Abstract
Doxorubicin (DOX) is an anthracycline antibiotic. Despite its unwanted side effects, it has been successfully used in tumor therapy. Given that oxidative stress and inflammatory factors are essential to cardiotoxicity caused by DOX, we assumed that alamandine, which enhances endogenous antioxidants and has anti-inflammatory effects, may prevent DOX-induced cardiotoxicity. Rats received DOX (3.75 mg/kg) i.p on days 14, 21, 28, and 35 (total cumulative dose = 15 mg/kg) and alamandine (50 μg/kg/day) via mini-osmotic pumps for 42 days. At the end of the 42-day period, we evaluated hemodynamic parameters, electrocardiogram, cardiac troponin I (cTnI), superoxidase dismutase (SOD), total antioxidant capacity (TAC), malondialdehyde (MDA), inflammatory cytokines (tumor necrosis factor-α (TNF-α), IL-1β, NF-κB), apoptosis markers (caspase 3), and histopathology of haemotoxylin- and eosin-stained cardiac muscle fibers were evaluated. DOX significantly increased QT, corrected QT (QTc), and RR intervals. Alamandine co-therapy prevented ECG changes. Alamandine administration restored DOX-induced disruptions in the cardiac muscle architecture and vascular congestion. Alamandine co-therapy also alleviated other effects of DOX, including cardiac contractility, decreased systolic and diastolic blood pressure, and increased left ventricular end-diastolic pressure. Moreover, alamandine co-therapy substantially decreased the elevation of oxidative stress markers, inflammatory cytokines, and caspase 3 in DOX-treated rats. The results suggest that alamandine reduced DOX-induced cardiotoxicity via antioxidant, anti-inflammatory, and anti-apoptotic activities.
Introduction
DOX is an anticancer drug widely used to treat a broad range of malignancies and solid tumors. 1 Although it has high antitumor efficacy, its toxicity limits its use in chemotherapy. 2 The toxic effects of DOX on cardiomyocytes are also a significant concern. DOX can cause congestive heart failure if it reaches a certain cumulative dose. 3 Different pathways are involved in the cardiotoxicity processes induced by DOX. A substantial amount of literature suggests that oxidative stress, inflammation, and apoptosis are also involved.4,5 Oxidative stress is an imbalance between reactive oxygen species (ROS) and antioxidants. DOX significantly decreases endogenous antioxidant levels, resulting in redox imbalances and increased oxidative stress. Oxidative disruptions to the myocardium (compared to other tissues) are likely attributed to reduced levels of antioxidant enzymes, including SOD. SOD is an enzyme that catalyzes toxic superoxide radicals so that they become natural molecular oxygen and hydrogen peroxide. SOD is effective at protecting cells from toxic oxygen metabolites. 3
Increased ROS production could be another mechanism of DOX-induced oxidative stress. 3 Generally, most ROS are produced in mitochondria. 3 DOX can be converted into semiquinone by ROS-producing mitochondrial enzymes. This DOX semiquinone radical generates hydrogen peroxide and superoxide radicals. 3
Lipid peroxidation is one DOX-induced cardiac toxicity mechanism. Malondialdehyde (MDA) is a low-molecular-weight aldehyde that can be formed by free radical attacks on unsaturated fatty acids in a lipid membrane. MDA levels are a valuable indicator of lipid peroxidation. 6
Exposure to DOX increases oxidative stress, thereby intensifying the expression of nuclear factor-kappa B (NF-κB) cells. As a result, more pro-inflammatory cytokines are released into the myocardium.7,8
NF-κB signaling is the converging point at which subsequent signal cascades are regulated, including IL-6, TNF-α, IL-1, and other inflammatory cytokines. 9 TNF-α is a pro-inflammatory cytokine that binds to TNF-R1 and TNF-R2 receptors. The TNF/TNF-R1 pathway is a critical element of the body’s inflammatory response to DOX. 10
IL-6 is a vital cytokine that controls cell proliferation, angiogenesis, and invasion. Increased IL-6 levels in serum or tumors are linked with aggressive cancer cell phenotypes and decreased survival in multiple cancer types. 11 Several studies have shown that DOX therapy increases inflammation and IL-6 levels.12,13
Evidence indicates that the extrinsic and intrinsic pathways of apoptosis are associated with DOX-induced cardiotoxicity. Caspase-3 plays a central role in the execution of apoptosis. 14 DOX significantly increases the levels of caspase-3—a biomarker of cardiac toxicity and myocardial infarction—in the serum levels of rats. 15
Compounds with potent antioxidant properties could effectively ameliorate DOX-induced cardiomyopathy.7,16 Alamandine is a new member of the angiotensin family that has exhibited significant cardioprotective effects in rats treated with isoproterenol. 17 This peptide is similar to Ang-(1–7) and can bind to the Mas-related G-coupled receptor known as member D (MrgD). 17
In previous research, alamandine increased the expression of several antioxidant proteins, such as SOD and catalase, in ventricles subjected to ischemia and reperfusion (I/R). It also decreased the expression of caspase-9, Bax, and caspase-3 in I/R ventricles. However, co-treatment with PD123319 or MrgD receptor antagonists reduced these effects. 18 Alamandine also reduced IL-1β and TNF-α levels in rat neonatal cardiomyocytes following increases brought on by LPS treatment. 19
Based on the antioxidant, anti-inflammatory, and cardioprotective effects of alamandine, the present study aimed to evaluate the protective effect of alamandine against DOX-induced cardiac toxicity through serological, biochemical, hemodynamic, and histopathological studies.
