Abstract
Background:
Isoflurane, one of the most commonly used inhalational anesthetics, is usually used in surgery patients and often causes long-term learning and memory impairment. The aim of this study was to explore the role of microRNA-204-5p (miR-204-5p) in isoflurane-induced learning and memory impairment in rats.
Methods:
The Morris Water Maze (MWM) test was used to estimate the spatial learning and memory abilities of laboratory rats. Enzyme-linked immunosorbent assay (ELISA) was used to determine interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor-α (TNF-α) concentrations in the hippocampal tissues. The expression level of miR-204-5p was determined by using quantitative reverse transcription polymerase chain reaction (qRT-PCR). The potential target genes of miR-204-5p were predicted and verified by the TargetScan and dual-luciferase reporter assay, respectively.
Results:
Isoflurane-induced rats showed significantly higher neurological function scores, higher escape latency and shorter time spent in the original quadrant. Isoflurane could significantly induce neuroinflammation, and the expression of miR-204-5p was increased in the hippocampal tissue of rats exposed to isoflurane. Moreover, downregulation of miR-204-5p attenuated the effect of isoflurane treatment on the escape latency and the time in the original quadrant, and inflammatory cytokines level was downregulated by inhibiting the expression of miR-204-5p. EphB2 was verified as a direct target gene of miR-204-5p.
Conclusion:
Downregulated miR-204-5p exerts protective effects against isoflurane-induced learning and memory impairment via targeting EphB2 and inhibiting neuroinflammation. MiR-204-5p could serve as a potential therapeutic target for the lightening of cognitive dysfunction induced by isoflurane.
Introduction
MicroRNAs (miRNAs/miRs), firstly discovered in the early 19th by Lee et al., 1 are single-stranded and non-coding RNA species consisting of approximately 18–25 nucleotides. MiRNAs can serve as important regulators of post-transcriptional gene expression. 2 Abnormal expression of miRNAs has been confirmed to be related to the occurrence of various diseases such as the nervous system, autoimmune system, and cardiovascular system diseases. MiRNAs are also involved in the regulation of cell growth, proliferation, differentiation, and degradation, the study of these molecules would enable us to better understand the pathogenesis of diseases. 3 The dysregulation of miRNAs has been reported to play a role in the protective mechanism of several neurological diseases.4–7 Recently, miR-204-5p is reported to play a vital role in neurodegenerative diseases, and involved in the occurrence and development of Parkinson’s disease. 8 In addition, in a study of sensorineural hearing loss (SNHL), miR-204-5p is also suggested to be involved in the functional recovery of impaired spiral ganglion neurons. All studies indicated the neuroprotective role of miR-204-5p. However, the role of miR-204-5p in neurological damage caused by isoflurane has not been studied.
Isoflurane is currently a widely used inhaled anesthetic in clinical applications, and its induced cognitive dysfunction is a common complication after major surgery. 9 The typical symptoms are their impairment in learning, writing and reading.10,11 Isoflurane can also cause a variety of pathophysiological processes, such as neuroinflammation, 12 excitotoxicity, 13 mitochondrial dysfunction, 14 and oxidative stress. 15 For example, Wang et al. showed that isoflurane-induced age-related hippocampal neuroinflammation via NLRP3 inflammasome activation. 16 It is worth noting that early isoflurane exposure may cause persistent learning deficits and cognitive dysfunction in children and rodents. 17 However, the pathogenesis of neurological damage caused by isoflurane is not fully understood. Herein, it is an increasingly urgent duty to understand the pathogenesis of isoflurane-induced neurological damage and discover candidate drugs for neuroprotection.
Here, the current study results indicated that miR-204-5p was upregulated in isoflurane-treated rats, and we hypothesized that downregulated miR-204-5p had a neuroprotective effect on isoflurane-induced cognitive dysfunction in rats. Moreover, it is predicted that the 3′-UTR of ephrin type B receptor 2 (EphB2) is the direct target of miR-204-5p. The down-regulated miR-204-5p may play a protective role in isoflurane-induced learning and memory impairment by targeting EphB2 and inhibiting neuroinflammation.
