Abstract
With the ban of conventional cigarettes from public spaces, electronic cigarette (E-cig) liquids have emerged as a nicotine replacement treatment for smoking cessation. However, consumers possess little knowledge of the ingredients and health effects of E-cig liquids following exposure. This study evaluated hair cell damage and developmental toxicities following gestational exposure to E-cig liquids. Zebrafish embryos were exposed to E-cig liquids at different concentrations (0.1%, 0.2%, and 0.4%). Embryonic developmental toxicity and hair cell damage was evaluated at 6 and 7 d, respectively, after fertilization. The average number of hair cells in the anterior lateral line (ALL) and posterior lateral line (PLL) following E-cig exposure was compared to that of the control. Morphological abnormalities and heart rate were evaluated. E-cig liquids significantly damaged the hair cells in the ALL, compared to the control (control; 52.85 ± 5.29 cells, 0.1% E-cig; 49.43 ± 7.70 cells, 0.2% E-cig; 40.68 ± 12.00 cells, 0.4% E-cig; 32.14 ± 20.75%; n = 29–40; p < 0.01). At high concentrations, E-cig liquids significantly damaged the hair cells in the PLL (control; 36.88 ± 5.43 cells, 0.1% E-cig; 33.06 ± 5.21 cells, 0.2% E-cig; 30.95 ± 8.03 cells, 0.4% E-cig; 23.72 ± 15.53%, n = 29–40; p < 0.01). No morphological abnormalities in body shape, somites, notochord, tail, and pectoral fin were observed; however, abnormalities were observed in the dorsal fin and heart rate at high concentrations. Thus, gestational exposure to E-cigs significantly damaged hair cells in a concentration-dependent manner and induced developmental toxicities to the dorsal fin and heart rate at high concentrations.
Introduction
With the increase in the regulation of conventional cigarettes and their ban from public spaces, electronic cigarettes (E-cigs) have emerged; they are advertised as nicotine replacement treatment for smoking cessation. E-cigs are battery-operated devices that vaporize nicotine, flavors, and other additives to simulate the conventional smoking experience. E-cigs can be categorized into three broad types: 1) disposable cigarette-like devices (cig-a-like); 2) mid-sized Vape Pens with a disposable tank; and 3) advanced personal vaporizers with a refillable tank.
Since their introduction into the market by a Chinese pharmacist, Hon Lik, in 2004, the global production and sale of E-cigs has grown considerably and is expected to continue in this manner. The expenditure on E-cig advertising and the use of E-cigs have been continuously increasing. 1 The consumption of E-cigs among the young has increased considerably over the recent years, primarily due to the different flavors available. 2,3
The toxicity of conventional cigarettes to gestational development and sensory hair cells is well described. 4,5 The consumption of conventional cigarettes during pregnancy induces toxicity to the conceptus at all stages of prenatal development, at birth, during infancy, childhood, adolescence, and throughout adult life. Apart from nicotine, the most well elucidated chemical in cigarette smoke, carbon monoxide and other toxic chemicals also play a role in inducing toxicity. As E-cigs deliver aerosolized nicotine in the form of vapor instead of smoke and as some E-cig solutions do not contain nicotine, it is generally considered that E-cigs are relatively less harmful than conventional cigarettes. However, the short-term and long-term safety of E-cigs remains unclear.
Several studies have addressed the toxicity of the different chemical components of E-cigs. E-cig liquids contain propylene glycol, nicotine, and flavoring chemicals, including diacetyl, cinnamaldehyde, acetoin, maltol, pentanedione and other flavors, in addition to flavor-enhancing chemicals. 6 The pulmonary system, which is directly affected by aerosolized nicotine and other flavoring additives, is postulated to be impaired by the use of E-cigs. The main component of E-cig liquids, 1,2-propanediol, also called propylene glycol, is the most abundant volatile compound in E-cig vapor and may also play a role in toxicity, as demonstrated by a previous in vitro study. 7
The toxic effect of E-cig usage during pregnancy has been also addressed; the results demonstrated that the nicotine consumed during the use of E-cigs is similar to that consumed during conventional cigarette smoking. 8 Numerous animal studies have confirmed the toxic effects of nicotine to the developing fetus; 9,10 the effects of nicotine exposure by E-cig usage during fetal development is suggested to be similar to those by conventional cigarette smoking. 8,11 However, the constituents and potential adverse effects of E-cigs are not well understood; 3,11,12 moreover, there is a dearth of scientific information pertaining to the hazards of these substances to gestational development and sensory hair cells.
