Abstract
The objective of this study was to evaluate the histopathological effect of gas explosion on rats, and to explore the metabolic alterations associated with gas explosion-induced acute blast lung injury (ABLI) in real roadway environment using metabolomics analyses. All rats were exposed to the gas explosion source at different distance points (160 m and 240 m) except the control group. Respiratory function indexes were monitored and lung tissue analysis was performed to correlate histopathological effect to serum metabolomics. Their sera samples were collected to measure the metabolic alterations by ultra-performance liquid chromatography-mass spectrometry (UPLC-MS). HE staining in lung showed that the gas explosion caused obvious inflammatory pulmonary injury, which was consistent with respiratory function monitoring results and the serum metabolomics analysis results. The metabolomics identified 9 significantly metabolites different between the control- and ABLI rats. 2-aminoadipic acid, L-methionine, L-alanine, L-lysine, L-threonine, cholic acid and L-histidine were significantly increased in the exposed groups. Citric acid and aconitic acid were significantly decreased after exposure. Pathway analyses identified 8 perturbed metabolic pathways, which provided novel potential mechanisms for the gas explosion-induced ABLI. Therefore, metabolomics analysis identified both known and unknown alterations in circulating biomarkers, adding an integral mechanistic insight into the gas explosion-induced ABLI in real roadway environment.
Introduction
Blast lung injury (BLI) is one of the main causes of death, and it is the focus of current blast injury protection research for its high-energy trauma with high mortality. BLI caused by blast waves is extremely critical in the prehospital setting, and hypotension is thought to be the main cause of death in such cases. 1 It is also characterized by respiratory disturbance and hypoxia. 2 A retrospective analysis of 132 cases of blast injury of lungs caused by gas explosion injuries showed that pulmonary explosive injury was characterized by severe internal injury, multiple injuries, high incidence of multiple injuries, and difficult diagnosis. 3,4 The lung tissues receive a direct mechanical injury from the shock wave, causing vascular permeability and infiltration. 5 Animal studies have shown that the injury of gas explosion can activate NF-kappaB, which has close correlation with the acute injury to lung. 6 –8 Therefore, research for seeking new treatments to attenuate excessive inflammation after severely burned injury is very necessary, especially in the acute phase. Several animal models have been developed in early researches, 9 but no mature model has been established because of various model limitations.
So far, some protective mechanisms of blast lung injury in humans and animal models were studied. Studies have demonstrated that blast limb trauma causes remote lung injury, which is likely associated with remarkable inflammatory response, oxidative stress, and depletion of protective mechanisms. 10 Although the effects and intervention studies of gas explosion-induced ABLI have been well studied, the underlying mechanisms explaining the gas explosion-induced ABLI and the effect of short-term exposure on the body metabolism level have not yet been completely elucidated. A possible reason is the limitation of relevant conditions, such as experimental apparatus. Thus, a new approach for studying the lung injury effects of short-term, acute exposure to gas explosion needs to be applied.
Metabolomics is defined as a new discipline for the qualitative and quantitative analysis of small molecular metabolites with relative molecular weights less than 1,000 in an organism or a cell. As an important part of system biology, metabolomics has a wide application prospect in the field of clinical medicine. 11 Recently, the technology and application of liquid chromatography-mass spectrometry (LC-MS) have made great progress in comprehensive metabolite profiling. In addition, ultraperformance liquid chromatography (UPLC) has advantages in many aspects such as wide analysis scope, strong separation ability, reliable qualitative analysis result, low detection limit and fast analysis time. 12 Compared with the genomic and proteomic studies, metabolomics analysis can screen out a large number of meaningful low molecular metabolites, which are often closely associated with many diseases. 13 In previously studies, the liver and kidney injury were evaluated using metabolomics. 14,15 The results showed that metabolomics can offer more injury information than the common injury assessment method.
In a word, metabolomics analysis is a useful tool for understanding the pathogenesis of gas explosion-induced ABLI. To examine the metabolite profiles changes of gas explosion-induced ABLI, we employed a non-targeted serum metabolomics analysis to identify differential metabolites related to ABLI caused by gas explosion, which will reveal a mechanistic basis for comprehending, ameliorating, or preventing gas explosion-induced adverse effects in the lung.
Materials and methods
Chemicals and reagents
High-performance liquid chromatography (HPLC)-grade formic acid was purchased from American Merck company (CAS: 64-18-6, LC-MS grade). HPLC-grade ammonium formate was purchased from American Sigma company (CAS: 540-69-2, 99.0% purity). HPLC-grade methanol was purchased from China WoKai company (CAS: 67-56-1, 99.0% purity). HPLC-grade acetonitrile was purchased from American Merck company (CAS: 75-05-8, 99.0% purity). Distilled water was filtered using a Milli-Q system (Arium® mini, Sartorius). Leucine enkephalin used in HPLC was purchased from Sigma–Aldrich (St. Louis, MO, USA). Other standards used in this study were purchased from Sigma (Sigma–Aldrich, St Louis, MO, USA, 99% purity). All other chemicals, reagents, and buffers were analytical-grade products from Amresco Llc. (Solon, OH, USA).
