Abstract
Butylated hydroxyanisole (BHA) has been widely used in the cosmetics, pharmaceutical, and food industries due to its antioxidant activity. Despite the antioxidant effects, reported adverse effects of BHA at the cellular level have made its use controversial. In this regard, this study was performed to elucidate the potential toxicity mechanism caused by BHA at the molecular level in zebrafish embryos. For this purpose, zebrafish embryos were exposed to BHA at levels of 0.5, 1, 5, 7.5 and 10 ppm and monitored at 24, 48, 72 and 96 hours. Survival rate, hatching rate and malformations were evaluated. We examined the potential for reactive oxygen species (ROS) production and apoptosis signalling accumulation in the whole body. Moreover, we evaluated histopathological and immunohistochemical (8-OHDG) characterization of the brain in zebrafish embryos at the 96th hour. We also examined apoptosis, histopathological and immunohistochemical (8-OHDG) characteristics in 96 hpf zebrafish larvae exposed to tertiary butylhydroquinone (TBHQ), one of the major metabolites of BHA, at doses of 0.5, 2.5, 3.75 and 5 ppm. Consequently, it has been considered that increased embryonic and larval malformations in this study may have been caused by ROS-induced apoptosis. After 96 h of exposure, positive 8-OHdG immunofluorescence, degenerative changes, and necrosis were observed in the brain of BHA and TBHQ-treated zebrafish larvae in a dose-dependent manner. BHA and TBHQ exposure could lead to an increase in 8-OHdG activities by resulting oxidative DNA damage. In particular, the obtained data indicate that the induction of ROS formation, occurring during exposure to BHA and/or multiple hydroxyl groups, could be responsible for apoptosis.
Introduction
In recent years, there has been a great deal of interest in antioxidants used as additives that protect foods against textural and colour changes, bad odour and aroma, loss of quality and shortening shelf life as well as lipid oxidation which leads to loss of nutritional value. 1,2 The antioxidants may be found naturally in foods (ascorbic acid, alpha-tocopherol, flavonoids and phenolic acids etc.), whereas others are produced synthetically, such as butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), tertiary butylhydroquinone (TBHQ) and the gallates. 3 BHA has been widely used in the cosmetic and pharmaceutical industries as well as in the food industry for protection of foods against lipid oxidation and free radical formations. 4,5 Although BHA has been listed in the Generally Recognized As Safe (GRAS) by the U.S. Food and Drug Administration (FDA), studies have shown that BHA has been associated with malignant and benign tumours in the anterior stomach of rats, 6 cell degeneration with diffuse granulitic infiltration in dog liver, 7 inhibition in corticosteroid hydroxysteroid 11-beta dehydrogenase 2 enzyme activity, 8 lower androgen production in Leydig cells by inhibiting of 17-alpha-monooxygenase and hydroxy-delta-5-steroid dehydrogenase enzyme activity in rats 9 and promotion the bladder carcinogenesis initiated by nitrosamine in rats. 10 On the other hands, it has been reported that BHA has potential anticancerogenic 11 and antioxidative effects 12 at the cellular level. However, it has induced the irreversible induction in cell proliferation capacity before induction of apoptosis in Vero cells, a mammalian continuous cell lines commonly used in molecular and cell biology research. 13 Further, tert-butylhydroquinone, the major metabolite of BHA, caused the formation of 8-hydroxy deoxyguanosine (8-OHdG) which is known an oxidative stress biomarker in the calf’s thymus. 14 Apoptosis, also called programmed cell death, occurs as a result of a homeostatic mechanism that provides a balance between decreasing and increasing cell populations in tissues that occur normally during development and ageing. 15,16 On the other hands, apoptosis is an important process that neutralizes the DNA damaged cells caused by the cellular imbalance of reactive oxygen species (ROS) and thus prevents carcinogenesis. 