Abstract
The aim of this study was to investigate the protective effects of fish omega-3 (n-3) fatty acids on doxorubicin (DOX)-induced acute cardiotoxicity. A total of 24 rats were divided into three groups: control, DOX-treated, and DOX treated with fish n-3 fatty acids. Control group received 0.4 ml/kg/day of saline intragastrically. The rats in the fish n-3 fatty acid-pretreated group were given 400 mg/kg/day fish n-3 fatty acids for 30 days by intragastric intubation. To induce acute cardiotoxicity, DOX (30 mg/kg) was injected intraperitoneally by a single dose and the rats were killed after 48 h. DOX treatment caused severe damage in heart tissues. Disorganization of myocardial muscle fibers, myofibrillar loss, and cardiotoxic myocardial fibers with cytoplasmic vacuoles were seen. Fish n-3 fatty acid-treated rats showed an improved histological appearance in the DOX-treated group. Our data indicate a significant reduction in the activity of terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling in cardiomyocytes of the DOX-treated group with fish n-3 fatty acids therapy. The DOX-treated with fish n-3 fatty acids group showed a significant decrease in malondialdehyde levels, and an increase in superoxide dismutase and glutathione peroxidase activities in comparison with the DOX-treated group. This study showed that fish n-3 fatty acids may be a suitable cardioprotector against acute toxic effects of DOX.
Introduction
The anthracycline antibiotic doxorubicin (DOX) is one of the most potent anticancer drugs, which is used in cancer treatment. 1 However, its clinical usefulness is limited by specific toxicities to cardiac tissues. 2 –4 Cardiotoxicity is a important limiting factor in cancer chemotherapy. 5 The cardiotoxic effects of DOX may occur immediately after a single dose or several weeks to months after repetitive DOX administration. 6 In previous studies, congestive heart failure, cardiac dysfunction, cardiomyopathy, cardiac muscle wasting, electrocardiographic changes, endothelial dysfunction, dysregulation of blood pressure and heart rate, and loss of contractility were seen after DOX administration. 7 –12 Histologically, DOX toxicity leads to disorganization of myocardial histology and disruption of myocyte structure, including damage to the microtubules, vacuolization, picnotic nuclei, edema, sarcomere disruption, dilatation of the sarcoplasmic reticulum, mitochondrial injury, increase in collagen content around vessels, and loss of myofibrils. 8,9,13 Serious cardiac damage and mortality were detected in DOX-treated patients, suggesting that DOX was selectively more toxic to cardiac muscle cells when compared with other cell types. 14 DOX-induced cardiac toxicity is related to cardiomyocyte apoptosis and necrosis, other forms of cell death such as autophagy and senescence/aging can also be related. 15
Numerous studies show that DOX-induced cardiomyopathy is principally due to increased production of reactive oxygen species (ROS), inflammation, and apoptotic cell death of myocardial cells. 16,17 Apoptosis finally causes cardiomyopathy and congestive heart failure through a systematic reduction in the amount of cardiomyocytes in myocardium. 18 The target organelle of DOX toxicity in cardiomyocytes is mitochondria wherein DOX accumulates with time. 19,20 Sawyer et al. 16 demonstrated that myocardial impairment caused by anthracyclines may involve myocyte apoptosis. Anthracycline-induced myocyte apoptosis is probably mediated by oxidative free radical formation.
Oxidative damage to membrane lipids and other cellular components is believed to be a important factor in DOX toxicity. 3,21 DOX binds and intercalates DNA, inhibits topoisomerase II activity, generates free radicals, and covalently binds to DNA after reductive metabolism. 22 DOX reactions lead to the formation of ROS such as superoxide anion, hydrogen peroxide, and hydroxyl radicals. 23 Numerous studies have demonstrated that DOX administration caused oxidative damage to myocardial tissue. 24 Cellular redox status is in balance under normal physiological conditions. 25 DOX leads to myocardial damage via the formation of free oxygen radicals. 3 ROS start free radical-mediated chain reactions, resulting in the conversion of membrane unsaturated fatty acids into lipid peroxides, which lead to disturbed integrity of cell membrane and consequently to the tissue injury. 26 Antioxidant enzymes like superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) detoxified effectively free oxygen radicals under healthy situations. However, when there is an imbalance between the production of oxidants and the defense systems of the organism, the initiation of lipid peroxidation and change in intracellular homeostasis happened, as seen in DOX-induced cardiotoxicity. 27 ROS can damage myocytes owing to decreased levels of antioxidant enzymes such as SOD, CAT, and GSH-Px. 28
Dietary fish oil may prevent the development and progression of cardiac dysfunction and heart failure. Omega-3 (n-3) is an essential fatty acid (EFA) found in large amounts in fish oil. It contains eicosapentaenoic acid (EPA, C20:5n-3) and docosahexaenoic acid (DHA, C22:6n-3). α-Linolenic acid (ALA) is found in vegetable sources such as soybean and linseed oil. EPA, DHA, and ALA are categorized as n-3 EFAs. 29 Several studies have demonstrated apparent beneficial effects of fish n-3 fatty acids on the plasma level of triglycerides, 30 the levels of LDL cholesterol, 31 the frequencies of cardiac arrhythmias and sudden death, 32 the progression of atherosclerotic plaques, 33 mortality from heart disease, 34 and the risk of ischaemic heart disease. 35 Numerous studies have proposed that n-3 polyunsaturated fatty acids (PUFA) may prevent the development and progression of heart failure. 36–38 In addition, previous studies have demonstrated that fish n-3 fatty acids have antioxidants, anti-inflammatory, and antiapoptotic properties. 39,40
The present study was planned to investigate the protective effects of fish n-3 fatty acids pretreatment on DOX-induced acute cardiotoxicity including histopathological changes, apoptosis, and oxidative stress in rat model of cardiomyopathy.