Materials and methods
Animals
Male Sprague-Dawley rats (weight = 200–250 g) were subjected to a 12-hour/12-hour light/dark cycle and were given standard rat chow and water ad libitum. All animal experiments followed the guidelines of the Fasa University of Medical Sciences Animal Ethics Committee for animal studies. These guidelines are based on Directive 2010/63/EU of the European Parliament, which protects animals used for scientific purposes. The study was approved by the Fasa University Animal Ethics Committee (IR.FUMS.REC.1397.014). For biochemical and hemodynamic studies, rats were anesthetized via an intraperitoneal sodium pentobarbital injection (50 mg/kg). The anesthesia level was evaluated using the toe-pinch reflex.
Experimental protocols and classification
A total of 35 animals were randomly divided into five groups of seven animals. These groups are described below. Group I (control group): The control group did not experience any surgical intervention. Group II (sham group): This group received normal saline for 42 days via mini-osmotic pumps (model 2006; ALZET Osmotic Pumps, CA, USA) placed subcutaneously between the scapulae. These rats also received normal saline intraperitoneally (i.p) (injection volume = 0.5 ml) on days 14, 21, 28, and 35. Group III (DOX group): This group received 3.75 mg/kg doxorubicin HCL (Tocris Bioscience) i.p on days 14, 21, 28, and 35 (total cumulative dose = 15 mg/kg). Group IV (ala group): These rats received alamandine (Phoenix Pharmaceuticals Inc., CA, USA) for 42 days via mini-osmotic pumps (infusion rate = 0.15 μl/h) (50 µg alamandine/kg/day). Group V (Ala + DOX group): Alamandine was administered by mini-osmotic pumps for 42 days (50 µg alamandine/kg/day) and DOX (3.75 mg/kg) i.p on days 14, 21, 28, and 35 to reach the (total cumulative dose = 15 mg / kg).
Biochemical, hemodynamic, and histopathological studies were conducted 7 days after the final DOX injection.20,21 Each animal was weighed at the beginning of the study and 1 week after the last dose of DOX.
Electrocardiogram
Electrocardiogram (ECG) traces were recorded on day 42 and before the rats were sacrificed. Needle electrodes were placed under the skin of the animals in the lead II position. ECG parameters (heart rate (HR), QT, corrected QT interval (QTc), PR interval, and RR interval) were recorded with PowerLabs and LabChart software (ADInstruments, Australia).
Hemodynamic parameters
After ECG recording, a polyethylene catheter was placed in the right carotid artery, leading to the left ventricle. The right femoral artery had also been cannulated. The catheters were attached to the pressure transducers that were used to measure left ventricular pressure and blood pressure. We recorded HR, systolic blood pressure (SBP), diastolic blood pressure (DBP), cardiac contractility (dP/dt), left ventricular systolic pressure (LVSP), and left ventricular end-diastolic pressure (LVEDP) using a 4-channel Power Lab physiological imaging system (ADInstruments, Australia).
Next, rats were euthanized with pentobarbital 150 mg/kg, i.p. Blood samples were then taken and centrifuged at 4000 rpm for 10 min. The serum was taken and maintained at –80°C before being used to assess biochemical parameters. Rats’ hearts were dissected and washed with PBS (10 mM PO43−, 137 mM NaCl and 2.7 mM KCl; pH = 7.4) and then dried and weighed on filter paper. The small part of the left ventricle was weighed and then homogenized in 10 mM phosphate-buffered saline (PBS; pH 7.4 containing 0.1% sodium dodecyl sulfate, 0.1 mM phenylmethylsulphonyl fluoride, cooled) at a buffer-to-tissue ratio of 5:1 (w/w). It was then centrifuged at 10,000×g for 20 min at 4°C. The supernatant was stored at −20°C until needed for analyzing oxidative stress parameters, antioxidants, and cytokines (which was done using the ELISA kit).
