Abstract
Objective:
Oxidative stress is one of the major mechanisms of cyclophosphamide (CPX)-induced toxicities. However, it is unknown how CPX induces oxidative stress. Based on the available information, we speculated that CPX could increase iron content in the tissues and then induce oxidative stress.
Method:
We tested this hypothesis by investigating the effects of CPX on iron and ferritin contents, expression of transferrin receptor 1 (TfR1), ferroportin 1 (Fpn1), iron regulatory proteins (IRPs), hepcidin, and nuclear factor erythroid 2-related factor-2 (Nrf2) in the liver and spleen, and also on reticulocyte count, immature reticulocyte fraction, and hemoglobin (Hb) in the blood in c57/B6 mouse.
Results:
We demonstrated that CPX could induce a significant increase in iron contents and ferritin expression in the liver and spleen, notably inhibit erythropoiesis and Hb synthesis and lead to a reduction in iron usage. The reduced expression in TfR1 and Fpn1 is a secondary effect of CPX-induced iron accumulation in the liver and spleen and also partly associated with the suppressed IRP/iron-responsive element system, upregulation of hepcidin, and downregulation of Nrf2.
Conclusions:
The reduced iron usage is one of the causes for iron overload in the liver and spleen and the increased tissue iron might be one of the mechanisms for CPX to induce oxidative stress and toxicities.
Keywords
Introduction
Cyclophosphamide (CPX) is one of the most commonly used chemotherapeutic drugs. Oral or intravenous administration of CPX, either used alone or combined with other drugs, has been found to be effective in treating not only hematological malignancies, such as lymphoma, multiple myeloma, and leukemia, 1,2 but also breast cancer and ovarian cancer. 3,4 Meanwhile, its ability to suppress the immune systems also leads to application in autoimmune diseases like severe aplastic anemia, lupus erythematosus, lupus nephritis, vasculitis, scleroderma lung disease, as well as posttransplantation graft versus host prophylaxis. 5 –9
Apart from its therapeutic effects, CPX is also known to possess a wide spectrum of adverse effects, including myelosuppression, neutropenia, lymphopenia, erythropenia, 10,11 thrombocytopenia, 12 and anemia 13,14 ; hemorrhagic cystitis and alopecia 15 ; cardiotoxicity 16 ; hepatotoxicity 17 ; kidney toxicity 17,18 ; and pulmonary toxicity. 19 Up to now, the mechanisms involved in the CPX-induced severe toxicities have not been fully elucidated. Understanding the causes behind these unwanted side effects is of prime importance as it is directly related to the prognosis and survival rate of chemotherapeutically treated patients. 16
Recently, oxidative stress has been suggested to be one of the major mechanisms of CPX-induced severe toxicities. 17,18 However, it is not completely known about how CPX induces oxidative stress. Iron is one of the hematopoietic raw materials 20 and also a major generator of reactive oxygen species (ROS) when it is increased abnormally in cells and tissues. The iron-induced ROS are capable of damaging biological molecules, leading to multiple organ injuries. 21 The reduced expression of erythropoietin 22 and abnormal hematopoietic function, 13,14 induced by CPX, could lead to a suppressed erythropoiesis and a reduced iron usage, and then iron overload in tissues or organs as found in CBS knockout (CBS−/−) mice. 23,24
Based on the above, we speculated that the increased iron content in the tissues, induced by CPX, may be one of the mechanisms for CPX to induce oxidative stress. In the present study, we tested this hypothesis by investigating the effects of a single dose of CPX on iron content and ferritin (Ft-L, ferritin light chain (Ft-L) and ferritin heavy chain (Ft-H)) expression in the liver and spleen. Also, we examined the expression of transferrin receptor (TfR1), ferroportin 1 (Fpn1), iron regulatory proteins (IRP1 and IRP2), hepcidin, and nuclear factor erythroid 2-related factor-2 (Nrf2) in the liver and/or spleen, and on reticulocyte count (RET), immature reticulocyte fraction (IRF), and hemoglobin (Hb) in the blood in c57/B6 mouse. The findings suggested that the reduced iron usage induced by CPX may be one of the causes for iron overload in the liver and spleen, while the increased iron in the tissues may be one of the novel mechanisms for CPX to induce oxidative stress and toxicities.
