Abstract
Quercetin is a natural flavonoid with potential anticancer properties without significant cytotoxicity in normal tissues. However, the effects of quercetin on Ehrlich ascites carcinoma (EAC) have not yet been clarified. The study aimed to show the antitumoral effect of quercetin through argyrophilic nucleolar organizer region (AgNOR) protein synthesis in mice carrying EAC. Thirty mice were used in the experiment (negative control (n = 6), tumor control (n = 8), quercetin 50 mg/kg (n = 8), and quercetin 100 mg/kg (n = 8) intravenously). The animals were euthanized on the 14th day, and the solid tumors were removed. Then, the total AgNOR area/nuclear area (TAA/NA) and average AgNOR number were calculated for each mice and the immunoreactivity of the factor VIII protein in tumor was evaluated. Tumor volumes and animal weights were statistically significant compared to the tumor control group (p < 0.05). Statistically significant differences were observed between the groups in terms of TAA/NA ratio (p < 0.05). Factor VIII expressions decreased in quercetin groups compared to control tumor tissue (p < 0.05). The current study showed that quercetin has an important function against cancer development.
Keywords
Introduction
It has been reported that quercetin-like natural plant flavonoids are beneficial to the body. 1 The term “vitamin P” is an old concept. It belongs to most flavonoid families. “Vitamin P” has also been used to describe the activity of some flavonoids, such as quercetin and myricetin. The antioxidant and antitumoral effects of plant extracts have been tested on many cancer models through proliferation, apoptosis, and modulation of angiogenesis pathways. Quercetin is one of the antioxidant flavonoids that can prevent active-free radicals from degrading low-density lipoproteins. 2 Flavonoids and their metabolites affect each stage of carcinogenesis through multiple signal transduction pathways. For this reason, it has been reported in studies that quercetin (lipid peroxidation inhibitor) can be beneficially used against cancer cells. In several studies, quercetin has been named as both an antioxidant and a prooxidant agent. In fact, the antioxidant or prooxidant activity of quercetin depends on the redox status of the cells and the quercetin concentration. Although it has been reported in studies that it promotes antioxidant and prooxidant properties against oxidative stress, similar to common anticancer drugs, such as doxorubicin, quercetin needs to be further investigated in the field of cancer. 3 –5 Quercetin has been reported in studies as a component that prevents the proliferation of several cell lines related to human breast cancer. Quercetin contributes to the prevention of tumor growth due to its prooxidant capability. “Vitamin P” was also used to define the activity of some flavonoids, including quercetin, myricetin, and rutin. Angiogenesis, the formation of new blood vessels from preexisting blood vessels, plays a crucial role in the growth, progression, and metastasis of tumors by meeting the oxygen, nutrient requirement of the growing tumor. It is now perceived as one of the promising strategies in cancer treatment. Recent studies have gained more attention due to the proangiogenic or antiangiogenic effects of quercetin. Some studies have reported the antiangiogenic effects of quercetin in some tumor types. 5,6 In studies conducted, it has been reported that quercetin has an anticancer feature with proapoptosis effect, inhibiting growth against cancer cell models and tumors. Again, considering the ability of quercetin to trigger multiple inflammations and carcinogenesis-related signaling pathways, including mitogen-activated protein kinase (MEK)/extracellular signal-regulated kinase (ERK), cat-catenin, signal transducer and activator of transcription 3 (STAT3), epidermal growth factor receptor (EGFR), phosphoinositide-3 kinases (PI3K), protein kinase B (AKT), and mammalian target of rapamycin (mTOR), and nuclear related factor 2 (Nrf2)/Kelch-like erythroid cell-derived protein with CNC homology [ECH]-associated protein 1 (Keap 1), it is capable of targeting multiple routes. The antitumor effect has been reported to show great promise. As a flavanone-type flavonoid found in human diet, it is mostly found in vegetables, tea, fruit, and wine. 7,8 Oxidative cell damage is known to be closely related to tumor formation, and the effects of various antioxidant plants (Viburnum opulus, curcumin, and Cornus mas L) on tumor formation have been reported in studies. 9 –11 In the literature, in various studies, quercetin was administered to mice at doses of 30, 40, 70, and 100 mg/kg. 12 –16 In this study, quercetin was investigated, which was tried in various types of cancer in different doses in Ehrlich ascites carcinoma (EAC) model. EAC first appeared as a spontaneous breast adenocarcinoma in a female mouse, and tumor fragments were transplanted from one mouse to the other subcutaneously and transformed into experimental tumors. Later, it was obtained in the form of growing liquid in the peritoneum of the mice, and this tumor was named EAC because of the formation of acid fluid in the peritoneum besides these cells. 11,17,18