Assessment of cardiac troponin I
Cardiac troponin I (cTnI) serum levels were measured using a sandwich ELISA kit (cat. no. HEA478Ra; Cloud-Clone Corp). Microplate wells were filled with 50 µl standard or sample, and then a prepared detection reagent was added and incubated for 1 h at 37°C. After washing, 100 µl of a substrate solution was added to each well and incubated for 10–20 min at 37°C. Then, 50 µl of a stop solution was added, and color changes were measured at 450 nm with a BioTek ELISA reader.
Assessment of inflammatory cytokines in cardiac tissue and serum
The sample levels of IL-1β, TNF-α, IL-6, and NF-kB were measured using an ELISA kit following the manufacturer’s protocol. A TNF-α assay kit (cat. no. CSB-E11987r) purchased from CUSABIO (Wuhan, China), an IL-1β assay kit (cat. no. SEA563Ra), an NF-κB assay kit (cat. no. SEB824Ra), and an IL-6 assay kit (cat. no. SEA079Ra) were obtained from Cloud-Clone Crop (Cloud-Clone Crop Technology co., Ltd., Wuhan, China). Cytokine levels in serum and cardiac tissues were measured using the quantitative ELISA sandwich technique. For the TNF-α analysis, the kit’s microplate was pre-coated with a TNF-α-specific monoclonal antibody. Briefly, 100 µl standard or sample was added to the appropriate microplate wells and then incubated for 2 h at 37°C. After incubation, 100 µl TNF-α -specific biotin-conjugated antibody was added to each well and incubated for 1 h at 37°C. After washing, 100 µl avidin conjugated with horseradish peroxidase (HRP) was added to each well and incubated for 1 h at 37°C. After washing, 90 µl TMB (3,3’,5,5’-Tetramethylbenzidine) substrate solution was added and incubated for 20 min at 37°C, and finally, the enzyme-substrate reaction was terminated by the addition of 50 µl stop solution. Then, color changes were measured spectrophotometrically at 450 nm with a BioTek ELISA reader. The TNF-α concentration was then calculated by comparing the OD of the samples to the standard curve. The assay protocols for IL-1β, NF-κB, and IL-6 were similar to those for TNF-α.
Assessment of oxidative stress markers in serum and cardiac tissue
The MDA assay kit (cat. no. A003-1) and SOD assay kit (cat. no. A001-3-2) were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China), and the TAC assay kit (cat. no. ZB-TAC-96A) was obtained from ZellBio GmbH (Germany).
MDA measurements were based on its reaction with thiobarbituric acid (TBA). A volume of 0.15 ml sample was transferred to the sample tube. A standard solution (0.15 ml) was added to the standard tube, and then 0.15 ml of dehydrated alcohol was added to another blank testing tube. A volume of 4 ml mixed reagent was then introduced into each of these tubes. The contents of each tube were mixed sufficiently by vortex and then incubated at 95°C for 40 min. After that, each specimen was cooled in lotic water and centrifuged at 3500 rpm for 10 min. Finally, the supernatant in each tube underwent colorimetric assay at 532 nm (zero was adjusted by distilled water).
The measurement of SOD was based on the WST-1 method. A volume of 20 µl double distilled water, 20 µl enzyme working solution, and 200 µl substrate application solution was added to the control wells. Next, 20 µl double distilled water, 20 µl enzyme dilution solution, and 200 µl substrate application solution was added to the control blank wells. After that, 20 µl sample, 20 µl enzyme working solution, and 200 µl substrate application solution was added to the measure wells. Following that, 20 µl sample, 20 µl enzyme dilution solution, and 200 µl substrate application solution was added to the measure blank wells. With all the contents now incorporated, the samples were mixed well and incubated at 37°C. The absorbance value was measured at 450 nm by a microplate reader. The SOD activity was calculated based on the degree of inhibition of this reaction.
We used a TAC assay kit to measure the total antioxidant capacity of the biological samples. As part of this process, 10 µl standard/ sample and 190 µl working chromogen reagent were added to the measure wells. OD1 was measured with a microplate reader at 490 nm. Then, 50 µl of the metal ion solution was added, and the samples were incubated at room temperature for 2 min. After the stop solution was added, OD2 was measured at 490 nm. The ΔOD values were calculated by subtracting the OD2 values from the OD1 values. The concentrations of the sample were calculated based on the equation y = mx + b, as determined by the weighted (1/x) linear regression of the standard curve.