Materials and methods
Animals and treatments
A total of 24 male Sprague-Dawley rats, weighing 300 ± 25 g and aged 16 weeks old, were used in this study. The animals were obtained from Trakya University Animal Care and Research Unit (Edirne, Turkey). Rats were fed on a standard rat chow and tap water ad libitum. In the windowless animal quarter, automatic temperature (22 ± 1°C) and lighting controls (light on at 7 a.m. and off at 7 p.m.: 12 h light/12 h dark cycle) were maintained. Humidity ranged from 50 to 55%. All animals received human care according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Sciences and published by the National Institutes of Health. The experimental study was approved by the Namik Kemal University Animal Ethical Committee (numbered as 2011/10).
Experimental design
The rats were randomly divided into the following three groups: (1) control, (2) DOX-treated, and (3) DOX treated with fish n-3 fatty acids. Each group contains eight animals.
Experimental procedures and sample collection
The rats in fish n-3 fatty acid-treated group were given 400 mg/kg fish n-3 fatty acids daily, for 30 days (New Life EFA S-1200, Eurocaps Limited, England), by intragastric intubation. 29,39,40 The fatty acid composition of the EFA S-1200 capsule (1200 mg) includes EPA (396 mg), DHA (264 mg), and other fatty acids (60 mg). The rats in control group were given 0.4 ml/kg saline by intragastric intubation. This application continued daily for a total of 30 days. To induce acute cardiotoxicity, single dose of DOX (Carlo Erba, Milan, Italy; 30 mg/kg body weight) was injected intraperitoneally and the rats were killed after 48 h. 41 No rats died during the experiment.
At the end of the experiment, the rats of all groups were anesthetized by intraperitoneal administration of 90 mg/kg of ketamine and 10 mg/kg of xylazine. All rats were killed under anesthesia and the heart tissues were evaluated for lipid peroxidation products, antioxidant enzymes, morphological appearance, and apoptosis.
Histopathological examination
Heart tissues were fixed in 10% neutral formalin and embedded in paraffin. Sections of 5 μm were obtained, deparaffinized, and stained with hematoxylin and eosin. The heart tissue was examined, evaluated, and photographed in random order under blindfold conditions with a standard light microscope (Nikon Optiphot 2, Nikon, Tokyo, Japan). The severity of changes was quantitated none (−) to severe (+++) based on the degree of cytoplasmic vacuolization, myocardial disorganization, and myofibrillar loss. The scoring system was as follows: (−) no damage, (+) mild, (++) moderate, and (+++) severe negative features.
Evaluation of cardiomyocyte apoptosis
Cardiomyocyte apoptosis was evaluated by terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay. The TUNEL method, which detects fragmentation of DNA in the nucleus during apoptotic cell death in situ, was employed using an apoptosis detection kit (TdT-Fragel™ DNA Fragmentation Detection Kit, Cat. No. QIA33, Calbiochem, Billerica, Millipore, USA). All reagents listed below are from the kit and were prepared following the manufacturer’s instructions. Heart tissue sections of 5-µm-thick were deparaffinized in xylene and rehydrated through a graded ethanol series as described previously. The sections were then incubated with 20 mg/ml proteinase K for 20 min and rinsed in Tris-buffered saline (TBS). Endogenous peroxidase activity was inhibited by incubation with 3% hydrogen peroxide. The sections were then incubated with equilibration buffer for 10–30 s and then with TdT-enzyme in a humidified atmosphere at 37°C for 90 min. The sections were subsequently put into prewarmed working strength stop/wash buffer at room temperature for 10 min and incubated with blocking buffer for 30 min. Each step was separated by thorough washes in TBS. Labeling was revealed using DAB, counter staining was performed using methyl green, and sections were dehydrated, cleared, and mounted. The average ratio of the total TUNEL-positive cardiomyocyte number was calculated from randomly selected 10 microscopic high-power fields for each rat in all the groups. This ratio represented the apoptotic index of the sample and was compared between groups.