Experimental animals
Twenty-four 8 weeks male Sprague-Dawley rats with an average weight of 200 ± 20 g were purchased from laboratory animal center of the third affiliated hospital of The Third Military Medical University. They were housed in a specific environment under controlled temperature (22 ± 2°C) and humidity of 50–60%, with a 12-h light/dark cycle for 1 week, with free access to food and water.
After adaptation for 7 days, rats were randomly chosen for the acute blast lung injury (ABLI) model exposed to explosion from a distance (n = 8/group): control group (nonparticipation) and ABLI group (160 meters and 240 meters away from the explosion source. Our preliminary experimental results have showed that this distance is reliable for developing a stable rat model of severe ABLI). All rats were transferred to the laneway environment and placed in a fixed cage with their chest pointed to the source of the explosion under sodium pentobarbital anesthesia before the gas explosion. Rats were transferred to a lab quickly in a controlled time for 30 min after the gas explosion from the ventilated roadway, and sacrificed under sodium pentobarbital anesthesia at 48th hour in a lab after gas explosion. The rats’ general health and behavior were observed every 2 hours in the lab. We performed an ignition explosion before the rats waked up under the gas explosion roadway in the Chongqing coal academy of China, which simulates gas explosion in a coal mine. Meanwhile, respiratory function tests were launched using by small animal lung function meter (WBP) before the rats were sacrificed at the 48th hour after gas explosion.
The researchers involved in this study were systematically and specifically trained before the experiment, including methods of anesthesia, placement, post-injury treatment, transfer, post-injury monitoring and execution. The ultimate goal of all training is to alleviate the pain and unnecessary pain of laboratory animals. All animal experimentation was performed in accordance with the Institute of Zoology Animal and Medical Ethics Committee of Xinxiang Medical University and was in accordance with the current Chinese legislation, in addition to international standards (NIH publications No 80-23 revised 1996).
Roadway parameters and fixtures
The roadway was 3 m in diameter and 300 m in length. According to the environment of gas explosion roadway, the volume of the mixed gas was 100 m3. Special gas with an oxygen concentration of 8−20% and gas concentration of 9−9.5% was selected. The ignition and explosion pressure in the center was 1.6−2.0 kg. Explosion parameters: gas capacity was 1.36 m3; maximum pressure value was 0.03864 MPa; maximum pressure value time was 0.699 s; at a flame front velocity of 162 m/s; at a peak temperature of 853°C.
Sample collection and processing
Rats were transferred to a lab and sacrificed under sodium pentobarbital anesthesia via intraperitoneal injection at 48 h after gas explosion. Blood samples were obtained from the abdominal aorta before the rats were sacrificed and prior to blood clotting. Serum was obtained by centrifugation of the blood at 3000 rpm for 15 min in a refrigerated centrifuge = (H1650-W, Eppendorf) and then immediately stored at −80°C. The lung of rats was extracted and histological observation was performed. The lung mass was weighed for damage assessment.
Before metabolomics analysis, the frozen serum samples were thawed at 4°C. After the samples had completely thawed, 200 µL of each sample was transferred into 1.5 mL centrifuge tubes, to which 800 µL of methanol (pre-cooled at −20°C) was added. The tubes were for 60 s using MixStar (QL-866, Vortex Mixer) and centrifuged for 10 min at 12,000 rpm 4°C. The supernatant from each tube was transferred into another 1.5 mL centrifuge tube and the samples were blow-dried by vacuum concentration. To dissolve the samples, 300 µL of methanol aqueous solution (4:1, 4°C) was added, and the samples were filtered through a 0.22 µm membrane (0.22 µm PTFE, Jin Teng) in order to obtain the prepared sample extracts for LC-MS. For the quality control (QC) of the samples, a mixture containing 20 µL from each prepared sample extract was prepared.
Conditions of chromatography and mass spectrometry
Chromatographic separation was accomplished in a Thermo Ultimate 3000 system equipped with an ACQUITY UPLC® HSS T3 (150 × 2.1 mm, 1.8 µm, Waters) column maintained at 40°C. The temperature of the autosampler was 8°C. Gradient elution of analytes was carried out with 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B) or 5 mM ammonium formate in water (C) and acetonitrile (D), at a flow rate of 0.25 mL/min. Injection of 2 µL of each sample was performed after equilibration. An increasing linear gradient of solvent B (v/v) was added as follows: 0−1 min, 2% B/D; 1−9 min, 2−50% B/D; 9−14 min, 50−98% B/D; 14−15 min, 98% B/D; 15−15.5 min, 98−2% B/D; 15.5−17 min, 2% B/D.
