Abstract
Diabetic cardiomyopathy (DCM) is one of the major complications of diabetes that causes mortality and morbidity in diabetic patients. Gastrodin (GSTD) is a bioactive phenolic glucoside component of an ancient Chinese herb Tianma (Gastrodia elata Bl.), which is widely used for cardiovascular and cerebrovascular diseases by ancient Chinese. Up to now, whether GSTD has a beneficial effect on DCM is unclear. Therefore, this study aimed to investigate the effect of GSTD on high glucose-induced injury in H9c2 rat cardiomyocytes and HL-1 mouse cardiomyocytes, and its underlying mechanisms. High glucose (33 mM) treatment caused cardiomyocyte toxicity, oxidative stress and apoptosis in both H9c2 and HL-1 cells. Under both normal (5.5 mM glucose) and high glucose conditions, GSTD showed protective effect against high glucose-induced cytotoxicity and promoted the nuclear translocation of Nrf2 in a concentration and time-dependent manner in H9c2 and HL-1 cells. Knockdown of Nrf2 expression using siRNA specifically targeting Nrf2 attenuated the protective effect of GSTD. Furthermore, GSTD promoted the nuclear translocation of Nrf2 via activating glycogen synthase kinse-3β (GSK-3β) signaling pathway. 4-benzyl, 2-methyl, 1, 2, 4-thiadiazolidine, 3, 5 dione (TDZD-8), an inhibitor of GSK-3β, inhibited the nuclear translocation of Nrf2 induced by GSTD, and attenuated the protective effect of GSTD as Nrf2 knockdown did. In summary, GSTD could protect against high glucose-induced cardiomyocyte toxicity via GSK-3β-mediated nuclear translocation of Nrf2.
Introduction
Diabetic cardiomyopathy (DCM) is one of the major complications of diabetes that causes mortality and morbidity in diabetic patients. 1 Previous studies have demonstrated that reactive oxygen species (ROS) plays a critical role in hyperglycemia-induced myocardial damage, which facilitates the development of myocardial infarction, cardiac hypertrophy, and even heart failure. Inhibition of ROS production is a potential therapeutic strategy for hyperglycemia-induced myocardial damage.2–5 Nuclear factor-erythroid 2-related factor (Nrf2) is a master regulator of redox status and cellular detoxification responses by inducing the expression of multiple downstream anti-oxidant genes, including heme oxygenase (HO-1), nicotinamide adenine dinucleotide phosphate-H (NADPH), quinineoxidoreductase-1 (NQO-1), glutamate-cysteine ligase catalytic subunit (Gclc), glutamate-cysteine ligase modifier subunit (Gclm), thioredoxin reductase 1 (Txnrd1) and peroxiredoxin 1 (Prdx1). 6 In unstressed cells, Nrf2 is maintained in the cytoplasm by binding to Kelch-like ECH-associated protein 1 (Keap1). In a stressed state, Nrf2 could be released to enter nucleus under the stimulation of ROS. 7 Moreover, Several signaling pathways reportedly regulate the nuclear translocation of Nrf2, such as p38/ mitogen-activated protein kinase (MAPK) signaling pathway,8,9 extracellular signal-regulated kinase (ERK) signaling pathway,10,11 glycogen synthase kinse-3 (GSK-3) signaling pathway.12,13 Recently, some studies highlighted the role of Nrf2 in the treatment of hyperglycemia-induced cardiac injury.14,15
The phenolic glucoside gastrodin (GSTD), a bioactive component of an ancient Chinese herb Tianma (Gastrodia elata Bl.), has been proven to possess anti-inflammatory16,17 and anti-oxidative18,19 properties. Recent studies showed that GSTD protected rat cardiomyocytes H9c2 from hypoxia-induced injury by up-regulation of microRNA-21. 20 GSTD could also protect cardiomyocytes from anoxia/reoxygenation injury via several mechanisms, including up-regulating 14-3-3η level, 20 inhibiting cell autophagy through the activation of mammalian target of rapamycin (mTOR) signaling, 21 and inhibiting mitochondrial apoptotic pathway. 22 However, whether GSTD possesses a protective effect on hyperglycemia-induced myocardial damage remains unclear.
In present study, we aimed to investigate the protective effect of GSTD on high glucose-induced cardiomyocyte toxicity and its underlying mechanisms. Our data showed that GSTD performed desirable cytoprotective effect on high glucose-treated H9c2 and HL-1 cells. GSTD protected against high glucose-induced cardiomyocyte toxicity via GSK-3β-mediated Nrf2 nuclear translocation. Thus, the present study provides a possibility for the use of GSTD in the prevention and treatment of DCM.