Zebrafish (Danio rerio) has several advantages for assessing developmental toxicity owing to its high fecundity rate and a well characterized developmental morphological scoring system. 13 –19 Zebrafish is known to be a good model to study the development and teratogenicity of organ systems and structures due to its inexpensive, rapid development, and transparency. 14,16,19
The hair cells in the inner ear of humans are important organs that recognize sound from the external environment and perceive the sense of rotation and movement. 20
Especially, zebrafish has been used as a highly efficient tool for assessing damage to hair cells as the mechanosensory neuromasts of zebrafish are structurally and functionally similar to the hair cells in the inner ear of humans. 21 –27 The hair cells of neuromasts, which are clusters of 5–20 hair cells, in the zebrafish lateral line are particularly efficient for confirming hair cell damage after exposure to ototoxic agents. 21,25 –28
This study aimed to evaluate the damage to hair cells and identify the associated developmental toxicities caused by the gestational exposure of zebrafish embryos to E-cig liquids.
Materials and methods
Zebrafish housing and chemicals
Zebrafish were fed and maintained in embryo medium, comprising 15 mM sodium chloride, 0.5 mM potassium chloride, 1 mM calcium chloride, 1 mM magnesium sulfate, 0.15 mM monopotassium phosphate, 0.05 mM ammonium phosphate, and 0.7 mM sodium bicarbonate, at a temperature of 28.5 ± 1°C in the zebrafish facility at Korea University Ansan Hospital, according to standard recommendations. 29 The number of days post-fertilization (dpf) was staged following fertilization. 30
For this study, we selected an E-cig solution comprising a mixture of the embryo medium and tobacco-flavored E-cig liquid devoid of nicotine (MAG7 Black label, Rayben). The E-cig solution was prepared at concentrations of 0.1%, 0.2%, 0.4%, 0.8%, and 1.0% by dissolving the E-cig solution in embryo medium. All the experiments were performed in accordance with the guidelines of the Animal Care Ethics Committee of Korea University Medical Center and the National Institutes of Health guidelines (IACUC No. KOREA 2018-0054).
Evaluation of developmental toxicity to embryos exposed to E-cigs
First, the LD50 was evaluated to determine the appropriate dose of the E-cig liquid for assessing developmental toxicity. The healthy embryos were transferred to a petri dish for evaluating the LD50. The E-cig solutions were added to the dishes at different concentrations of 0.1%, 0.2%, 0.4%, 0.8%, and 1.0% and incubated at a temperature of 28.5 ± 1°C for 6 d. The mortality rate was expressed as the percentage of the number of dead embryos, compared to the total number of embryos at 6 d (n = 60–91 embryos for each of the E-cig solutions at different concentrations) (Figure 1).

Schematic timeline of the study. The embryos were exposed to E-cig liquids at 4 hpf. The mortality, developmental toxicity, and damage to hair cells were assessed at 3, 6, and 7 dpf, as per the study schedule.
The morphological changes were assessed on the basis of the morphological scoring system for zebrafish (0: no evident structures, 1: multiple abnormalities, 2: two or more abnormalities, 3: only one abnormality, and 5: entirely normal), suggested by Panzica-Kelly and coworkers, to study the morphological changes in body shape, somites, notochord, tail, dorsal fins, pectoral fins, and heart at 6 dpf (n = 48–87 for each of the E-cig solutions at different concentrations). 14 The ratio of any abnormal morphological change was also assessed.