The ESI-MSN experiments were executed on the Thermo Q Exactive Focus mass spectrometer with spray voltage of 3.8 kV and −2.5 kV in positive and negative modes, respectively. Sheath gas and auxiliary gas were set at 45 and 15 arbitrary units, respectively. The capillary temperature was set at 325°C. The analyzer scanned over a mass range of m/z 81−1,000 for a full scan at a mass resolution of 70,000. Data-dependent acquisition (DDA) MS/MS experiments were performed with HCD scan. The normalized collision energy was 30 eV. Dynamic exclusion was implemented to remove unnecessary information regarding the MS/MS spectra. In addition, the precision and repeatability of the PLS-DA was evaluated using a representative pooled QC sample. In this study, QCs were injected at regular intervals (i.e., for every 10 test samples) throughout the analytical run. The method were validated by the reduplicate analysis of six injections of the same QCs and six parallel samples prepared using mixing equal volumes of the serum samples from five control rats and five ABLI rats, respectively. Meanwhile, serum standard solutions of lower (1 ng/mL), medium (20 ng/mL) and higher (400 ng/mL) concentrations were prepared as QCs by the same procedure for methodological validation purpose.
Data processing, metabolite identification and metabolic pathway analysis
The UPLC/Q-TOF-MS data were processed using the MarkerLynx Application Manager 4.1 SCN 714 (Waters, Milford, MA, USA). MarkerLynx was employed for peak finding, peak alignment, and reporting of the mass, retention time, and intensity of the peaks in each sample. All these data were introduced into the EZinfo 2.0 software (Umeå, Sweden) for multivariate statistical analysis. Unsupervised principal component analysis (PCA) was first used for all samples to visualize the general separation. EZinfo was further performed to highlight the difference between groups using partial least squares-discriminant analysis (PLS-DA). Pareto-scaling was used prior to multivariate statistical analysis to avoid chemical noise. Moreover, a default 7-fold cross-validation and testing with 100 random permutations were performed using SIMCA-P software (version 14.0; Umetrics AB, Umeå, Sweden) to avoid overfitting of PLS-DA models.
For biomarker identification, the ions, which were mostly responsible for the variances, were identified based on Variable Importance in the Projection (VIP) values (>1.5) and statistical tests (P < 0.05) in the PLS-DA model were regarded as potential biomarkers. The probable formulas for the potential biomarkers were first identified according to accurate mass measurement (mass error < 30 PPM) and by considering the relative intensities of the isotope peaks based on high-resolution MS spectra. Then, the MassFragmentTM application manager (MassLynx v4.1, Waters Corp., USA) was used to facilitate the MS/MS fragment ion-analysis process using chemically intelligent peak-matching algorithms. Briefly, the UPLC/MS/MS product ion spectrum of potential biomarkers were matched with the structural data of metabolites acquired from the Human Metabolome Database (HMDB) or ChemSpider, using the Mass Fragment software (Waters Corp.) according to some parameters, such as deviation from double bond equivalent (DBE) and i-fit value (the isotopic pattern of the selected ion). Finally, the biomarkers were further identified using standard samples based on accurate mass, RT, and matching MS spectra of the unknown with the standard MS spectra in the database, such as the Human Metabolome Database (HMDB, http://www.hmdb.ca), Metlin (http://metlin.scripps.edu/), and MassBank (http://www.massbank.jp/).
The implicated pathways associated with the biomarkers were described using databases, including HMDB (http://www.hmdb.ca) and Kyoto Encyclopedia of Genes and Genomes (KEGG, http://www.genome.jp/kegg/). In addition, other implicated pathways of biomarkers were further interpreted based on previous literatures.
Statistical analysis
Data were presented as the mean ± SD. One-way analysis of variance (one-way ANOVA) or nonparametric Kruskal−Wallis test was used for analyzing the differences among groups. When statistical differences were indicated, LSD test or Dunnett T3 was used as a post hoc test. All P values were subjected to a 2-tailed test and a P value < 0.05 was considered significant. All statistical analysis in this study was performed using with SPSS version 17.0 (SPSS, Beijing Stats Data Mining Co. Ltd, Beijing, China).
Results
Gas explosion-induced ABLI caused lung tissues damage
In this study, the main injuries observed in rats exposed to gas explosion were ABLI, while burns were found to be not obvious. Compared with the control group (Figure 1(A)), gas explosion exhibited an obvious effect on the lung of rats in the ABLI groups, as reflected by obvious pulmonary congestion and oedema (Figure 1(B) and (C)), and increased levels of dry and wet ratio of lung tissues in the ABLI groups (Figure 1(D)), indicating that the rat model of ABLI induced by gas explosion was established successfully.