Assessment of apoptotic markers
Cardiac tissue was collected from all groups a few min after the rats were sacrificed. The homogenates of the fresh hearts were then prepared and centrifuged at 10,000 g for 10 min at 4°C. The volume of supernatant was measured, and the total protein content was calculated by the Bradford method. 22 Briefly, bovine serum albumin was used as a standard to determine protein concentration. A volume of 1 ml of Bradford reagent (BioRad, Cambridge, UK) was added to 20 μ of sample or standard. The absorbance value was measured by a spectrophotometer at 595 nm after 5 min. After this, the fresh heart homogenates were used to measure caspase-3 in the heart tissue using the assay kit (cat. no. SEA626Ra, Uscn. Life Science Inc.). Briefly, 100 µl standard or sample was added to each well and incubated for 1 hour at 37°C. After aspiration, 100 µl of prepared detection reagent was added and incubated for 1 hour at 37°C. After washing, 90 µl of the substrate solution was added and incubated for 15 min at 37°C. After the addition of the stop solution, color changes were measured at 450 nm using a BioTek ELISA reader.
Histological examination of the myocardium
The apical parts of the tissue sections were fixed for 48 h in 10% neutral buffered formalin (cat. no. HT501128; Sigma-Aldrich). These tissue samples were then dehydrated by being passed through graded ethanol concentrations (cat. no. 51976; Sigma-Aldrich). The dehydration sequence was as follows: 70% ethanol for 1 h, 80% ethanol for 1 h, 90% ethanol for 1 h, 95% ethanol for 1 h, and two incubation steps in 100% ethanol for 1 h each. Xylene (cat. no. 108298; Sigma-Aldrich) was used for cleaning. Finally, samples were placed in paraffin wax at 63°C. Subsequently, the paraffin blocks were cut into 5-μM thick sections with a rotary microtome (HM 325; Thermo Scientific USA) and stained with the hematoxylin-eosin.
Hematoxylin-eosin staining was performed using the standard process, starting with immersion in three xylene bottles to complete dewaxing. Then, sections were re-hydrated with two incubation steps in 100% ethanol (2 min for each step), 70% ethanol (2 min), and three incubation steps in deionized water (3 min). Samples were then stained with a hematoxylin solution (cat. no. H3136; Sigma-Aldrich) for 30 min, followed by a 1-min rinse in deionized water. Acid alcohol was used for destaining.
Following that, the samples were counterstained in an eosin solution (cat. no. E4009; Sigma-Aldrich) for 4 min. Finally, sections were dehydrated again with two incubation steps in 95% ethanol and two incubation steps in 100% ethanol (2 min for each step). We used tap water to wash samples between all steps except the last ones. Finally, the cleaning process was finished using xylene. The slides were placed on a 24 × 60 glass cover with an entellan adhesion solution. The slides in each category were microscopically analyzed using an Olympus BX-53 microscope, and all histopathological changes (e.g., inflammatory cell infiltration, hemorrhaging, and necrosis of myofibers) were examined. 23 The severity of these pathological lesions was ranked as follows: score 0 was considered to be normal; score 1 as borderline; score 2 as mild; score 3 as moderate and score 4 as severe.
Data and statistical analyses
Statistical analyses were done using GraphPad Prism software. All data are expressed as mean ± standard deviation (SD). When the data were representative of a normal distribution, one-way analysis of variance (ANOVA) was used to compare all groups, followed by Tukey’s post-hoc test. Score analysis of myocardial damage, was done using the non-parametric ANOVA (Kruskal-Wallis test) with Dunn’s post-hoc test. A p value of ≤ 0.05 was considered to be statistically significant for all tests.
Because the measured parameters were not affected by saline injection for the sham group, only the exposure of the control group were compared with the results of the remaining groups.
Results
Impact of doxorubicin and alamandine co-administration on cardiac performance indices
Table 1 shows the effect of DOX and alamandine co-administration on cardiac performance indices. The SBP and DBP of the rats treated with DOX were significantly reduced when compared with the control group (p = 0.004 and p = 0.013, respectively). Compared with the DOX group, the co-administration with 50 μg/kg/day alamandine for 42 days yielded a significant increase in SBP (p = 0.016) but no significant increase in DBP. The dp/dtmax, dp/dtmin (p < 0.001), and LVSP (p = 0.005) of rats treated with DOX were significantly reduced when compared with the control group. Meanwhile, the LVEDP of DOX-treated rats increased (p < 0.001). Treatment with alamandine (50 μg/kg/day for 42 days) plus DOX increased dP/dtmax (p < 0.001), dP/dtmin (p = 0.006) and LVSP (p = 0.043) and decreased LVEDP (p < 0.001) when compared to the DOX group. Alamandine treatment alone ( 50 μg/kg/day for 42 days) had no significant effect on arterial blood pressure, dP/dtmax, dP/dtmin, LVEDP, or LVSP when compared to the control group.