17 The antioxidant defence system and oxidative stress have in perpetual balance in the living organisms as a consequence of normal homeostatic mechanism. One of the main sources of oxidative stress is the increased ROS that are also produced by xenobiotics taken into the body. 18,19 ROS is also produced in very small quantities for under normal physiological conditions during aerobic metabolism. When the level of ROS exceeds the level that the cellular antioxidant defence system can tolerate, it leads to oxidative stress with reduced cellular antioxidant capacity. 19,20 The detoxification of the ROS is achieved by the activation of enzymatic and nonenzymatic antioxidants both intracellularly and extracellularly. External factors such as smoking, 21 alcohol consumption, 22 UV radiation, 23 exposure to environmental pollutants 24 and over/wrong-dose food additives, 19,25 may disrupt the balance between the produced and detoxified ROS. Studies have been shown that excessive ROS have been implicated in the activation of various cellular signalling pathways including cell survival and/or cell death processes such as autophagy and apoptosis. 15,26 Under oxidative stress, the increase in hydroxyl radicals which are the most important oxygen-free radicals, leads to the formation of radical adducts. The resulting radical adducts lead to an electron abstraction by activating guanine from DNA nucleobases, resulting in the formation of C8-hydroxyguanine (8-OHGua) or its nucleoside, 8-OHdG. 25,27
Due to the toxicological data on the BHA, it has been accepted as ‘possibly carcinogenic to humans (2B)’ by the International Agency for Research on Cancer (IARC). 28 However, it is not included in United States Environmental Protection Agency (EPA) Cancer Classification. On the other hand, BHA is accepted as ‘Reasonably Anticipated to Be a Carcinogen’ in the National Toxicology Program (NTP) Carcinogen List. 29 FDA 30 reported that although there is insufficient evidence that BHA is a public health hazard when used at current levels, additional studies are needed for its effects on different enzymes and metabolic systems. It is largely believed that the root cause of many chronic diseases is free radicals and resultant oxidative stress induced by xenobiotics such as food additives. 31
Zebrafish (
Material and methods
Zebrafish maintenance and embryo treatment
AB strain zebrafish (
Apoptosis detection assay
Apoptotic cells were detected in 10 larvae selected randomly from each group exposed to BHA (control, 0.5, 1, 5, 7.5 and 10 ppm) and TBHQ (control, 0.25, 0.5, 2.5, 3.75 and 5 ppm) at 96 hpf using acridine orange (AO) staining, a nucleic acid selective metachromatic dye that interacts with DNA and RNA by intercalation or electrostatic attractions. 38,41 Larvae were rinsed three times with PBS and incubated in 5 mg/mL concentration and 2 mL amount of AO for 30 min in the dark at 28°C, followed by three times rinses in PBS. 42 Stained larvae were examined using fluorescence microscopy (Zeiss, Discovery V12, Germany). The fluorescence intensity of larvae was measured and quantified using ZEN 2 (blue edition) software (Zeiss, Germany) with excitation and emission wavelengths of 488 and 525 nm respectively.
Reactive oxygen species (ROS) detection assay
ROS were detected in 10 larvae selected randomly from each group exposed to BHA at 96 hpf. The compound chloromethyl-20,70-dichlorodihydrofluorescein diacetate (CM-H2DCFDA, Calbiochem) was used to monitor the accumulation of reactive oxygen species in zebrafish larvae. This molecule reacts with many different ROS, and it is a general indicator of oxidative stress (Invitrogen). Fluorescent DCF was formed through ROS oxidation. Selected larvae were washed with ultra-pure H2O three times, and excess water was removed. 43 Larvae were incubated in 1 μg/mL CM-H2DCFDA for 2 h under dark condition and washed again with ultra-pure H2O three times, then immobilized in 3% methylcellulose. 43 Images of live samples were captured using a fluoresce stereo microscope (Zeiss, Discovery V12, Germany) with excitation and emission wavelengths of 495 and 527 nm respectively.