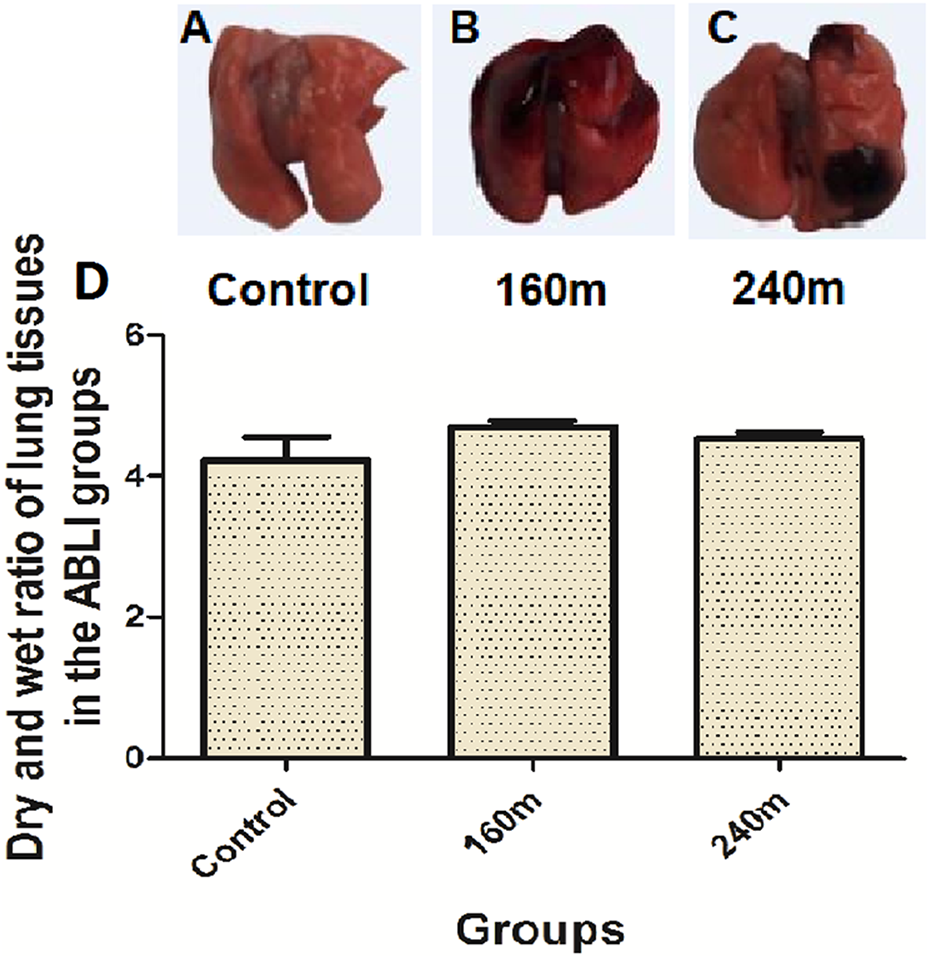
Changes in lung appearance (A, B and C) and dry and wet ratio of lung tissues (D) after gas explosion. (A): Control, control group; (B): 160 m, close-distance gas explosion group; (C): 240 m, far-distance gas explosion group. Data are expressed as mean ± SD (n = 8). (*) P < 0.05, significantly different from the control.
Gas explosion-induced ABLI resulted in pathological changes on lung tissues
The histopathological changes of lung tissues in the control rats were not found (Figure 2(A)), whereas the lungs of the gas explosion-induced ABLI rats revealed histopathological abnormalities, such as injury of vascular endothelium and lung epithelial cells, lung vacuoles, lung collapse were observed in near-distance group (Figure 2(B)), which were more pronounced in near-distance group than far-distance group (Figure 2(C)) 48 hours after the gas explosion.

The histopathologic changes of lung 48 hours in the control and ABLI groups after gas explosion. (A): Control, control group rat; (B): 160 m, close-distance gas explosion group rat; (C): 240 m, far-distance gas explosion group rat. ABLI: acute blast lung injury group. HE stain, magnification, 200×.
To further prove the histopathological results of lung injury induced by gas explosion were reliable, a pulmonary score was conducted to rats according to the criteria for pathology of lung injury, and the effects of close-distance and long-distance exposure to the gas explosion source were analyzed. Through the above analysis, we found that rats that received close-distance exposure to the gas explosion source obtain a high pulmonary histopathological score, and received long-distance exposure to the gas explosion source obtain a low pulmonary histopathological score.
Gas explosion-induced ABLI led to respiratory function changes in rats
Respiratory function assay is a useful and sensitive method for measuring physiological function changes in lung. The respiratory function index, including respiratory frequency (f), tidal volume (TVb), minute volume (MVb), enhanced pause (Penh), pause (PAU), peak inspiratory flow (PIF), 50% of expiratory flow (EF50), provide a clear indication of the functional changes of the lungs over a wide range of injury levels. The results of respiratory function index of in each ABLI group are shown in Table 1. The above-mentioned parameters were not statistically significant change, but with an increase or decrease trend, in group 160 m compared with the control group (p > 0.05). However, the TVb, PIFb significantly increased, and Penh, PAU significantly decreased in group 240 m compared with the control group (p < 0.05).
Alterations of lung function indexes in rats after 48h of the gas explosion (n = 8, x ± s).