Hemodynamic parameters of DXR-induced heart failure rats when treated with alamandine compared to the control group.

Data are expressed as mean ± SD; n = 7 for each treatment group. Ala, alamandine; DOX, doxorubicin;
SBP, systolic blood pressure; DBP, diastolic blood pressure; +dP/dtmax, maximal positive derivative of ventricular pressure; (−dP/dtmax), maximal negative derivative of ventricular pressure; LVSP, left ventricular systolic pressure; LVEDP, left ventricular end-diastolic pressure.
*p < 0.05, **p < 0. 01, ***p < 0.001 compared to the control.
#p < 0.05, ##p < 0. 01, ###p < 0.001 compared to the DOX group.
Effect of doxorubicin and alamandine co-administration on ECG parameters in rats
As shown in Table 2 and Figure 1, DOX administration caused ECG abnormalities—as presented by QT and QTc prolongation, bradycardia, and the lengthening of the R-R interval—when compared to the control group. Co-treatment with alamandine significantly decreased DOX-induced QT interval prolongation (p = 0.036).
ECG parameters of DXR-induced heart failure rats when treated with alamandine compared to the control group.

Data are expressed as mean ± SD; n = 7 for each treatment group. Ala, alamandine; DOX, doxorubicin; HR, heart rate; bmp, beat per minute; QTc; Corrected QT Interval.
*p < 0.05, ***p < 0.001 compared to the control; #p < 0.05, compared to the DOX group.

Electrocardiogram (ECG) analysis of the anesthetized rats. The averaging view of ECG is shown on the right.
Effect of doxorubicin and alamandine co-administration on body weight, heart weight, and heart weight to body weight ratio
Table 3 shows the mean body weights of the four groups of rats over the 42-day experimental period. Pretreatment weights were not significantly different across groups. However, after 42 days, the average body weight in the DOX-treated group decreased (p < 0.001) when compared to the control group. The average body weight of the DOX + alamandine group increased when compared to the DOX-treated group (p < 0.001). No significant differences between the groups’ heart weight and heart weight to body weight ratio were detected.
Body weight, heart weight and heart weight to body weight ratios in different groups.

Data are expressed as mean ± SD; n = 7 for each treatment group. Ala, alamandine; DOX, doxorubicin; BW, body weight; HW, heart weight.
**p < 0. 01, ***p < 0.001 compared to the control; ###p < 0.001, compared to the DOX group.
Impact of doxorubicin and alamandine co-administration on serum cardiac troponin
DOX increased cTnI levels when compared with the control group (5.15 ± 0.90 vs. 1.95 ± 0.50 (pgl/mL), respectively; p < 0.001). Almandine significantly decreased cTnI levels in the DOX + alamandine group compared to the DOX group (3.34 ± 1.12 vs. 5.15 ± 0.90 (pgl/mL), respectively; p = 0.014).
Effect of doxorubicin and alamandine co-administration on oxidative stress markers (MDA, SOD, and TAC) in serum and cardiac tissue
DOX administration resulted in a marked increase in cardiac and serum MDA (p < 0.001) and a significant decrease in serum SOD (p = 0.015), cardiac SOD (p = 0.002), and serum TAC (p = 0.002) relative to the control group. Co-treatment with alamandine reduced these effects (Figure 2).

Oxidative stress markers in the doxorubicin-induced cardiotoxicity in rats. (A) Serum total antioxidant capacity (TAC). (B) Serum malondialdehyde (MDA). (C) Serum superoxidase dismutase (SOD). (D) Myocardial MDA. (E) Myocardial SOD. (Data are mean ± SD, n = 7. Comparisons were made using the one-way ANOVA with Tukey’s post hoc tests. **p < 0. 01, ***p < 0.001 versus respective control; #p < 0.05, ##p < 0. 01 versus respective DOX group.)
Effect of doxorubicin and alamandine co-administration on inflammatory cytokines
TNF-α, IL-1β, IL-6, and NF-κB are pro-inflammatory mediators. TNF-α, IL-1β, IL-6, and NF-κB levels increased in the DOX group when compared to the control group. Rats co-treated with alamandine presented significantly fewer inflammatory cytokines than the DOX group (Figure 3).

