Abstract
Lithium is one of the most powerful and commonly used medications for the treatment of various psychiatric diseases, especially bipolar disorder. However, it has a narrow therapeutic index with toxic effects on various organs. There are several case reports of lithium-induced arrhythmia and ischemia. The current work aimed to study the toxic effects of lithium on the heart of adult albino rats and its molecular mechanisms and the ameliorating effect of N-acetyl cysteine (NAC). Sixty adult male Wistar albino rats were classified into four groups; control, NAC-treated received NAC 500 mg/kg/week dissolved in 1 ml 0.9% sodium chloride intraperitoneal, lithium-treated received 52.5 mg/kg/day of lithium carbonate dissolved in 1 ml 0.9% sodium chloride orally by gavage, and lithium-and-NAC-treated (group IV) received lithium and NAC in the previous doses. After 12 weeks, the rats of group III showed a significant accumulation of ascites and a decrease in the mean arterial blood pressure and electrocardiographic (ECG) findings of ischemia and arrhythmia. In addition, there was an elevation in cardiac biomarkers creatine kinase MB (CK-MB), cardiac troponin I (cTnI), and several histological lesions with a significant increase in the area % of Van Gieson, endothelial nitric oxide synthase (eNOS), and 8-hydroxy-2′-deoxyguanosine (8-OHdG) immunoreaction. There was significant upregulation of microRNA-1, microRNA-21 (miRNA-21), and microRNA-29 (miRNA-29). MiRNA-21 was strongly positively correlated to the area % of 8-OHdG, while miRNA-29 was strongly positively correlated to the area % of Van Gieson staining. NAC significantly improved the cardiotoxic effects of lithium. Being a nontoxic and safe antioxidant, NAC can be used to ameliorate lithium-induced cardiac injury.
Introduction
Lithium (meaning Stone in Greek) was discovered in 1811 by Arfwedson. 1 Lithium carbonate (Li2CO3), a salt of lithium, is a psychiatric medication that has been used effectively in the acute and long-term treatment of bipolar affective disorders for seven decades. Since its discovery, lithium was used to treat urinary calculi and gout with minimal success, until Cade 2 reported its antimanic effect. 3
Lithium has many other therapeutic uses including schizoaffective disorder, impulse control disorder, attention deficit disorder, eating disorder, aggression, refractory depressive disorder, long-term prophylaxis of cluster headache, certain subsets of alcoholism, and dermatological disorders such as eczematoid dermatitis, genital herpes, and seborrheic dermatitis. 4
It is water soluble and readily absorbable after oral administration where peak level is reached after 2–4 h. 5 It poorly binds to low- and high-molecular-weight plasma proteins, although it strongly binds to very low-molecular-weight legends and passes very slowly from extracellular to intracellular compartment so it requires about 6–8 days to reach steady-state concentration and therapeutic blood level. 6 Lithium distributes almost uniformly in the body organs and accumulates against a concentration gradient in different tissues like the brain, kidney, thyroid, liver, muscles, and bone. About 95% of absorbed Li2CO3 is excreted in urine, 4–5% in sweat, and about 1% in feces. 5
Despite its remarkable efficacy, lithium has a narrow therapeutic index and leads to various side effects and often toxicity involving various body organs. 7 Its decreased excretion and gradual accumulation is a common event that can be predisposed by impaired renal function, volume depletion, drug interactions, and concurrent illnesses, such as congestive heart failure and cirrhosis. 8
Lithium affects a wide range of cellular functions through inhibiting the production of inositol, and it also affects the protein kinase C signaling pathway and inhibits glycogen synthase kinase 3. 9
Although there are several human reports demonstrating that lithium induces cardiotoxicity, 10 –15 there are minimal systemized studies of lithium-induced cardiac effects and their underlying mechanisms. The cardiotoxic effects of lithium range from simple electrocardiogram (ECG) abnormalities to dysrhythmias, cardiomyopathy, and even acute myocardial infarction (AMI). 16 Shah et al. demonstrated that therapeutic doses of lithium can result in duration-dependent histopathological changes in the heart of albino rats. 17 Several studies indicated that part of lithium toxicity is mediated by oxidative stress. 18 –20 Moreover, Allagui et al. detected that lithium can downregulate genes coding for antiapoptotic gene BAG-1 causing toxicity since these proteins are essential for cell survival. 21
N-acetyl cysteine (NAC), the precursor of glutathione (GSH), increases the cellular content of GSH, stabilizes cell membranes, scavenges reactive oxygen species (ROS), protects cell viability, and prevents ROS-induced apoptosis. 22 Several studies have reported that the antioxidant properties of NAC can provide cardiac protection through the scavenging of free radicals. 23 The cardioprotective effect of NAC has been proved against several toxins as cisplatin 24 and cyclophosphamide. 25
The aim of this work is to study the toxic effects of lithium on the heart of adult male Wistar albino rats and the possible ameliorative role of NAC against these cardiac changes.