Histopathological assay
The 96 hpf zebrafish larvaes were fixed in 4% paraformaldehyde. After routine alcohol and xylene procedure, they were blocked in the paraffin. Sections with a thickness of 4 µm from the blocks were left on the flotation bath for just long enough to flatten then promptly were picked up on a slide to dry polysine microscope slides. One of every five sections were taken and eight sections were collected from each larva in total. Hematoxylin-eosin staining method was applied to sections taken on normal slides. The brain was examined in samples belonging to each group under ×20 magnification under light microscope. (Olympus BX51, DP72). Three sections of the whole body of each animal (n = 10) were also scored the histopathological findings by another veterinary pathologist for blinded scoring. Score determination in histopathological assay was based on the severity as well as the number of slides out of the total in which the histological changes were observed with absent (−), minimal (+), mild (++), moderate (+++), severe (++++) degeneration and necrosis in neurons.
Immunofluorescence assay
Immunofluorescence signals were detected in 10 larvae selected randomly from each group (compounds-treated and control) at 96 hpf. These larvae were fixed in 4% paraformaldehyde. Routine tissue follow-up and embedded in paraffin blocks. The sections taken were dipped in 3% H2O2 for 10 min to block the endogenous peroxidase activity after deparaffinization. Then the slides were immersed in an antigen retrieval solution (pH 6.0) and heated in a microwave for 15 min to unmask antigens. Protein block were dripped onto the tissues to prevent non-specific binding. 8-OhdG (sc-66036, Santa Cruz, EU, mouse monoclonal antibody) primary antibodies were applied to the sections at a dilution of 1/100 and were incubated at room temperature for 1 h. Then, the immunofluorescent antibody (ab6785, Abcam, Goat Anti-Mouse IgG H&L (FITC)) was dropped on the sections at a dilution of 1/50 and kept in the dark and washed with water at room temperature for 45 min. Sections were examined under laser scanning confocal microscope (Zeiss LSM 710) at magnification of 10× with excitation and emission wavelengths of 495 and 519 nm respectively. Three sections of the whole body of each animal (n = 10) were also scored the intensity of the immunopositivity by another veterinary pathologist for blinded scoring. Sections were evaluated as absent (−), minimal (+), mild (++), moderate (+++) and severe (++++) according to their immune positivity.
Statistical analysis
Statistical analysis of the data obtained in the study was done using SPSS (SPSS Inc., Chicago, IL, USA) program. Descriptive statistics including mean and standard deviation were calculated. A one-way analysis of variance (ANOVA) with post hoc Tukey studentized range honestly significant difference (HSD) test was used to identify differences in all compound-treated groups.
For the analysis of the differences of the semiquantitatively obtained data in the histopathological examination, Kruskal–Wallis (a nonparametric test) was used. Mann–Whitney U test was also applied for the comparison of groups.
Results
Survival and hatching rates of embryo/larvae
To evaluate developmental toxicity effects of BHA and TBHQ on zebrafish embryo/larvae, the survival and hatching rates have been determined. As shown in Figure 1 (upper graph), survival rate decreased slightly in both BHA and its metabolite in a dose depended manner, but the decrease in survival rate at high doses was significant in both compounds (p < 0.05). There were no significant differences between BHA exposed and control groups at 48 and 72 hpf but significantly delay at 72 and 96 hpf in the groups exposed to BHA of 5 and 10 ppm dose in hatching rate (p < 0.05). On the other hand, according to the hatching results, it has been observed that its metabolite does not affect hatching as much as BHA (Figure 1, mid and lower graphs).

Comparative survival rate of 96 hpf zebrafish embryos exposed to BHA and TBHQ (Upper graph). Hatching rate of zebrafish embryos exposed to BHA and TBHQ for 96 hpf (mid and lower graphs). The hatching rate results showed a strong inhibition of hatching rate after embryos exposed to higher BHA concentrations (5, 7.5 and 10 ppm). Data are expressed as means ± S.D. from three independent experiments. (*p < 0.05; ANOVA, Tukey’s test) (n = 3; 40 embryos for each n).