Materials and methods
Animals
This animal research was authorized by the Ethics Committee of the Experimental Animal Center of Jinan Third People’s Hospital Hospital (Ethical Approval Number: 201719). Animals were fed under the principles outlined in the Guide for the Care and Use of Laboratory Animals of the Institute for Laboratory Animal Research. Sixty-four male Sprague-Dawley rats (Age, 1 week; Weight, 18–22 g) were obtained from the Animal Center of Chinese Academy of Sciences. All rats were fed in a regulated environment (24 ± 2°C, 45–65% humidity, and a 12 h light/dark cycle) with free access to food and water.
Experimental groups
All rats excluded control group was exposed to 0.75% isoflurane for 6 h according to the previous study. 18 In brief, rats were anesthetized in a preheated, humidified anesthetic chamber with 0.75% isoflurane for 6 h, and the gas composition of the anesthetic chamber was monitored with the application of a calibrated Datex Capnomac Ultima (Datex Instrumentarium Corp., Helsinki, Finland).
The rats were randomly divided into four groups (n = 16 in each group): i, sham control group, rats were exposed in regular air without contact with isoflurane at any time; ii, isoflurane group, rats were exposed to 0.75% isoflurane for 6 h; iii, isoflurane + miR-NC group, rats were injected miR-204-5p negative control antagomir prior to isoflurane exposure; iv, isoflurane + miR-antagomir group, rats were injected miR-204-5p antagomir prior to isoflurane exposure. After experiments, eight rats in each group were sacrificed, and the hippocampus tissues were collected for qRT-PCR. The remaining rats were fed until day 14 (P14), and used for the following neurological function score and Morris water maze test.
Estimation of neurological function score
Rats were fed until day 14 (P14). To assess the neurological function of rats, we investigated the balance, touch, vision, abnormal behavior, muscle mass, sensation and motion of rats, and estimated using a modified Neurological Severity Score (mNSS). Neurological function is scored between 0 and 18, with 0 indicating no deficits and 18 indicating maximum deficits.
Morris water maze test
Rats were fed until day 14 (P14). To assess the learning and memory function of rats, a Morris water maze test was performed using a method earlier described. 19 In brief, the rats in each group were forced to perform the hidden-platform training using a pool (80 cm height and 120 cm diameter) for five consecutive days with four trials daily. Four quadrants were created in the apparatus, and the platform was depth at 30 cm under the water. The releasing position was selected randomly, and animals were placed into the pool facing the wall of the pool. If they failed to find the submerged platform within 120 s of swimming, the rats will be put on the platform passively and stayed for 15 s. Then we removed the platform on the 6th day, and a probe trial was performed. The rats were allowed to swim freely in the pool for 60 s and the escape latency and time in the original quadrant of rats were recorded and analyzed by a video imaging analysis system.
Quantitative real-time PCR (qRT-PCR)
RNA was separated and extracted from the hippocampus tissue samples using TRIzol reagent (Cat no. 15596018; Invitrogen; Thermo Fisher Scientific, Inc.) according to the experimental instructions. A miScript RT kit (Cat no. 218161; Qiagen GmbH) was used for reverse transcription. To investigate miR-204-5p expression levels, the qRT-PCR assay was performed using SYBR green I Master Mix kit (Cat no. 4385610; Invitrogen; Thermo Fisher Scientific, Inc.). Using the gene sequence database GenBank and the miR database BASE, the primer sequences of miR-204-5p and U6 were designed by using Primer 5.0 primer design software and the sequences were recorded in Table 1. The cycle conditions were as follows: initial denaturation at 95°C for 5 min; 30 cycles of 95°C for 30 s, 60°C for 30 s and 72°C for 20 s; and a final extension at 72°C for 10 min. U6 was used as an endogenous control quantification of the relative expression of miR-204-5p which was calculated by using the 2−ΔΔCt method.
Primer sequences information used to amplify miR-204-5p and U6.