Materials and methods
Chemicals
Li2CO3, CAS number 554-13-2, was obtained from Sigma-Aldrich, Egypt, in the form of white crystalline powder.
NAC, CAS number 616-91-1, was obtained from Sigma-Aldrich, Egypt, in the form of white to off white powder.
Animals
Sixty adult male Wistar albino rats of average weight between 170 g and 200 g were obtained from the animal house of the Faculty of Medicine, Zagazig University. Before commencing the experiment, the animals were subjected to 10 days period of passive prelimination to adapt themselves to the new environment, to ascertain their physical well-being, and to exclude diseased animals. Food was offered in equal amounts to all rats in each cage, water was offered in separate clean containers.
All rats received care in compliance with the guidelines of the Medical Research Ethics Committee of Zagazig University, Egypt.
Study protocol
Rats were randomly divided into four groups: Group I (control group): this group is divided into three subgroups: Subgroup Ia (negative control group): 10 rats received water and food for 12 weeks to measure the basic parameters. Subgroup Ib (positive control 1): 10 rats, each rat received 1 ml 0.9% NaCl/day orally by gavage for 12 weeks. Subgroup Ic (positive control 2): 10 rats, each rat received 1 ml 0.9% NaCl/day orally by gavage for 12 weeks, in addition to intraperitoneal injection of 1 ml 0.9 NaCl at the start of each week. Group II (NAC-treated group): 10 rats, each received NAC 500 mg/kg/week dissolved in 1 ml 0.9% NaCl intraperitoneal for 12 weeks.
24
Group III (lithium-treated group): 10 rats, each received 52.5 mg/kg/day of Li2CO3 (1/10 LD50) dissolved in 1 ml 0.9% NaCl orally by gavage for 12 weeks.
26
Group IV (lithium-and-NAC-treated group): 10 rats, each received 52.5 mg/kg/day of Li2CO3 dissolved in 1 ml 0.9% NaCl orally by gavage for 12 weeks, at the start of each week of Li2CO3 treatment rats were given 500 mg/kg NAC dissolved in 1 ml 0.9% NaCl intraperitoneal.
At the end of the study, all rats were subjected to: Physical examination: The mortality rate, body weight, physical activity, and presence of ascites were recorded. Measurement of mean arterial blood pressure (MABP): MABP was measured in millimeters of mercury (mm Hg) using a noninvasive blood pressure (BP) monitor (NIBP 250, with a serial number 21202-180, BIOPAC System, Inc., Goleta, California, USA). ECG: The overnight fasted (8–10 h) rats were anesthetized with urethane (1.5 gm/kg intraperitoneal)
27
and placed on a suitable rodent surgical table or a flat movable surface. The surface was not electrically conductive. Three bipolar ECG leads were used in electrocardiography. Positive, negative, and reference ECG electrodes were placed at the left foreleg, right foreleg, and left thigh, respectively, to record ECG using PowerLab 4/20 (data acquisition system) with MLT844 physiological pressure transducer with clip-on BP domes (AD Instruments Pty Ltd, Australia). Calibration of the voltage (millivolts) was performed, and the results were automatically calculated based on the calibration value Biochemical analysis: Blood samples were collected from the retro-orbital plexuses of the anesthetized rats using a capillary glass tube according to the method described by Schemere (1967). The collected blood was left for spontaneous coagulation and serum was separated by centrifugation of blood 3000 r/min for 10 min and stored at −20°C for later estimation of: Cardiac troponin I (cTnI) level (ng/ml): cTnI was measured calorimetrically by enzyme-linked immunosorbent assay (ELISA) using Life Diagnostics, rat cTnI ELISA kit (cat. No. CTNI-2-HS) according to the manufacture’s instruction where cTnI reacts with and becomes sandwiched between the solid phase and horseradish peroxidase (HRP)-conjugated antibodies producing color. The concentration of cTnI is proportional to color absorbance measured at 450 nm. Creatine kinase MB (CK-MB) level (ng/ml): CK-MB was measured by ELISA using rat CK-MB ELISA kit (cat. No KT-12247) from Kamiya Biomedical Company according to the kit instructions. The enzyme–substrate reaction produces color which can be measured spectrophotometrically at a wavelength of 450 nm ± 10 nm. Measurement of microRNA-1 (miRNA-1), microRNA-21 (miRNA-21), and microRNA-29b (miRNA-29b) expression in cardiac tissue using quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR): After obtaining blood samples, rats were killed and part of the left ventricle of each rat was obtained and immediately stored in liquid nitrogen at −80°C. The tissue samples were homogenized in 1 ml of TRIzol™ Reagent (Invitrogen, Carlsbad, California, USA) per 50–100 mg of tissue using a tissue homogenizer.