Materials and Methods
Chemicals
Unless otherwise stated, all chemicals including mouse monoclonal anti-
Animals and treatment
c57/B6 mice (7–8 weeks old) were purchased from the Sippr-BK Experimental Animal Center, Shanghai, China, and were housed in stainless steel cages at 21 ± 2°C with a relative humidity of 55–60% and alternating 12-h periods of light (07:00–19:00) and dark (19:00–07:00) cycles. All mice were allowed to acclimate for 1 week before initiation of any experiment. Mice were randomly assigned to receive intraperitoneal injection of NS or CPX (160 mg/kg). Animals were euthanized afterward at prespecified time points (Figure 1). After being anesthetized, mice received cardiac perfusion of 30 mL NS. Liver and spleen tissues were removed, rinsed in NS, dried, cut into appropriate sizes, and prepared for Western blot, RNA extraction, and tissue iron measurement. For the measurement of serum iron, RET, IRF, and Hb, blood samples were collected into normal Eppendorf tubes. Serum aliquots were collected after centrifuging, and the relevant measurements were conducted as described previously.
25
All mice received humane care according to the

A scheme on the animal protocol.
Western blot
Tissues were homogenized in RIPA buffer and centrifuged at 13,000 r/min under 4°C for 15 min. The supernatant from each tube was transferred into another tube for protein concentration tests using BCA protein assay kit. Aliquots containing 30 μg protein from each well were loaded and run on a single track of 12% SDS-PAGE under reducing conditions and subsequently transferred onto a PVDF membrane (Bio-Rad, Hercules, California, USA). The membrane was cut into blots according to the molecule weights of target proteins and blocked for 2 h under room temperature using 5% nonfat milk. Blots were subsequently washed and incubated overnight under 4°C with primary antibodies: antirat TfR1 (1:500), antimouse Fpn1 (1:1000), anti-Ft-L (1:2000) and anti-Ft-H (1:2000), anti-IRP1 and anti-IRP2 (1:1000), and
Total RNA extraction and quantitative RT-PCR
Total RNA was extracted from tissue samples using TRIzol reagent, and cDNA was synthesized using the AevertAid first-strand cDNA synthesis kit, both performed in accordance with the instructions of the manufacturers. Real-time polymerase chain reaction (PCR) was conducted using Fast Start Universal SYBR Green Master and Light Cycler96 (Roche). Primer sequences included mouse hepcidin sense: 5′-GCACCACCTATCTCCATCAACA-3′, antisense: 5′-TTCTTCCCCGTGCAAAGG-3′; mouse Nrf2 sense: 5′-CGAGATATACGCAGGAGAGGTAAGA-3′, antisense: 5′-GCTCGACAATGTTCTCCAGCTT-3′. The cycle threshold value of each target gene was normalized to that of the
Nonheme iron concentration in mouse liver and spleen
Hepatic and splenic iron concentrations were determined, as previously described. 30 Tissues were homogenized with 20 mM 4-(2-hydroxyethyl)-1-piperazine ethane sulfonic acid, digested with nitric acid, and measured using a graphite furnace atomic absorption spectrophotometer (PerkinElmer, Waltham, MA, USA).
Statistical analysis
Statistical analyses were performed with GraphPad prism. Data were presented as mean ± standard error of the mean. The difference between the means was determined by Student’s
Results
CPX induced a significant reduction in RET, IRF, and Hb in the blood and an increase in iron levels in the serum, liver, and spleen in mice.
We first investigated the effects of CPX on RET, an important indicator of erythropoiesis, IRF, a predictor of erythropoietic recovery, 31,32 Hb in the serum and iron contents in the serum, liver, and spleen. CPX was found to induce a time-dependent reduction in RET (Figure 2(a);) and IRF (Figure 2(b)) during the period from day 0 to 5 (RET) or day 3 (IRF). The RET at days 1, 3, and 5 was significantly lower than that at day 0, however, that at day 11 was higher than that at day 0. In the case of IRF, the values only at days 1 and 3, but not day 5, were significantly lower than those at day 0, while the values at day 5 do not differ from those at day 0. The significant increase in IRF was found at day 11. There were no differences in RET and IRF between days 30 and 0. CPX induced a significant increase in serum Hb at day 1 and the reduction at days 3, 5, and 11, and has no effect at day 30 (Figure 2(c)). The iron content in the serum (Figure 2(d)), liver (Figure 2(e)), and spleen (Figure 2(f)) was found to increase with time, peaked at day 3 (serum and liver) or 5 (spleen), and then decreased gradually in CPX-treated mice. There were no differences in iron content at days 30 and 0 in the serum (Figure 2(d)), liver (Figure 2(e)), and spleen (Figure 2(f)).

