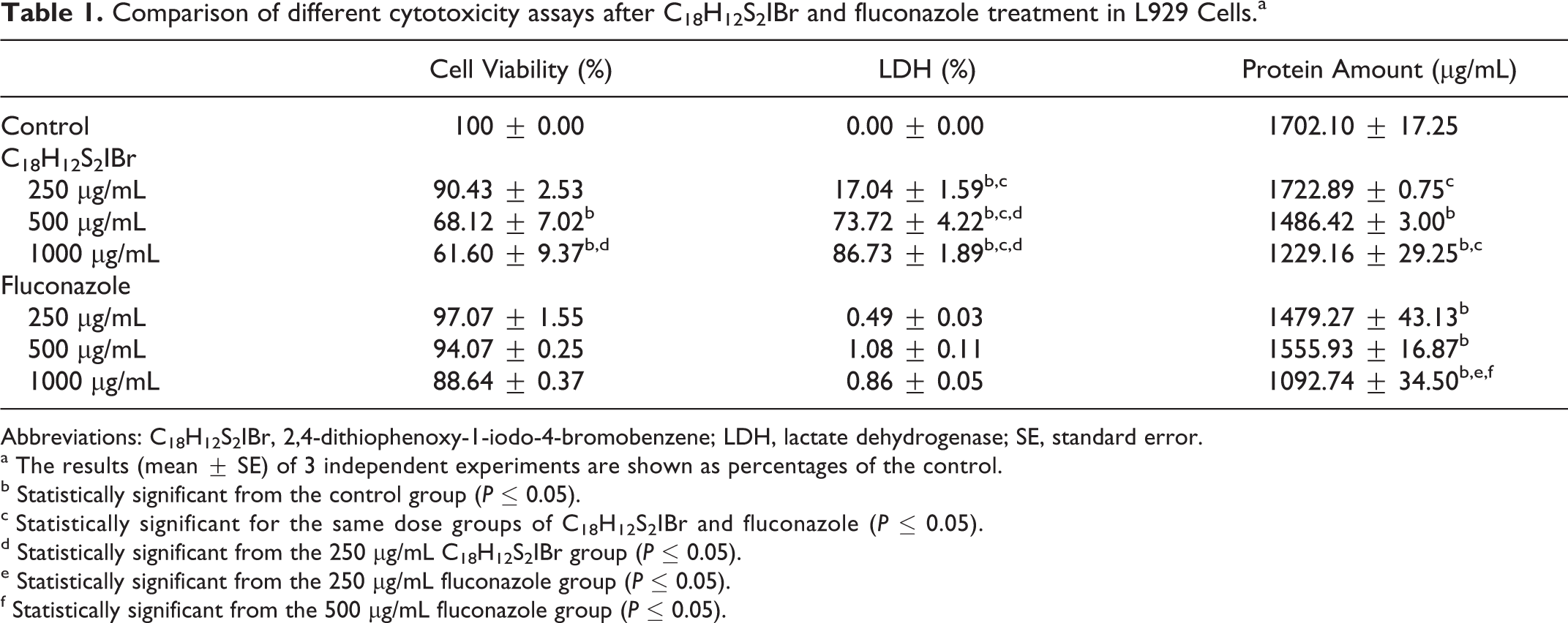
Abbreviations: C18H12S2IBr, 2,4-dithiophenoxy-1-iodo-4-bromobenzene; LDH, lactate dehydrogenase; SE, standard error.
a The results (mean ± SE) of 3 independent experiments are shown as percentages of the control.
b Statistically significant from the control group (P ≤ 0.05).
c Statistically significant for the same dose groups of C18H12S2IBr and fluconazole (P ≤ 0.05).
d Statistically significant from the 250 µg/mL C18H12S2IBr group (P ≤ 0.05).
e Statistically significant from the 250 µg/mL fluconazole group (P ≤ 0.05).
f Statistically significant from the 500 µg/mL fluconazole group (P ≤ 0.05).
Lactate Dehydrogenase (LDH) Cytotoxicity Assay
According to LDH assay, cytotoxicity increased significantly in all dose groups at 24 hours incubation time when compared with the control group. LDH cytotoxicity increased in a dose-dependent manner (Table 1).
Protein Determination Assay
According to the BCA protein assay results, protein levels in 500 µg/mL and 1000 µg/mL decreased significantly in C18H12S2IBr and fluconazole groups when compared to the control group (Table 1). Protein levels in all fluconazole dose groups decreased significantly when compared to the control group. Additionally, protein levels decreased statistically in 250 µg/mL and 1000 µg/mL fluconazole groups when compared with the same doses of C18H12S2IBr groups (Table 1).
