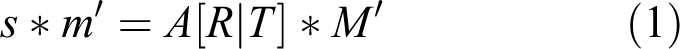Abstract
Background and objectives:
Zebrafish are commonly used as experimental animals in otolaryngology studies. However, the behavioral characteristics of these fish are not well known, especially those related to the vestibular system. The goal of this study was to evaluate behavioral changes in zebrafish due to toxicity in the balance system.
Materials and methods:
Zebrafish were exposed to 1000 μM cisplatin for 6 h. We, then, periodically monitored swimming depth, total swimming distance, peak swimming velocity, and mean swimming velocity of the fish for approximately 21 days.
Results:
Total swimming distance (p < 0.0001), peak swimming velocity (p = 0.0063), and mean swimming velocity (p < 0.0001) in the cisplatin-administered group were significantly decreased when compared with control fish.
Conclusion:
Our findings demonstrate that cisplatin can alter the locomotion behavior of zebrafish.
Introduction
Recent studies on ototoxicity are focused more on prevention and hair cell regeneration. 1,2 Rodents have been commonly used as an ototoxic animal model by which ototoxic effect is evaluated in terms of hearing, nystagmus, and posture measurements. 3 Although these parameters sensitively reflect the degree of ototoxicity, they still fail to eliminate the possible errors in the intricate steps of experiments. 4,5 Moreover, the need for an evaluation model of ototoxic effect on lifestyle with adjustment of environmental factors has already been raised but no successful efforts have yet been reported. 5
Zebrafish are used as simple experimental animal models for vertebrate development. 6–8 Among wild-type zebrafish, phonotypes of mutants with dysplasia of the inner ear organs, including the otolithic organs, are well known. 7 However, only a few studies of behavioral changes in zebrafish associated with drug-induced toxicity to the balance system have been conducted and these studies are very complicated (e.g. measuring eye deviation, rotation, or posture). 8–12 Therefore, we implemented a simple and objective method to observe behavioral changes in zebrafish by exploiting the habits of these animals to dwell at certain depths in a water tank. 13
The body balance system in fish consists of the vestibule of the ear and the lateral line system. The lateral line system has neuroepithelia with ciliated hair cells similar to the vestibular sensory epithelia in mammals, and it plays a role in the maintenance of body posture. The aim of this study is to detect locomotor behavioral effects on zebrafish, induced by cisplatin toxicity to the balance system.
Cisplatin is well known as a potent platinum anticancer drug that has been commonly used to treat lung, head, and neck cancers in otolaryngology. Even at low doses, 14 platinum-based anticancer drugs generate reactive oxygen species similar to aminoglycosides due to melanin binding at the stria vascularis in the cochlea. 15,16 After inducing the lateral line system toxicity in zebrafish, we periodically tracked swimming depth, total swimming distance, peak swimming velocity, and mean swimming velocity for approximately 21 days. Throughout this work, we were able to assess the behavioral effects of cisplatin.
Materials and methods
Animal management
Among 100 wild-type AB zebrafish (Danio rerio, Homeplus, Seoul, South Korea) purchased, which were adults older than 6 months, 29 were ultimately selected for further behavioral study. We maintained the zebrafish in carbon-filtered tap water at 28°C with a 12-h light/12-h dark cycle. The experimental water tank (Hyunjin Precision, Seoul, Korea) was made of polymethyl methacrylate with an inner diameter of 50 mm, an outer diameter of 60 mm, and a height of 300 mm. We moved the zebrafish to an experimental water tank 24 h prior to the start of each experiment to allow them to acclimatize to the experimental conditions. We tracked the position of the zebrafish each day at 7:00 a.m. for 1 h. We performed all experiments at a constant room temperature of 25°C, 50% humidity, and luminance of 100 lux. All animal experiments were carried out according to the Gachon University (Incheon, Korea) Institutional Animal Care and Use Committee (GIACUCR-001).