CPX significantly reduced RET, IRF, and Hb in the blood and elevated iron levels in the serum, liver, and spleen in mice. C57/B6 mice were treated with CPX (160 mg/kg) by intraperitoneal injection and then euthanized at days 0 (control, no CPX treatment), 1, 3, 5, 11, and 30 after CPX treatment. (a) Reticulocyte count (RET%: the percentages of reticulocytes in blood), (b) immature reticulocyte fraction (IRF%: the ratio of immature reticulocytes to total reticulocytes), and (c) Hb (g/L) in the blood and iron content in the (d) serum, (e) liver, and (f) spleen of mice were measured, as described in the “Methods” section. The values were presented as mean ± SEM of four to seven independent experiments. *
CPX induced a significant increase in the expression of ferritins in the liver and spleen
We also examined the effects of CPX on the expression of Ft-L and Ft-H proteins in the liver and spleen. Treatment with CPX for 3 (Figure 3(b) and (g)) and 5 (Figure 3(c) and (h)) days led to a significant increase in the expression of Ft-H and Ft-L proteins in the liver and for 3 (Figure 3(l) and (q)), 5 (Figure 3(m) and (r)), and 11(Figure 3(n) and (s)) days in the spleen. There were no significant differences in the expression of Ft-L and Ft-H between the control and CPX-treated mice at days 1 (Figure 3(a) and (f)), 11 (Figure 3(d) and (i)), and 30 (Figure 3(e) and (j)) in the liver and at days 1 (Figure 3(k) and (p)) and 30 (Figure 3(o) and (t)) in the spleen.

CPX induced a significant increase in the expression of ferritin in the liver and spleen. C57/B6 mice were treated with 0 (control) or 160 mg/kg of CPX by intraperitoneal injection and then euthanized at days 1, 3, 5, 11, and 30 after CPX treatment. The expression of ferritin in the liver ((a–e) Ft-L; (f–j) Ft-H) and spleen ((k–o) Ft-L; (p–t) Ft-H) was measured by Western blot analysis. The values were presented as mean ± SEM of four to seven independent experiments. *
CPX induced a significant reduction in the expression of TfR1 and Fpn1 in the liver and spleen
In most types of cells, iron balance is mainly dependent on the expression of TfR1 (importer) and Fpn1 (exporter). The expression of these two proteins is also regulated by cell iron content. To find out the possible mechanisms involved in the increased iron content in the serum and tissues, we then investigated the effects of CPX on the expression of iron uptake-protein TfR1 and iron release-protein Fpn1 in the liver and spleen. In the liver, the expression of TfR1 and Fpn1 was found to be significantly lower in mice at days 3 (Figure 4(b) and (g)) and 5 (Figure 4(c) and (h)) after CPX treatment than the corresponding control, while that in the mice at days 1 (Figure 4(a) and (f)), 11 (Figure 4(d) and (i)), and 30 (Figure 4(e) and (j)) after CPX treatment did not differ from that in the corresponding control. In the spleen, the significant differences in the expression of TfR1 and Fpn1 between the CPX mice and the control were found at days 1 (Figure 4(k) and (p)), 3 (Figure 4(l) and (q)), 5 (Figure 4(m) and (r)), 11 (Figure 4(n) and (s)), and 30 (Figure 4(o) and (t)).