Malformations of embryo/larvae
The typically malformations including pericardial edema (PE), yolk sac edema (YSE), body malformations (BM) (including tail malformation, short tail and head malformation) and curved body axis (CBA) were examined in BHA and TBHQ exposed groups and control groups at 24, 48, 72 and 96 hpf (Figures 2–3). There were no significantly differences between control groups and groups exposed to BHA up to 5 ppm, whereas significant increases in malformations were found in groups exposed to BHA 7.5 and 10 ppm when compared the control and dosed groups in terms of all malformations (p < 0.05) (Figure 3A). Similarly, TBHQ, one of the important metabolites of BHA, has been found to cause significant malformations at high doses (p < 0.05) (Figure 3C). Another interesting result is that BHA causes malformations earlier than its metabolite (Figures 3B, 3D).
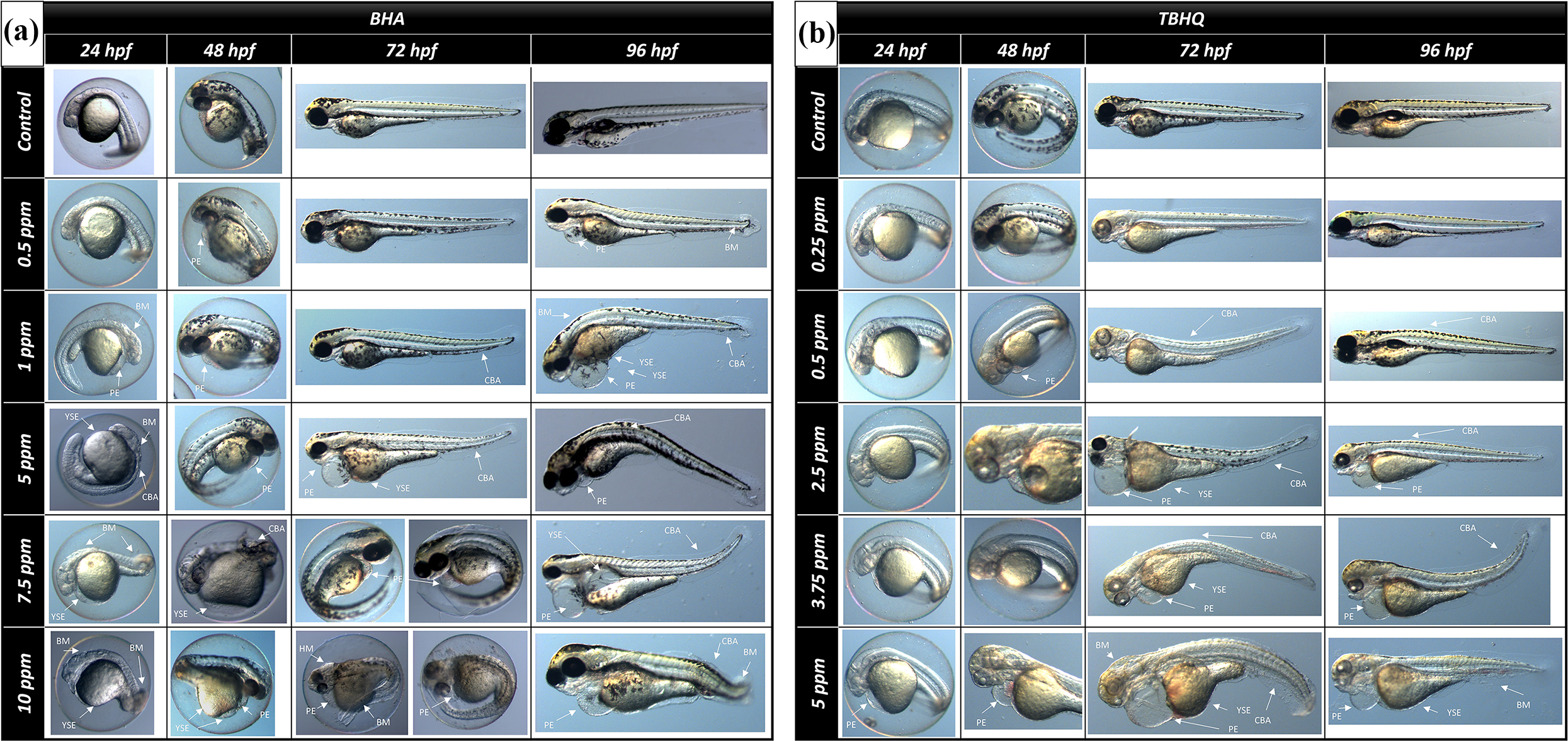
Microscopic images of embryos exposed to BHA (A) and TBHQ (B). YSE: yolk sac edema; PE: pericardial edema; CBA: curved body axis; BM: body malformation.