Arranging the water tank and webcam
The water level in the experimental water tank was parallel to the horizontal surface on which the water tanks were placed, as measured with a digital apparatus. We monitored six experimental water tanks simultaneously and separated the water tanks from one another with pieces of opaque cardboard to minimize the visual interference between water tanks. The distance between each experimental water tanks and the webcam was 1200 mm. We used a white foam board (HF®, 1T, 600 × 900 mm, Hyunjin Art, Goyang, South Korea) as a background and placed three 20-W fluorescent lights (FL20SSEX-D/18, OSRAM Sylvania, Danvers, Massachusetts, USA) 30 cm above the foam board to illuminate the 6 experimental water tanks at equal intensities. We measured background light to ±10 lux with an illuminometer (DT-8820, Bright Medi-Weld Appliances, Mumbai, Maharashtra, India) placed 30 cm in front of the experimental water tanks.
Image recording and processing
We saved our videos on an IBM-compatible computer (Core™ 2 Quad CPU Q6600 at 2.40 GHz; Intel®, Santa Clara, California, USA) with 2 GB DDR3 RAM (Samsung, Seoul, South Korea) and a Windows® XP SP3 operating system (Microsoft®, Redmond, Washington, USA). We captured video images using a webcam (PLEOMAX® PWC-2200; Samsung, Seoul, South Korea) and Windows Movie Maker (Microsoft). We used the following camera settings: black-and-white mode, 60 Hz flicker, and γ = 0.18, contrast = 1.08, brightness = 16, white balance = auto, USB bandwidth = auto, frame rate = 1 frame/s, and time limit = 3600 s. We saved the videos as *.wmv files to maximize image quality. We performed image processing and data analysis using Visual Studio 6.0, Open CV v1.0 (Microsoft) and Excel 2007 (Microsoft).
Camera calibration
At the front of the experimental water tanks, we attached clear film (CG-3300, 3M, Maplewood, Minnesota, USA) with a lattice of 10-mm black dots printed on it. We considered each of the theoretical spots (X, Y, and Z) as defining the actual spatial coordinates. We used matrix A, the camera matrix, to calibrate information related to the camera alone. We calibrated the focal distance using matrix A. We calibrated the camera for three-dimensional (3D) movement using [R|t], which accounted for the effects of factors unassociated with the camera itself. Thus, we minimized the distortion caused by projecting the 3D information into two dimensions (2D) by obtaining the matrix information through calibration prior to data analysis (equations (1) and (2)).
In this case, s means an arbitrary scale factor, m′ comprises the 2D points projected on the camera’s image plane (u, v), A is the intrinsic parameter matrix for the camera, R is the rotational matrix applied to coincide with the principal axis with Z-axis, t is the camera translation to origin of the X-Y-Z coordinate, and M comprises the 3D points in the real world (X, Y, Z).
Toxicity induction to the balance system
The cisplatin (Cisplan® 10 mg/20 mL; Dong A Pham., Seoul, South Korea) used in this study is a colorless, odorless solution with a half-life of 30–100 h. We previously determined the concentration of cisplatin required to induce ototoxicity by performing small-scale pilot studies using fluorescence microscopy with mutant zebrafish (Brn3C: enhanced-green fluorescent protein (EGFP)). 17 In additional pilot studies, we exposed 19 zebrafish to different concentrations of cisplatin over different periods of time and then determined the optimal exposure time and concentration needed to damage the neuromasts or hair cells of the adult zebrafish, while minimizing mortality using scanning electron microscopy (SEM) confirmation. 18,19
For the initial screen, we selected only 29 zebrafish originally obtained for this study that maintained a constant swimming depth of greater than 500 s. Consistency in behavioral test results may not reflect different behavioral characteristics from one subject to another. Therefore, to evaluate more accurate behavioral characteristics, we selected subjects that were more consistent in terms of their behavioral habits.
