Abstract
Postoperative cognitive dysfunction, which is associated with a wide range of cognitive functions including working memory, long-term memory, information processing, attention, and cognitive flexibility, is a major clinical issue in geriatric surgical patients. The aim of the current study was to determine the protective role and possible mechanisms of salidroside against isoflurane-induced cognitive impairment. Sprague Dawley rats were randomly assigned to five groups and were treated with or without salidroside before isoflurane exposure. Open-field and fear conditioning tests were conducted to evaluate the cognitive function of the rats. Moreover, the hippocampus tissues were obtained for biochemical analysis. The results showed that the isoflurane anesthesia decreased the freezing time to context significantly at 48 h after the isoflurane exposure in the fear conditioning test. Salidroside could ameliorate isoflurane-induced cognitive dysfunction. Further analysis demonstrated salidroside markedly suppressed the release of tumor necrosis factor-α and interleukin-1β. Moreover, salidroside reversed the decreased activity of choline acetyltransferase, superoxide dismutase, glutathione peroxidase, and content of acetylcholine, as well as the increased activity of acetylcholine esterase and content of malondialdehyde in hippocampal tissue of isoflurane-exposed rats. According to the results, we concluded that that salidroside has a protective effect against isoflurane-induced cognitive dysfunction by inhibiting excessive inflammatory responses, decreasing oxidative stress, and regulating the cholinergic system.
Introduction
Postoperative cognitive dysfunction (POCD), which is associated with a wide range of cognitive functions including working memory, long-term memory, information processing, attention, and cognitive flexibility, is a major clinical issue in geriatric surgical patients. 1,2 It is self-limiting in most patients, but in some patients, it is long-term or even permanent. 3 Isoflurane, a common inhalation anesthetic, has been postulated as a potential cause of POCD, however, its upstream mechanism remains elusive. 4,5
Recently, neuroinflammation has come to be recognized as an important underlying mechanism in general anesthetics-induced cognitive impairments. 6,7 Animal studies showed that anesthesia with isoflurane in neonatal mice could result in excessive expression of pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-1β in the hippocampus, which may contribute to cognitive impairment. 8 Lin et al. 9 have also demonstrated that increased expression of inflammatory cytokines and reduced neuronal density in hippocampus is probably involved in the isoflurane-induced cognitive impairment. In addition, the oxidative stress has been implicated in many aspects of neurodegenerative diseases, including Alzheimer’s disease (AD), POCD, and many other disorders. 10 Therefore, anti-inflammatory and antioxidant therapies might become the two most important strategies for the prevention and treatment of POCD.
Salidroside, the major active ingredient isolated from Rhodiola crenulata, possesses multiple pharmacological properties including antiaging, antifatigue, anti-oxidative, anti-cancer, and anti-inflammatory effects. 11 –13 Previous study demonstrated salidroside can inhibit beta amyloid (Aβ)-induced oxidative stress and inflammatory in rat hippocampus. 11 In vitro studies also revealed the ability of salidroside to reduce the cerebral ischemia-reperfusion injury in rat by its antioxidant activity. 12 These findings indicate a neuroprotective effect of salidroside. Therefore, we hypothesize salidroside protects against isoflurane-induced cognitive dysfunction via its anti-inflammatory and anti-oxidative property. The current study was carried out to test this possibility using rat pups.
Materials and methods
Fifty male Sprague Dawley rats (22 weeks old; 260–360 g) were obtained from the Animal Center of Suzhou Aiermaite Technology Co. Ltd. (SPF grade, Certificate No. SCXK20150004). Animals were housed 2 weeks to adapt to the environment. They were maintained in a standard condition with room temperature of 22 ± 1°C and 50 ± 10% humidity, under a 12-h light–dark cycle (light from 07:00–19:00), and ad libitum access to food and water. The study protocol was approved by Institutional Animal Care Committee of Binzhou Medical College and all procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals.
Isoflurane exposure and treatment regimen
Rats were randomly assigned to five groups: (1) control group; (2) isoflurane alone group; and (3) 60, 120, or 180 mg kg−1 salidroside + isoflurane group. The doses of salidroside were used based on our preliminary experiments. At 30 min before isoflurane exposure, the rats in salidroside + isoflurane group were administered orally with salidroside. Briefly, salidroside was first diluted with saline. Then, control and isoflurane alone group were administered with an equal volume of physiological saline. To induce general anesthesia, the rats were placed in a tightly sealed plastic chamber (length, 30 cm; width, 43 cm; and height, 14 cm) within a thermostatic bath. The concentration of isoflurane at 1.5% was flushed in a gas mixture of 30% O2 and 70% N2. Control experiments were performed in the same manner, except no isoflurane was added when flushing the chamber. The gas mixture was delivered at a flow rate of 2 L min−1. The concentrations of isoflurane, O2 and CO2 in the chamber were continuously monitored using a Capnomac ULTIMA monitor (Datex, Helsinki, Finland).