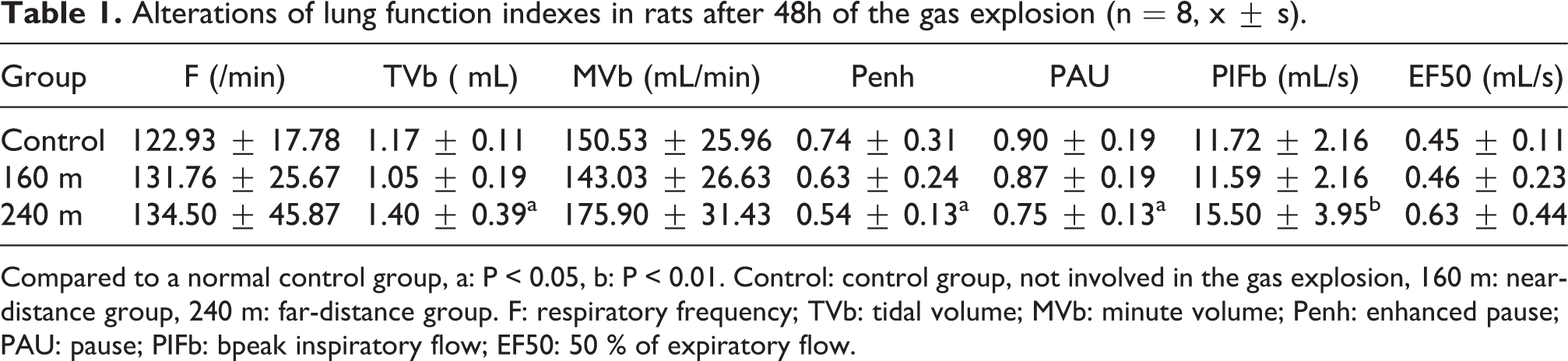
Compared to a normal control group, a: P < 0.05, b: P < 0.01. Control: control group, not involved in the gas explosion, 160 m: near-distance group, 240 m: far-distance group. F: respiratory frequency; TVb: tidal volume; MVb: minute volume; Penh: enhanced pause; PAU: pause; PIFb: bpeak inspiratory flow; EF50: 50 % of expiratory flow.
Quality assessment and methodological validation of the metabolomics platform based on gas explosion-induced ABLI
Generally, the smaller difference has positive correlation with the stability of the method and the better quality of the data. We observed a dense distribution of the QC samples, which is presented in the diagram of principal component analysis (PCA) score plots. The clustered and reproducible QC samples shown in supplemental file 1 A and C (https://figshare.com/s/acd53092653114b59993) indicated that the system is stable. For the purpose of quality control (QC), quality assurance (QA) is usually carried out to remove the characteristic peak of the poor repetitive features in the QC sample in order to obtain a higher quality data set. In this study, the proportion of characteristic peaks of QC samples with RSD < 30% could reach about 70%, which suggested that the precision and repeatability of the metabolomics platform was acceptable and sufficient to ensure the data quality for further metabolomics analysis (see supplemental file 1 B and D, https://figshare.com/s/acd53092653114b59993).
UPLC/Q-TOF-MS fingerprinting and multivariate analysis of gas explosion-induced ABLI rats’ serum samples
In this study, all rat serum samples from hours 48 collected were analyzed by UPLC/Q-TOF MS/MS in ESI+ and ESI− for serum metabolic profiling. Representative positive and negative chromatograms of total ion chromatography (TIC) of serum obtained from the standard and ABLI groups showed that the metabolites got successfully separation on the UPLC T3 column (Figure 3).

Representative positive (A, C) and negative (B, D) TIC chromatograms of serum at 48 hour obtained from the experimental sample (A, B) and test standard (C, D). TIC: Total Ion Chromatography.
To understand the subtle metabolic changes and to characterize the metabolite profile of the control and ABLI groups, unsupervised PCA and supervised PLS-DA models constructed based on the two groups were performed to identify the changes in metabolite profiles. As shown in the PCA score plot in Figure 4, the data plots of the two treated groups were thoroughly separated from those of the control group. The resulting score plot of PLS-DA (R2Y = 0.99 and Q2 = −0.1, Figure 5(A) and (B); R2Y = 0.94 and Q2 = 0.02, Figure 5 (C) and (D)) in the positive mode and negative (R2Y = 0.95 and Q2 = −0.23, Figure 6(A) and (B); R2Y = 0.97 and Q2 = −0.15, Figure 6 (C) and (D)) mode for the ABLI (160 m and 240 m) and the control groups gradually formed two separate clusters in the PLS-DA and OPLS-DA (see supplemental file 2, https://figshare.com/s/01c5038c05470b9c2e51) plots at the 48th hour after gas explosion. To evaluate the possibility of error that the present PLS-DA model might generate, one hundred times of the permutation test for PLS-DA were applied. All R2Y and Q2 values to the left were lower than the original points to the right (Figure 5 and Figure 6), showing that the PLS-DA models were valid. These results suggested that ABLI could lead to serum metabolite alterations in the rat model established in our experiment. Based on these results, the serum metabolite profiling of the rats was alleviated as the distance of the gas explosion increased. The aforementioned serum metabolic profiling supply people with valuable information for us to resolve for the diagnosis of gas explosion-induced ABLI.

PCA score plot derived from the UPLC/ESI-Q-TOF/MS of serum obtained from the control and ABLI groups. A: positive ion mode; B: negative ion mode. Control: (control group, red round); J160: (close-distance group, blue square); Y240: (long-distance group, yellow triangle). PCA: principal components analysis. ABLI: acute blast lung injury group. Each data point represents one subject.