Biochemical analysis
Determination of GSH-Px activity
Prior to dissection, heart tissues were rinsed with a phosphate-buffered saline (PBS) solution, pH 7.4, to remove any red blood cells and clots. Heart tissues were homogenized using a homogenizer in 10 ml of cold buffer (50 mM Tris-HCl, pH 7.5, 5 mM EDTA, and 1 mM DTT) per gram tissue. Tissue homogenates were centrifuged at 10,000g for 15 min at 4°C. The supernatant was removed for assay and stored at −80°C. Supernatant was centrifuged for 15 min at 10,000g to obtain postmitochondrial fraction. The GSH-Px activity was examined in the postmitochondrial fraction of these homogenates. The GSH-Px activity was determined spectrophotometrically using a commercially available assay kit obtained from Cayman Chemical Company (Ann Arbor, Michigan, USA, Cat. No. 703102). The GSH-Px activity was measured by the method of Paglia and Valentine. 42 Results were expressed as units per gram of tissue protein (U/g protein).
Determination of SOD activity
Prior to dissection, heart tissues were rinsed with PBS solution, pH 7.4, to remove any red blood cells and clots. Heart tissues were homogenized using a homogenizer in 10 ml of cold 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid buffer, pH 7.2, containing 1 mM ethylene glycol tetraacetic acid, 210 mM mannitol, and 70 mM sucrose per gram tissue. Tissue homogenates were centrifuged at 1500g for 5 min at 4°C. The supernatant was removed for assay and stored at −80°C. The supernatant was centrifuged for 15 min at 10,000g to obtain postmitochondrial fraction. The SOD activity was examined in the postmitochondrial fraction of these homogenates. The SOD activity was determined spectrophotometrically using the commercially available assay kit obtained from Cayman Chemical Company (Ann Arbor, Cat. No. 706002). The SOD activity was determined according to the method of Sun et al. 43 The SOD activity was expressed as units per milligram of tissue protein (U/mg protein).
Determination of MDA level
All tissues were weighed and homogenized with 0.15 M potassium chloride solution and 10% homogenates (w/v) of these tissues were prepared. Tissue homogenates were centrifuged in a cold centrifuge at 600g for 10 min at 4°C. Malondialdehyde (MDA), as an end point of lipid peroxidation, was calculated by detecting absorbance of thiobarbituric acid reactive substances at 532 nm. 44 The MDA levels were expressed as MDA nanomoles per milligram of protein.
Determination of protein
The bicinchoninic acid method was used to determine the amount of protein in samples. 45
Statistical analysis
All statistical analyses were performed using PASW® Statistics 18 for Windows (SPSS Inc., Chicago, IL, USA). All data were presented as median (min–max). Distribution of the groups was analyzed using the Shapiro–Wilk test. Because the number of rats in each group was eight, the results showed abnormal distribution, so the data were considered to be nonparametric. Differences between two groups were analyzed using the Mann–Whitney U test. p < 0.05 was considered statistically significant.
Results
Histopathological changes
Histopathological changes in the heart tissue of the control, DOX-treated, and DOX treated with fish n-3 fatty acids rats are reported in Table 1. On histopathological examination of heart tissues, the control group exhibited myocardium fibers with normal structure and regular morphology of myocardial cell (Figure 1(a)). On the other hand, there were severe cardiac damages including disorganization of myocardial muscle fibers, myofibrillar loss, and cardiotoxic myocardial fibers with cytoplasmic vacuoles in heart tissues of DOX-treated group (Figure 1(b)). Fish n-3 fatty acid-treated animals showed an improved histological appearance in the DOX-treated group (Figure 1(c)).