Additionally, the genesis of the swimbladder, which arises from an outgrowth of the endoderm of the foregut, similar to the mammalian lung, and body length were assessed at 3 dpf and 6 dpf in each of the groups receiving different concentrations of E-cig solutions (n = 58–90 embryos for each of the E-cig solutions at different concentrations). 31,32 The genesis of the swimbladder was assessed by the following scoring system: 0: no evident structures, 1: partially inflated (if not enough inflated to the control group), and 2: fully inflated (if enough inflated to the control group).
Evaluation of cardiac development of the embryos exposed to E-cigs
The heart rate was first assessed by counting the number of heartbeats within 10 s of visual inspection and extrapolating to heartbeats per minute (n = 30–35 embryos for each of the E-cig solutions at different concentrations).
Secondly, we assessed the differences in the heart area using Flk-transgenic zebrafish (n = 10 embryos for each of the E-cig solutions at different concentrations). 33,34 At 6 dpf, the Flk-transgenic zebrafish larvae that were treated with the E-cig solutions were placed in 3% methylcellulose to characterize the development of the heart in the embryos exposed to E-cig. The difference between the systolic and diastolic heart area was measured using Image J software from the images of the Flk-transgenic zebrafish captured at a magnification of 12.4X. Only the section of the image that showed the maximum heart diameter was used for analyzing the heart area; such sections from individual zebrafishes that met this requirement were repeatedly measured five times and the mean difference in heart area was calculated.
Evaluation of hair cell toxicity induced by the gestational exposure to E-cig solutions
The zebrafish embryos, exposed to E-cig liquids at different concentrations (from 4 hours post-fertilization (hpf) to 144 hpf), were washed thrice with embryo medium and anesthetized using tricaine (3-aminobenzoic acid 0.4 g/ethyl ester; 100 mL; pH 7, adjusted using Tris buffer) for 5 min, as described previously. 18,35,36 The embryos were then mounted onto a depression slide in methylcellulose. The average number of hair cells in two main regions, the anterior lateral line (ALL) system, including the supraorbital [SO1 and SO2], otic [O1], and occipital [OC1] nerves, and the posterior lateral line (PLL) system, including the neuromasts of the trunk and tail of zebrafishes (p 3–6), was determined using a fluorescence microscope at 7 dpf (LSM5 PASCAL; Carl Zeiss, Germany). 9,17,18,25,27 The hair cells were identified by our two experts, and they were evaluated in all experimental and control conditions (n = 29–40 for each of the concentrations)
Statistical analyses
All the data are represented as the mean ± standard deviation, when applicable. Descriptive analysis was used to evaluate the mortality rate. The continuous variables were compared across the different experimental groups by independent t-tests or Mann-Whitney U tests, as appropriate, after determining the normality at baseline with the Kolmogorov-Smirnov test. One-way analysis of variance (ANOVA) was performed with the data obtained from the repeated experiments (post hoc test: Tukey honestly significant difference test) to compare the number of hair cells or heart rate when exposed to different concentrations of the E-cig solution. Fisher’s exact test was performed to evaluate any differences in the morphological changes among the groups when exposed to different concentrations of the E-cig solution. A two-sided P value <0.05 was considered statistically significant. All the data were analyzed with SPSS software, version 20.0 (SPSS, Chicago, Illinois, USA).
Results
Developmental toxicity induced by gestational exposure to E-cig solutions
The mortality rate was significantly increased at higher concentrations of the E-cig solution (p < 0.001, linear-by-linear association test) (Figure 2). The survival rate of the control and 0.1%, 0.2%, 0.4%, 0.8%, and 1.0% E-cig groups was 96% and 93%, 93%, 53%, 3%, and 0%, respectively, at 6 dpf. Analyses of zebrafish larvae at 6 dpf revealed that the LD50 of the E-cig solution was 0.4%. Therefore, we’ve adopted the 0.1%, 0.2%, and 0.4% E-cig groups for further analysis.