The positive PLS-DA score plot and permutation test for PLS-DA derived from the UPLC/ESI-Q-TOF/MS of serum obtained from the control and ABLI groups. A and B: the control and close-distance group; C and D: the control and far-distance group. Control: (control group, red round); J160: (close-distance group, blue square); CY240: (long-distance group, yellow triangle). PLS-DA: partial least-squares discriminant analysis. ABLI: acute blast lung injury group. Each data point represents one subject. The R2Y value represents the goodness of fit of the model, and the Q2 value represents the predictability of the models.

The negative PLS-DA score plot and permutation test for PLS-DA derived from the UPLC/ESI-Q-TOF/MS of serum obtained from the control and ABLI groups. A and B: the control and close-distance group; C and D: the control and far-distance group. Control: (control group, red round); J160: (close-distance group, blue square); CY240: (long-distance group, yellow triangle). PLS-DA: partial least-squares discriminant analysis. ABLI: acute blast lung injury group. Each data point represents one subject. The R2Y value represents the goodness of fit of the model, and the Q2 value represents the predictability of the models.
Gas explosion-induced ABLI disturbed potential biomarkers in serum and metabolic pathway recognition
A number of related possible metabolites, which played vital roles in the separation, were screened based on the PCA analysis. The variable importance in the projection (VIP) from positive and negative loading plots is a major parameter for screening potential biomarkers. A variable is considered important for the model and as highly significant biomarker only when its VIP is above 1.5. Accordingly, five ions in serum from the positive mode were initially identified, while four ions in serum from the negative mode were initially identified. To elucidate the elemental composition of each biomarker, 9 biomarkers were confirmed by comparing their retention times and MS/MS fragmentation patterns using Q-TOF MS/MS. They were also identified by searching several free online databases. All biomarkers were determined based on exact mass, isotopic distribution, and mass spectra fragmentation patterns using MassFragment software. The m/z and retention time, postulated identity, elemental composition structural formula, and mass fragment information of identified biomarkers in this experiment are shown in Table 2. The identification figures, box figures, and bar figures of these metabolites identified in rat serum in the control and ABLI groups at the 48th hour after gas explosion are shown in supplemental files 3 (https://figshare.com/s/fa56e180966456710374), 4 (https://figshare.com/s/501dd603ee6fc5508e0d) and 5 (https://figshare.com/s/11f80de1e64e0b28dd8e), respectively. In a word, significant metabolomics alterations were observed in the serum of the rats, including increases in 7 metabolites and decreases in 2 metabolites (see supplemental file 6, https://figshare.com/s/8e0d9f638e3ed1ba5638). Compared with the control group, the intensity of 2-aminoadipic acid, L-methionine, L-alanine, L-lysine, L-threonine were significantly increased in the positive ion mode, but a significant decrease in the intensity of cholic acid and L-histidine was observed, whereas citric acid and aconitic acid were significantly increased in the negative ion mode in the short- and long- distance groups at 48 hours after gas explosion (P < 0.05).
Potential serum biomarkers of ABLI induced by gas explosion in UPLC/Q-TOF-MS positive and negative ion mode.

a Ions identified by comparison to the standard chemicals. bBiomarkers identified by the Human Metabolome Database (HMDB) and confirmed using exact mass data and MS fragmentation. ↑, above control levels; ↓, below control levels (P < 0.05 or P < 0.01).
Abbreviations: UPLC/Q-TOF-MS—ultra-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry.
The metabolic changes were mainly involved in amino acid, and biotin metabolism, primary bile acid biosynthesis, TCA cycle, lysine degradation, nitrogen metabolism, aminoacyl-tRNA biosynthesis, glyoxylate and dicarboxylate metabolism, glycine, serine and threonine metabolism, selenoamino acid metabolism, histidine metabolism, and cysteine and methionine metabolism (see supplemental file 7, https://figshare.com/s/fd60bbe4757134510009), which revealed a mechanistic basis for comprehending, ameliorating, or preventing gas explosion-induced adverse effects in the lung.
Discussion
Gas explosion-induced lung injury has a complex pathological process, individual reactions to gas explosion accidents depend on the intensity of the explosion and the distance from the source of the explosion. Numerous studies have demonstrated that the damage effects of gas explosion both existed in animal models and in humans. 16 –18 Epidemiological studies have suggested that gas explosion was related to multiple respiratory disorders, such as pulmonary fibrosis and chronic obstructive pulmonary disease. 3 However, the pathological mechanism underlying gas explosion-induced ABLI is still not completely clarified. Therefore, it is worth investigating the potential effects of gas explosion to ABLI in real roadway environment. Metabolomics analysis is a useful tool for understanding the pathogenesis of gas explosion-induced ABLI. 13 To examine the metabolite profiles changes of gas explosion-induced ABLI, we employed a non-targeted serum metabolomics analysis to identify differential metabolites affected by gas explosion.