Malformation degree of zebrafish embryos exposed to BHA (A,B) and TBHQ (C,D). Data are expressed as means ± S.D. from three independent experiments (*p < 0.05; ANOVA, Tukey’s test). YES: Yolk sac edema; PE: pericardial edema; BM: body malformation; CBA: curved body axis.
ROS and apoptosis detection
We have used a fluorescent ROS indicator, CM-H2DCFDA, to determine whether the BHA causes production of ROS in the live larvae at 96 hpf. This indicator produces a green fluorescent when oxidized by any ROS. We detected the accumulation of fluorescence signal of ROS in through the whole body, especially the head region, in groups exposed to BHA (Figure 4). No fluorescence signalling was detected in control and 0.5 ppm groups while mild signalling was found in 1 and 5 ppm groups. Moreover, the signal was significantly increased in 7.5 and 10 ppm groups exposed to BHA (p < 0.05) (Figure 4).

CMH2DCFDA staining in vivo production of ROS in 96 hpf larvae exposed to BHA. (A: Control, B: 0.5 ppm, C: 1 ppm, D and H: 10 ppm, E and G: 7.5 ppm, F: 5 ppm). The immunopositively reaction of larvae was measured and quantified using ZEN 2 (blue edition) software. The immunopositivity were graded as absent (−), mild (+), moderate (++) and severe (+++).
Acridine orange (AO) that is a fluorescent marker of binding to DNA/RNA was used to determine whether exposure to BHA and TBHQ would lead to cause in cellular death in whole live larvae at 96 hpf (Figure 5). According to results, it would be said that exposure to both BHA and TBHQ have been resulted significantly increase in overall cellular death in a dose-dependent manner (p < 0.05).

Apoptotic cells were determined using acridine orange staining of BHA (A) and TBHQ (B) exposed larvae at 96 hpf. Whole larvae cell death images detected by fluorescence microscope. The fluorescence intensity of larvae was measured and quantified using ZEN 2 (blue edition) software. Data are expressed as means ± S.D. from three independent experiments. (*p < 0.05; ANOVA, Tukey’s test) (n = 3; 10 larvae for each n).
Histopathological findings and oxidative DNA damage detection
Histopathological examination showed that a normal histological structure of the brain was in the control and 0.5 ppm groups for BHA, and control and 0.25 ppm groups for TBHQ (Figure 6A, 6B). When the brain tissues of zebrafish exposed to BHA at doses 1, 5, 7.5 and 10 ppm were examined minimal, mild, moderate and severe levels of degeneration and necrosis were observed in the neuropils, respectively (Figure 6A). The severity of the changes was greater at the 10 ppm administered dose. In the brain tissues of zebrafish exposed to TBHQ at doses 0.5, 2.5, 3.75 and 5 ppm, minimal, mild, moderate and severe levels of degeneration and necrosis were detected in neuropils, respectively (Figure 6B). This difference observed between groups in terms of degeneration and necrosis was found statistically significant (p < 0.05).