Among the selected zebrafish, we treated 18 zebrafish of these animals with 1000 μM cisplatin solution for 6 h at room temperature to induce ototoxicity and called them as cisplatin-administered group. Treatment process was carried out in a dark room to prevent photodissociation of cisplatin. The other 11 zebrafish were moved to an experimental water tank without cisplatin administration as the sham group. The same swimming behavior-tracking experiment was repeated on 1, 3, 5, 7, 14, and 21 days after cisplatin administration in both groups.
Fluorescence microscopy
We previously reported that these transgenic zebrafish (Brn3C: EGFP) produce green neuromasts that are visible by fluorescence microscopy without any staining. Essentially, the number of these neuromasts are similar to the number of hair cells when compared with wild-type zebrafish. 17 In this report, we prepared the cisplatin solution by adding pure cisplatin powder (Cat No. p4394, Sigma-Aldrich Co., St Louis, Missouri, USA) to embryo medium (1 mM magnesium sulfate, 120 μM monopotassium phosphate, 74 μM disodium phosphate, 1 mM calcium chloride, 500 μM potassium chloride, 15 μM sodium chloride, and 500 μM sodium bicarbonate in distilled water). We exposed age-matched adult transgenic zebrafish (Brn3C: EGFP) to 1000 μM cisplatin for 6 h. We rinsed the zebrafish three times in embryo medium and anesthetized them with tricane (3-aminobenzoic acid 0.4 g/100 mL ethyl ester, pH 7 adjusted with Tris buffer) for 5 min. We mounted the zebrafish in methylcellulose (Sigma-Aldrich, St Louis, Missouri, USA) on a depression slide to examine the hair cells within the lateral line neuromasts by fluorescence microscopy (Imager M1; Carl Zeiss, Jena, Germany). We selected neuromasts from the head and body of the zebrafish. We counted the total number of hair cells in the zebrafish by fluorescence microscopy.
Scanning electron microscopy
We performed SEM on both experimental groups to confirm the damaging of neuromasts or hair cells in same experimental environment. We prefixed the zebrafish by immersion in 0.1 M phosphate buffer containing 2% glutaraldehyde and postfixed the animals for 2 h in phosphate-buffered saline containing 1% osmic acid. We treated the samples with a graded series of ethanol and t-butyl alcohol, dried them in a freeze dryer (ES-2030; Hitachi, Tokyo, Japan), coated them in platinum using an ion coater (IB-5, Eiko Engineering Co., Tokyo, Japan) and observed them with field-emission SEM (S-4700; Hitachi, Tokyo, Japan). 20
Tracking algorithm
We analyzed data from the videos of the zebrafish that had been recorded over 1 h and compared the mean total swimming length, peak swimming velocity, and mean swimming velocity values. Before analysis, we obtained binary images B(i,j) by subtracting current images from previous ones consecutively to track the position of the zebrafish using equation (3).
In this formula, Ft(i,j) is the current image, Ft−1(i,j) is the previous image, t is the number of frames, (i, j) denotes the image coordinates, and T is the threshold value.
Using equation (4), we estimated the location of the zebrafish
In this formula, N and M represent the size of the image.
We calculated the total swimming distances using the Pythagorean theorem (equation (5)).
In this formula, Lt
is the moving distance of a zebrafish between t frame and t−1 frame, L
total is the total swing distance for 3600 s, and
V peak is the peak velocity of the zebrafish for 3600 s, and max(Lt−Lt−1 ) is the longest moving distance between each consecutive frames. V mean is the average swimming velocity of the zebrafish for 3600 s.