28
mirVana™ miRNA Isolation Kit from Ambion, Inc was used for microRNA (miRNA) isolation according to manufacturer’s instructions. The RNA quantity and purity were determined by the Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, California, USA). Estimation of miRNA-1, miRNA-21, and miRNA-29b was performed by qRT-PCR on complementary DNA produced from 200 ng of the whole RNA using mirVana qRT-PCR miRNA Detection Kit (Ambion, Inc). The used primers were designed by the Primer-BLAST tool (https://www.ncbi.nlm.nih.gov/tools/primer-blast/index.cgi) according to the miRNA sequence obtained from GenBank as given in Table 1. All specimens were run in duplicates using the master mix described in the kit instructions in ABI 7500 Real-Time PCR System (Applied Biosystems, Foster City, California, USA). PCR amplification consisted of 40 cycles of denaturation at 95°C for 3 min, annealing at 95°C for 15 s, and extension at 60°C for 30 s. The cycle threshold (Ct) values were determined by the SDS 2.0.1 software (Applied Biosystems) supplied by the thermal cycler manufacturer. U6 primers were used as an internal control for RNA template normalization where the relative gene expression was calculated according to the
Sequence, forward, and reverse of the primers for miRNA-1, miRNA-21, miRNA-29, and the control U6 primer.

MiRNA: microRNA.
Histopathological and immunohistochemical study: Parts from left ventricle of each rat were obtained and fixed in 10% neutral formalin, embedded in paraffin, and sectioned at a thickness of 5 mm, then these sections were subjected for staining by the hematoxylin and eosin stain for examining the overall morphology and Van Gieson stain for inspection of collagen fibers according to the method described by Bancroft and Layton. 30
Four-micrometer thick sections of formalin-fixed, paraffin-embedded heart tissue from each rat were obtained then subjected to dewaxing, rehydration, and blocking of the endogenous peroxidase activity by incubation with hydrogen peroxide 0.1% for 10 min. Antigen retrieval was performed in a microwave by 10 mm citrate buffer (pH 6.0) for 10 min and then cooled to room temperature. Then, incubation with the primary antibody was performed overnight at 4°C, followed by HRP-labeled secondary antibodies for 30 min at room temperature. The primary antibodies used were 8-hydroxy-2′-deoxyguanosine, 8-OHdG (15A3), mouse monoclonal antibody (catalog no. sc-66036, Santa Cruz Biotechnology, Inc., Dallas, Texas, USA) for 8OHdG and nitric oxide synthase 3 (C-6), mouse monoclonal antibody (catalog no. sc-376542, Santa Cruz Biotechnology, Inc., USA) for endothelial nitric oxide synthase (eNos).
The sections were incubated with diaminobenzidine tetrahydrochloride and counterstained with hematoxylin. Mouse brain extract (catalog no. sc-2253) was used as a positive control. Negative controls were done using the same tissue with the omission of the primary antibody. 31
Sections were viewed using an Olympus microscope (C5060-AUD, 5H01155, Japan) and images were captured by a digital camera (Canon PowerShot A620, UK).
Two independent pathologists examined the slides; the examination was done blindly to the groups and other results of the study. The reported results were the mean of results of the two pathologists.
Morphometric study: The mean area percentage (area %) of the collagen fibers between cardiac muscle fibers in Van Gieson stained sections and the area % of the positive immune reaction in 8-OHdG and eNOS immune stained sections were calculated in the Image Analysis Unit, Pathology Department, Faculty of Dentistry, Cairo University using the image analyzer computer system Leica Qwin 500 (Leica Ltd, Cambridge, UK). Ten readings were obtained from five sections for each rat of the randomly selected five rats out of each group. The total magnification of 400× was used.