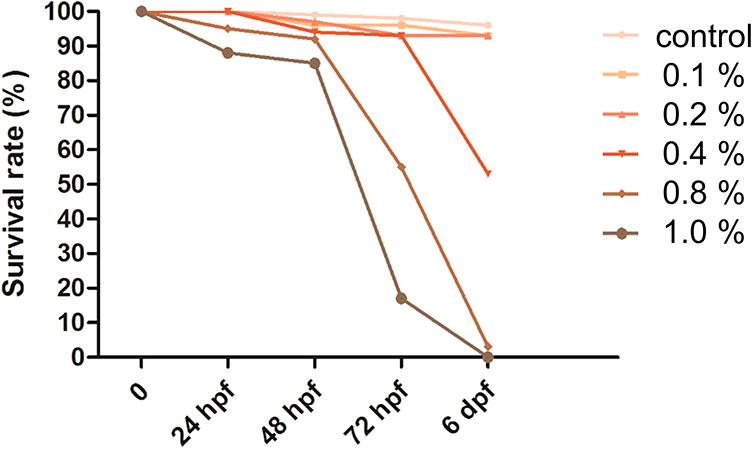
The mortality rate following gestational exposure to E-cig solutions. The mortality rate increased at high concentrations of the E-cig solutions. At 6 dpf, the survival rate of the control and the groups exposed to 0.1%, 0.2%, 0.4%, 0.8%, and 1.0% E-cig solutions was 96%, 93%, 93%, 53%, 3%, and 0%, respectively (p < 0.001, Fisher’s exact test).
The morphological changes were assessed on the basis of the zebrafish morphological scoring system suggested by Panzica-Kelly and coworkers and from the ratio of abnormal morphological changes depicted in Table 1. The ratio of abnormal tail development was higher, being 13.1% and 14.6%, when the concentrations of the E-cig solution were 0.2% and 0.4%, respectively; however, no significant differences were observed in the abnormal developmental ratio among the groups exposed to different concentrations of the E-cig solution.
Morphologic changes of 6 dpf zebrafish with the exposure of electronic cigarettes substrates exposure assessed by Panzica-Kelly et al.

The results of the analysis of swimbladder genesis is depicted in Figure 3. Overall, 17.1% (60/350) of the zebrafish at 3 dpf exhibited complete swimbladder genesis and 67.5% (237/350) of the zebrafish at 3 dpf did not show any swimbladder development. At 3 dpf, the rate of complete swimbladder genesis decreased as the concentration of the E-cig solution increased. At 6 dpf, the overall rate of complete swimbladder genesis was 75.1% (139/185); 23.2% (43/185) of the surviving zebrafish at 6 dpf did not show any swimbladder development. The rate of complete swimbladder genesis was negatively associated with a higher concentration of the E-cig solution; all surviving zebrafish at 6 dpf showed swimbladder agenesis when the concentration of the E-cig solution was 0.4%. Analyses of body length revealed that there were no significant differences among the groups (data not shown).

Swimbladder genesis in the groups exposed to E-cig solutions. (a) At 3 dpf, the rate of complete swimbladder genesis decreased as the concentration of E-cig solution was increased. (b) At 6 dpf, the rate of complete swimbladder genesis was negatively associated with the concentration of the E-cig solution; all surviving zebrafish at 6 dpf exposed to 0.4% E-cig solution showed agenesis of the swimbladder (Fisher’s exact test, p < 0.001).
Evaluation of heart development of the embryos exposed to the E-cig solutions
The heart rate significantly decreased following E-cig exposure at all concentrations, comparison to the control group (p < 0.001, one-way ANOVA, Figure 4(a)). The average heart rate was 125.1 ± 15.3 and 107.8 ± 7.5, 114.9 ± 12.1, and 112.6 ± 9.0 heart beats/min in the control and 0.1%, 0.2%, and 0.4% E-cig groups. However, no differences were observed in the average heart rate among the groups exposed to various concentrations of the E-cig solution (p > 0.05, one-way ANOVA with Tukey’s post-hoc test).