Statistical analysis
We used Statistical Package for Social Sciences v18.0 (SPSS Inc., Chicago, Illinois, USA) for statistical analysis. Data are presented as box and whisker plots created with Prism® 4 (Graphpad Software Inc., La Jolla, California, USA). The mean values of continuous variables for the cisplatin-administered and sham groups were compared with independent two-sample t tests after performing Levene’s test for variance equality and the Kolmogorov–Smirnov test for normality. A Mann–Whitney test was also used to compare the central tendency of continuous variables that did not follow a normal distribution. Bonferroni’s correction was used for correcting multiple comparison problems. A repeated measures analysis of variance (RM ANOVA) was used to test for statistical differences of any between-subjects effect (e.g. difference of two groups) and within-subjects effect (e.g. replicate experiments). The Greenhouse–Geisser method was applied to correct the sphericity of the covariance. The values of p ≤ 0.05 were considered statistically significant.
Results
A histological evaluation was performed in the current study to monitor the changes of hair cells in the neuromasts in the sham and cisplatin-administered groups. Hair cells in the neuromasts of the body were partially lost in the cisplatin-administered group (Figures 1 and 2). SEM results showed that the sham group had intact kinocilia and stereocilia bundles of hair cells. However, these structures had been transformed or were missing in the cisplatin-administered group (Figure 3). The cumulative time that one zebrafish swam at a certain depth reached a peak at higher water levels according to comparisons made between before and after cisplatin administration (Figure 4). Before administration with cisplatin, the zebrafish mainly stayed at high water levels that changed after administration with 1000 μM cisplatin.

Fluorescent microscopy images (×10 magnification) of neuromasts present in adult transgenic zebrafish (Brn3C: EGFP) trunk lateral lines before ((a) to (c)) and after ((d) to (f)) 6 h of administration with 1000 μM cisplatin. As shown in panels (a) to (c), neuromasts were identified using fluorescence microscopy. There were fewer visible and representative neuromasts shown in panels (d) to (f). EGFP: enhanced-green fluorescent protein.
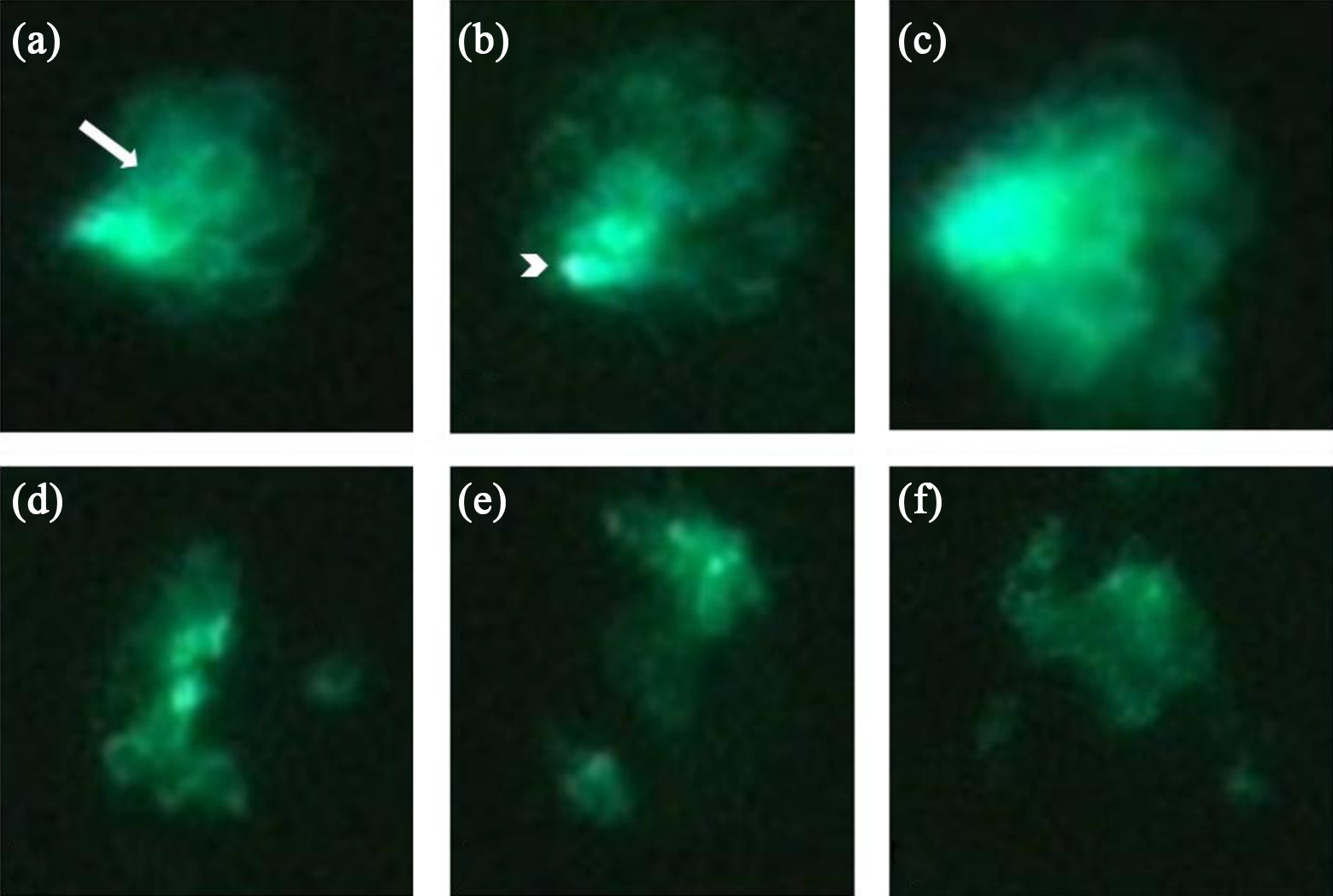