CPX induced a significant reduction in the expression of TfR1 and Fpn1 in the liver and spleen. C57/B6 mice were treated with 0 (control) or 160 mg/kg of CPX by intraperitoneal injection and then euthanized at days 1, 3, 5, 11, and 30 after CPX treatment. The expression of TfR1 and Fpn1 in the liver ((a–e) TfR1; (f–j) Fpn1) and spleen ((k–o) TfR1; (p–t) Fpn1) was measured by Western blot analysis. The values were presented as mean ± SEM of four to seven independent experiments. *
CPX enhanced the expression of hepcidin and reduced the expression of IRP2 and Nrf2 in the liver
To explore the underlying mechanisms of how CPX treatment modulated the expression of iron transport proteins in the liver, we investigated the effects of CPX on the expression of hepcidin and IRPs in the liver because mammalian iron metabolism is regulated systemically by hepcidin and cellular by IRPs. 33,34 As compared with that at day 0, the expression of hepcidin mRNA is significantly increased at days 1, 3, and 5 after CPX treatment and then reduced, and there were no differences in the expression of hepcidin mRNA between day 0 and day 11 or 30 (Figure 5(a)). In the cases of the expression of IRPs, IRP2 content was significantly lower in CPX-treated mice than those in the controls (Figure 5(d)), while IRP1 expression remained unchanged (Figure 5(c)) at day 3 after CPX treatment. We also examined the expression of Nrf2 because impaired Nrf2 expression has been demonstrated to reduce Fpn1 transcription and then diminish iron egress in mouse and human cells. 35,36 It was found that Nrf2 mRNA expression was significantly lower in CPX-treated mice than that in the controls at day 3 after CPX treatment (Figure 5(b)).
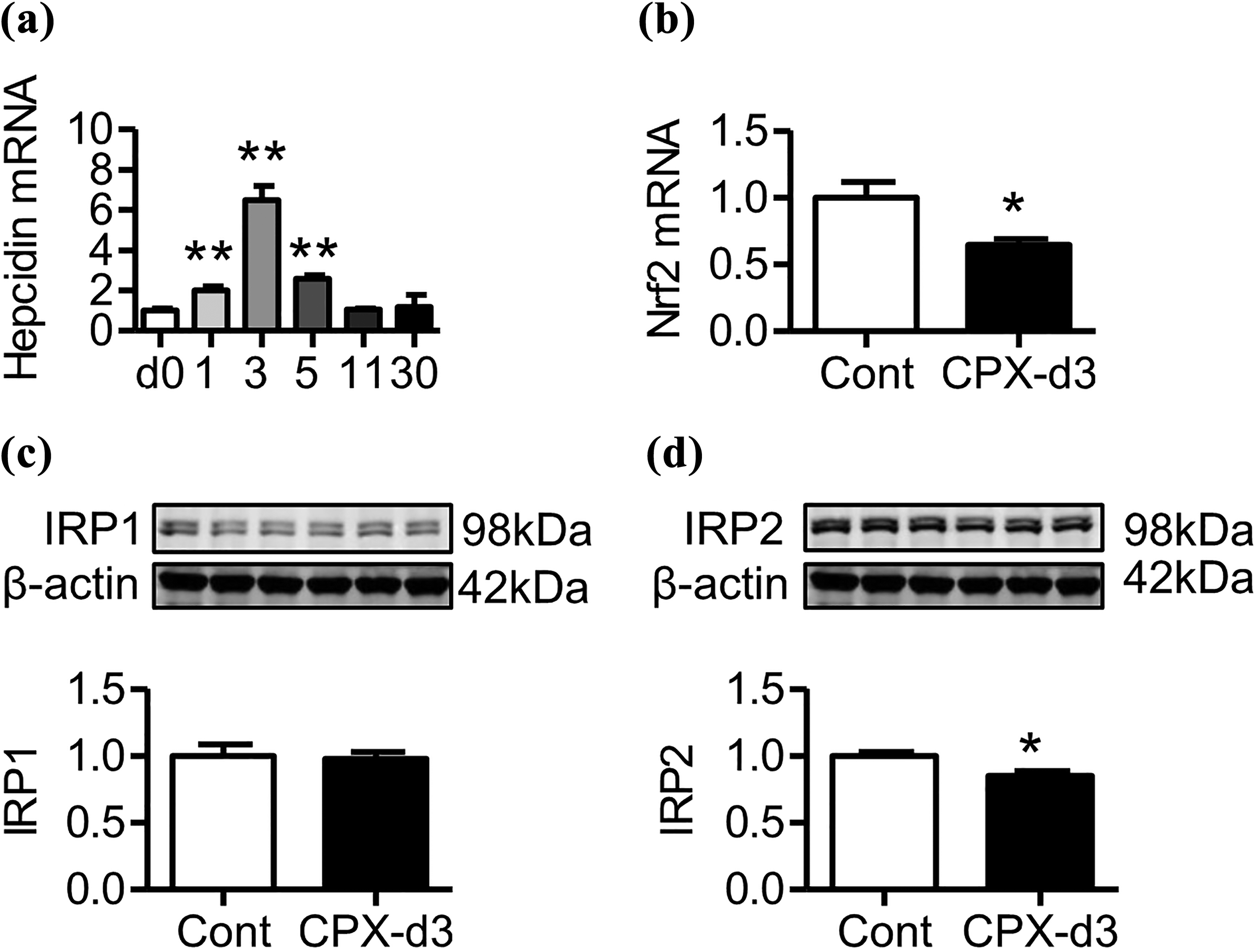
CPX significantly upregulated hepcidin mRNA and downregulated Nrf2 mRNA and IRP2 protein in the liver of mouse. (a) C57/B6 mice were treated with 0 (control) or 160 mg/kg of CPX by intraperitoneal injection and then euthanized at days 1, 3, 5, 11, and 30 after CPX treatment. The expression of hepcidin mRNA in the liver was measured by RT-PCR analysis. (b–d) C57/B6 mice were treated with 0 (control) or 160 mg/kg of CPX by intraperitoneal injection and then euthanized at day 3 after CPX treatment. The expression of (b) Nrf2 mRNA, (c) IRP1, and (d) IRP2 proteins in the liver was determined by RT-PCR or Western blot analysis, respectively. The values were presented as mean ± SEM of four to seven independent experiments. *
Discussion