Statistical analysis: Data were collected, tabulated, and managed using Statistical Package for Social Science version 16 (SPSS Inc., Chicago, Illinois, USA). Quantitative data were expressed as a mean ± standard deviation; differences among the groups were analyzed through a one-way analysis of variance (ANOVA) followed by the Turkey post hoc test for intergroup comparisons difference. χ2 was used to estimate the statistical significance of the categorical variables. A p value of less than 0.05 was considered statistically significant.
Results
There was no statistically significant difference between the negative control subgroup (Ia), the positive control subgroup (Ib), and the positive control subgroup (Ic) regarding all the measured parameters so the negative control one was used in the statistical comparison to other groups of the study.
Physical examination
The reported mortality at the end of the study was one rat from each of the control, and lithium-and-NAC-treated groups and two rats from the lithium-treated group with no significant difference between the groups (Table 2). The physical examination of rats of each group at the end of the study revealed that rats of the lithium-treated group were apparently weaker, sicker, and lethargic. As given in Table 2, there was a slight increase in the body weight of lithium-treated group when compared to other groups, although this difference was not statistically significant. In addition, there was a significant accumulation of ascites in the lithium-treated group when compared to other groups.
Statistical comparison among different studied groups of the mortality rate and ascites by χ2 test and body weight and MABP by one-way ANOVA with Tukey HSD post hoc test.

MABP: mean arterial blood pressure; ANOVA: analysis of variance; SD: standard deviation.
a Versus negative control.
b Versus NAC-treated group.
c Versus lithium-treated group.
d Significant.
MABP and ECG
As shown in Table 2, the MABP was significantly lower in the lithium-treated group when compared to the control group and the NAC-treated group. In addition, the concurrent NAC treatment with lithium led to a significant improvement of the MABP.
Regarding the ECG recording of rats, the control group shows normal ECG recording. The lithium-treated group showed ischemic changes in the form of depressed ST segment, decreased the amplitude of R wave, and narrow PR interval. In addition, rhythm disturbances in the form of sinus tachycardia, extrasystoles, and ventricular dysrhythmia were also recorded. The rats treated by combined lithium and NAC showed normal ECG (Figure 1).

(a) The ECG tracing of the control group showing normal findings, (b) to (e) show the abnormal ECG findings detected in lithium-treated group where (b) shows depressed ST segment, (c) shows increased heart rate, decreased amplitude of R wave, and narrow PR interval, (d) shows abnormal rhythm in the form of atrial extrasystoles, and (e) shows ventricular arrhythmia. (f) The normal ECG finding of lithium-and-NAC-treated group. ECG: electrocardiogram.
Biochemical results
Table 3 shows that cTnI was significantly higher in the lithium-treated group when compared to control, NAC-treated, and lithium-and-NAC-treated group. The Turkey HSD post hoc test revealed that the lithium-and-NAC-treated groups are significantly higher than the control (p < 0.05) and the NAC-treated group (p < 0.01).
Regarding CK-MB levels, Table 3 shows that the mean values of CK-MB were significantly higher in the lithium-treated group when compared to control, NAC-treated and lithium-and-NAC-treated group. There was no significant difference between the lithium-and-NAC-treated group and the control or NAC-treated group.
Statistical comparison among different studied groups of the cTnI level (ng/ml) and CK-MB level (ng/ml) by one-way ANOVA with Tukey HSD post hoc test.