8-OHdG immunostaining was performed to detect oxidative DNA damage. While immunopositivity was not observed in the control and 0.5 ppm groups, minimal, mild, moderate and severe signalling was observed in zebrafish larvae exposed to BHA at doses of 1, 5, 7.5 and 10 ppm, respectively (Figure 6A). Similarly, 8-OHdG immunopositivity was not observed in control and 0.25 ppm groups, whereas exposed to TBHQ at doses 0.5, 2.5, 3.75 and 5 ppm had minimal, mild, moderate and severe signalling, respectively (Figure 6B). This difference observed between groups in terms of immunopositivity was found statistically significant (p < 0.05).
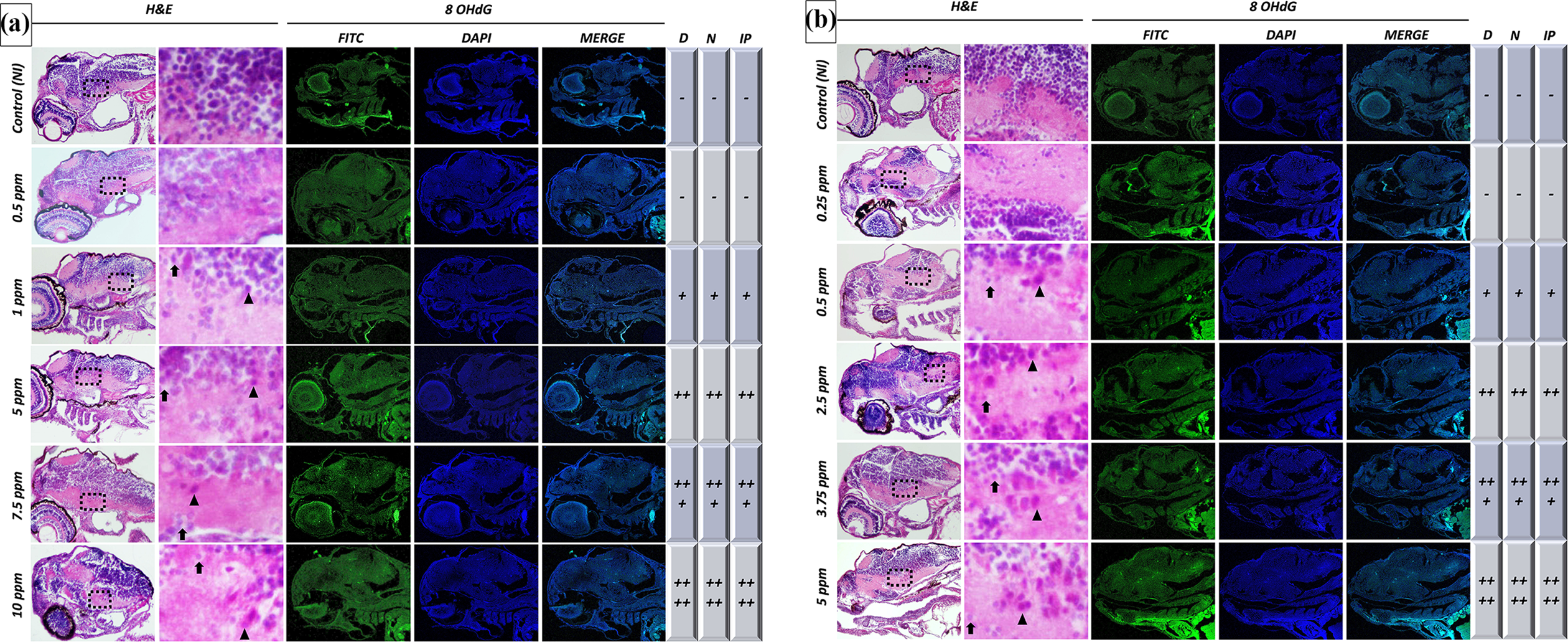
Histological findings and immunofluorescence reaction for 8-OHdG in brain tissues of zebrafish larvae exposed to BHA (A) and TBHQ (B). The first column contains pictures of histopathological findings, and the second column contains detailed pictures of these findings (necrosis and degeneration). ( ▽ : necrosis; → : degeneration). The third, fourth and fifth columns contain images of the immunopositive reaction for 8 OHdG. The last three columns contain scoring of all findings (D: degeneration, N: necrosis, IP: immunopositivity). The findings were graded as absent (−), minimal (+), mild (++), moderate (+++), severe (++++).