The present study is the first to perform a comprehensive evaluation of the effects of CPX on the contents of RET, IRF (erythropoiesis) and Hb in the blood, and on iron levels and expression of proteins (or peptides) involved in iron storage, transport, and regulation in the liver and spleen of mice. The liver and spleen were picked because they are two important organs for iron metabolism and homeostasis. The liver is the major site for the production of proteins that maintain systemic iron balance and is also critical for the mobilization of iron from hepatocytes to the circulation to meet metabolic requirements, while the spleen (splenic macrophages) is a major site for iron recycling in conjunction with those of the liver.
As we expected, treatment with a single dose of CPX induced a significant increase in iron content in the liver, spleen, and serum all at days 1–5 after CPX injection. The iron content in these organs and serum all increased with time, peaked at day 3 or 5, and then decreased and returned to control (day 0) levels at day 11. Also, CPX induced a remarkable enhancement in the expression of iron storage proteins, Ft-L and Ft-H, in the liver and spleen. The levels of both Ft-L and Ft-H at days 3 and 5 in the liver and at days 3, 5, and 11 in the spleen in CPX mice were significantly higher than those in control mice and returned to control level at day 11 in the liver and at day 30 in the spleen. Because iron is a major generator of ROS, 37 the increased iron would lead to an increase in the ROS formation, and then damage biological molecules, such as lipids, carbohydrates, proteins, and nucleic acids and multiple organs. This may be one of the mechanisms for CPX to induce oxidative stress and severe toxicities (Figure 6).

A hypothetical scheme for the mechanisms involved in the oxidative stress and toxicities induced by CPX. CPX could inhibit erythropoiesis and Hb synthesis, inducing a reduction in iron usage and then an increase in iron content in the liver and spleen. The increased tissue iron may be one of the mechanisms for CPX to induce oxidative stress and toxicities. The reduced expression in TfR1 and Fpn1is a secondary effect of CPX-induced iron accumulation in the liver and spleen and also associated partly with the suppressed IRP/IRE system, upregulation of hepcidin, and downregulation of Nrf2. Nrf2: nuclear factor erythroid 2-related factor 2; IRP: iron regulatory protein; CPX: cyclophosphamide; Hb: hemoglobin; IRE: iron-responsive element.