The gathering of the exposed groups and the control group were divided completely via PLS-DA (Figure 5(A) and (B), Figure 6(A) and (B)). Thus, the metabolic profiles were altered after the gas explosion at 48 h. Moreover, the PLS-DA plots for the exposed groups gradually deviated from the control group with decreasing explosive distance. In the current study, our KEGG pathway analyses have identified 8 metabolic pathways that were significantly impacted by exposure to gas explosion. Nine metabolites in serum were screened by UPLC/Q-TOF/MS analysis. The changes in the intensities of these metabolites in each group in the positive and negative ESI modes are showed in Table 2. It is noteworthy that all of these impacted pathways were related to metabolism of amino acids. As shown in Table 2, the proteinogenic amino acids impacted by gas explosion include L-alanine, L-methionine, L-threonine, L-lysine and L-histidine. Close exposure to gas explosion significantly increased their circulating levels. Interestingly, although L-alanine, L-methionine and L-threonine belong to different classes of amino acids and have different biological functions, they have been shown to correlate with energy metabolism. Therefore, these impacts on amino acid metabolism by gas explosion may also reflect impairment of glucose homeostasis. However, further studies are still needed to determine whether these increases in circulating L-alanine, L-methionine and L-threonine are indicative of impaired glucose homeostasis in the context of gas explosion-induced ABLI.
The tricarboxylic acid cycle (TCA) is the metabolic pathway for the three major nutrients (sugars, lipids, and amino acids), and the most effective way to oxidize sugar or other substances to obtain energy. The first step in the TCA is the conversion of glucose or fatty acids to acetyl-CoA, followed by the condensation of acetyl-CoA with oxaloacetic acid to citric acid. 19,20 Aconitic acid and Citric acid are both derived from the TCA and are integral components of energy production. 21 Citric acid metabolism contributes to energy production by providing a major alternative pathway for nicotinamide adenine dinucleotide (NAD +) regeneration and allowing acetyl phosphate to yield acetate/adenosine triphosphate. Increased serum concentrations of citric acid and aconitic acid in our study indicated that enhanced TCA and energy generation to compensate for wound healing in injured lung tissue. Histidine is one of the 22 proteinogenic amino acid, and has anti-oxidant, anti-inflammatory and anti-secretory properties. 22 The efficacy of L-histidine in protecting inflamed tissue is attributed to the capacity of the imidazole ring to scavenge reactive oxygen species (ROS) generated by cells during acute inflammatory response. 22 Histidine, when administered in therapeutic quantities is able to inhibit cytokines and growth factors involved in cell and tissue damage (US patent 6150392). Asthma patients exhibit increased serum levels of histidine over normal controls. 23 Histidine appears to suppress pro-inflammatory cytokine expression, possibly via the NF-κB pathway, in adipocytes. 24 Recent studies have demonstrated that histidine is able to inhibit cytokines and growth factors involved in cell and tissue damage (US patent 6150392). Increased levels of L-histidine in this study suggested that gas explosion could induce inflammatory response to ALI, and L-histidine supplementation could inhibit cytokines and growth factors from cell and lung tissue damage. A study have indicated that ulinastatin has positive therapeutic effects on ALI in rats with severe burn-blast combined injuries through its good regulating effects on coagulation and fibrinolytic disorders. 25
Cholic acid is a major primary bile acid produced in the liver, and is considered to be the least hepatotoxic. 26 The liver toxicity of bile acids appears to be due to their ability to peroxidate lipids and to lyse liver cells. In our study, we observed that close exposure to the source of the gas explosion resulted in increased serum levels of cholic acid, suggesting that gas explosion might not only influence the cell energy balance by impairing glucose tolerance but also cause liver dysfunction after the gas explosion-induced ABLI. The L-methionine has effects on DNA methylation in rat liver during the early stages of hepatocarcinogenesis. 27 Increasing levels of L-methionine in the ABLI groups indicated that the immune system was impaired by the gas explosion-induced DNA methylation in the liver and might further affect the metabolism. Threonine is an essential amino acid in humans, which can also probably promote cell immune defense function. Increasing serum L-threonine concentrations causes neurological dysfunction in experimental animals. Such accumulation affects the neurotransmitter balance, which may have consequences for the immune system after the gas explosion-induced ABLI. A study have shown that apoptosis plays an important role in the occurrence and development of acute lung injury (ALI) in rabbits by participating in lung injury and promoting the progression of ALI. 17
The last marker analyzed to understand the metabolomics of gas explosion-induced ABLI was an L-lysine, which belongs to the class of foundational amino acids. L-lysine plays important roles in the immune system in the liver, and acts as an anti-inflammatory to the entire nervous system, especially the central nervous system. The increased serum concentration of L-lysine suggested that systematic inflammation occurred in the nervous system followed by close range exposure to the gas explosion-induced ABLI. In this study, we also found that the serum 2-aminoadipic acid concentration was significantly increased in gas explosion-induced rats compared with control rats, which was consistent with the results previous studies showing that increased serum 2-aminoadipic acid concentrations are associated with multiple types of peripheral nervous system, further indicated that gas explosion could induce ABLI via destroying the nervous immune system. A study have suggested that shock wave injury can cause inflammation, oxidative stress and apoptosis of lung tissue in mice, and chitosan oligosaccharide (COS) may have a protective effect on ALI. 28 Studies have suggested that elevated level of N-terminal modified poly (L-lysine)-antibody conjugate as a carrier for targeted gene delivery in mouse lung endothelial cells. 29 Early peritoneal dialysis can attenuate pulmonary edema and inflammation, and further prevent BLI after the injury. 30 Research has also shown that glycyl-l-histidyl-l-lysine-Cu possesses a protective effect in ALI by inhibiting excessive inflammatory responses through the suppression of NF-κB p65 and p38 MAPK signaling in vitro. 31 Another result showed that the explosive lung injury would be further reduced for the effectively inhibition of systemic inflammatory responses. 32 L-lysine acetylsalicylate improves the safety of paraquat formulation in rats by increasing its elimination and preventing lung injury.