Materials and methods
Materials
GSTD (purity >98%) was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Dulbecco’s modified Eagle’s medium (DMEM), dimethyl sulfoxide (DMSO), fetal bovine serum (FBS) and trypsin-EDTA were purchased from Gibco (Grand Island, NY). Plates, antibiotics, and chemicals used for cell culture were purchased from Difco Laboratory (Detroit, MI, USA). 4-benzyl, 2-methyl, 1, 2, 4-thiadiazolidine, 3, 5 dione (TDZD-8) was purchased from MedChemExpress (New Jersey, USA).
Cell culture
H9c2 rat cardiomyocytes and HL-1 mouse cardiomyocytes obtained from American Type Culture Collection (ATCC, Rockville, MD, USA) were cultured in DMEM containing 1.5 g/l NaHCO3 and 5.5 mM glucose, supplemented with 10% FBS, 100 U/ml penicillin, and 100 U/ml streptomycin under 95% air/5% CO2 at 37°C. The culture medium was changed every 2 days, and cells were subcultured once a week. The medium was changed to serum-deprived medium, and cells were washed with serum-free DMEM 24 h before experiments and replanted in the 96-well plates. A phosphate buffer saline (PBS, pH 7.2) was used to adjust the cell number to 105/ml for various experiments and analyses.
3-(4, 5-Dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay
MTT assay was performed to examine cell viability. Briefly, H9c2 and HL-1 cells were incubated with 0.25 mg MTT/ml for 3 h at 37°C. The amount of MTT formazan product was determined by measuring absorbance at 570 nm (630 nm as a reference) using a microplate reader (Bio-Rad, Hercules, CA, USA). Cell viability was expressed as a percentage of control groups.
Lactate dehydrogenase (LDH) assay
The plasma membrane damage of H9c2 and HL-1 cells was evaluated by measuring LDH variation in culture supernatants. Culture supernatants (50 μl) were collected from each well. LDH activity (U/l) was analyzed by a colorimetric LDH assay kit (Sigma Chemical Co., St Louis, MO, USA).
Western blot
Total protein was obtained using radio-immunoprecipitation assay (RIPA) buffer (Beyotime, Shanghai, China) supplemented with the protease inhibitor cocktail (Sigma-Aldrich). In certain experiments, cytoplasmic and mitochondrial proteins were extracted using Cell Mitochondria Isolation Kit according to the manufacturer’s instructions, nuclear and cytoplasmic proteins were extracted using Nuclear and Cytoplasmic Protein Extraction Kit (Beyotime) according to the manufacturer’s instructions. Protein concentrations were measured using BCA protein assay kit (Beyotime). The protein samples were separated by SDS-PAGE and blotted onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). Next, the membranes were incubated at 4°C overnight with primary antibodies against p-p70S6k (Ser371), p70S6k, p-mTOR (Ser2448), mTOR, Cleaved Caspase-3, Cytochrome C, Nrf2, p-GSK-3β, GSK-3β, p-ERK1/2, ERK1/2, p-p38 MAPK, p38 MAPK (Cell Signaling Technology, Beverly, MA, USA), Bcl-2, Bax, β-Actin, COXⅣ, NF-κB, Lamin B1 (Proteintech, Wuhan, China), followed by horseradish peroxidase-conjugated secondary antibodies. The immunoblot signals were visualized by enhanced chemiluminescence detection kit (Amersham Pharmacia Biotec, Buckinghamshire, UK). The gray-scale values were quantified by Image-J software (NIH, Bethesda, MD, USA).
Detection of GSH and ROS
Cells were washed twice with PBS (pH 7.2), then were scraped from the plates and followed by homogenizing in PBS containing 0.5 mM butylated hydroxytoluene to prevent further oxidation of sample. The homogenate was centrifuged at 3000 g for 20 min at 4°C, and the supernatant was used for these assays according to the manufacturer’ s instructions. GSH or GSSG concentration (ng/mg protein) was quantified by a commercial colorimetric assay kit (OxisResearch, Portland, OR, USA). ROS level was determined using an oxidation sensitive dye, 2′, 7′-dichlorofluorescein diacetate (DCFH-DA) (Sigma, St. Louis, Missouri, USA). Cells were incubated with 50 μM dye for 30 min and washed with PBS. Fluorescence value was measured by using a fluorescence microplate reader (Molecular Devices, USA) at excitation and emission wavelengths of 485 and 530 nm, respectively. Relative fluorescence unit (RFU) was the difference in fluorescence values obtained at time 0 and 5 min. Results are expressed as RFU/mg protein.
Measurement of SOD and CAT activities
Total SOD and CAT activities were measured using a Total Superoxide Dismutase Assay Kit with NBT (Beyotime Biotechnology, Nanjing, Jiangsu, China) and a Catalase Assay Kit (Beyotime Biotechnology, Nanjing, Jiangsu, China), respectively. The assays were performed according to the instructions provided by the manufacturer.