Discussion
Although BHA has been listed as Generally Recognized As Safe (GRAS), European Food Safety Administration (EFSA)
44
has determined this food additive to have an acceptable daily intake (ADI) value of 1 mg/kg bw day for both adult and children population based on possible endocrine disrupting and carcinogenic effects. Due to the conflicts on the usage of BHA, it is important that to obtain more informative and reliable data from in vivo studies on potential adverse effects of this food additive.
45
In this regard, zebrafish (
The obtained data show that BHA and/or TBHQ causes malformations, mortality and delayed/blocked hatching rates in a dose-dependent manner in zebrafish embryos during the embryonic developmental stages. Different levels of body malformations including pericardial edema (PE) which is the most common abnormality, yolk sac edema (YSE), body malformation (BM) and curved body axis (CBA) were observed in the embryos exposed to both compounds. Developmental toxicity studies on zebrafish suggested that one of the main reasons for the malformations, especially the occurrence of PE, may have been due to the response to the toxic environment. 48 –50 Moreover, PE often occurred in developing embryos exposed to toxic agents has been interpreted as an indicator of a defective cardiovascular system in zebrafish. 48,49 Hatching is another sensitive indicator for assessing the toxicity of potential toxic agents, and delay or failure at this stage may be due to the fact that the embryo cannot break the chorion because of the external or internal factors. 51 The observed malformations in the present study may partly or wholly affect the embryos ability to break chorion with decreased hatching rate in a dose-dependent manner. Interestingly, however, we found that the rate of hatching of the metabolite does not affect BHA as much. This may be explained by the fact that BHA may have an effect on delaying hatching with pathways other than the metabolite of TBHQ.
Also, previous studies introduced that another main reason for malformations is apoptosis. 25,50,52 There is a correlation between the mechanism of apoptosis induction with overall tissue damages. 53 According to our results, the accumulation of fluorescence signal from apoptotic cells in the whole body by acridine orange staining was increased in a dose-dependent manner at the end of exposure time to BHA. In a study, it has been introduced that over intake of BHA has capable of inhibiting cell growth and inducing apoptosis by tyrosine phosphorylation-dependent signal transduction pathway. 54 Similarly, Labrador and colleagues suggested that BHA caused induction of apoptosis in isolated rat hepatocytes and human tumour cell lines by way of caspase activation and direct release of cytochrome C. 13 One of the possible pathways of toxicity caused by BHA may be non-conjugated and tert-butylhydroquinone (TBHQ) caused by degradation of BHA via induction or inhibition of human BHA-specific phase-I and -II metabolizing enzymes or biological accumulation of BHA and/or metabolites in the body. 55 Indeed, several studies have shown that TBHQ has cytotoxic effects on mouse 3T3 cells at high concentrations, 56 lethal effects on rat thymocytes induced by hydrogen peroxide (H2O2) at sublethal concentrations (30 μM or less), 57 cytotoxic effects on both A549 and human umbilical vein endothelial cells (HUVEC cells) in a dose- and time-dependent manner, 58 Furthermore, the increasing of ROS and direct interactions of TBHQ with chromatin are two important possible causes of genotoxic effect and apoptosis induction. 58 In this study, it was also evaluated and confirmed that TBHQ, the metabolite of BHA, which we consider as a hypothesis, may lead to induction of apoptosis.