At 6 h after exposure, six rats from each group were euthanized by overdose Nembutal for hippocampal harvesting. Then the hippocampus was dissected for biochemical analysis. And the rest of rats were subjected to behavior study to determine their cognitive function after 48 h.
The hippocampal levels of IL-1β and TNF-α
The hippocampus was lysed in ice bath for 30 min. Then, the supernatant was collected after centrifugation (10,000 rpm, 4°C, 10 min). The levels of TNF-α and IL-1β were measured using enzyme-linked immunosorbent assay (ELISA) kits (Nanjing Jiancheng Co., Nanjing, China) according to the manufacturer’s instructions.
Quantification of AchE, ChAT, Ach, SOD, GSH-Px, and MDA
The homogenate from hippocampus was centrifuged and the supernatant was used for the assays of acetylcholine esterase (AchE), choline acetyltransferase (ChAT), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and malondialdehyde (MDA) levels, according to the manufacturer’s instructions, respectively (Nanjing Jiancheng Co., Nanjing, China). The contents of acetylcholine (Ach) in hippocampus were measured using an ELISA kit according to the manufacturer’s instructions.
Open-field test
At 48 h after isoflurane exposure, open-field test was performed to determine the emotional responses and exploration activity to a novel environment. 13 The test was carried out in a white opaque plastic chamber (40 cm × 40 cm × 40 cm). Each rat was gently placed in the center of the arena, then the movement was recorded for 5 min with a video tracking system. At the end of each test, the arena was carefully cleaned with 75% alcohol to avoid the presence of olfactory cues.
Fear conditioning
One hour after the open-field test, rats were subjected to fear conditioning test. Each rat was placed into a conditioning chamber wiped with 70% alcohol and allowed to explore for 3 min. Then a 30-s tone-foot shock (tone: 2000 Hz, 85 db) was delivered followed by a 2-s foot shock (0.7 mA). When it was finished, the mouse stayed in the chamber for another 30 s and then returned to the home cage. Twenty-four hours later, the rat was placed back to the chamber for 8 min in the absence of tone and shock. The amount of time with freezing behavior was recorded in an 8 s interval. Two hours later, the animal was placed in a test chamber that had different context and smell from the first test chamber and allowed to explore for 3 min. Then the training tone was delivered for an additional 3 min and the freezing behavior was scored. Freezing behavior was defined as the absence of all visible movement except for respiration, and it was recorded and expressed as the percentage of the observation period. At the end of each test, the chamber was carefully cleaned with 75% alcohol to avoid the presence of olfactory cues.
Statistical analysis
Statistical analysis was implemented using SPSS18.0 for windows. All data were reported as the mean ± SD, and differences between groups were evaluated by one-way analysis of variance followed by Tukey’s multiple range test. The level of statistical signification was set at p < 0.05.
Results
Salidroside attenuated the isoflurane-induced cognitive impairment in rats
Open-field test revealed that the ambulatory distances and time spent in the center among the five groups had no significant differences (Figure 1(a) and (b)), suggesting that all rats had no physical or emotional depression in a novel environment. The fear conditioning test was used to access the hippocampus-dependent memory. As shown in Figure 1(c), isoflurane-exposed rats displayed lower freezing time to context compared with control rats, which was reversed by chronic administration of salidroside. There was no significant difference in the freezing time to tone among the five groups (Figure 1(d)).