Measurement of mitochondrial membrane potential (MMP)
The MMP was monitored using the Rh-123. After 24 h incubation in normal medium with or without treatment, the H9c2 and HL-1 cells were changed to serum-free medium containing 10 μM Rh-123 and incubated for 30 min at 37°C. The cells were then collected and the fluorescence intensity was analyzed by flow cytometry (FACScan, Becton Dickinson, USA).
Preparation of cytoplasmic and mitochondrial protein
For immunoblot analysis, the extraction and isolation of nuclear protein was performed using the Cell Mitochondria Isolation Kit (Beyotime, Shanghai, China). After treatment with indicated drugs, the cells were washed with PBS and digested with trypsin-EDTA Solution. Then the cells were centrifuged at room temperature for 5–10 min. Pre-cooled PBS was used to gently resuscitate the cell precipitate. A small number of cells were taken for counting. The remaining cells were centrifuged at 600 g at 4°C for 5 min to precipitate the cells and then removed the supernatant. Added 1–2.5 ml mitochondrial separation reagent or mitochondrial separation reagent with PMSF added before use to 20–50 million cells, gently suspended the cells, and placed in the ice bath for 10–15 min. Transferred the cell suspension to a suitably sized glass homogenizer and homogenized about 10–30 times. The homogenate was centrifuged at 600 g at 4°C for 10 min. Carefully transferred the supernatant to another tube and centrifuged at 11,000 g at 4°C for 10 min. Removed the supernatant carefully to another tube, the supernatant was cytoplasm and the precipitation was the isolated cell mitochondria.
Preparation of cytoplasmic and nuclear protein
For immunoblot analysis, the extraction and isolation of nuclear protein was performed using the Nuclear and Cytoplasmic Protein Extraction Kit (Beyotime, Shanghai, China). According to the manufacturer’s instructions, after treated with indicated drugs, the cells were washed with 1 ml ice-cold PBS, then collected and centrifuged for 5 min at 1200 rpm at 4°C and the pellet was dissolved with cytoplasmic protein extraction agent A supplemented with 1 mM phenylmethylsulfonyl fluoride (PMSF). After 5 s vortex, the tubes were incubated for 12 min on ice in order to promote lysis. Then the cytoplasmic protein extraction agent B was added, vortexed for 5 s and incubated on ice for 5 s. The samples were then centrifuged for 5 min at 15,000 g at 4°C and the supernatant was immediately frozen for further analysis. The pellet was resuspended in nuclear protein extraction agent supplemented with 1 mM PMSF. After 15 times vortex for 30 min and 15,000 g centrifuging for 10 min, the supernatants containing the nuclear extracts were obtained.
RNA extraction and real-time reverse transcriptase polymerase chain reaction (RT-qPCR)
After treatment, total RNA was isolated from H9c2 and HL-1 cells using RNA isolater Total RNA Extraction Reagent (Vazyme Biotech Co., Ltd) and reversely transcribed into cDNA using HiScript II Q Select RT SuperMix for qPCR (Vazyme Biotech Co., Ltd) according to the supplier’s protocol. Real-time PCR was performed with gene specific primers (Table 1, Thermo Fisher Scientific Inc.) with 18 s as internal control. The reactions were performed in an ABI Prism® 7900 High-Throughput RealTime PCR System (Applied Biosystems, Foster City, CA, USA). The comparative threshold cycle (CT) method was used for relative quantification of target gene expression, which was plotted as fold of control.
Primer sequences in quantitative RT-PCR.

RNA interference
siRNAs targeting Nrf2 were purchased from GenePharma (Shanghai, China). The sequences of these three siRNAs are as follows: siRNA-1, 5′-CCGAAUUACAGUGUCUUAA-3′; siRNA-2, 5′-CUCGCAUUGAUCCGAGAUA-3′; siRNA-3, 5′-CAAGGAGCAAUUCAAUGAA-3′. The transfection was performed using Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) in Opti-MEM medium (Gibco BRL, Grand Island, NY, USA) for 4–6 h. Then, the medium was replaced with fresh DMEM medium containing 10% FBS. After another 24 h culture, cells were harvested, RNA samples were prepared for analyzing Nrf2 mRNA expression using RT-qPCR and protein samples were prepared for analyzing Nrf2 protein expression using Western Blot.
Statistical analysis
Statistical evaluation was done by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. P value less than 0.05 was considered to be statistically significant.
Results
The effect of GSTD on high glucose-induced cytotoxicity in H9c2 and HL-1 cardiomyocytes
In order to investigate whether GSTD (molecular structure is showing in Figure 1(a) could protect H9c2 and HL-1 cardiomyocytes against high glucose (33 mM glucose)-induced injury, MTT assay was performed to detect the effect of GSTD on cell viability. The result showed that GSTD rescued high glucose-caused decrease in cell viability of H9c2 and HL-1 cells in a concentration-dependent manner (Figure 1(b)). The release of LDH, a marker of myocardial injury, was also inhibited by GSTD under high glucose condition (Figure 1(c)). The mTOR signaling pathway, which is closely related with growth and survival in mammal, 23 was also rescued by GSTD under high glucose condition (Figure 1(d)). These results indicate that GSTD could protect H9c2 and HL-1 cells against high glucose-induced cytotoxicity.