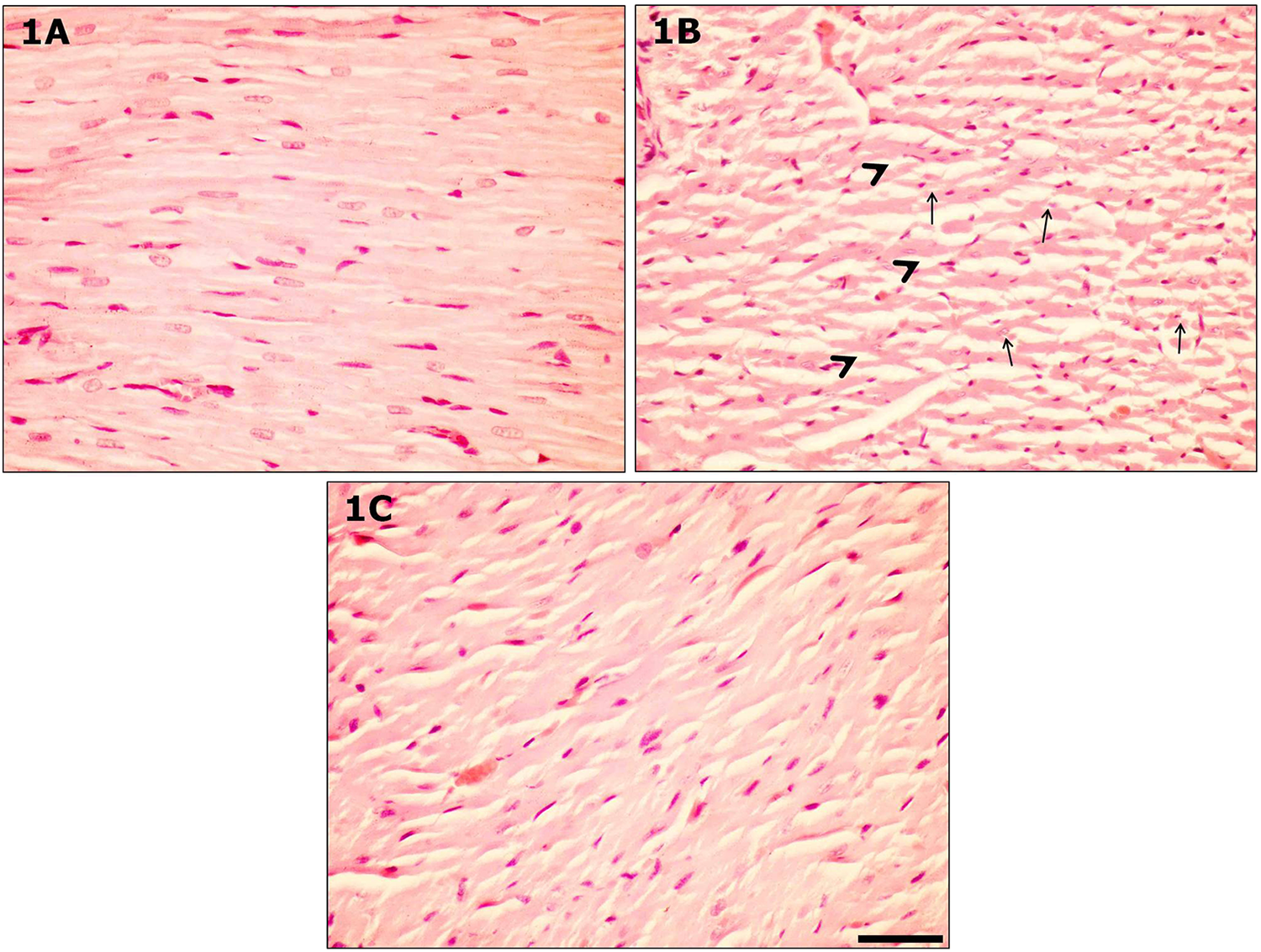
Histological section of the heart tissue in rat treated with DOX (b) shows severe lesions (myocardial fibers with cytoplasmic vacuoles, disorganization of myocardial muscle fibers, and myofibrillar loss) in comparison with DOX treated with fish n-3 fatty acids group (c) and control group (a). (Arrows: cytoplasmic vacuoles, arrowhead: myofibrillar loss; hematoxylin and eosin stain, magnification = ×40, scale bar = 50 μm.) DOX: doxorubicin; n-3: omega-3.
Cardiomyopathy scores in groups A, B, and C.a

DOX: doxorubicin; n-3: omega-3; Group A: control; Group B: DOX-treated group; Group C: DOX treated with fish n-3 fatty acids.
a The scoring system was as follows: (−) no damage, (+) mild, (++) moderate, and (+++) severe.