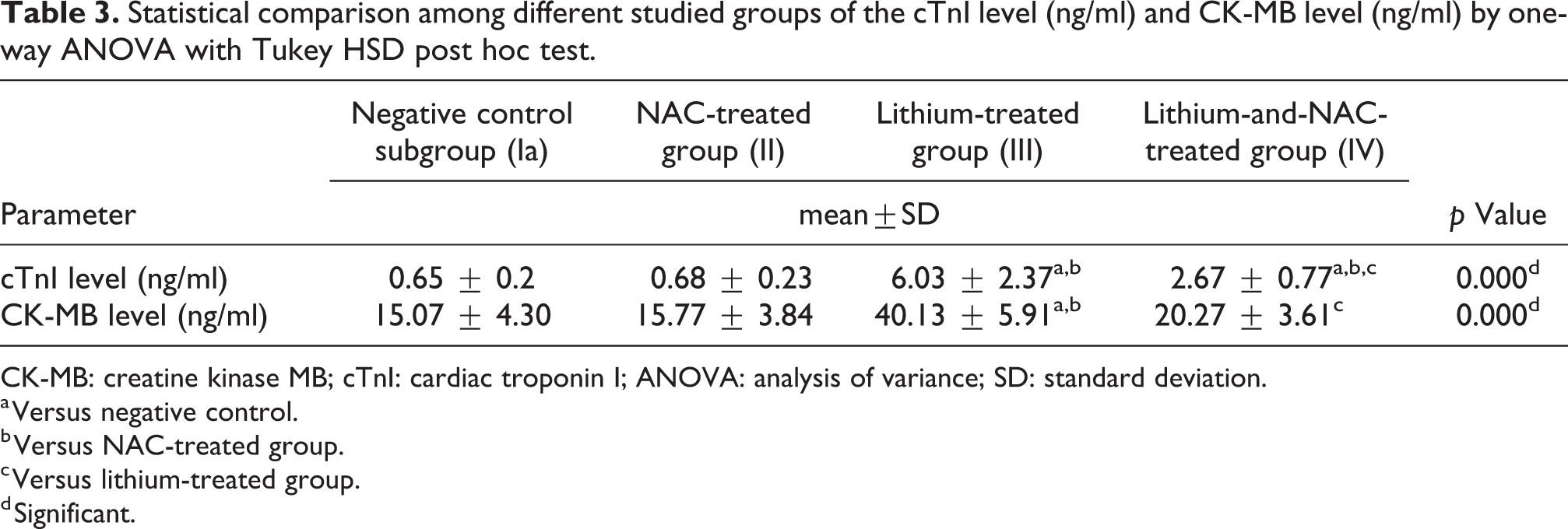
CK-MB: creatine kinase MB; cTnI: cardiac troponin I; ANOVA: analysis of variance; SD: standard deviation.
a Versus negative control.
b Versus NAC-treated group.
c Versus lithium-treated group.
d Significant.
MiRNA-1, miRNA-21, and miRNA-29b expression in cardiac tissue
In Table 4, the lithium-treated group shows a significant fold increase in miRNA-1, miRNA-21, and miRNA-29 relative gene expression with mean values of 2.7, 3.6, and 3.5, respectively. The lithium-and-NAC-treated group showed a significant difference when compared to both NAC-treated and lithium-treated group (p < 0.05).
Statistical comparison among different studied groups of the miRNA-1, miRNA-21, and miRNA-29 gene expression by one-way ANOVA with Tukey HSD post hoc test.a

MicroRNA: miRNA; ANOVA: analysis of variance; SD: standard deviation.
a Results are expressed as mean fold change ± SD.
b Versus NAC-treated group.
c Versus lithium-treated group.
c Significant.
Histopathological and immunohistochemical results
In the current work, the hematoxylin-and-eosin-stained left ventricular tissue sections of the control and NAC-treated groups showed the normal appearance of the branching cardiac fibers which contain acidophilic cytoplasm and central vesicular oval nuclei. The spaces between them are narrow containing fibroblasts and blood capillaries, while the stained sections from the lithium-treated group showed several abnormalities in the form of separation between the cells with congested capillaries surrounded by acidophilic homogenous material and cellular infiltration. Hyaline degeneration with degenerated cardiomyocytes, pyknotic nuclei, and vacuolation of the cells was also appearing. The co-treatment with NAC in group IV led to a marked improvement in these abnormalities where the sections appeared nearly normal apart from a minimal separation between the cells and few pyknotic nuclei as shown in Figure 2.

(a) The left ventricular myocardium of the control group showing normal branching cardiomyocytes with acidophilic cytoplasm, d, and central vesicular oval nuclei, n, and the spaces between cells are narrow containing fibroblasts, f, and blood capillaries. (b) Section from the left ventricular myocardium of lithium-treated group showing separation between the cells (star) with congested capillaries, c, surrounded by acidophilic homogenous material (arrow) and cellular infiltration, f. (c) Another section from the lithium-treated group showing congested capillaries, c, with hyaline degeneration, h, degenerated cardiomyocytes, d, pyknotic nuclei, p, and vacuolation of the cells, v. (d) Section of the left ventricular myocardium of lithium-and-NAC-treated group showing that the cardiomyocytes preserve the normal architecture except for a few muscle fibers with small dark nuclei, p, and minimal separation (star) (H&E ×400). NAC: N-acetyl cysteine; H&E: hematoxylin and eosin.
As shown in Figure 3, the Van Gieson-stained sections of the control group showed fine few collagen fibers between cardiac myocytes while in the lithium-treated group there was clearly observed an increase in the collagen fibers in the interstitial spaces. Group IV concurrently treated with lithium and NAC showed fewer collagen fibers. These findings were approved and confirmed by the morphometric measurement of area % of the collagen fibers between cardiomyocytes which was significantly higher (p < 0.01) in the lithium-treated group (55.12 ± 12.22) when compared to the control (12.75 ± 1.3), NAC- (14.65 ± 3.1), and lithium-and-NAC-treated groups (18.27±3.9). There was no statistically significant difference between the lithium-and-NAC-treated group and control or NAC-treated group (p > 0.05).