The heart rate and heart area altered following E-cig exposure. (a) The heart rate significantly decreased following E-cig exposure at all concentrations in comparison to that of the control (*p < 0.001, one-way ANOVA: 125.1 ± 15.27, 107.8 ± 7.47, 114.9 ± 12.10, and 112.6 ± 8.98 heart beats/min for the control, 0.1%, 0.2%, and 0.4% E-cig groups, respectively). (b) The heart area significantly decreased in the group that was exposed to 0.2% E-cig solution; however, the heart area significantly increased following exposure to 0.4% E-cig solution in comparison to that of the groups exposed to 0.1% and 0.2% E-cig solutions (**p < 0.01, Kruskal-Wallis test with Dunn’s multiple comparison test).
The heart area of the Flk-transgenic zebrafishes receiving different concentrations of the E-cig solution was assessed; the results are depicted in Figure 4(b). The heart area significantly decreased in group that was exposed to 0.2% E-cig solution, compared to the control; however, the heart area significantly increased in the group exposed to 0.4% E-cig solution in comparison with that in the groups exposed to 0.1% and 0.2% E-cig solution (n = 10, p < 0.01, Kruskal-Wallis test with Dunn’s multiple comparison test) (Figure 5).
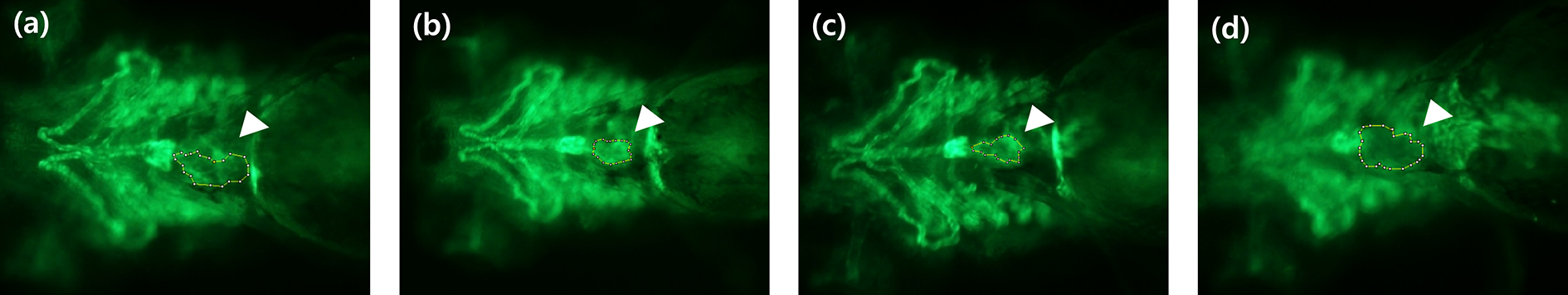
Assessment of the heart area of Flk1 transgenic zebrafish models. The white arrow heads indicate the heart area of flk1 transgenic zebrafish models. (a) Control and groups exposed to (b) 0.1%, (c) 0.2%, and (d) 0.4% E-cig solution.
Hair cell damage induced by gestational exposure to E-cig solutions
The relationship between the concentration of the E-cig solution (0.1%, 0.2%, and 0.4%) and hair cell damage was evaluated in ALL and PLL. Analysis of hair cell damage in the ALL revealed that exposure to 0.2% and 0.4% E-cig solutions for 7 days significantly reduced the average number of hair cells on the ALL, compared to the control group (52.9 ± 5.3 cells, n = 40, p < 0.001, one-way ANOVA with Tukey’s post-hoc test, Figure 6). Additionally, there was a significant difference among the groups exposed to different concentrations of E-cig solutions with regard to hair cell damage (0.1% (n = 35): 49.4 ± 7.7 cells, 0.2% (n = 40): 40.7 ± 12.0 cells, 0.4% (n = 29): 32.1 ± 20.8 cells, p < 0.01).