TUNEL Assay
TUNEL assay was performed in order to determine apoptosis in L929 cells treated with C18H12S2IBr and fluconazole. The TUNEL-positive cells were counted randomly in all groups, and no significant differences were observed between the groups. According to the results, treatment with C18H12S2IBr and fluconazole did not induce apoptosis in L929 cells (Figure 1).
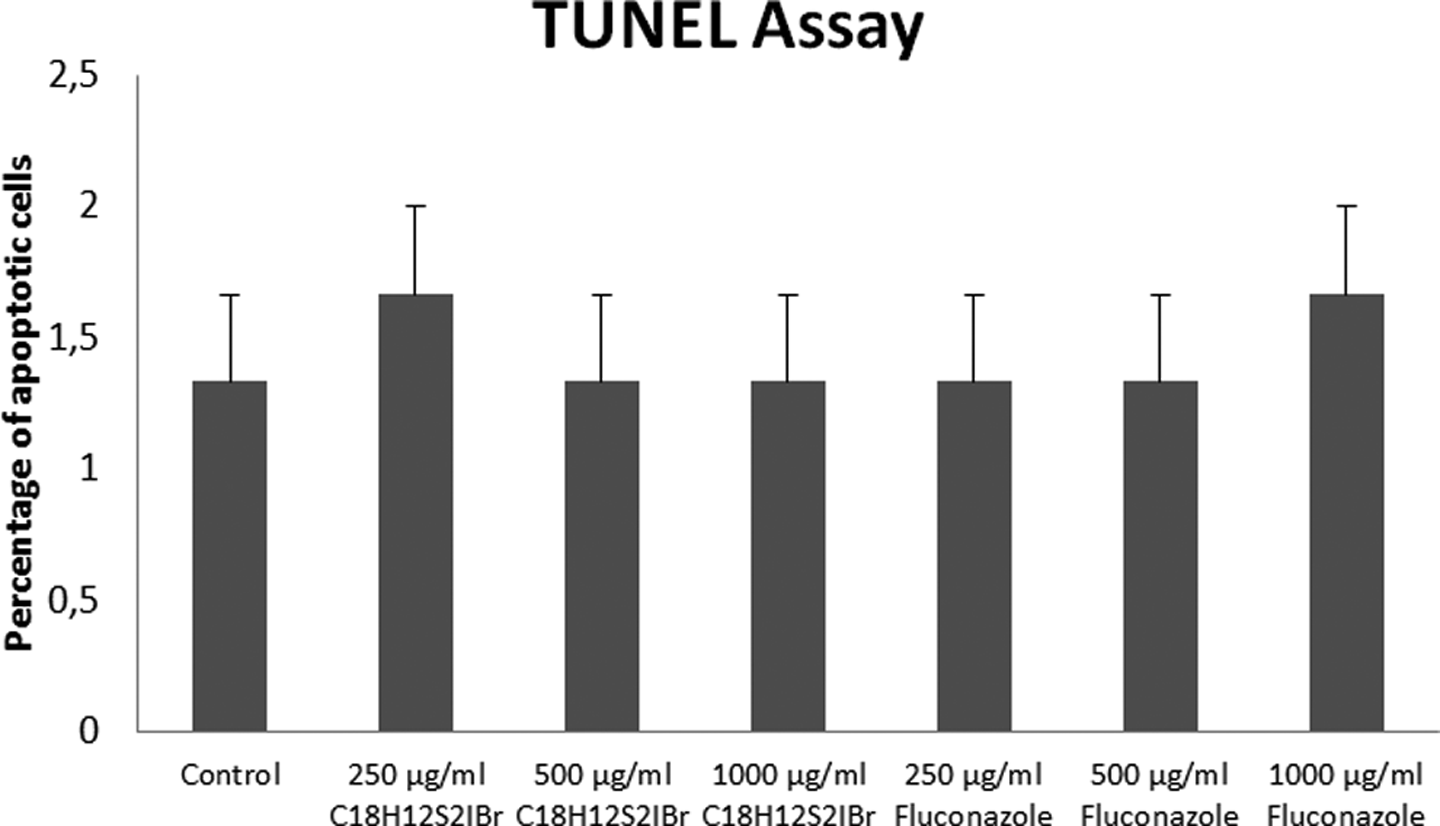
Results of TUNEL assay in L929 cells treated with C18H12S2IBr and fluconazole for 24 hours.