Evaluation of cardiomyocytes apoptosis
Apoptotic cells in the heart tissues were identified by TUNEL staining. In the control group, only a few TUNEL-positive cells were observed (Figure 2(a)). However, in the DOX-treated group, the amounts of TUNEL-positive cardiomyocytes were quantitatively higher (Figure 2(b)) compared to the control group. Pretreatment of fish n-3 fatty acids noticeably reduced the reactivity and the amount of TUNEL-positive cardiomyocytes (Figure 2(c)). Figure 3 shows the apoptotic index for cardiomyocytes in the control, DOX-treated, and DOX treated with fish n-3 fatty acids. The percentage of apoptotic cardiomyocytes was significantly increased by DOX (p < 0.001), but was diminished by fish n-3 fatty acid pretreatment (p < 0.001).

TUNEL detection of apoptotic cells in control (a), DOX-treated (b), and DOX treated with fish n-3 fatty acids (c) groups. Pretreatment of fish n-3 fatty acids markedly reduced the reactivity and the number of cardiomyocytes apoptosis. Nuclei were counterstained with methyl green. (Arrow: TUNEL positive cardiomyocytes, scale bar = 50 μm.) DOX: doxorubicin; n-3: omega-3; TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling.
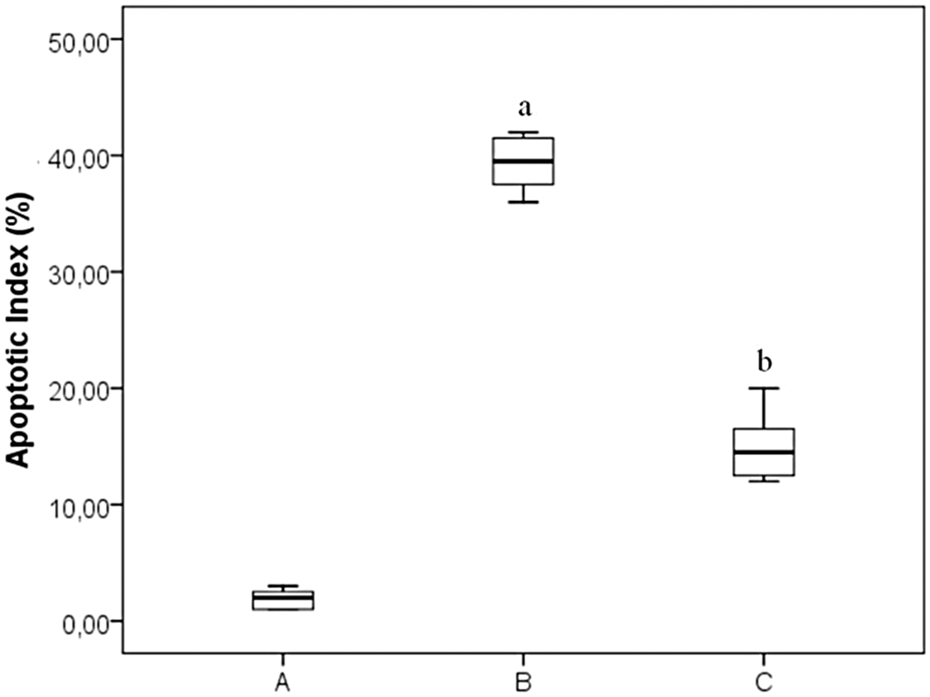
Quantitative analysis of TUNEL results. (A) Control, (B) DOX-treated, and (C) DOX-treated with fish n-3 fatty acids group. The apoptotic index (TUNEL-positive cell ratio) was significantly higher in DOX-treated group cardiomyocytes in comparison to control group. The apoptotic index was significantly decreased in the DOX-treated with fish n-3 fatty acids group when compared to DOX-treated group. ((a): p < 0.001 compared with control group; (b): p < 0.001 compared with DOX-treated group). DOX: doxorubicin; n-3: omega-3; TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling.
Biochemical results
The biochemical results of heart tissues are illustrated in Table 2. The activities of GSH-Px and SOD in heart tissues were lower in DOX-treated group than the control group (p < 0.05). Fish n-3 fatty acid pretreatment increased the activities of GSH-Px (p < 0.001) and SOD (p < 0.05) in DOX treated with fish n-3 fatty acids group compared to the DOX-treated group. The levels of MDA in the heart tissues were significantly increased in DOX-treated group in comparison with the control group (p < 0.05). DOX treated with fish n-3 fatty acids produced a significant decrease in MDA levels in comparison with DOX-treated group (p < 0.05).