(a) The hair cells in the four neuromasts in the ALL (SO1, SO2, O1, and OC1) decreased in the groups exposed to the E-cig solutions, as revealed by fluorescence microscopy (40X at 7 dpf). (b) Exposure to 0.2% and 0.4% E-cig solutions for 7 days significantly reduced the average number of hair cells in comparison to that of the control (*p < 0.001, one-way ANOVA with Tukey’s post-hoc test).
Analysis of hair cell damage on the PLL revealed that exposure to 0.2% and 0.4% E-cig solutions for 7 days (0.2% (n = 40): 31.0 ± 8.0 cells, 0.4% (n = 29): 23.7 ± 15.5 cells) significantly reduced the average number of hair cells on the PLL, in comparison to that of the normal control (36.9 ± 5.4 cells, n = 40, p < 0.01, one-way ANOVA with Tukey’s post-hoc test, Figure 7) and that of the group exposed to 0.1% E-cig solution (33.1 ± 5.2 cells, n = 35, p < 0.01).
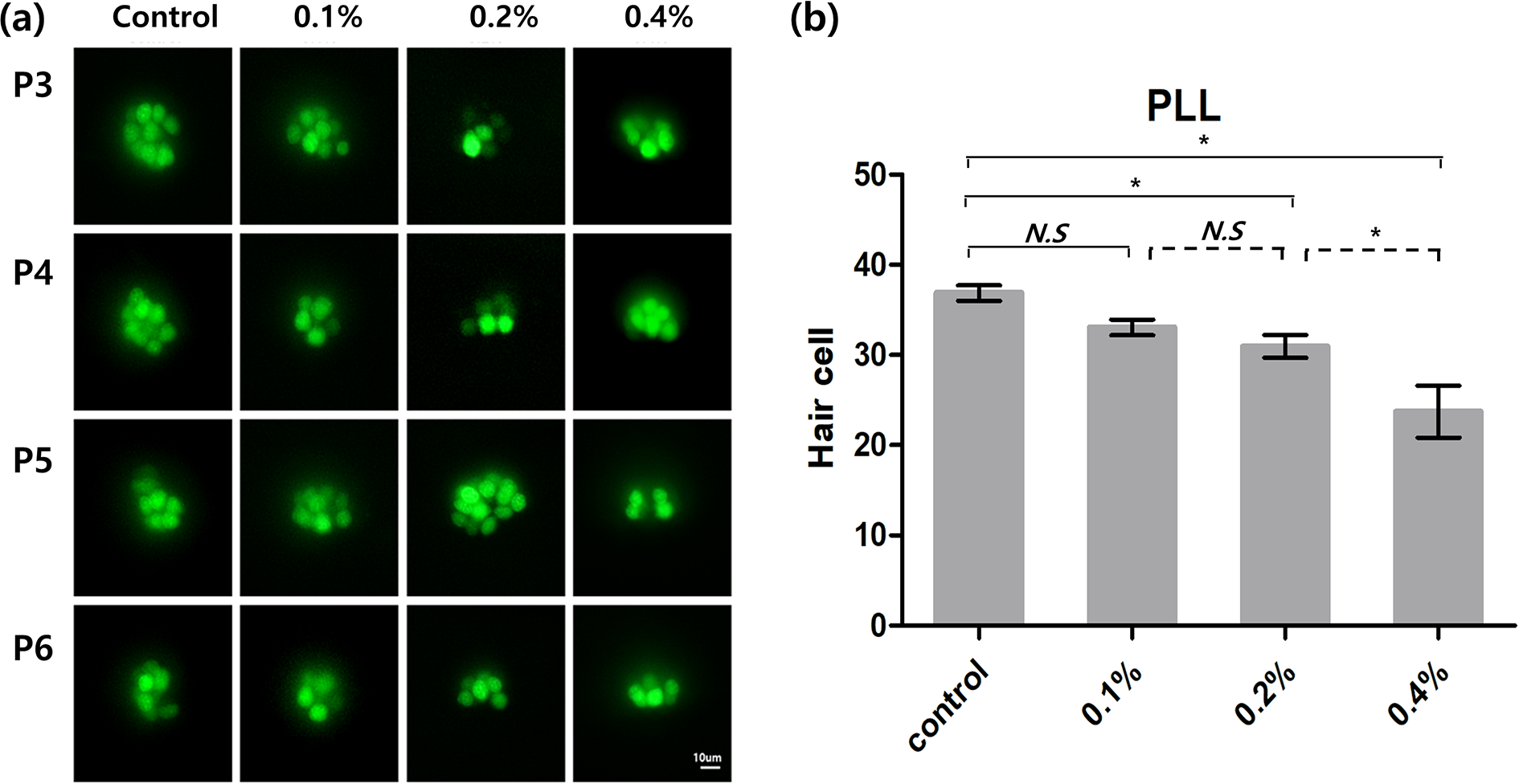
(a) The hair cells in the four neuromasts in the PLL (P3, P4, P5, and P6) decreased in the groups exposed to E-cig solutions, as revealed by fluorescence microscopy (40X at 7 dpf). (b) Exposure to 0.2% and 0.4% E-cig solutions for 7 days significantly reduced the average number of hair cells in comparison to that of the control (**p < 0.01, one-way ANOVA with Tukey’s post-hoc test).
Discussion
Our results demonstrated that gestational exposure to E-cig solutions caused loss of hair cells during development and was associated with various developmental anomalies and mortality. The mechanisms underlying the loss of hair cells following E-cig exposure was unclear as the E-cig solution used here was a liquid mixture containing nicotine, propylene glycol, glycerin, and flavorings, which made it difficult to distinguish the effects of each chemical. Moreover, the composition ratios of the different compounds differ for each E-cig solution. Among these chemicals, the effects of E-cig flavorings on inflammatory and oxidative responses is well elucidated; previous in vitro and animal studies have suggested possible mechanisms of hair cell loss following E-cig exposure. 