EAC is used as an acid form or solid form depending on the purpose. If the acid fluid containing the tumor cell is injected into the experimental animal by intraperitoneal (ip) method, the acid form is obtained; if it is injected by the subcutaneous (sc) method, the solid form is obtained. 19 EAC cells grow in suspension in the peritoneal cavity of mice and do not adhere to artificial surfaces in vitro experiments. It can be understood that the acid started to form 4–6 days after the passage and 5–12 cc acid is formed. 20 EAC cells grow in the peritoneal cavity of the mouse in two phases following inoculation. These phases are the proliferation phase, in which the number of cells increases exponentially and the plateau phase, followed by which the number of cells remains almost constant. 19 Studies have shown that after ip injection of 1 × 106 EAC cells, the number of cells increased exponentially until the ninth day, and the plateau phase was entered from the 9th and 10th days. 20 –22 EAC is an undifferentiated tumor. The original of this tumor, which has a high transplantable rate, does not show regression and has a fast proliferation and a short survival time, leading to 100% death and does not have tumor-specific transplantation antigens in terms of immunology, and is also hyperdiploid. 9,10 Nucleolar organizer regions (NORs) are ribosomal gene regions on chromosomes. These regions consist of ribosomal DNA (rDNA) and proteins, and some of which have argyrophilic properties. These regions are copied to ribosomal RNA, which is converted into anterior ribosomes in nucleoli and mature ribosomes in the cytoplasm. 11 When these areas are active, they can be stained with silver. Depending on silver, these proteins are called argyrophilic NOR (AgNOR)-related proteins, and the silver staining method is most suitable for indicating nucleoli in interphase nuclei. 23 There are several studies on the importance of interphase AgNOR in tumor pathology for prognostic and diagnostic definitions of different types of cancer. 11,21,22,24
In the literature, there have been no studies on the application of different doses of quercetin, angiogenesis, and AgNOR protein determination on the EAC model. This study aims to investigate the antitumoral effect of quercetin in different doses on the EAC solid tumor model experimentally created in male mice of Balb/c type.
Methods
Subject type, number, and distribution used in the research
The stage of the study on experimental animals was made in accordance with the decision dated May 15, 2019, and numbered 19/115 obtained from the Animal Experts Local Ethics Committee of Erciyes University. In this study, Balb/c type, 8–10 week-old male mice weighing 25–30 g, was used. During the study, the mice were housed in specially prepared, automatically conditioned rooms with a constant temperature of 21°C and 12 h of light/dark periods. To obtain enough EAC cells, the stock mouse was first formed before the groups were created in the study. EAC cells from stock mice were used for solid tumor formation.
Experimental groups
EAC and quercetin injections were applied to the experimental groups, as specified in the experimental model (Table 1). The weights of the animals were measured from the first day of the experiment until the day they were euthanized. At the same time, the solid tumor area was palpated and checked daily, and tumor diameters were measured with an electronic caliper after the tumor started. To measure tumor volumes, the formula used was Tumor volume (mm3) = Width2 × Length × 0.52. 25
Creation of experiment groups.

PBS: phosphate-buffered saline; EAC: Ehrlich ascites carcinoma; sc: subcutaneous; ip: intraperitoneal.
Formation of the stock mice
EAC Erciyes University Genome and Stem Cells were available. The EAC cells that were kept at −80°C were used in forming the stock animals. The stock cells were thawed at room temperature and were administered to stock animals as 0.1 ml ip at the joint of the left rear leg and the stomach area. It was expected that ascites tumors would occur in the stock animal within 6–7 days. The 1 × 106 EAC cells in the ascites fluid were drawn with the help of an injector from the stock animal. It was administered to the neck area in a subcutaneous manner to form a solid tumor.
Histological analysis
Tissues taken at the end of the experiment were fixed in a 10% formaldehyde solution. After the detection process, the tissues were dehydrated by passing through increasing alcohol series (50%, 70%, 80%, 96%, and 100%). Tissues that were made transparent with xylene were embedded in paraffin. Hematoxylin and eosin staining was applied to 5-μm-thick sections taken from paraffin blocks and then closed with a closing solution (Entellan®, Merck, Minneapolis, MN, USA). The prepared slides were examined under an Olympus BX53 Light Microscope incekaralar Ankara Turkey.
Immunohistochemical analysis
Factor VIII staining is one of the methods used in determining the increasing angiogenesis in the tumor tissue. For this reason, we used this method in the present study of ours. The immunoreactivity of the factor VIII protein in tissues was determined using the avidin–biotin peroxidase method. In summary, after deparaffinization of sections taken at a thickness of 5 μm, citrate buffer was used to open the epitopes (pH 6.0). The slides were then taken into a 3% hydrogen peroxide solution in methanol to prevent endogenous peroxidase activity. Ultra V block solution was applied to prevent nonspecific staining. The staining kit Cat number was BS-10048RpolyclonalAC03185423. The sections were then incubated overnight at 4°C with primary antibodies. Secondary streptavidin-Horseradish peroxidase (HRP) and 3,3 diaminobenzidine (DAB) chromogens were biotinylated, respectively, and then sections were counterstained with Gill’s hematoxylin. The sections were dehydrated by passing through increasing alcohol series and closed with a concealer called entellan. The sections were then examined with an Olympus BX53 Light Microscope. The evaluation of the immunoreactivity levels was done with ImageJ Program 1.46r. A total of 10 different areas were evaluated for each slide.