Inflammation in the doxorubicin-induced cardiotoxicity in rats. (A) The serum content of nuclear factor kappa B cell (NF-κB). (B) The serum content of interleukin 6. (C) Serum interleukin-1 beta (IL-1β). (D) The serum content of the tumor necrosis factor alpha (TNF-α). (E) Myocardial IL-1β. (Data are mean ± SD, n = 7. Comparisons were made using the one-way ANOVA with Tukey’s post hoc tests. **p < 0. 01, ***p < 0.001 versus respective control; #p < 0.05, ##p < 0. 01 versus respective DOX group.)
Effect of doxorubicin and alamandine co-administration on apoptotic marker caspase-3 in cardiac tissue
DOX increased caspase-3 in the serum (p = 0.011) and cardiac tissue (p < 0.001) compared with the control group, and alamandine co-treatment reduced these effects (Figure 4).
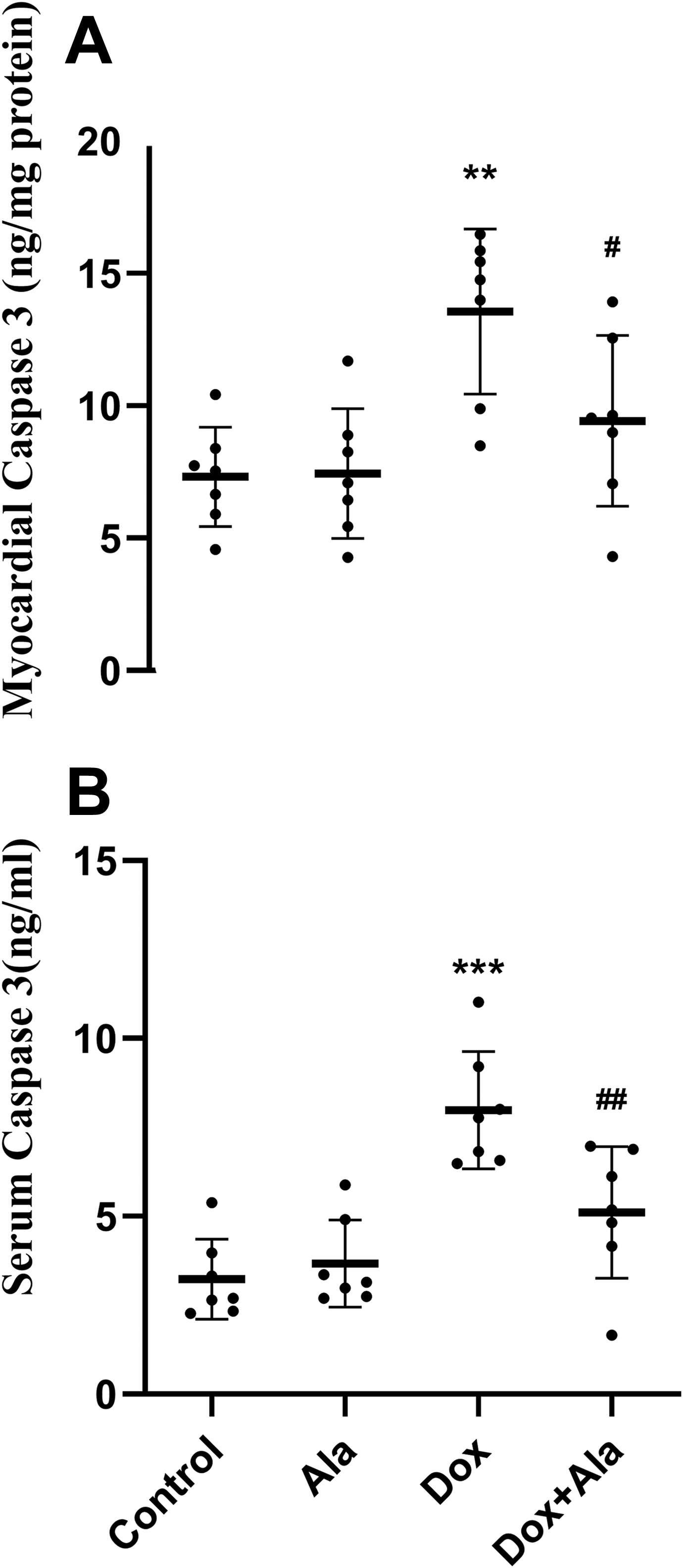
Apoptosis parameters in the doxorubicin-induced cardiotoxicity in rats. (A) The myocardial content of caspase 3. (B) The serum content of caspase 3. (Data are mean ± SD, n = 7. Comparisons were made using the one-way ANOVA with Tukey’s post hoc tests. **p < 0. 01, ***p < 0.001 versus respective control; #p < 0.05, ##p < 0. 01 versus respective DOX group.)