GSTD protected against high glucose-induced cytotoxicity in H9c2 and HL-1 cells. (a) Molecular structural of GSTD. (b) H9c2 and HL-1 cells were treated with GSTD at the indicated concentrations for 24 h under normal condition (5.5 mM glucose), namely control group and high glucose (33 mM glucose) condition. Cell viability was detected by MTT assay. (c) LDH assay kit was used to detect LDH content in medium of H9c2 and HL-1 cells after GSTD treatment as above mentioned. (d) The expression of p-p70S6 k (Ser371), p70S6 k, p-mTOR (Ser2448) and mTOR in H9c2 and HL-1 cells after GSTD treatment as above mentioned was detected by Western Blot, with β-actin as a loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; #P < 0.05 vs glucose (33 mM) group; ##P < 0.01 vs glucose (33 mM) group.
GSTD inhibited high glucose-induced oxidative stress in H9c2 and HL-1 cardiomyocytes
Oxidative stress plays a critical role in high glucose-induced myocardial injury. 24 Thus, whether GSTD could protect against high glucose-induced oxidative stress was checked. As expected, the results showed that GSTD increased GSH level (Figure 2(a)) and decreased ROS level (Figure 2(b)) under high glucose condition in H9c2 and HL-1 cells. The activity of antioxidant enzymes, superoxide dismutase (SOD) and catalase (CAT) was also increased after GSTD treatment under high glucose condition in H9c2 and HL-1 cells (Figure 2(c) and 2(d)). Therefore, these results showed that GSTD could protect against high glucose-induced oxidative stress in H9c2 and HL-1 cells. ROS is a common inducer of mitochondrial apoptosis. To test whether GSTD could protect against high glucose-induced mitochondrial apoptosis, flow cytometry with Rh-123 staining was performed to detect mitochondrial membrane potential (MMP) and Western Blot was used to detect the expression of anti-apoptotic protein Bcl-2, pro-apoptotic protein Bax, the apoptotic marker protein Cleaved Caspase-3 and the release of Cytochrome C from mitochondria to cytosol. Results showed that GSTD increased MMP in a concentration-dependent manner under high glucose condition (Figure 3(a)). Bcl-2 expression was up-regulated while Bax expression was down-regulated by GSTD treatment. Moreover, GSTD decreased the expression of Cleaved Caspase-3 expression (Figure 3(b)). Upon GSTD treatment, the expression of Cytochrome C was increased in mitochondria while it was decreased in cytosol, suggesting that the release of Cytochrome C from mitochondria to cytosol was inhibited by GSTD under high glucose condition (Figure 3(c)). Taken together, these results indicate that GSTD could protect against high glucose-induced mitochondrial apoptosis. In summary, GSTD could protect against high glucose-induced cytotoxicity in H9c2 and HL-1 cells.

GSTD inhibited high glucose-induced oxidative stress in H9c2 and HL-1 cells. (a, b) H9c2 and HL-1 cells were treated with GSTD with above mentioned concentrations. GSH level was detected using a commercial colorimetric assay kits, and ROS level was detected using an oxidation sensitive dye, DCFH-DA. (c, d) Total SOD and CAT activities were measured using a Total Superoxide Dismutase Assay Kit with NBT and a Catalase Assay Kit after GSTD treatment with above mentioned concentrations for 24 h. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; #P < 0.05 vs glucose (33 mM) group; ##P < 0.01 vs glucose (33 mM) group.