Survivin ELISA
Survivin is an inhibitory protein that regulates apoptosis and cell cycle. Neither drug had an effect on survivin levels (Figure 2).
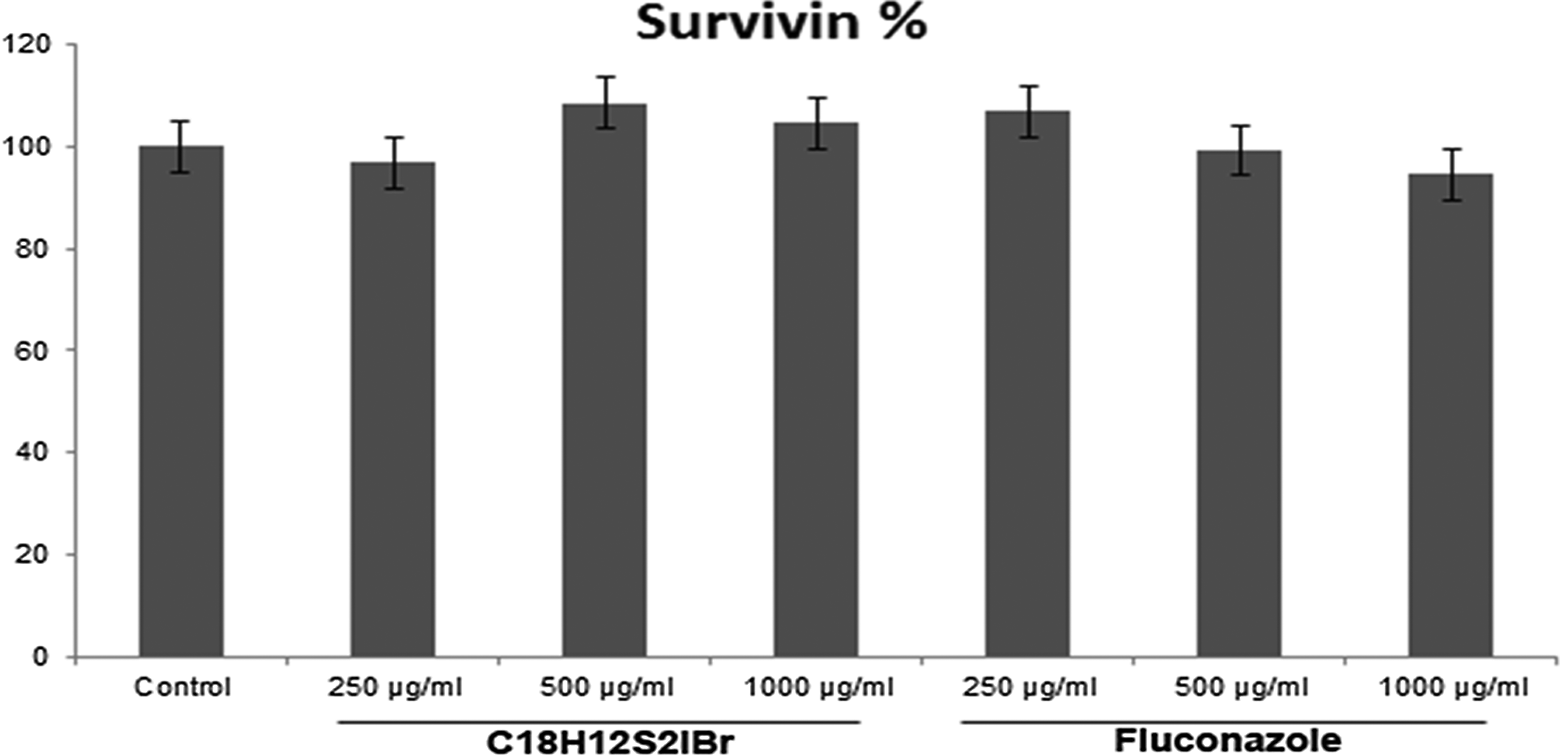
Percentage of survivin % in L929 cells treated with C18H12S2IBr and fluconazole for 24 hours.
Metabolic Activity Assay
As cell viability decreased due to the increase in C18H12S2IBr concentration, the amount of glucose left in the culture medium increased and lactate production decreased accordingly (Table 2). The alterations in 500 µg/mL and 1000 µg/mL C18H12S2IBr treatment groups were found to be statistically significant from the control group. Moreover, the glucose and lactate concentrations were significantly different in the C18H12S2IBr group when compared to the same concentrations of fluconazole (Table 2).