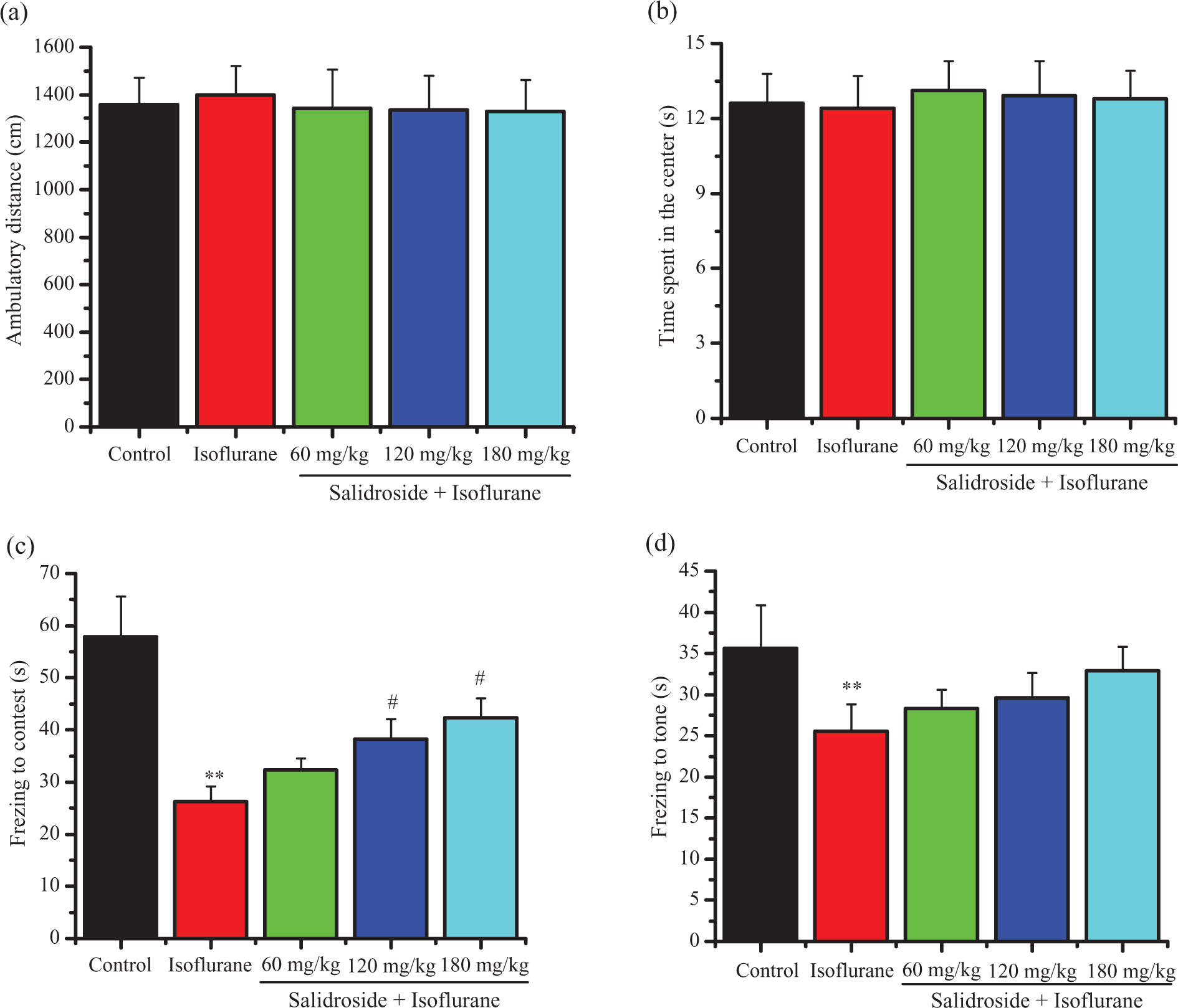
The effects of salidroside on the behaviors of isoflurane-exposed rats in the open field and fear conditioning tests. (a) Ambulatory distances in the open-field test; (b) time spent in the center in the open-field test; (c) freezing time to context in fear conditioning tests; and (d) freezing time to tone in fear conditioning tests. Data are presented as mean ± SD. **p < 0.01 versus control group; #p < 0.05, ##p < 0.01 versus isoflurane alone group. SD: standard deviation.
Salidroside down-regulates TNF-α and IL-1β levels in hippocampus
To determine the anti-inflammatory property of salidroside, we measured levels of inflammatory mediators including TNF-α and in hippocampus. As illustrated in Figure 2, the levels of TNF-α and IL-1β in isoflurane alone group were markedly increased in contrast with the control group, whereas salidroside significantly prevented the rise of both TNF-α and IL-1β in a dose-dependent manner.

Salidroside down-regulates (a) TNF-α and (b) IL-1β levels. Data are presented as mean ± SD. **p < 0.01 versus control group; #p < 0.05, ##p < 0.01 versus isoflurane alone group. ) TNF- α: tumor necrosis factor; IL-1β: interleukin-1 β
Effect of salidroside on the levels of GSH-Px, SOD, and MDA in hippocampus
To evaluate the effect of salidroside on isoflurane-induced oxidative stress, we detected the SOD activity, GSH-Px activity, and MDA content. As demonstrated in Figure 3, isoflurane exposure induced significant decreases of GSH-Px and SOD activities (Figure 3(a) and (b)) and increase of MDA content (Figure 3(c)). Administration of salidroside significantly increased the activity of GSH-Px and SOD and decreased the content of MDA.