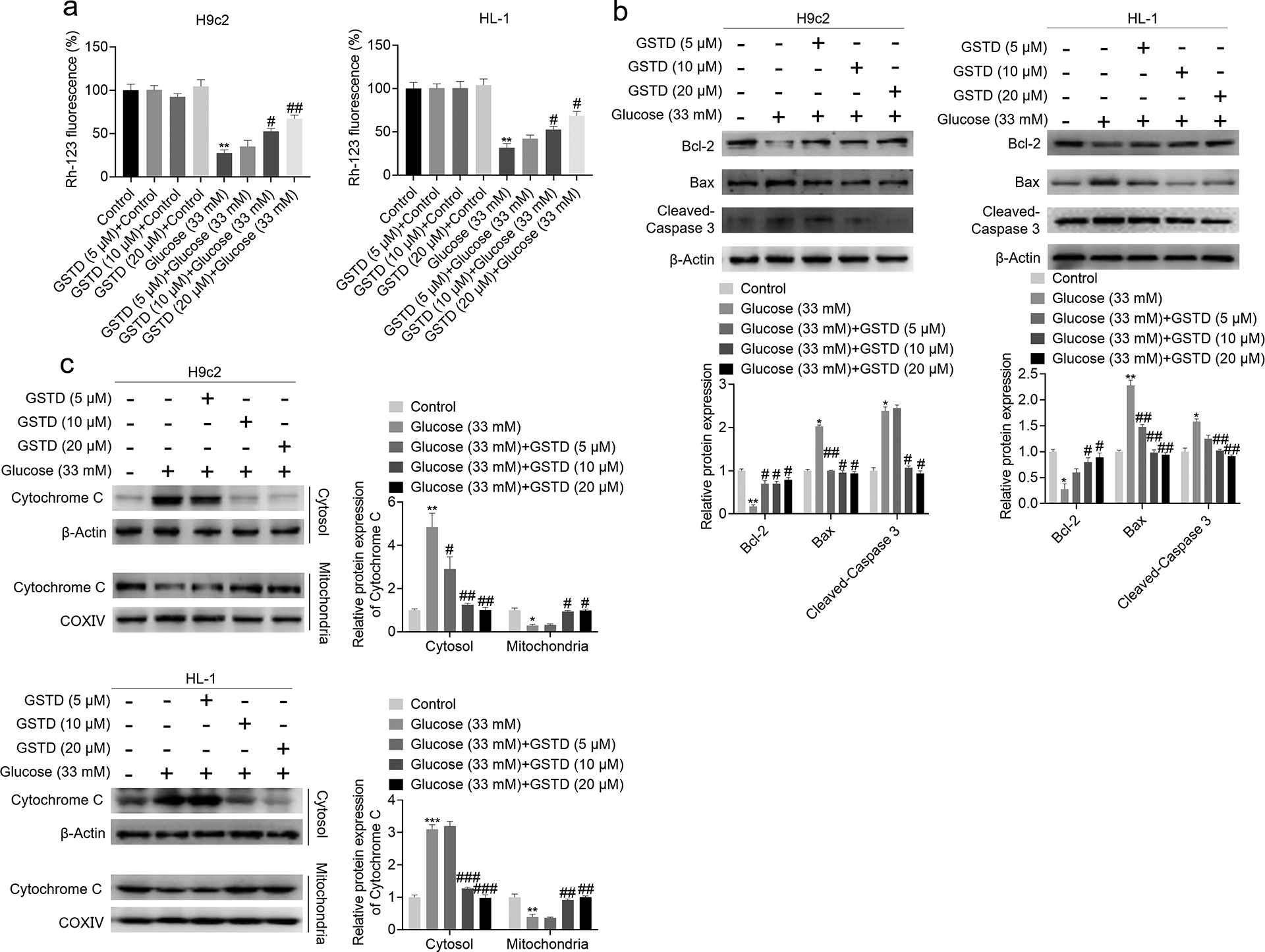
GSTD prevented high glucose-induced apoptosis in H9c2 and HL-1 cells. (a) H9c2 and HL-1 cells were treated with GSTD with above mentioned concentrations for 24 h. Cells were stained with 10 μM Rh-123 and the fluorescence intensity was analyzed by flow cytometry. (b) H9c2 and HL-1 cells under high glucose condition were treated with GSTD with above mentioned concentrations for 24 h, cells were collected and the expression of Bcl-2, Bax and Cleaved Caspase 3 was analyzed by Western Blot, with β-actin as a loading control. (c) H9c2 and HL-1 cells under high glucose condition were treated with GSTD with above mentioned concentrations for 24 h, cells were collected and cytoplasmic and mitochondrial protein was extracted using a Cell Mitochondria Isolation Kit. Then the expression of Cytochrome C in mitochondria and cytosol was detected by Western Blot, with COXIV and β-actin as a loading control respectively. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; #P < 0.05 vs glucose (33 mM) group; ##P < 0.01 vs glucose (33 mM) group.
GSTD promoted the nuclear translocation of Nrf2 in H9c2 and HL-1 cells
To investigate whether Nrf2 (nuclear factor-E2-related factor 2) and Nuclear factor κB (NF-κB), the two most important transcription factors for inducing a set of antioxidant and detoxification enzymes,25,26 were involved in the cytoprotective effect of GSTD. Total protein expression and the nuclear translocation of Nrf2 and NF-κB after GSTD treatment under high glucose condition were checked by Western Blot. The result showed that neither total Nrf2 expression nor total NF-κB expression was changed by GSTD treatment (Figure 4(a)). However, GSTD promoted the nuclear translocation of Nrf2 in a concentration-dependent manner but had no effect on the nuclear translocation of Nrf2 under high glucose condition (Figure 4(b)). Next, H9c2 and HL-1 cells were treated with 10 μM GSTD for 12, 24 and 48 h under high glucose condition, it was observed that GSTD promoted the nuclear translocation of Nrf2 in a time-dependent manner (Figure 4(c)). Moreover, the mRNA expression of four target genes of Nrf2, Gclc, Gclm, Txnrd1 and Prdx1 was all up-regulated by GSTD (Figure 4(d–g)). Taken together, these results demonstrate that GSTD could promote the nuclear translocation of Nrf2 in H9c2 and HL-1 cells under high glucose condition.