37,38 Previously, human monocytic cell types, MM6 and U937, were exposed to the flavoring chemicals present in commonly used E-cigs; treatment of the cells with flavored e-liquids without nicotine induced cytotoxicity in a dose-dependent manner. The exposure to flavored e-liquids induced the secretion of interleukin 8 (IL-8), which is a chemokine and a pro-inflammatory cytokine, 37 from the monocytic cells in a dose-dependent manner in comparison with groups that were not exposed to flavored e-liquids. Other studies have also demonstrated that the secretion of IL-8 is significantly induced by flavoring chemicals in E-cigs. 8,39 Our results are similar to those of previous in vivo studies that investigated the toxicity of E-cigs on hair cells following gestational exposure in zebrafish.
Our results demonstrated that embryonic mortality was increased in a dose-dependent manner following exposure to E-cig solutions. The main component of E-cig solutions, 1,2-propanediol, also called propylene glycol, is the most abundant volatile compound in E-cig vapor and could play a role in the early developmental toxicity in zebrafish. These results are consistent with those of an earlier study, which reported that exposure to 1,2-propanediol, identified as the main component of E-cig liquids, significantly increase the mortality rate in a dose-dependent manner. 7 Massarsky and coworkers 7 exposed zebrafish embryos to 2.5% 1,2-propanediol at 6 to 72 hpf and observed that the mortality rate was below 5% in all the treatment groups; however, at a concentration of 5%, 1,2-propanediol induced severe deformities, and at 10% 1,2-propanediol caused 100% mortality. Lahnsteiner also reported mortality following exposure to 1,2-propanediol; they demonstrated that the LD0 (maximum dose at which there are no significant effects on mortality) of 1,2-propanediol, when the embryos were exposed for 48 h starting at 1 hpf, was 0.75%. The duration of exposure was different in each study, which could affect the dose at which 1,2-propanediol was observed to induce toxicity, however, it was clear that 1,2-propanediol induces zebrafish mortality in a dose-dependent manner following exposure at the embryonic or larval stage. Although the physiologically available dose of 1,2-propanediol that is actually taken up by the embryos or larvae is unknown, the results might explain the importance of evaluating the toxicity of 1,2-propanediol.