Fluorescent microscopy images (×40 magnification) of hair cells within a neuromast present in adult transgenic zebrafish (Brn3C: EGFP) trunk lateral lines before ((a) to (c)) and after ((d) to (f)) 6 h of administration with 1000 μM cisplatin. As shown in panels (a) to (c), hair cell stereocilia bundles from a neuromast were identified using fluorescence microscopy. There were fewer visible hair cells in a representative neuromast shown in panels (d) to (f). Kinocilia and stereocilia bundles had been eliminated. The arrow in A indicates stereocilia of the hair cells. The arrowhead in B indicates hair cell bodies. EGFP: enhanced-green fluorescent protein.

Morphological changes of hair cells after cisplatin administration. (a) SEM analysis of a representative neuromast from an untreated adult zebrafish showing intact kinocilia and stereocilia bundles of the hair cells. (b) A damaged neuromast from an adult zebrafish 6 h after administration with 1000 μM cisplatin. Hair cells of the cisplatin-administered zebrafish were destroyed. Kinocilia and stereocilia loss or fusion was also observed. The arrow in panel A is pointing to kinocilia. SEM: scanning electron microscopy.

The cumulative time at swimming depth in one zebrafish before and 1, 3, 7, and 21 days after administration with 1000 μM cisplatin. Before administration with cisplatin, the zebrafish stayed at high water levels, whereas the time at water level of zebrafish was changed after administration with 1000 μM cisplatin.
Analysis of between-subjects effects by RM ANOVA demonstrated that the cisplatin-administered group had a significantly reduced total swimming distance compared with the sham group (F(1, 27) = 206.41, p < 0.001). Analysis of within-subjects effects showed that each group had significant interaction effects (F(3.3, 88.4) = 4.94, p = 0.009]. Individual analysis of within-subjects effects revealed significant differences in total swimming distance on days 1 and 21 of measurement (F(1, 27) = 6.33, p = 0.018), but a significant interaction effect was not observed (F(1, 27) = 2.55, p = 0.12; Figure 5). When comparing data for the cisplatin-administered and sham groups recorded after day 14 (Figures 6 and 7

Comparison of total swimming distance between the sham and cisplatin-administered groups according to time after cisplatin administration. Total swimming distance of the cisplatin-administered group was significantly decreased (p < 0.0001). Overall, swimming distances of the two groups differed significantly, although no differences were observed at the first and last time points. Interactions between “between-subjects effects” and “within-subjects effects” were statistically significant (F(3.3, 88.4) = 4.94; p = 0.009).