The results of biochemical analysis (GSH-Px, SOD, and MDA) in groups A, B, and C in heart of rats.a
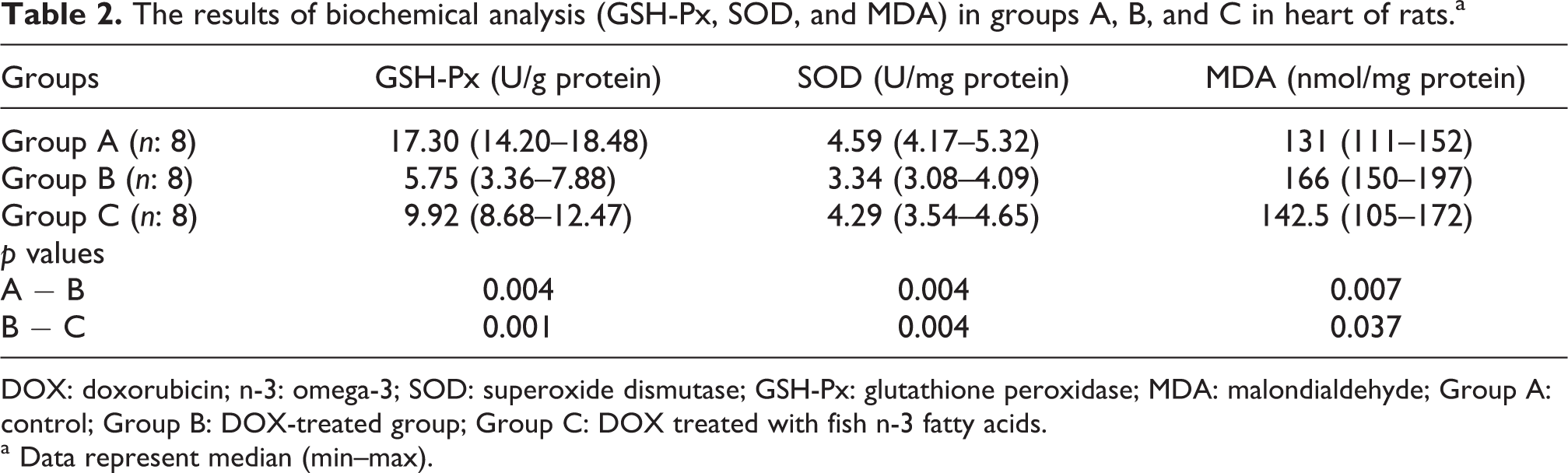
DOX: doxorubicin; n-3: omega-3; SOD: superoxide dismutase; GSH-Px: glutathione peroxidase; MDA: malondialdehyde; Group A: control; Group B: DOX-treated group; Group C: DOX treated with fish n-3 fatty acids.
a Data represent median (min–max).
Discussion
DOX is an effective anthracycline, anticancer agent with a wide spectrum of activity treating malignancies. 13 Anticancer therapy generally destroys the physiological homoeostasis during the cancer treatment in different organs. 46 Clinical usefulness of DOX is restricted by specific toxicities to cardiac tissues. 2 –4 Therefore, much attention has been focused lately on the protective effects of antioxidants and naturally occurring substances against DOX-induced cardiotoxicity. 47 DOX-induced cardiotoxicity may present as either acute or chronic cardiomyopathy. DOX-induced acute cardiotoxicity is occurring after high dose DOX administration, while DOX-induced chronic cardiotoxicity is dose dependent. 15
The objective of the present study was to examine the protective effect of fish n-3 fatty acids against DOX-induced acute cardiotoxicity by studying the changes in histological levels, the number of apoptotic cardiomyocyte, MDA levels, and activities of SOD and GSH-Px. Several studies have exhibited protective effects of some antioxidants against DOX-induced cardiotoxicity. 13,41,48 In this study, to evaluate the effects of fish n-3 fatty acids on DOX-induced acute cardiotoxicity, a single dose of DOX (30 mg/kg) induced severe acute cardiac toxicity, characterized by oxidant injury similar to the reports in the literature. 41 Treatment with DOX has been reported to cause cardiomyopathy in various animal studies. 12,47 DOX-induced myocardial damage was also confirmed by myocardial histopathological changes like those reported in other studies. 41,47
In this study, the rats in fish n-3 fatty acid-treated group were given 400 mg/kg fish n-3 fatty acids by intragastric intubation and this application continued daily for a total of 30 days. It has been reported that 0.6% of body weight (bw)/day dose of fish oil (8 weeks intragastrically) attenuated DOX-induced cardiac dysfunction. 38 Numerous studies have shown a beneficial effect of fish n-3 fatty acids on anthracycline-induced cardiotoxicity. Schjott et al. 49 declared that EPA and DHA decreased anthracycline-induced cardiotoxicity. Schjott et al. 49 concluded that EPA and DHA could be useful during epirubicin infusion. In a study, it was concluded that an oil containing γ-linolenic acid and dexrazoxane had cardioprotective properties against DOX-induced cardiotoxicity. 50 Teng et al. 38 observed that cardiac function and histological changes greatly improved after treatment with n-3 PUFA. Some hypotheses have been suggested to describe the mechanism responsible for the beneficial effect of fish n-3 fatty acids. It could be mediated by the replacement of anthracycline-peroxidized fatty acids in lipid including structures such as membranes, 50,51 or by alterations in eicosanoid metabolic pathways, 49,52 or by restoration of an imbalance of the cytokine network. 38 In this study, it was observed that histological changes, antioxidant status, and apoptosis markedly reduced with fish n-3 fatty acid treatment. Therefore, we speculate that fish n-3 fatty acids reduce DOX-induced acute cardiotoxicity, the effects that might be associated with antioxidant and antiapoptotic properties.