Histopathological findings
The control group exhibited normal myocardium architecture. The heart sections of the DOX group presented significant injuries in the cardiac tissue as evidenced by vascular congestion, infiltration of inflammatory cells, disrupted cardiac muscle architecture, loss of muscular striations, hydropic degeneration, focal apoptosis, and coagulative necrosis.
The histopathological findings showed that alamandine alone had no significant effect on cardiac fibers. Alamandine co-treatment for 42 days (50 μg/kg/day) protected the cardiac cell architecture from the harmful effects of DOX, keeping it almost as it was before DOX treatment (Figure 5).

(A) The histological sections of the rats’ heart: (A) Control group, (B) DOX-treated group, (C) Alamandine group, (D) DOX + Alamandine group. Inflammatory cell (arrowhead), disruption of cardiac muscles architecture (arrow) and, vascular congestion (star). (Hematoxylin-eosin, ×400). (E) The histopathological score analysis of the heart tissue damage showing the effect of DOX with/without alamandine. The score 0 signifies normal; 1 is borderline; 2 is mild; 3 is moderate; 4 is severe. (Data are mean ± SD, n = 7. Comparisons were made using the non-parametric ANOVA (Kruskal-Wallis test) with Dunn’s post-hoc test. ***p < 0.001 versus respective control; #p < 0.05 versus respective DOX group.)
Discussion
DOX-induced cardiotoxicity can be present at any treatment stage. Oxidative stress, inflammation, and apoptosis play primary roles in DOX-induced cardiomyopathy. 24 Alamandine has antioxidant, anti-inflammatory, and anti-apoptotic activities. 25 Therefore, alamandine could also defend against DOX-induced cardiotoxicity. The purpose of this study was to determine the cardioprotective effects of alamandine on DOX-induced cardiotoxicity in rats. Also, to identify the underlying mechanisms that may have been involved.
DOX-induced cardiotoxicity was evaluated by measuring cardiac performance indexes, oxidative stress markers (MDA, SOD, and TAC), inflammatory cytokines, and serum cTnI, along with a histopathological analysis of the heart tissue. Our study reveals that DOX-treated rats experienced increased levels of oxidative stress markers (MDA, SOD, and TAC).
Oxidative stress is a significant factor in the pathophysiology of DOX cardiotoxicity. Compounds with antioxidant properties counteract the clinical manifestations of cardiotoxicity induced by chemotherapeutic agents. Coenzyme Q10, as an intracellular antioxidant, can benefit heart function during chemotherapy in pediatric cancer patients. 26 Compared to other organs, the heart has less superoxide dismutase, glutathione peroxidase, and catalase, which might explain the heart’s high DOX toxicity. 27
In this study, alamandine co-administration with DOX significantly decreased MDA and increased SOD and TAC levels. These findings were consistent with the results of previous research in which alamandine enhanced antioxidant protein expression in ventricles exposed to ischemia-reperfusion injury. These effects were decreased by co-treatment with either PD123319 or MrgD receptor antagonists. 18
Inflammatory cascades can be triggered by oxidative stress. These cascades are mainly mediated by the NF-κB transcription factor.28,29 This transcription factor is essential in regulating inflammatory processes. 30 Several studies have reported the involvement of the NF-κB transcription factor in the pathogenesis of heart failure. 31 NF-κB activation increases the production of cytokines and chemokines.
Our results show increases in the DOX group’s inflammatory cytokines (i.e., IL-6, IL-1, TNF-α, and NF-κB) in comparison to the control group. The results also indicate that alamandine co-administration reduced these effects. This finding corroborates previous research that demonstrated the anti-inflammatory effects of alamandine. 19 It has been shown that an increase in LPS-induces IL-1β and TNF-α levels in mice cardiac tissues attenuated by alamandine. Moreover, alamandine decreases the gene expression of pro-inflammatory factors such as TNF-α, CCL2, and IL-1α induced by transverse aortic constriction in mice. 32
Oxidative stress evoked by DOX leads to cardiomyocytes apoptosis. 17 Various signaling pathways are involved in this type of apoptotic death, including caspase-3 activation. 33 In this study, an increase in caspase-3 activation was observed in the DOX group when compared to the control group. This activity significantly decreased when alamandine was co-administrated with DOX, indicating the cardiac protection effect of alamandine (by reducing the apoptosis rate in the heart). This is in agreement with the results produced by Byung et al., who reported that alamandine decreases caspase-3 expression in cardiac tissue after ischemia-reperfusion injury. 18
Increased oxidative stress triggers many signaling pathways, including caspase-3. 17 Therefore, alamandine’s ability to reduce oxidative stress could decrease caspase-3 levels. In the present study, DOX increased cTnI levels in the serum. cTnI is a biomarker for cardiotoxicity in cancer patients. 34 The alamandine co-administration group exhibited decreased serum cTnI, which indicates a decrease in DOX-induced cardiac toxicity.