GSTD promoted the nuclear translocation of Nrf2 in H9c2 and HL-1 cells. (a) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with GSTD at the indicated concentrations (5, 10, 20 μM) for 24 h, the expression of Nrf2 and NF-κB was detected by Western Blot with β-actin a loading control. (b) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with GSTD with above mentioned concentrations for 24 h, then nuclear proteins were extracted using a Nuclear and Cytoplasmic Protein Extraction Kit. Nuclear expression of Nrf2 and NF-κB was detected by Western Blot with lamin B1 as a loading control. (c) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 10 μM GSTD for 12, 24, 48 h, then nuclear proteins were extracted using a Nuclear and Cytoplasmic Protein Extraction Kit. Nuclear expression of Nrf2 and NF-κB was detected by Western Blot with lamin B1 as a loading control. (d–g) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with GSTD with above mentioned concentrations for 24 h, total RNA was extracted and RT-qPCR was performed to detect the mRNA expression of Gclc, Gclm, Txnrd1 and Prdx1. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs GSTD (0 μM) group; **P < 0.01 vs GSTD (0 μM) group; ***P < 0.001 vs GSTD (0 μM) group. In Figure 4(c), **P < 0.01 vs time (0 hrs) group; ***P < 0.001 vs time (0 hrs) group.
Nrf2 knockdown attenuated the protective effects of GSTD on high glucose-induced cytotoxicity and oxidative stress in H9c2 and HL-1 cells
To investigate the role of Nrf2 in the protective effect of GSTD on high glucose-treated H9c2 and HL-1 cells, Nrf2 knockdown was performed. Three RNA interference sequences target Nrf2 (siNrf2#1, siNrf2#2, siNrf2#3) and one scrambled negative control (NC) sequence were designed and transfected into H9c2 and HL-1 cells. The knockdown efficiency of Nrf2 was checked by RT-qPCR and Western Blot. Compared with blank control group (Control), NC showed no influence on Nrf2 mRNA expression, while siNrf2#2 showed the highest knockdown efficiency among these three interference sequences in both H9c2 and HL-1 cells (Figure 5(a)). The knockdown efficiency at protein level was consistent with the knockdown efficiency at mRNA level (Figure 5(b)). Thus, siNrf2#2 was picked to address our following experiments. When Nrf2 expression was inhibited by siNrf2#2, the nuclear translocation of Nrf2 was significantly inhibited after GSTD treatment under high glucose condition (Figure 5(c)). The protective effect of GSTD on cell viability and the inhibitory effect of GSTD on LDH release under high glucose condition were also inhibited by Nrf2 knockdown (Figure 5(d–e)). Moreover, Nrf2 knockdown also attenuated the inhibitory effect of GSTD on high glucose-induced oxidative stress. As shown in Figure 5(f–g), Nrf2 knockdown decreased GSH level and increased ROS level upon GSTD treatment under high glucose condition. The enhancement of SOD activity and CAT activity by GSTD under high glucose condition was also reversed by Nrf2 knockdown (Figure 5(h–i)). In conclusion, these results indicate that Nrf2 mediates the protective effects of GSTD on high glucose-induced cytotoxicity and oxidative stress in H9c2 and HL-1 cells.

Nrf2 knockdown attenuated the inhibitory effects of GSTD on high glucose-induced cytotoxicity and oxidative stress in H9c2 and HL-1 cells. (a, b) Three RNA interference sequences target Nrf2 (siNrf2#1, siNrf2#2, siNrf2#3) and one scrambled negative control (NC) sequence were designed and transfected into H9c2 and HL-1 cells, the knockdown efficiency of Nrf2 was checked by RT-qPCR and Western Blot. (c) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 20 μM Nrf2 siRNA#2 for 4 h, then maintained for 24 h for Nrf2 knockdown. Then the cells were treated with 10 μM GSTD for 24 h, nuclear proteins were extracted and nuclear expression of Nrf2 was detected by Western Blot with lamin B1 as a loading control. (d) H9c2 and HL-1 cells under normal condition (5.5 mM glucose), namely control group and high glucose (33 mM glucose) condition were treated with 20 μM Nrf2 siRNA#2 and 10 μM GSTD as above mentioned, cell viability was detected by MTT assay. (e) H9c2 and HL-1 cells under normal condition (5.5 mM glucose), namely control group and high glucose (33 mM glucose) condition were treated with 20 μM Nrf2 siRNA#2 and 10 μM GSTD as above mentioned, LDH content in medium was detected using a LDH assay kit. (f, g) H9c2 and HL-1 cells under normal condition (5.5 mM glucose), namely control group and high glucose (33 mM glucose) condition were treated with 20 μM Nrf2 siRNA#2 and 10 μM GSTD as above mentioned, intracellular GSH and ROS level were detected by a commercial colorimetric assay kit and DCFH-DA respectively. (h, i) H9c2 and HL-1 cells under normal condition (5.5 mM glucose), namely control group and high glucose (33 mM glucose) condition were treated with 20 μM Nrf2 siRNA#2 and 10 μM GSTD as above mentioned, total SOD and CAT activities were measured using a Total Superoxide Dismutase Assay Kit with NBT and a Catalase Assay Kit. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; #P < 0.05 vs glucose (33 mM) group; ##P < 0.01 vs glucose (33 mM) group.