In this study, we showed that DOX-induced severe morphological changes in heart tissues. Disorganization of myocardial muscle fibers, myofibrillar loss, and cardiotoxic myocardial fibers with cytoplasmic vacuoles were seen. These results corroborate the results of previous studies, putting forward that DOX treatment causes important morphological changes in heart tissues. 13,41 Frequent DOX administration has been demonstrated to cause cardiomyopathic changes in patients 53 and in a variety of animal models. 9 In numerous studies, disorganization of myocardial histology, loss of striation, severe disruption of mitochondrial fine structure, picnotic nuclei, edema, increase in collagen content around vessels, fibrosis around arteries, and an increase in inflammatory cells have been seen in DOX-treated animals. 6,13,48 Santos et al. 54 demonstrated that DOX administration led to extensive vacuolization in the cytoplasm of myocardial cells. Saad et al. 55 showed that acute DOX-induced cardiac damage and also observed marked edema, disorganized myocardial fibers, and necrosis. Myofibrillar loss is a marker for DOX cardiotoxicity. 56 In the present study, the histopathological findings verified myocardial lesions such as cell vacuolization and loss of the myofibrils were obvious in DOX-treated rats. Fish n-3 fatty acid-treated rats showed an improved histological appearance in the DOX treated with fish n-3 fatty acids group.
MDA is a secondary lipid peroxidation product, and it can be used as an indicator of cell membrane injury. 57 SOD is a metalloenzyme that catalyzes the dismutation of the superoxide anion to molecular oxygen and hydrogen peroxide and thus form a crucial part of the cellular antioxidant defense mechanism. 58 GSH-Px catalyzes the reduction of hydroperoxides, containing hydrogen peroxide, by reduced glutathione and functions to protect the cell from oxidative damage. 59 Antioxidants are molecules that can prevent or reduce the extent of oxidative destruction of biomolecules. 60 The amount of SOD and GSH-Px present in cellular and extracellular environments is critical for the protection of diseases connected to oxidative stress. Oxidative stress can directly cause cell damage, cell apoptosis, and autophagy. 61,62 Under healthy situations, the oxidant–antioxidant system is in balance, but when there is an imbalance, tissue damage can appear. Anthracyclines are causative factors of the degeneration and may alter the oxidant–antioxidant status to favor oxidants. 48 In this study, acute administration of DOX caused an increase in MDA levels and a decrease in SOD and GSH-Px enzyme activities as compared to control group. In addition, DOX treated with fish n-3 fatty acid groups induced a significant decrease in MDA levels and an increase in SOD and GSH-Px activities compared to DOX-treated group. It is well known that DOX increases oxygen free radicals and causes lipid peroxidation in the myocardium. 24,28 However, our previous study exhibited that acute DOX administration caused oxidative stress in heart tissue as indicated in the literature. 41 Numerous studies demonstrated that DOX significantly decreased SOD and GSH-Px activities 63 and increased MDA levels in myocardial tissue. 6,63,64 Our results are in agreement with literature data on acute cardiotoxicity after DOX administration. 41,47,55,64 The toxicity of DOX is believed to be mediated through several mechanisms including free lipid peroxidation, radical formation, mitochondrial damage, and iron-dependent oxidative damage to biological macromolecules and membrane. 46 Increased formation of oxygen free radicals and superoxide damage cells and their organelles by lipid peroxidation and extensive oxidation of cellular components during DOX-induced cardiotoxicity. This process explains the pathological picture of DOX cardiotoxicity characterized by disruption of heart mitochondrial and sarcoplasmic reticular formation in myocardial compartments. 3,28 Free radical scavengers and iron chelators might supply cardiac protection by preventing the formation of the exceedingly reactive hydroxyl radical and by scavenging radicals that have been formed. 6 Fish n-3 fatty acids have been demonstrated to possess antioxidants, anti-inflammatory, and antiapoptotic properties. 39,40 Previous studies have demonstrated that fish n-3 fatty acids exhibit antioxidant properties in various oxidant situations that lead to tissue damage. 