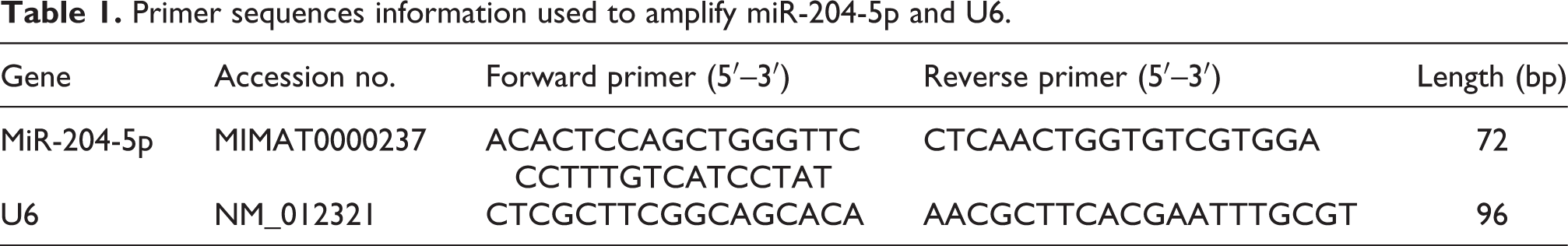
Enzyme-linked immunosorbent assay (ELISA)
Levels of IL-6 (Cat no. E02I0006), TNF-α (Cat no. E02T0008), and IL-1β (Cat no. E02I0010) in the hippocampus tissues were detected by the specific enzyme-linked immunosorbent (ELISA) kits (Blue Gene Biotech company, Shanghai, China) according to the manufacturer’s recommendations.
Dual-luciferase reporter assay
Using TargetScan 7.2 to predict candidate target gene of miR-204-5p, the 3′-untranslated region (3′-UTR) of EphB2 was found to have the complementary sequence of miR-204-5p as shown in Figure 5(A). To evaluate their interaction, a dual-luciferase reporter assay was performed as mentioned in previously reporting with modified. 20 In short, the HEK293T cell was co-transfected with wild-type (WT) or mutant-type (MT) EphB2 3′-untranslated region luciferase vector together with miR-204-5p mimic, miR-204-5p inhibitor or their negative control (miR-NC) using Lipofectamine 3000 Reagent (Invitrogen). The transfection lasted for 24 h, then the luciferase activity of the cells in each group was assessed by a luciferase assay kit (Cat no E1550; Promega, Madison, WI, USA). Experiments were run at least three times.
Statistical analysis
GraphPad Prism 7.04 software was used for analyzing all statistical data. Data were reported as mean and SD. Student’s t-test and one-way ANOVA analysis were applied for the significance test of differences between two groups or among multiple groups. P < 0.05 was considered to be statistically significant.
Results
Isoflurane-induced cognitive dysfunction in rats
The neurological examination and Morris water maze test were performed to assess the effect of isoflurane on neurological impairment in rats. As shown in Figure 1(A), the neurological function score in isoflurane-induced rats was significantly higher than that in the control group (P < 0.001). Compared with rats of the control group, the escape latency of rats during Morris water maze tests was significantly prolonged in the isoflurane-treated rat group (P < 0.001, Figure 1(B)), showing that the spatial learning ability of the rats was impaired. The duration of staying in the original safe-platform quadrant was significantly decreased in isoflurane-induced group compared with the control group (P < 0.001, Figure 1(C)), illustrating an impairment in the memory ability of the rats.