Van Gieson-stained sections of the left ventricle of (a) the control group showing few fine collagen fibers between cardiac myocytes (arrows), (b) the lithium-treated group showing increased collagen fibers in the interstitial spaces between cardiac myocytes (arrows), and (c) the lithium-and-NAC-treated group showing few fine collagen fibers between cardiac myocytes (arrows) (Van Gieson ×400). (d) Column chart showing the comparative magnitude of the mean values of the area % of collagen fibers in the studied groups. NAC: N-acetyl cysteine.
The immunohistochemical (eNOS)-stained sections of the control group showed mild positive immunoreaction and also those of the lithium-and-NAC-treated group while eNOS-stained sections of the lithium-treated group showed strong positive immunoreaction as shown in Figure 4. Statistical comparison of the area % of the positive eNOS immunoreaction showed that the lithium-treated group (47.18 ± 11.13) was significantly higher than (p < 0.01) the control (19.21 ± 4.29), NAC- (18.86 ± 3.84), and lithium-and-NAC-treated groups (26.01 ± 6.46). There was no statistically significant difference between the lithium-and-NAC-treated group and control or NAC-treated groups (p > 0.05).

Immunohistochemical (eNOS) stained section of the left ventricle of (a) the control group showing positive immune reaction in the cytoplasm of cardiac fibers (arrow), (b) the lithium-treated group showing strong positive immune reaction in the cytoplasm of cardiac fibers (arrow), and (c) the lithium-and-NAC-treated group showing positive immune reaction in the cytoplasm of cardiomyocytes (arrow) (eNOS ×400). (d) Column chart showing comparative magnitude of the mean values of the area % of positive eNOS immune reaction in the studied groups. eNOS: endothelial nitric oxide synthase.
In Figure 5, the 8-OHdG immunohistochemical-stained sections of the control group and NAC-treated group showed minimal positive immunoreaction with area % 8.97 ± 2.09 and 9.35±3.11, respectively, in contrast to the lithium-treated group which showed strong positive nuclear and cytoplasmic immunoreaction with area % 51.02 ± 7.41 while the lithium-and-NAC-treated group shows mild immunoreaction with area % 19.84 ± 4.40. ANOVA with Turkey HSD post hoc test showed that area % of positive immunoreaction was significantly higher in the lithium-treated group when compared to control, NAC-treated, and lithium-and-NAC-treated groups (p < 0.01). In addition, the area % of lithium-and-NAC-treated group was significantly higher in comparison to the control and NAC-treated groups (p < 0.01).

Immunohistochemical (8-OHdG)-stained section of the left ventricle of (a) the control group showing negative immunostaining, (b) the lithium-treated group showing strong positive nuclear (star) and cytoplasmic immune staining (arrow), and (c) the lithium-and-NAC-treated group showing weak immunostaining (8-OHdG ×400). (d) Column chart showing the comparative magnitude of the mean values of the area % of positive 8-OHdG immune reaction in the studied groups. 8-OHdG: 8-hydroxy-2′-deoxyguanosine.
Discussion
Lithium (Li) is an appropriate mood stabilizer metal for the therapy of bipolar disorder (BD), due to its effect in ameliorating depression and mania and reducing the risk of suicide. However, there are some clinical concerns regarding its safety as it has a narrow therapeutic index requiring regular checking of the serum level. 32
The current study was designed to analyze the lithium-induced cardiotoxicity in male rats, the underlying molecular mechanisms, and the amelioration of these toxic effects by NAC.