The overall heart rate decreased following the exposure to E-cig solutions. The mechanisms underlying the loss of hair cells following exposure to E-cig solutions are ambiguous; these results differed from common knowledge, as nicotine is one of the chemicals known to stimulate the sympathetic stimulus and increase the heart rate in human subjects. A previous study reported that the transient exposure of zebrafish to nicotine at the embryonic stage delays the development of secondary spinal motoneurons. 40 The authors also reported that nicotine induces pathfinding errors in the axons of secondary motoneurons. As we observed the toxicity of the E-cig solution to zebrafish at the gestational stage, the delay of spinal motoneuron development could be associated with the decrease in the heart rate and requires further investigation.
We identified the deformities of the heart in Flk transgenic zebrafish, wherein the vascular endothelial growth factor receptor gene is a well-established gene for studying cardiac deformities. 33 The results of heart area analysis were somewhat ambiguous. We observed that the heart area decreased following exposure to 0.2% E-cig solution and increased following exposure to 0.4% E-cig solution; these results were dissimilar to those of previous studies, which demonstrated that the pericardial area increases following exposure to 1,2-propanediol in a dose-dependent manner. 7,41 The underlying mechanism by which E-cig affects the heart area could not be determined because we used an E-cig solution, which was a mixture of embryo medium and a tobacco-flavored e-liquid devoid of nicotine. The different unknown compounds in the E-cig solution could have affected the development of the heart and it was difficult to elucidate the mechanism by which the deformities of the heart, as observed in Flk transgenic zebrafish, had developed.
In addition to the heart rate, the development of the swimbladder was also affected following exposure to the E-cig solution. Previous studies have reported pericardial edema and decreased heart rate following exposure to E-cig solutions, 7,41 leading to the inference that blood circulation is impaired following exposure to E-cig solutions. As the normal peripheral circulation is known to be important for the development of a swimbladder, 31,42 the impairment of blood circulation could have led to the delay in swimbladder development. Our observations, that the rate of complete swimbladder genesis was negatively associated with the concentration of E-cig solutions and that the overall heart rate was generally unaltered following exposure to the E-cig solution, support the notion that exposure to the E-cig solution affected the development of the swimbladder due to possible impairment of the normal peripheral circulation.
This study has several limitations. First, the results of this study were derived using a zebrafish model, and, therefore, the mechanism of toxicity of E-cig liquids would be different from that in human subjects. Secondly, this study did not elucidate which chemicals in the E-cig solution were toxic to the hair cells, as an E-cig solution containing a mixture of different chemicals was used herein. Therefore, the limitations of this study lie in the fact that only observational and descriptive strategies were employed to assess the damage to the hair cells and to study the developmental toxicity associated with gestational exposure to E-cig solutions in a zebrafish model. Direct exposure of the cells of target organs to E-cig liquids may provide meaningful toxicological data. However, data from this observational study data can elucidate the possible harmful effects of E-cig and establish further research avenues.
In conclusion, we assessed the toxicity due to gestational exposure of zebrafish models to E-cig solutions; the results demonstrated that E-cig liquids caused significant damage to the hair cells, depending on the concentration of the E-cig solution. Developmental toxicities to the swimbladder and heart rate were also observed following exposure to the E-cig solution at high concentrations.
Footnotes
Acknowledgments
I am grateful to my brothers, Dr. Seung Hyun Cho and Eun Joong Kim at Soo ENT Clinic.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Research Foundation of Korea (NRF, 2015R1C1A1A01054397) and also supported by Korea University Research Grant.