L929 cells were treated either with C18H12S2IBr or fluconazole. The concentration of glucose and lactate (mmol/L) in the culture medium was measured.a
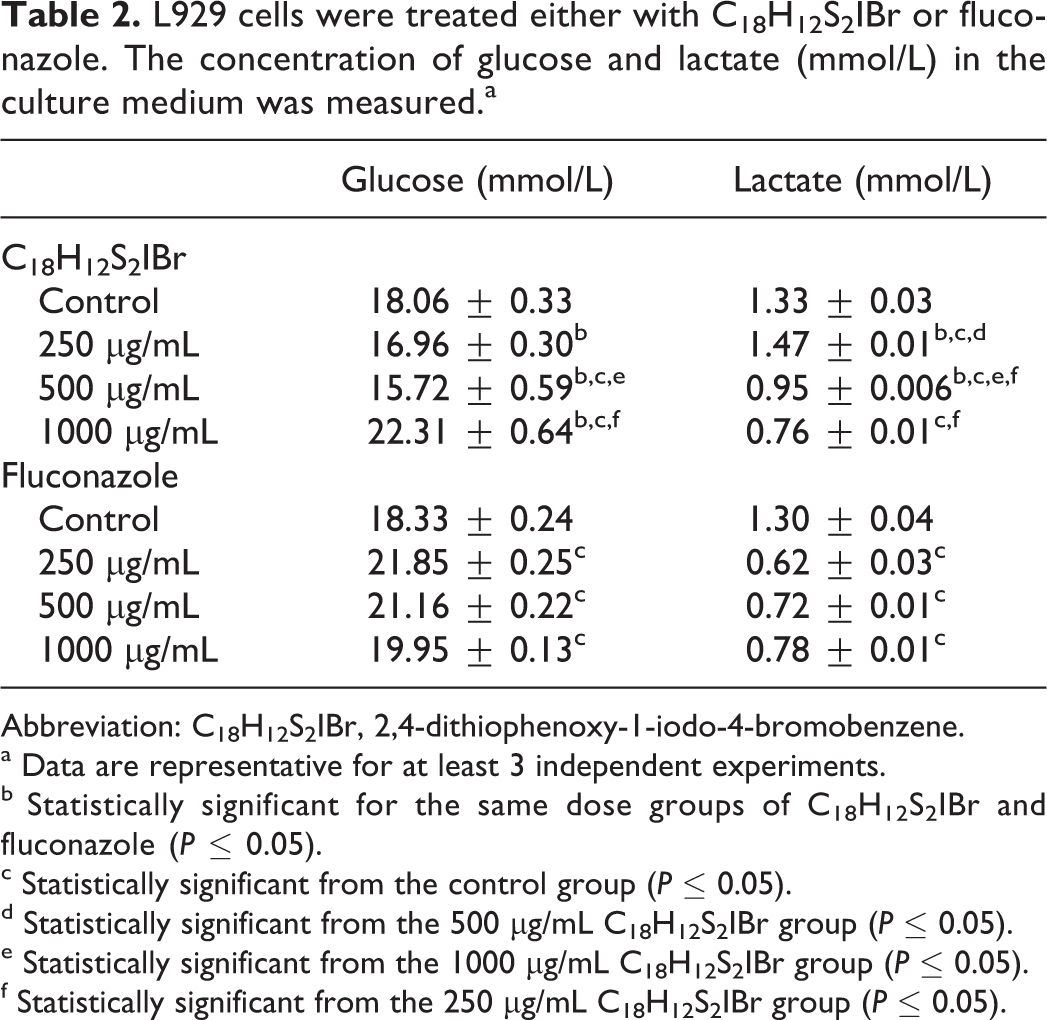
Abbreviation: C18H12S2IBr, 2,4-dithiophenoxy-1-iodo-4-bromobenzene.
a Data are representative for at least 3 independent experiments.
b Statistically significant for the same dose groups of C18H12S2IBr and fluconazole (P ≤ 0.05).
c Statistically significant from the control group (P ≤ 0.05).
d Statistically significant from the 500 µg/mL C18H12S2IBr group (P ≤ 0.05).
e Statistically significant from the 1000 µg/mL C18H12S2IBr group (P ≤ 0.05).
f Statistically significant from the 250 µg/mL C18H12S2IBr group (P ≤ 0.05).