The effect of isoflurane on neurological damage in rats. (
Effects of isoflurane on neuroinflammation
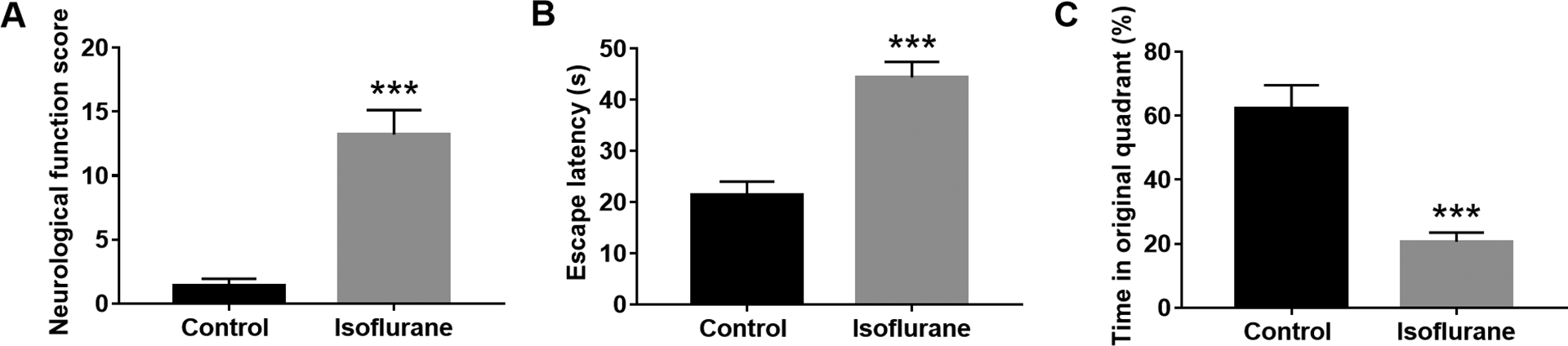
The isoflurane-induced learning and memory impairment is often accompanied by an inflammatory response. 12 The expression levels of IL-6, IL-1β and TNF-α were measured to analyze the role of neuroinflammation in cognitive handicap. The result showed that, in the hippocampus of rats, the protein levels of IL-6, TNF-α, and IL-1β were increased significantly after exposure to isoflurane (P < 0.001, Figure 2).
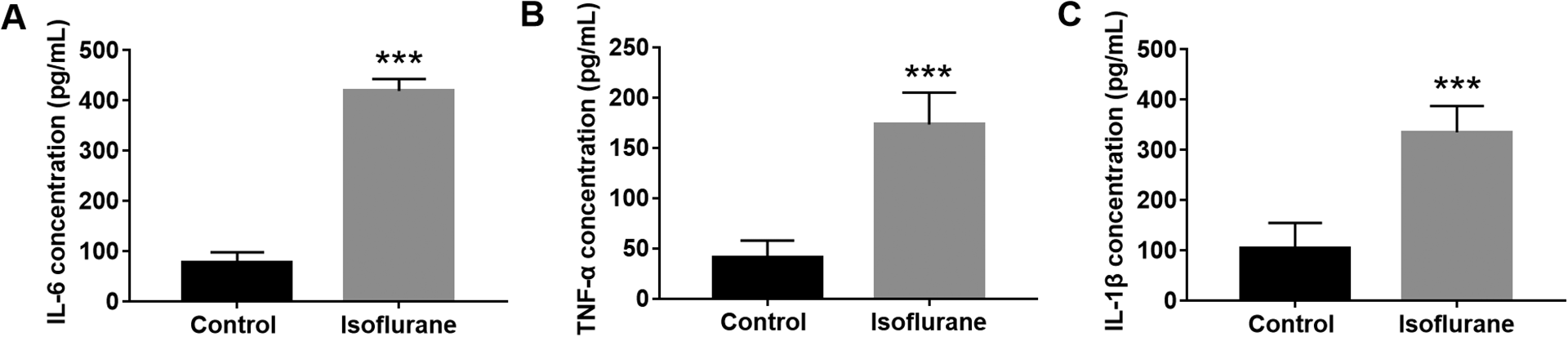
The concentration of IL-6 (
Dysregulation of miR-204-5p in isoflurane-induced rats
The expression level of miR-204-5p was detected by qRT-PCR assay in the hippocampus of both the control group and isoflurane group. It was found that the expression of miR-204-5p was upregulated significantly in rats exposed to isoflurane compared with the control group (P < 0.01, Figure 3), indicating that the upregulated miR-204-5p might be related to the neurological function impairment induced by isoflurane.