GSTD promoted the nuclear translocation of Nrf2 via activating GSK-3β signaling pathway
Next, we aimed to investigate the signaling pathway through which GSTD promoted the nuclear translocation of Nrf2. GSK-3β, ERK1/2 and p38 MAPK signaling pathways are reported to regulate the nuclear translocation of Nrf2.9,10,27 Thus, the effect of GSTD on these signaling pathways were checked. GSTD significantly increased the phosphorylation of GSK-3β but had no obvious influence on ERK1/2 and p38 MAPK, suggesting that GSK-3β signaling pathway was activated by GSTD (Figure 6(a)). We next checked the stimulative effect of GSTD on GSK-3β signaling pathway under high glucose condition. The result showed that high glucose (33 mM glucose) suppressed the phosphorylation of GSK-3β compared with control (5.5 mM glucose), and GSTD rescued the phosphorylation of GSK-3β under high glucose condition (Figure 6(b)). The nuclear translocation of Nrf2 was also increased by GSTD under high glucose condition in a concentration-dependent manner (Figure 6(c)). To test whether GSK-3β signaling pathway mediated the nuclear translocation of Nrf2, a GSK-3β inhibitor TDZD-8 was employed to inhibit GSK-3β activity. Upon 10 μM TDZD-8 treatment, the nuclear translocation of Nrf2 by GSTD treatment was decreased (Figure 6(d)). Moreover, TDZD-8 attenuated the protective effect of GSTD on cell viability and promoted LDH release in H9c2 and HL-1 cells upon GSTD treatment under high glucose condition (Figure 6(e–f)). Increased GSH level and decreased ROS level by GSTD treatment were also blocked by TDZD-8 (Figure 6(g–h)), as well as the enhancement of SOD activity and CAT activity by GSTD (Figure 5(i–j)). Taken together, these results demonstrate that GSTD promotes the nuclear translocation of Nrf2 via activating GSK-3β signaling pathway.

GSTD promoted the nuclear translocation of Nrf2 via activating GSK-3β. (a) H9c2 and HL-1 cells under normal (5.5 mM glucose) were treated with GSTD at indicated concentrations (5, 10, 20 μM) for 24 h, the expression of p-GSK-3β, GSK-3β, p-ERK1/2, ERK1/2, p-p38 MAPK, p38 MAPK was detected by Western Blot. (b) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with GSTD at indicated concentrations (5, 10, 20 μM) for 24 h, the expression of p-GSK-3β and GSK-3β was detected by Western Blot. (c) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with GSTD as above mentioned for 24 h, nuclear protein was extracted and the nuclear expression of Nrf2 was detected by Western Blot. (d) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 10 μM GSTD, 10 μM TDZD-8 and 10 μM GSTD combined with 10 μM TDZD-8 for 24 h, nuclear proteins were extracted and the nuclear expression of Nrf2 was detected by Western Blot with lamin B1 as a loading control. (e, f) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 10 μM GSTD or 10 μM TDZD-8 or 10 μM GSTD combined with 10 μM TDZD-8 for 24 h, cell viability was detected by MTT assay and LDH content in serum was detected using a LDH assay kit. (g, h) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 10 μM GSTD, 10 μM TDZD-8 and 10 μM GSTD combined with 10 μM TDZD-8 for 24 h, intracellular GSH and ROS level were detected by a commercial colorimetric assay kit and DCFH-DA respectively. (i, j) H9c2 and HL-1 cells under high glucose (33 mM glucose) condition were treated with 10 μM GSTD, 10 μM TDZD-8 and 10 μM GSTD combined with 10 μM TDZD-8 for 24 h, total SOD and CAT activities were measured using a Total Superoxide Dismutase Assay Kit with NBT and a Catalase Assay Kit. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; ***P < 0.001 vs control group; #P < 0.05 vs glucose (33 mM) group; ##P < 0.01 vs glucose (33 mM) group.