Discussion
In the present study, the comparative cytotoxic effects of the newly synthesized antifungal agent C18H12S2IBr and a well-known antifungal drug fluconazole were evaluated in mouse fibroblast L929 cells. It has been reported that L929 cells represent a good cell model for evaluating the chemical toxicity in vitro. 15
In order to test drugs and to predict human toxicity, in vitro cytotoxicity assays can be useful. Different cytotoxicity assays can give several results about the tested compound depending on the type of assay. 16 Carrying out multiple markers for cytotoxicity is crucial for revealing early reversible or late irreversible effects of cytotoxic compounds. 17
In this study, L929 cells were cultured in the different concentrations of fluconazole and C18H12S2IBr for 24 hours and cytotoxicity was determined with the MTT assay, the LDH leakage assay, and by measuring cellular protein content.
It is important to emphasize that lower concentrations of C18H12S2IBr (10, 25, 50, 100, and 150 µg/mL) did not induce cell cytotoxicity in L929 cells after 24 hours incubation (data not shown). But in this study, the two highest concentrations of C18H12S2IBr (500 and 1000 µg/mL) investigated showed 32% and 39% decrease in cell viability according to MTT assay. Although the concentrations chosen were high compared to allowed doses for fluconazole, results would represent good correlation for repetitive exposure of this newly synthesized antifungal agent. No statistically significant changes were observed in all fluconazole concentrations compared to control. Similarly, in a study conducted by Rodriguez and Acosta, hepatocytes were exposed to 50 µmol/L to 1.0 mmol/L fluconazole for 0.5 to 6 hours and no significant decrease in cell viability was observed in MTT assay. 18
LDH release into the cell culture media increased in all C18H12S2IBr dose groups in a dose-dependent manner. However, there were no statistical differences in fluconazole dose groups. LDH is an intracellular enzyme and release of this enzyme into the culture media is a useful cytotoxicity marker that represents cell membrane damage. 19 Increase in LDH release in C18H12S2IBr-treated L929 cells revealed that this drug alters cell membrane properties. In a study conducted by Abe et al, 0.5 to 2.0 µg/mL fluconazole did not decrease cell viability or increase LDH release into the culture medium. 20 However, in the present study, fluconazole treatment doses were higher than the mentioned study and LDH leakage increased.
The proliferation rate of the cells decreases as a result of cytotoxicity and brings about the reduction in protein content of the cultured cells. 9 In the present study, the amount of total protein decreased in response to fluconazole and C18H12S2IBr treatment in a dose-dependant manner. 2,4-Dithiophenoxy-1-iodo-4-bromobenzene (250 µg/mL and 1000 µg/mL treatment groups) had higher protein levels than the same concentrations of fluconazole.
In this study, the TUNEL assay was used to assess apoptotic cell death. There were no statistically significant changes in the number of apoptotic cells among control, C18H12S2IBr, and fluconazole treatment groups.
The fundamental functions of survivin are the inhibition of apoptosis and regulation of cell division. 21 According to our results, survivin levels did not change among groups. The unchanged number of apoptotic cells was parallel to the unchanged survivin levels.
When developing new antifungal agents, it is important to focus on drug resistance. The clinical value of currently used fungicides is limited because of the emergence of drug resistance. 22 The dysregulation of apoptosis and overexpression of survivin were found to be one of the contributors to develop drug resistance. 23 Due to increased levels of survivin, overexpression of antiapoptototic protein Bcl-2 and decreased expression of apoptotic protease-activating factor were observed. These are other mechanisms, which indicate the dysregulation of apoptosis. 24 2,4-Dithiophenoxy-1-iodo-4-bromobenzene and fluconazole treatment neither cause changes in apoptosis nor survivin level, so we may propose that they seem not to have a potential to cause drug resistance by this particular mechanism. The gaps should be filled in this issue.
Glucose consumption and lactate production altered after L929 cells were treated with high doses of C18H12S2IBr and fluconazole, indicating the changed metabolic activities. These results are in accordance with the cell viability results. As the cell number decreased, glucose concentration increased and lactate concentration decreased in the culture medium.
We investigated for the first time the effect of fluconazole and newly synthesized C18H12S2IBr on glucose and lactate metabolism and also survivin expression in L929 cells. In both cytotoxicity and cell death analyses, the lowest dose of C18H12S2IBr and fluconazole responded similarly. Because of its established antifungal activity and similar toxicity to an antifungal agent currently in clinical use, C18H12S2IBr may provide a good alternative treatment for fungal infections. This is a preliminary in vitro study, and further investigations should be performed in different mammalian cells both in vitro and in vivo.
Footnotes
Acknowledgements
The authors want to thank to Hacettepe University Scientific Research Projects Coordination Unit for providing devices used in this study (Project no: 012D12601004, 013D07601, 013D11601003).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