The expression of miR-204-5p in isoflurane-treated rats. Compared with the control group, the expression level of mir-204-5p in the hippocampus of rats treated with isoflurane increased significantly. ***P < 0.001.
miR-204-5p attenuated the cognitive dysfunction and neuroinflammation induced by isoflurane
Based on the up-regulated expression of miR-204-5p in rats exposed to isoflurane, the effects of miR-204-5p on the learning and memory impairment and neuroinflammation induced by isoflurane were further explored by regulating miR-204-5p expression in vivo. The expression of miR-204-5p increased significantly after isoflurane treatment, while miR-204-5p antagomir could significantly reduce the expression of miR-204-5p (Figure 4(A), F = 9.893, P < 0.001). As shown in Figure 4(B), the down-regulated miR-204-5p significantly reduced the neurological function score in isoflurane-induced group (F = 28.07, P < 0.001). Moreover, downregulation of miR-204-5p can significantly attenuate the effect of isoflurane treatment on the escape latency (Figure 4(C), F = 45.52, P < 0.001) and the time in the original quadrant (Figure 4(D), F = 80.31, P < 0.001).

The effect of miR-204-5p on isoflurane-induced neurological impairment. (
To further investigate the effect of miR-204-5p on isoflurane-induced neurological impairment in rats, the inflammatory cytokines (IL-6, TNF-α, and IL-1β) levels in the hippocampus were performed by ELISA. The expression level of inflammatory cytokines was increased significantly after inducing by isoflurane. After miR-204-5p antagomir transfection, the levels of IL-6 (F = 27.51, P < 0.001), TNF-α (F = 18.6, P < 0.001)), and IL-1β (F = 20.44, P < 0.001)) induced by isoflurane were all downregulated significantly in the hippocampus of rats (Figure 4(E)).
EphB2 is the target gene of miR-204-5p
EphB2 was predicted to be the putative binding target of miR-204-5p via Targetscan7.2 (Figure 5(A)). The relative luciferase activity was performed by dual-luciferase reporter assay to confirm the relation of miR-204-5p and EphB2. The miR-204-5p inhibitor significantly enhanced the luciferase activity of EphB2-Wt vector, while the activity of EphB2-Mut reporter vectors had little change. Meanwhile, miR-204-5p mimic significantly decreased the luciferase activity of EphB2 wildtype luciferase reporter vector in cells compared to control group (Figure 5(B), F = 77.05, P < 0.001), while had no effect on EphB2 mutant luciferase reporter vector. The above results indicated the binding ability between miR-204-5p and EphB2.

EphB2 is a functional target of miR-204-5p. (
Discussion
Isoflurane-induced cognitive dysfunction has attracted more and more attention in recent studies. And miRNAs have shown the potential therapeutic effect on neurological diseases. In this study, we showed for the first time that miR-204-5p downregulation attenuated isoflurane-induced learning and memory impairment and neuroinflammation in neonatal rats. Moreover, the protective effect of miR-204-5p downregulation after isoflurane exposure might be achieved by targeting EphB2 and inhibiting neuroinflammation.
A growing number of studies have shown that regulation of miRNAs expression exhibits neuroprotective effects in several diseases involved nerve injuries, such as miR-15a/16-1, miR-214, 21 and miR-203. 22 These dysregulated miRNAs play a vital role in development of neuro-related diseases and its role in isoflurane-induced learning and memory impairment have attracted more and more attention for their therapeutic potential. For instance, Qi et al. demonstrated that vitexin had protective effects against isoflurane-induced neurotoxicity by inhibiting the expression of miR-409. 23 Work by Cao et al. demonstrated that isoflurane preconditioning-induced neuroprotection may involve increased expression of miR-203. 22 Based on previous studies, we established isoflurane-induced rat nerve injury model and evaluated the rat’s neurological function. 18 Experimental results demonstrated that isoflurane treatment caused learning and memory impairment in rats, implying that the rat nerve injury model induced by isoflurane was successfully established. Notably, it was found that miR-204-5p was upregulated by isoflurane treatment, demonstrated that miR-204-5p might be involved in the isoflurane-induced neurological impairment.