Discussion
DCM is a serious complications of diabetic patients. In our study, we employed an in vitro high glucose-induced cytotoxicity model cardiomyocytes to investigate the protective effect of GSTD, a phenolic glucoside from an ancient Chinese herb Tianma (Gastrodia elata Bl.). Our results demonstrated that GSTD displayed desirable protective effect on high glucose-treated H9c2 and HL-1 cardiomyocytes, including enhancing cell viability, inhibiting oxidative stress and mitochondrial apoptotic pathway-mediated apoptosis. Further studies showed that these effects were achieved by GSK-3β-mediated nuclear translocation of Nrf2, the most important transcription factor for antioxidant enzymes transcription. Knockdown of Nrf2 using siRNA or inhibition of GSK-3β signaling pathway using a GSK-3β inhibitor TDZD-8, both attenuated the protective effect of GSTD on high glucose-treated H9c2 and HL-1 cardiomyocytes. Thus, we provide a possibility for GSTD in the prevention and treatment of DCM.
GSTD is obtained from a Chinese herb Tianma (Gastrodia elata Bl.), which is widely used by ancient Chinese people for the prevention and treatment of cardiovascular and cerebrovascular diseases. Previous study reported that GSTD protected H9c2 cells from hypoxia-induced injury by up-regulation of microRNA-21 and attenuation of the inflammatory response.20,28 Thus, it is anticipated that GSTD could possess a protective effect on DCM. But experiments are needed to demonstrate it. High glucose-induced cell damage model is widely used for in vitro study of diabetic complications, including endothelial dysfunction, 29 renal mesangial cell injury 30 and myocardial damage.24,31 Thus, we employed high glucose-induced cardiomyocyte injury model for our study, and the results showed that GSTD performed expected protective effect on H9c2 and HL-1 cardiomyocytes.
Oxidative stress was reported to play an important role in DCM. 32 Thus ROS inhibition emerges as a promising strategy for the prevention and treatment of DCM. 33 In H9c2 and HL-1 cells, high glucose also triggered oxidative stress with increased ROS level, decreased GSH level and decreased SOD, CAT activities. GSTD inhibited the oxidative stress caused by high glucose in H9c2 and HL-1 cells. In addition, ROS is the common inducer of mitochondrial apoptosis. A previous study also indicated that hyperglycemia-related myocardial apoptosis was partially mediated by mitochondrial Caspase-3 activation. 34 Bcl-2 is an anti-apoptotic molecule, and Bax and Caspase-3 are pro-apoptotic molecules. When mitochondrial apoptotic pathway is activated, MMP decreases and Cytochrome C is released from mitochondria to cytosol. 35 After GSTD treatment, it was found that MMP was maintained and Cytochrome C was kept in mitochondria. Moreover, the expression Bcl-2 was increased while the expression of Bax and Cleaved Caspase-3 was decreased. These results strongly suggest that the ROS-mediated mitochondrial apoptotic pathway is inhibited by GSTD.
Nrf2 and NF-κB are the two most important transcription factor for the expression of antioxidant enzymes in mammalian cells.36,37 Thus we tested the effect of GSTD on the nuclear translocation of these two transcription factors. We found that the nuclear translocation of Nrf2 was increased by GSTD. Nrf2 is normally kept in cytoplasm and binds to the Kelch domain of Keap1 through the ETGE and DLG motifs of its Neh2 domain. However, under stressed condition, Nrf2 is released to transfer into nucleus for antioxidant genes transcription. 7 Several signaling pathways were reported to regulate this process, including GSK-3β, ERK, and p38 MAPK signaling pathways.38–40 Thus, which signaling pathway mediated the nuclear translocation of Nrf2 was investigated. The result showed that it was GSK-3β signaling pathway. Furthermore, both knockdown of Nrf2 using siRNA and inhibiting GSK-3β signaling pathway using a GSK-3β inhibitor TDZD-8 significantly attenuated the protective effect of GSTD on H9c2 and HL-1 cells. Therefore, we clarified the role of Nrf2 and GSK-3β in the protective effects of GSTD on high glucose-induced cytotoxicity in H9c2 and HL-1 cells.
Our study also has its limitations. The protective effect of GSTD tested in vivo could strengthen the conclusion. This part will be addressed in our future study. In addition, the detailed mechanism of how GSTD activates GSK-3β signaling pathway could be clarified. For example, chemical proteomics could be used to explore the direct target of GSTD. In summary, we demonstrate that GSTD protects against high glucose-induced cytotoxicity in H9c2 and HL-1 cells via GSK-3β-mediated nuclear translocation of Nrf2, and the study provides a possibility for GSTD in the prevention and treatment of DCM.
Footnotes
Author contributions
Li-Ming Sun cooperated designed the study and its technical strategies and drafted the initial manuscript and reviewed and finalizing it as submitted. Zheng Dong and Lv Bian conducted experiments, statistically analyzed data and reviewed the initial manuscript. Yi-Lian Wang helped to revise the initial manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financially supported by Key Research Projects of Bengbu Medical College (BYKY17183).