The current study demonstrated a slight increase in the body weight of lithium-treated group when compared to other groups. This body weight increase can be partially explained by lithium-induced increase in the leptin level, a hormone secreted from adipocytes, controlling diet intake and energy balance and providing the hypothalamus with information on the amount of body fat, 33 or can be also explained by the edema and ascites caused by the lithium-induced cardiotoxicity with increased pressure in the hepatic veins. Weight gain is a well-known undesirable side effect of continuing lithium therapy which remarkably restricts treatment compliance 34 ; however, this weight gain was not statistically significant in the current work. This can be referred to the observation reported by Baptista et al. who stated that lithium-induced excessive weight gain is observed in both male and female patients, but only in female rats 35 ; moreover, the decline in the general health of rats in the form of weakness, lethargy, and sickness which appeared in lithium-treated group, in accordance with Strbe et al. led to decrease in the feeding pattern of rats. 36
The assessment of MABP in the current study revealed a significant decrease in the MABP in lithium-treated rats in comparison to other groups in accordance with Abdel-Zaher and Abdel Rahman who explained this finding by decreasing the intracellular levels of adenosine triphosphate in cardiac and vascular tissue through the activation of adenosine triphosphate-sensitive potassium channels. 37 In the study performed by Badziuk et al., they found that the oxidative stress conditions are more favorable than normoxia for the activation of the ATP-sensitive potassium channels and they documented that elimination of ROS by NAC prevents the activation of these channels, 38 and this can explain the improvement of the MABP in lithium-and-NAC-treated group.
Regarding the ECG recording of rats, the lithium-treated group showed rhythm disturbances including sinus tachycardia and sinus extrasystoles in accordance with Steckler who reported that lithium reversibly targets the sinus node. 39 Lithium-induced sinus node dysfunction has even been reported in its therapeutic range. 40 In addition, the current study reported ventricular dysrhythmia in the lithium-treated group in accordance with Chen et al. 15 Various ECG changes have been reported in lithium overdose such as junctional bradycardia, complete heart block, and first-degree atrioventricular block. 41 –43 The current work showed also that lithium caused a depressed ST segment, decreased amplitude of R wave, and narrow PR interval. Numerous mechanisms might explain the lithium-related ECG changes. First, lithium competes with sodium, potassium, calcium, and magnesium ions, affecting fluids and salts balance. It interacts with Na/K pump inducing change in the physiology of cellular membrane, decreases the intracellular potassium, replacement of the intracellular calcium, and hypercalcemia. This leads to a reduction in the depolarization rate and electrical impulse propagation and induces various ECG changes especially those related to the process of myocardial repolarization such as nonspecific ST segment and T wave changes. 44
The cardiomyocyte injury in lithium-treated rats was also confirmed by the significant increase in cTnI and CK-MB. Cardiac muscle fibers injury leads to intramyocardial cTnI proteolysis and release of cTnI. 45 Investigational records in a pig model of myocardial ischemia verified cTnI release and distribution into the circulation from apoptotic cardiomyocytes as a different justification for clinical circumstances with increased cTnI without any other proof for myocardial necrosis. 46 CK-MB typically located in the cellular compartment and leaks out into circulation during myocardial injury due to the breakdown of contractile elements and sarcoplasmic reticulum. 47
The histopathological examination of the heart specimens of rats involved in the study revealed that the lithium-treated group showed several lesions in the form of separation between cells, hyalinization with disarranged, degenerated cardiac fibers containing pyknotic nuclei, and cellular aggregates that may refer to inflammatory infiltrate. These findings run in parallel to those observed by Shah et al. who reported that these changes were duration dependent. 17 Similar histopathological changes were also reported by Saad et al. 48 In addition, sections from the lithium-treated group showed congested capillaries surrounded by acidophilic homogenous material that may refer to fibrosis; this was confirmed in Van Gieson-stained sections where there was an increase in the collagen fibers in this group and there was a statistically significant increase in the area % of collagen fibers in the morphometric study. The fibrosis in cardiac tissues of rats after chronic lithium therapy was also reported by Moradi et al. who referred this atrial fibrosis to the increase in the expression of collagen I, alpha 1 gene. 49 Moreover, Nikolić-Kokić et al. 50 reported similar morphological and antioxidant enzymes function changes in rat heart treated with other atypical antipsychotics including clozapine, ziprasidone, and sertindole.