Effect of salidroside on the levels of (a) GSH-Px, (b) SOD, and (c) MDA in hippocampus. Data are presented as mean ± SD. **p < 0.01 versus control group; #p < 0.05, ##p < 0.01 versus isoflurane alone group. GSH-Px: glutathione peroxidase; SOD: superoxide dismutase; MDA: malondialdehyde.
Effect of salidroside on the levels of Ach, AchE, and ChAT in hippocampus
Cholinergic system plays a crucial role in cognitive function, so we verified the effect of salidroside administration on Ach content, AchE, and ChAT activities in hippocampus. As shown in Figure 4, the isoflurane alone group showed a significant increase of AchE activity and decrease of Ach and ChAT levels in hippocampus, compared with control group, while these effects were significantly reversed after treatment with salidroside.

Effect of salidroside on the levels of (a) Ach, (b) AchE, and (c) ChAT in hippocampus. Data are presented as mean ± SD. ** < 0.01 versus control group; #p < 0.05, ##p < 0.01 versus isoflurane alone group. SD: standard deviation Ach: acetylcholineAchE: acetylcholine esterase; ChAT: choline acetyltransferase.
Discussion
This study demonstrates that salidroside offers a protective role against isoflurane-induced cognitive dysfunction. This effect was identified by the improvement in the performance in open-field and fear conditioning test. And the mechanisms are associated with inhibition of inflammation parameters and regulation of antioxidant enzymes.
Increasing studies have shown isoflurane anesthesia induced short-term and/or long-term cognitive impairments. 14,15 In our study, the rat had lower freezing time to context in the fear conditioning test after the isoflurane anesthesia, verifying that the isoflurane anesthesia leads to hippocampus-dependent cognitive impairment, whereas salidroside pretreatment attenuated isoflurane-induced cognitive impairment.
It has been demonstrated that the increase in proinflammatory cytokines in the hippocampus is an important risk factor in cognitive decline. 16,17 Considerable evidences also show that the release of proinflammatory cytokines within the hippocampus interferes with cognitive function and development of long-term potentiation. 18,19 Our data showed that isoflurane exposure elevated the levels of TNF-α and IL-1β in vivo and resulted in the induction of neuroinflammatory processes. However, the releases of these pro-inflammatory mediators were inhibited by salidroside administration.
Furthermore, oxidative stress in the brain is considered to be another important cause of cognitive impairment diseases. 20 Some enzymes, such as SOD and GSH-Px, provide cellular protection against damage from oxygen-derived free radicals. 21 SOD was regarded as the first line of the antioxidant defense system converts superoxide anion to hydrogen peroxide. It can be further metabolized by GSH-Px, which is considered as a second defensive line of oxidative stress. 22 The MDA is another well-known indicator of lipid peroxidation under oxidative stress. In our study, with MDA significantly increased, SOD and GSH-Px significantly reduced after salidroside pretreatment, suggesting lipid peroxidation in damage localization increased, it is also the result of increased free radicals. However, salidroside pretreatment ameliorate these abnormalities. These results suggest that amelioration of isoflurane-induced cognitive dysfunction by salidroside may be associated with the inhibition of hippocampal oxidative stress.
It is reported that cholinergic changes in the hippocampus are in relation to spatial learning and memory impairment. 23 Ach, a key transmitter in brain, plays crucial role in learning and memory processes. AchE is essential to normal function of nervous system, since it rapidly terminates the action of Ach released into the synapse. 24 ChAT, a cholinergic marker, is also involved in the synthesis of Ach. Li et al. 23 reported that the cognitive dysfunction is correlated with decline in ChAT level and loss of cholinergic neurons. Zhang et al. 11 reported salidroside significantly inhibited the increase of AChE activity and the decrease of ACh level induced by Aβ1–40. In the present study, isoflurane exposure resulted in a significant increased activity of AchE and decreased activity of ChAT, as well as a significant decrease in the content of Ach in hippocampus of rats, while salidroside pretreatment markedly attenuated these changes. These results suggest that the role of salidroside on the activity of ChAT, AchE and the level of Ach may be involved in the improvement of isoflurane-induced cognitive impairment.
In conclusion, we demonstrated that salidroside had a significant effect on improving the isoflurane-induced cognitive deficits in rats. This effect might be associated with the excellent abilities of salidroside on modulating inflammatory mediators, oxidative stress, and the cholinergic system. These results suggested that salidroside might have a potential to be a novel method for the clinical treatment of cognitive dysfunction.
Footnotes
Author contribution
Lisheng Liang and Zhikun Ma are co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