Comparison of peak swimming velocity between the sham and cisplatin-administered groups 14 days after cisplatin treatment. The peak swimming velocity of the cisplatin-administered group was significantly decreased compared with the untreated sham animals, **p = 0.0063.
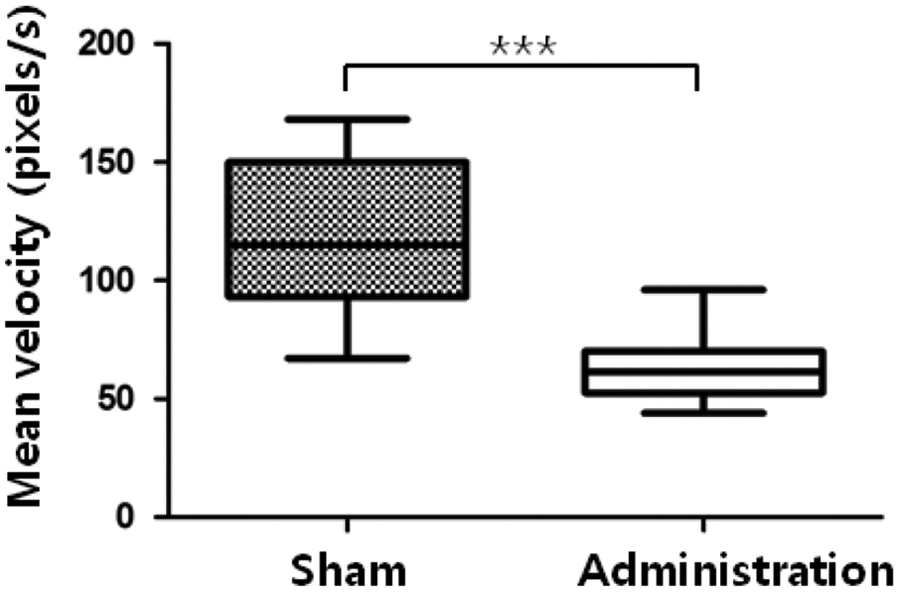
Comparison of mean swimming velocity between the sham and cisplatin-administered groups 14 days after cisplatin treatment. The mean swimming velocity of the cisplatin-administered group was significantly decreased compared with the untreated sham zebrafish, ***p < 0.0001.
Discussion
According to the present study, cisplatin may induce damage to the hair cells of neuromasts in the lateral line, which leads to significant behavioral changes of the zebrafish. This study may be the first report introducing an objective measurement method for behavioral changes due to cisplatin-induced toxicity of the balance system.