In this study, the final body weight of the DOX-treated group decreased when compared to the control group. The heart weight of the DOX group, however, did not differ from that of the control rats, perhaps due to the cardiac congestion induced by DOX. The cardiac weight of the alamandine co-administration group did not significantly change. This might be because cardiac muscle repair was associated with reduced vascular congestion, as indicated by the histopathological findings.
In this experiment, DOX-induced cardiac dysfunction was detected based on cardiac function parameters. These parameters include decreased systolic and diastolic pressure, LVSP, dP/dtmax, and dP/dtmin, combined with increases in LVEDP and all confirmed histopathological features of the heart (Table 1 and Figure 5).
Alamandine treatment improved cardiac function parameters. Alamandine’s cardioprotective effects have been reported in previous research. 35 Different mechanisms might be involved in alamandine’s cardioprotective effects against the adverse effects of DOX, such as the inhibition of oxidative stress and suppression of inflammatory cytokines.
The electrocardiographic report showed some abnormalities associated with DOX cardiotoxicity, including increased QT and QTc interval duration, bradycardia, and R-R interval prolongation. Prolonged QTc and R-R intervals indicate delayed repolarization and a negative chronotropic effect. 36 Mantawy et al. showed that DOX-treated rats undergo several ECG changes, such as bradycardia and prolonged R-R and QTc intervals, when compared to the control group. Pretreatment with chrysin, a natural flavonoid with antioxidant and anti-inflammatory properties, significantly improved such ECG abnormalities. 37 In another study, Elberry et al. showed that DOX-treated rats present several ECG abnormalities such as bradycardia, prolonged ST and QT intervals, and ST-segment depression. Such ECG changes were improved by cranberry extract pretreatment. 38
ECG abnormalities were attenuated by alamandine co-treatment, and left ventricular function was maintained. Anticancer chemotherapy-related ECG abnormalities include QT interval prolongation and torsade de pointes, especially when the treatment plan involves anthracyclines or arsenic trioxides. 39
Notable aspects of EGGs in rats include lack of an isoelectric interval between the QRS complex and the T wave and short QT intervals. Significant evidence shows that EGGs taken from rats and mice have a large amplitude of K+ repolarizing currents, particularly the transient outward K+ current, Ito. Previous research also identified alterations in Ito’s contribution to ventricular repolarization as a significant potential factor for EGG abnormalities in humans. 40 A distinguished Ito current has been recorded in ventricular myocytes.41–43 Kv1.4, Kv4.2, and Kv4.3 are three k+ cloned channels that might generate distinct Ito phenotypes.41,43–46 TNF-α is upregulated in various cardiovascular diseases, including congestive cardiac failure and myocardial infarction. 47
Increased action potential duration (APD) and a downregulated transient outward Ik+ current (Ito) were observed previously in mammalian heart failure ventricular myocytes.42,48,49 Decreased outward currents and APD prolongation could lead to ECG abnormalities. 50
TNF-α can lead to a significant decrease in Ito density, modify its inactivation, and down-regulate Kv4.2 protein expression. Exposure to TNF-α from rat ventricular myocytes induces electrophysiological changes, including reduced Ito and APD prolongation, owing to inducible nitric oxide synthase (iNOS) induction and oxidant species generation. 51 These factors suggest that TNF-α plays a crucial role in the cardiotoxicity-related molecular mechanism of DOX. Alamandine co-administration reduced DOX-induced QT prolongation, which might be related to alamandine-induced reductions in TNF-α. Figure 6 shows an overview of the effects of alamandine on DOX-induced cardiotoxicity.

Graphic abstract summarizing the effect of alamandine on doxorubicin-induced cardiotoxicity.
In conclusion, alamandine significantly reduces DOX-induced cardiotoxicity in rats by modulating antioxidant status, apoptosis, and inflammatory cytokines. Histopathological and electrocardiographic findings from this study also indicate that alamandine protects against cardiotoxicity induced by DOX.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