65,66 It was demonstrated that fish n-3 fatty acid treatment led to a decrease in lipid peroxidation in rat prefrontal cortex ischaemia–reperfusion injury. 66 Another study showed that increased SOD and GSH-Px enzyme activities and decreased MDA levels were detected in the rats administered n-3 fatty acids, while exposed to formaldehyde in prefrontal cortex. 39 An increase in free radical formation has been reported in DOX-induced cardiotoxicity, 3 therefore a contribution of indirect scavenger action of fish n-3 fatty acids in preventing DOX-induced acute cardiotoxicity seems to be probable. Our study shows that prophylactic application of fish n-3 fatty acids protects heart tissue effectively from DOX-induced oxidative injury. The treatment with fish n-3 fatty acids prevented lipid peroxidation and oxidative stress induced after acute DOX administration in the myocardium. In addition, the present study with fish n-3 fatty acid pretreatment resulted in decreasing the MDA levels. We thought that fish n-3 fatty acids prevented oxidant injury in a different way, in addition to its scavenger action. The activities of antioxidant enzymes were increased in fish n-3 fatty acid-treated rats, which means that fish n-3 fatty acids could elevate the antioxidant enzymes as well as their scavenging effect.
Previous studies have reported that apoptotic cell death is related to oxidative stress, which results from either an overproduction of ROS or a decrease in the antioxidant status. 67 DOX is a cytotoxic drug known to cause cardiac damage via several mechanisms, containing free radical generation, apoptosis, and autophagy. Cardiomyocyte apoptosis is one of the crucial events in DOX-induced heart failure. 61,62,68 The anthracycline daunorubicin treatment in myocytes leads to apoptotic cell death, as characterized by caspase activation and internucleosomal DNA degradation. 16 Cardiomyocyte apoptosis has extensive implications, particularly with regard to myocardial function, because loss of cardiomyocytes could begin or aggravate heart failure. 69 The anthracycline daunorubicin-induced myocyte apoptosis is mediated by oxy radical chemistry. 16 In our study, the number of cardiomyocytes apoptosis was quantitatively higher in the DOX-treated group and pretreatment of fish n-3 fatty acids noticeably reduced the reactivity and the number of cardiomyocytes apoptosis. In a study, transgenic mice after DOX injection demonstrated severe myocardial injury and a significant increase in TUNEL-positive cardiomyocytes, thus leading to severe cardiac dysfunction and increased mortality in transgenic mice. Zhang et al. 70 suggested that extensive apoptosis might be the major contributor to the decreased survival and cardiac dysfunction observed in transgenic mice after DOX injury.
Cardiac dysfunction is the most severe side effect of DOX therapy. Previous studies demonstrate that cardiomyocyte death through apoptosis, necrosis, and other forms is a primary contributor to the development of DOX-induced cardiomyopathy. 15 Our study suggested that the mechanism of the antiapoptotic and protective effect of fish n-3 fatty acids against acute DOX injury may include direct free radical scavenging activity. From previous studies, fish n-3 fatty acids consumption is known to be associated with heart failure. 36 –38 It has been reported that γ-linolenic acid had cardioprotective properties against DOX-induced cardiotoxicity. 50 Some hypotheses have been suggested to explain the mechanism responsible for the beneficial effect of fish n-3 fatty acids. Fish n-3 fatty acids could be mediated by the replacement of anthracycline-peroxidized fatty acids in lipid-containing structures such as membranes, 50,51 by recovery of an imbalance of the cytokine network 38 or by alterations in eicosanoid metabolic pathways. 49,52 Recent studies have shown that dietary supplementation with n-3 PUFA from fish oil have the potential to attenuate the detrimental effect of a number of cardiac dysfunctions. 36 –38
In conclusion, our data exhibit that fish n-3 fatty acid pretreatment protects against DOX-induced acute cardiotoxicity in rat model of cardiomyopathy. This is a result of the antioxidant and antiapoptotic properties of fish n-3 fatty acids.
Footnotes
Authors’ Note
This study was presented at the 4th International Symposium of Clinical and Applied Anatomy (4th ISCAA), June 28—July 1, 2012, Ankara, Turkey.
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This study was supported by Namik Kemal University Scientific Research Projects Unit (Project Number: NKUBAP.00.20.AR.11.05).