To further investigate the role of miR-204-5p in isoflurane-induced neurological function impairment, the level of miR-204-5p was regulated by transfection in vivo. It was found that miR-204-5p downregulation protected against isoflurane-induced neurological function impairment. Consistently, miR-204-5p has been reported to be involved in the mechanism of spatial learning and memory impairment caused by exposure to fluorine and aluminium during the embryonic stage, which was associated with the downregulation of the BDNF-TrkB pathway in the hippocampus. 24 Another study by Liu et al. recruited HT22 cells, and demonstrated that downregulation of miR-204-5p inhibited sevoflurane-induced neuron cell apoptosis and promoted cell proliferation. 25 Furthermore, inhibition of miR-204-5p alleviated sevoflurane-induced oxidative injuries in HT22 cells. 25 All evidence demonstrates the neuroprotective effect of miR-204-5p downregulation, which supported our present results. However, in the current study, the role of miR-204-5p in oxidative stress, neuron cell apoptosis, and autophagy was not investigated, which should be explored in future studies.
Accumulating evidence suggested that isoflurane-induced neuronal dysfunction and cognitive impairment were highly associated with neuroinflammation by elevating the expressions of pro-inflammatory cytokines.11,26 As Cao et al. reported that suppressing HIF-1α protein expression attenuated the neuroinflammation in the hippocampus by disturbing NF-κB signaling pathway activity and decreasing TNF-1α after 4-h isoflurane exposure. 11 In our study, we detected the protein levels of IL-6, TNF-α, and IL-1β in the hippocampus. The results showed that the expression level of inflammatory cytokines in the isoflurane-induced rats was significantly higher than that in the control group, indicating that neuroinflammation has a potential role in isoflurane-induced nerve injury. miR-204-5p has been widely reported to be involved in several diseases’ progression through regulating inflammatory response, such as PD, atherosclerosis, and sepsis.8,27,28 Therefore, we further explored the effect of miR-204-5p on the levels of inflammatory cytokines in isoflurane-treated rats. As expected, the downregulation of miR-204-5p significantly reversed the increase of IL-6, TNF-α and IL-1β protein levels induced by isoflurane. Herein, it is concluded that the neuroprotective effect of miR-204-5p antagomir on isoflurane-induced neurological impairment might be achieved through anti-inflammatory effects.
The EphB2 is a member of the membrane-bound Ephrin (Eph) family of receptor tyrosine kinases (RTKs). 29 It contains a hydrophobic transmembrane domain, a cytoplasmic kinase domain and an N-terminal branch involved in ligand binding. 30 EphB2 receptor is involved in synaptic development and maturation. 31 Accumulating evidence has indicated that EphB2 plays an important role in neuropsychiatric diseases, such as Alzheimer’s disease, 32 pain after peripheral nerve injury, 33 and anxiety. 31 For instance, Dou et al. suggested that EphB2 plays a critical role in synaptic development, and its potent activation by NAP suggests that ADNP may mediate synaptic development partly by activating EphB2. 34 Zhang et al. demonstrated that EphB2 was a critical factor that contributes to the vulnerability to stress and might be a potential target for the treatment of depression. 35 Additionally, a recent study validated that age-associated increase in miR-204 level and age-related loss of EphB2 might contribute to cognitive deficits by accelerating this deterioration. 36 In this study, bioinformatics analysis predicted the 3’-UTR of EphB2 to be a potential binding site for miR-204-5p, and a dual-luciferase reporter was performed to further confirm, which was consistent with previous findings. 3 Therefore, we deduced that downregulation of miR-204-5p might protect against isoflurane-induced learning and memory impairment via targeting EphB2. Consistently, in aged mice, EphB2 has been suggested to be essential for normal memory formation, which supported our hypothesis. 37 In future, further studies are needed to clarify the potential mechanism about the involvement of miR-204-5p in isoflurane-induced neurological impairment, and its potential role in neuron cell apoptosis, oxidative stress, and autophagy should also be examined.
Taken together, it is concluded that downregulation of miR-204-5p expression might play protective effects against isoflurane-induced learning and memory impairment via targeting EphB2 and inhibiting neuroinflammation. Therefore, miR-204-5p may serve as a potential therapeutic target for the treatment of cognitive dysfunction induced by isoflurane.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