Reticulocytes are newly produced immature red blood cells to be released from bone marrow to blood. 38,39 RET is an important indicator of erythropoiesis, which is the same as Hb synthesis, depending on the status of iron utilization by bone marrow. 40 IRF has been identified as a predictor of erythropoietic recovery. 31,32 The values of RET, IRF, and Hb in mice were demonstrated to be significantly reduced all at days 1, 3, 5, and 11 and all returned to the day 0 level after CPX injection. These findings were consistent with what was previously reported 41,42 and also provided further evidence that CPX can notably inhibit erythropoiesis and Hb synthesis and induce a prominent reduction in iron usage (Figure 6), which may be one of the curses to induce iron overload in the liver and the spleen.
To find out whether the CPX-induced increase in iron content in the liver and spleen is associated with its effects on the expression of cellular iron-uptake protein TfR1 and iron-release protein Fpn1, we also examined the changes in the expression of these two proteins in CPX-treated mice. In most types of cells, the expression of TfR1 and Fpn1 is a key to cellular iron balance 43 and also regulated by cellular iron contents. 44 We found that the expression of both TfR1 and Fpn1 is significantly lower, rather than higher, at days 3 and 5 in the liver of CPX-treated mice than that in controls and returns to control level at day 11. In the spleen, the expression of both TfR1 and Fpn1 is lower not only at days 3 and 5 but also at days 11 and 30 than that in controls and does not return to the control level during the period of our observation. These findings showed that the significantly reduced expression in TfR1 and Fpn1 may be mainly due to the increased iron content, namely, a secondary effect of iron accumulation in the liver and spleen, and also implied that restitution of TfR1 and Fpn1 expression in the liver is previous to that in the spleen in the CPX-treated mice (Figure 6). The causes or mechanism for the different recovery patterns are unknown and need to be investigated further.
It has been well-documented that the expression in TfR1 and Fpn1 is regulated by not only IRP/iron-responsive element (IRE) system but also hepcidin. Hepcidin and the IRP/IRE system are integrated together to precisely control the expression of TfR1 and Fpn1 under normal conditions. Under iron-replete conditions, the suppression of the IRP/IRE pathway, as found in the present study and by Mahmoud and Al Dera 45 and Miyazawa et al., 46 is able to promote ferritin mRNA transcription and downregulate TfR1 expression. 47 In addition, the reduced expression in TfR1 may partly be caused by increased hepcidin level because TfR1 has been found to be directly inhibited by hepcidin in different types of cells. 48,49 Fpn1 is a receptor for hepcidin, internalized and subsequently degraded after binding with hepcidin. 34 The reduced expression of Fpn1 may be mainly due to the CPX/iron-overload-induced increase in the expression of hepcidin (Figure 6). Also, the reduced expression of Nrf2 induced by CPX, as was found in the present study and by others, 45,46,50 might also be associated with the reduction in Fpn1 expression, although it is currently unknown how CPX reduces Nrf2 expression. Fpn1 contains an antioxidant-responsive element (ARE) at position 7 KB of its promoter sequence, and binding of Nrf2 to ARE facilitates its expression. 35 Therefore, CPX-induced Nrf2 downregulation may have contributed to the lowering of Fpn1 protein expression (Figure 6). This possibility needs to be investigated further.
Conclusion
In summary, we demonstrated for the first time that a single dose of CPX could induce a significant increase in iron content and ferritin expression in the liver, spleen, and/or serum and suggested that the increased iron in the tissues may be one of the mechanisms for CPX to induce oxidative stress and severe toxicities. Therefore, CPX combined with iron chelator clinically may be able to reduce tissue iron content, oxidative stress, and toxicities induced by CPX (Figure 5). We also provide further evidence that CPX can notably inhibit erythropoiesis and Hb synthesis and induce a prominent reduction in iron usage and the reduced iron usage may be one of the causes for iron overload in the liver and spleen (Figure 6). The findings also imply that the significantly reduced expression in TfR1 and Fpn1 is a secondary effect of CPX-induced iron accumulation in the liver and spleen and associated partly with the suppressed IRP/IRE system, the increased expression of hepcidin, and the downregulation of Nrf2.
Footnotes
Author note
YS and YJC contributed equally to this work.
Author contributions
YL and ZMQ conceived, organized, and supervised the study; YS, YJC, and JZ performed the experiments and/or contributed to the analysis of data. YL and ZMQ prepared and wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (81471108 and 31571195) and postdoctoral fellowship of Fudan University.