In the current work, there was a strong positive eNOS immunoreaction with a significant increase in its area % in the lithium-treated group when compared to other groups. Nitric oxide (NO) is synthesized from its precursor
miRNAs have been shown to regulate various cellular functions, including cell proliferation, migration, differentiation, and apoptosis. 61 –63 The current work detected a statistically significant overexpression of miRNA-1. MiRNA-1 is considered the most abundant miRNA in cardiac muscle. 64 Yang et al. reported that overexpression of miR-1 exacerbates arrhythmogenesis through posttranscriptionally repressing potassium channel subunit gene and gap junction protein connexin 43. 65 Moreover, Tang et al. reported an inverse relationship between the level of miRNA-1 and antiapoptotic B-cell lymphoma 2 protein expression in cardiomyocytes of the ischemia/reperfusion rat model concluding that miRNA-1 regulates cardiomyocyte apoptosis through the posttranscriptional suppression of Bcl-2. 66 Cardiomyocyte apoptosis is an additional trigger of arrhythmias. 67 Also, Wang et al. reported that miRNA-1 aggravates the cardiac oxidative stress through posttranscriptional repression of the antioxidant system including superoxide dismutase 1, glutamate-cysteine ligase catalytic, and glucose-6-phosphate dehydrogenase; they reported that overexpression of miRNA-1 in cardiomyocytes of neonatal Wistar rats exhibits higher ROS levels and lower resistance to H2O2-induced oxidative stress. This oxidative stress plays an important role in cardiovascular diseases. 68
In this work, there was also a significant overexpression of miRNA-21 in the lithium-treated group compared to other groups. Previous studies also showed its relationship to apoptosis of cardiomyocytes. 69 The fold increase in relative miRNA-21 gene expression was strongly positively correlated to the area % of 8-OHdG (r = 0.92 and p = 0.0003) denoting that oxidative stress induces upregulation of miRNA-21 running in parallel to the study by Tu et al. 70 In addition, there was a strong positive correlation between the fold increase in relative gene expression of miRNA-21 and area % of eNOS positive immunoreaction (r = 0.89 and p = 0.0013). A similar finding was previously reported by Yin et al., who stated that ischemic preconditioning induces overexpression of miRNA-21 that leads to the up-regulation of eNOS protein. 71
The current work showed a significant increase in the miRNA-29 gene expression in the hearts of lithium-treated rats, there was a positive correlation between miRNA-29 and the mean area % of Van Gieson staining (r = 0.91 and p = 0.0005). Cardiac fibrosis occurs in numerous heart diseases, including ischemia and infarction, cardiomyopathies, and myocarditis. Cardiac fibrosis increases the risk of ventricular dysfunction and arrhythmias. 72 Although the molecular mechanisms of cardiac fibrosis remain poorly elucidated, some studies suggest that altered expression of miRNA-29 is associated with cardiac fibrosis. van Rooij et al. demonstrated that miR-29 is expressed preferentially in the fibroblast population of the myocardium and encodes proteins involved in fibrosis, including multiple collagens, fibrillins, and elastin. 73 Also, Sassi et al. reported that global genetic deletion of miRNA-29 or antimiRNA-29 infusion prevents cardiac hypertrophy and fibrosis and improves cardiac function. 74
The NAC co-treatment with lithium in group IV significantly improved the cardiotoxic effects of lithium manifested by a significant reduction in ascites accumulation and decrease in ECG abnormalities and biochemical parameters of cardiac injury CK-MB and cTnI. Regarding the histopathological changes, NAC improved the pathologies induced by lithium and restored the tissue architecture to near normal apart from slight separation of fibers and few small dark nuclei. In addition, the NAC significantly reduced the Van Gieson, e-NOS, and 8-OHdG immunoreaction. The cardioprotective effect of NAC was previously reported by Wang et al. who recorded that NAC reduces the oxidative ischemic-reperfusion injury-induced oxidative stress and cardiac apoptosis and improves the function of mitochondrial permeability transition pores. 75 The current work proved that NAC treatment in combination with lithium reduced significantly the expression of miRNA-1, miRNA-21, and miRNA-29 in comparison to lithium-treated group. Sticking to this finding, Yamamoto et al. reported that NAC can blunt the expression of miRNA-21 that was associated with an increase in 8-OHdG in response to controlled diesel exhaust. 76 Yildirim et al. also demonstrated that intervention with NAC as an antioxidant treatment resulted in significant cardioprotection against diabetes-induced injury, mediated through miR-1 and its target protein junction, which is involved in the development of diabetic cardiomyopathy. 77 NAC also reduced the expression of miR-21 and miR-29b in mice treated with Cryptosporidium parvum as an inducer of inflammation. 78
Conclusion and recommendations
Lithium is a very powerful and commonly used medication for the treatment of various psychiatric diseases, especially BD. However, it has a very narrow therapeutic index. The current work proved that lithium treatment resulted in significant accumulation of ascites and a decrease in the MABP together with elevation in cardiac biomarkers CK-MB, cTnI, and several histological lesions with a significant increase in the area % of Van Gieson, eNOS, and 8-OHdG immunoreaction. There was significant upregulation of miRNA-1, miRNA-21, and miRNA-29.NAC improved the cardiotoxic effects of lithium. NAC, being a nontoxic and safe antioxidant with generally mild adverse effects, can be used to ameliorate lithium-induced cardiac injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