Behavioral changes are influenced by various factors, such as locomotion, emotion, learning, and memory. Sometimes the interpretation of behavioral changes is not easy, which is still challenging for experiments of fish. Zebrafish are widely used for various inner ear studies such as sensory hair cell regeneration, drug ototoxicity, and vestibular science in the otolaryngology field. In particular, disruption of the balance system can have significant effects. For example, mice with bilateral vestibulopathy show slow reaction velocity, short step, and a wide-base gait. 21 The vestibular-end organs of zebrafish consist of three semicircular canals and two otolithic organs similar to mice. One difference between zebrafish and mammals is that a giant otoconia forms a large polycrystalline mass, and the saccular, lagena, and macular neglecta in fish are involved in hearing but not in balance. 11,22 Moreover, the lateral line on zebrafish responds to low-frequency stimuli, such as water flow, and affects locomotion functions such as schooling and prey detection. 23 In particular, hair cells of the neuromasts in the lateral line play critical roles in such behaviors. 24 – 26 The lateral line consists of an anterior and posterior lateral line system, each containing eight neuromasts. The posterior lateral line system is influenced by fin position, morphology, and habitat. 25 –27 Ototoxic medications such as aminoglycosides and platinum-based anticancer drugs can destroy the lateral lines, semicircular canals, and otolithic organs simultaneously. 24,28 Therefore, comparative biologists have utilized aminoglycosides to experimentally ablate the lateral line system to examine its role in behavior. 24
Locomotion, an important behavioral determinant, can be influenced by several factors, such as proprioception, vestibular function, and visual cues. However, as higher level locomotion regulation center is not yet clear in vertebrates, vestibular function is used to determine behavioral patterns. Zebrafish tend to inhabit a certain depth within a water tank. 13 Since the vestibular system including otolithic organs is used to sense gravity, it can also influence swimming depth. 12 Therefore, acute changes in the balance system can induce behavioral changes, including altered swimming depths similar to mammals. Several techniques have been used to evaluate vestibular function in disease-specific animal models. These methods include eye tracking, 9 –11,29 measuring looping behavior 11,30 using fluorescent materials, 25,31 –33 and monitoring spike generation of the vestibular afferent nerve via patch clamps with a fixed body position and water flowing at a constant velocity. 31,33 In the present study, we repeatedly recorded the swimming depths of zebrafish in water tanks for 1 h. We calculated the total swimming distance, peak swimming velocity, and mean swimming velocity to create histograms of swimming behavior. The water tank contained a 3D space with an inner diameter of 50 mm. Spatial distortion can occur when data recorded in 3D space are plotted on a 2D plane. We, therefore, performed a camera correction to overcome this distortion.
Platinum-based anticancer drugs are associated with irreversible bilateral ototoxicity similarly to aminoglycosides that was used traditionally in ototoxic study. 14 While cisplatin inhibits cellular reproduction at the DNA level, aminoglycoside acts as a protein synthesis inhibitor. 34 The action mechanisms of these two representative ototoxic agents are not fully understood yet but are known to cause changes in internal signaling by free radicals or subsequent permanent sensory and neural cell damages, such as cell death due to denaturalization of mechanotransduction channels. 35
To confirm ototoxicity in zebrafish, a fluorescent vital dye, 2-(4-(dimethylamino)styryl)-N-ethylpyridinium iodide, was commonly used to stain zebrafish hair cells of neuromasts in the lateral line in vivo, and morphometric analysis was performed to quantify ototoxicity. 17 In this study, to determine the ototoxic concentration of cisplain, we used transgenic zebrafish (Brn3C: EGFP) which produce green neuromasts that are visible by fluorescence microscopy without any staining. To create a bilateral balance deficit model for animals, this method can be helpful to determine the proper concentration and exposure time for hair cell damage quickly and effectively. The cisplatin-induced hair cell damage occurs in a concentration-dependent fashion. 36 The conventional concentration of cisplatin for ototoxicity screening was 50–100 μM with which desired subjects were treated for 1 h. 28,37 However, we selected a high, but nonlethal, concentration of cisplatin to assure loss of superficial neuromasts in the posterior lateral line. 17 This loss was confirmed using SEM. 22,31 Ototoxicity occurs with different degrees of susceptibility to ototoxic drugs and to the hair cells within canal and superficial neuromasts. 24 Therefore, we used the posterior lateral line system to achieve cisplatin-induced ototoxicity due to the fact that it is more vulnerable to cisplatin-induced toxicity without selection bias.
Most ototoxicity-related studies using the lateral line of zebrafish have documented hair cell counts without a functional evaluation except for a stereotypical startle response to assess the impact of hair cell damage on hearing function. 34 In this study, we suggested a new functional evaluation method for balance that represents ototoxicity in zebrafish.
Conclusion
Our results demonstrate that cisplatin administration can change the locomotion behavior of zebrafish. The lateral line system is a factor that influences zebrafish behavior.
Footnotes
Authors’ Note
The authors alone are responsible for the content and writing of the paper.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (NRF-2010-0011858).