In this study, we found that rats exposed to gas explosion at different distance points (160 m and 240 m) resulted in severe ABLI and changes in serum metabolism. Our results suggest that gas explosion has developmental consequences affecting not only the lungs but also metabolism. To assess these effects, we first compared the different pathological effects of gas explosion groups with the control group. Severe lung injury was observed when rats were set in different distance points of gas explosion, indicating with certainty that gas explosion can induce ALI. With the exposure distance increased, the degree of lung injury is reduced. The pulmonary histopathological scoring results were consistent with the pathological and general observation results. In addition, these results were also consistent with the above-mentioned metabolite analysis. Moreover, we have found that the gas explosion in the shock tube significantly induced the degeneration and desquamation of type II alveolus epithelia in our previous publications. 6 Another study also showed that dexamethasone-loaded hemostatic nanoparticles could alleviate the physiological deprivation caused by blast injury and reduce lung injury damage. 33
The present study provides a deep insight into the metabolic effect of gas explosion through the metabolomics analysis of the serum in a gas explosion rat model. However, several limitations should be noted. Firstly, the present study did not investigate the development metabolic effects of gas explosion-induced ABLI, which requires the metabolomics analysis of serum at a series of time points. Secondly, this study did not determine the components of gas explosion responsible for these metabolic effects. In a word, the current study is a little small to apply to a clinical human population for some potentially limited of just one animal study.
Conclusions
In a word, the UPLC-MS-based metabolomics platform could be a powerful tool for the metabolomics screening of serum metabolite candidates upon gas explosion. The gathering of the exposed groups and the control group were clustered completely via PCA and PLS-DA. The metabolomics identified 9 significantly metabolites different between the control- and ABLI rats. 2-aminoadipic acid, L-methionine, L-alanine, L-lysine, L-threonine, cholic acid and L-histidine were significantly increased in the exposed groups. Citric acid and aconitic acid were significantly decreased after exposure. The identified biomarkers not only reflect well-known adverse health effects of gas explosion such as inflammation and impairment of glucose homeostasis, but also provide novel pathophysiological changes and molecular mechanisms for the gas explosion-induced ABLI. These alterations in biomarkers in the aforementioned pathway are potential biological mediators and biomarkers for gas explosion-induced ABLI. In order to restore the perturbed or discriminated metabolites, some possible treatment or intervention strategies are proposed as follows: 1) Supplement patients with a single or multiple biomarkers already identified in this study to reverse possible adverse reactions; 2) Take clinical drug intervention, including traditional Chinese medicine and western medicine, for the perturbed biomarkers; 3) Intervention is performed with key proteins or enzymes that regulate these discriminated metabolites; 4) Some other effective clinical intervention measures should be taken in time, such as early peritoneal dialysis, early fluid therapy or complement inhibition, to further reverse these metabolic changes. After further validation of the identified biomarkers in gas explosion-induced ABLI population, we expect that these biomarkers might be useful for clinical diagnosis and treatment strategies of gas explosion-induced ABLI in the future.
Supplementary material
Supplemental Material, Supplementary_files - Gas explosion-induced acute blast lung injury assessment and biomarker identification by a LC-MS-based serum metabolomics analysis
Supplemental Material, Supplementary_files for Gas explosion-induced acute blast lung injury assessment and biomarker identification by a LC-MS-based serum metabolomics analysis by X Dong, S Yao, W Wu, J Cao, L Sun, H Li, H Ren and W Ren in Human & Experimental Toxicology
Footnotes
Abbreviations
Acknowledgments
Author’s contributions
R.W., C.J. and W.W. designed the study and wrote the protocol. D.X., Y.S. and L.H. established the animal model of ABLI and wrote the manuscript. D.X. and Y.S. helped with sample preparation and interpretation of the study. S.L. and R.H. performed the statistical analysis and helped with measurement of experimental indicators. All authors contributed to and have approved the final manuscript.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All experimental protocols and procedures were approved by the Animal Care and Use Committee of Xinxiang Medical University (Xinxiang, China).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (grant number: U1904209).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