Excess exogenous ROS produced by xenobiotics can disrupt balance being between produced and detoxified ROS by the antioxidant system under normal physiological conditions. 59,60 Higher ROS levels are associated with oxidative damage to DNA, fatty acids, and other cellular components. 61 The ROS-induced oxidative stress by causing damage at the cellular level is implicated in carcinogenesis, atherosclerosis, neurodegeneration, diabetes, diseases associated with ischaemia, ageing, etc. 62 –64 The present work demonstrated that ROS increased in a dose-dependent manner by using CM-H2DCFDA. The imbalance of ROS levels during development lead to cellular damage on embryos by increasing levels of lipid peroxide, protein oxidation, and DNA damage. It is reported that there is a direct relationship between increased H2O2 production and damaged embryos, which suggests that ROS can induce apoptosis in the embryos. 65
Apoptosis occurs normally during development and ageing and as a homeostatic mechanism to maintain cell populations in tissues. 16 In this point, to clarify the main reason of apoptosis whether occurs as a result of the homeostatic mechanism or DNA damage resulted of imbalance of ROS levels, 8-OHdG has been used as a biological marker. As known, 8-OHdG, a product of oxidized DNA or RNA, is a good marker of oxidative cellular damage. 66 According to our results, BHA has caused 8-OHdG immunofluorescence positivity in brain tissues in zebrafish larvae, in a dose-dependent manner except 0.5 ppm exposure. Hydroxyl radicals known to cause damage to basic biomolecules and resulting in ROS end-occlusion lead to the formation of hydroxylation-causing 8-OHdG or 8-OHG at the C-8 position of deoxyguanosine or guanosine residues in DNA or RNA. 67 In this regard, 8-OHdG has been a potential biomarker used to establish carcinogenesis due to its relatively easy occurrence and promutogenic properties. 27,66 For this reason, it appears that 8-OHdG formation is associated with ROS production, indicating that ROS may lead to 8-OHdG formation. 67 This evidence suggested that the increase in tissue 8-OHdG activity may have emerged as an increased oxidative stress response. Nagai, Okubo 14 reported that TBHQ, which resulted in oxidative demethylation of BHA, significantly increased the level of 8-OHdG in the calf thymus. Another study 68 reported that BHA was a potent inducer of oxidative DNA damage in glandular stomach epithelial cells and that levels of 7-hydro-8-oxo-2-dioxiguanosine (8-oxoDG), which is known to be activated by the keto-enol tautomerism of 8-OHdG, was elevated depending on the prolongation of the exposure time to BHA. It is thought that there may be a physiological relationship between 8-OHdG activity in the larvae of zebrafish exposed to BHA and histopathological findings in tissues. To confirm this hypothesis, it has been observed that 8-OHdG fluorescence intensity increase in a dose-dependent in zebrafish larvae exposed to TBHQ. In our study, degenerative changes and necrosis in histopathologically observed brain tissues of zebrafish exposed to BHA except 0.5 ppm exposure. Similar effects were observed in TBHQ exposed groups in a dose-dependent manner.
Conclusion
BHA is a synthetic antioxidant substance widely used in the food, cosmetic and pharmaceutical industries. BHA can be transformed into different and more toxic compounds by some mechanisms. However, it enters the biological process directly through the consumption of food products and various drugs, leaving negative effects on human health at high doses. On the other hand, the evaluation of the standards related to its use by different institutions and organizations causes confusion. Eventually, there is a conflict on the usage of BHA in literature. In the present study, the potential developmental toxicity mechanism of BHA was tried to be clarified at the molecular level by using zebrafish which is one of the important model organisms widely used in toxicity studies. Taking all these results into account, it has been envisaged that BHA and/or TBHQ, which are activated due to BHA metabolism in the body, will likely trigger the induction of ROS formation and thus induce apoptosis. Finally, we suggested that the BHA usage should be well-controlled in all field especially food industries. In addition, more studies may be necessary and useful to change the attitudes of industries towards the use of BHA as a harmless preservative, which may lead to more cautious application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially supported by the Scientific and Technological Research Council of Turkey (TUBİTAK, Project no: 218O058), thus the authors thank TUBITAK for this financial support.