AgNOR staining
Five micrometer sections were taken from the slides from tissues embedded in 10% neutral formalin and paraffin. The sections were deparaffinized, rehydrated, and stained. AgNORs were stained (silver nitrate was supplied as a powder from Sigma Aldrich, USA (Cat number: 209139) according to the method proposed by the International AgNOR Quantitative committee, as described by Ploton et al. 26 Silver-stained samples were evaluated under a light microscope (Eclipse E-600, Nikon, Japan) using an image analysis system (NIS Elements Nikon, Japan). Fifty nuclear AgNOR protein images were evaluated per sample. The average AgNOR number and total AgNOR area (TAA)/total nuclear area (NA) ratio were calculated for each core.
Statistical analysis
SPSS for Windows® 23.0 (SPSS, Chicago, Illinois, USA) was used to analyze the data. The results are presented as the mean ± standard deviation of replicates. The data in all the experiments were analyzed for statistical significance using one-way analysis of variance (ANOVA). Post hoc analyses were conducted to compare Tukey test parameters in multicomponent comparisons. Tukey test or Dunn–Bonferroni test was used for multiple comparisons. For intergroup comparisons, the Kruskal–Wallis analysis was used. For factor VIII between the groups, ANOVA or the Kruskal–Wallis was used. The p value <0.05 was considered statistically significant. Another program used was GraphPad Prism for Windows (version 7.00, La Jolla, California, USA).
Results
Change of body weights of experimental groups
When the data on the daily body weights of the animals belonging to the groups during the experiment (14 days) were examined, it was determined that there was an increase in the weight in the tumor control group and the quercetin-administered groups, and this increase was higher in the tumor control group compared to the other groups (p < 0.05; Figure 1).
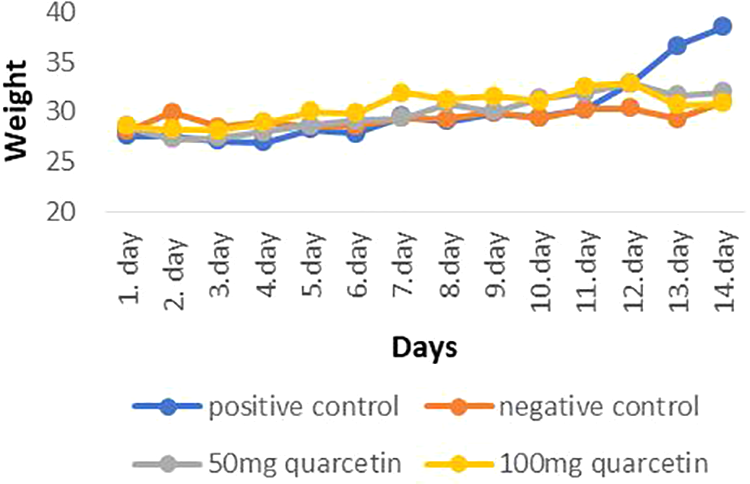
Mean weight changes in the control and experimental groups. When compared with the treatment groups, the average weight of the tumor control group appears to be significantly higher (p < 0.05).
Tumor volume change of experimental groups
Tumor measurement was started to be measured from the seventh day in the tumor control group, while these measurements could be measured from the eighth day in the treatment groups. On the last day of the experiment, tumor volumes were measured as 4602.91 mm3 in the tumor control group, 1297.76 mm3 in the 50 mg/kg quercetin group, and 2087.36 mm3 in the 100 mg/kg quercetin group. All tumor sizes in all animals were compared volumetrically. When the treatment groups and the tumor control group were compared, the tumor volume increase was found statistically significant (p < 0.05) and the findings of this comparison are presented in Table 2.
Comparison of the solid-tumor volumes between the groups.a

a Values measured in mm3.
b Compared with tumor control group, p < 0.05.
Histological and Immunohistochemical findings
Positive and negative staining was observed in vascular endothelial cells in the sections of control and treatment groups with immunohistochemical staining for factor VIII. In general, it was determined that from place to place, mitosis occurred, nucleus size increased, nucleus circumference became irregular, and cytoplasmic area decreased at the solid tumor tissues in all tumor groups. The presence of a large number of various inflammatory cells (neutrophils, lymphocytes, etc.) observed in necrotic areas supports the presence of necrosis. As seen in Figure 2, it is observed that as the dose of quercetin increased, necrosis of the dead tumor cells also increased. Necrotic cells are seen separately in lighter colors, breaking their relationship with each other. Factor VIII immunoreactivity results are shown in Figure 3. When the groups were compared with each other, it was determined that factor VIII expression increased in the 100-mg quercetin group (p < 0.05).

H&E images (star: necrosis, arrow: tumor cells) of (a, d, g) control, (b, e, h) 50 mg quercetin, and (c, f, i) 100 mg quercetin groups. Zoom: (a–c) ×20, bar = 50 μm; (d–h) ×40, bar = 20 μm. H&E: hematoxylin and eosin.
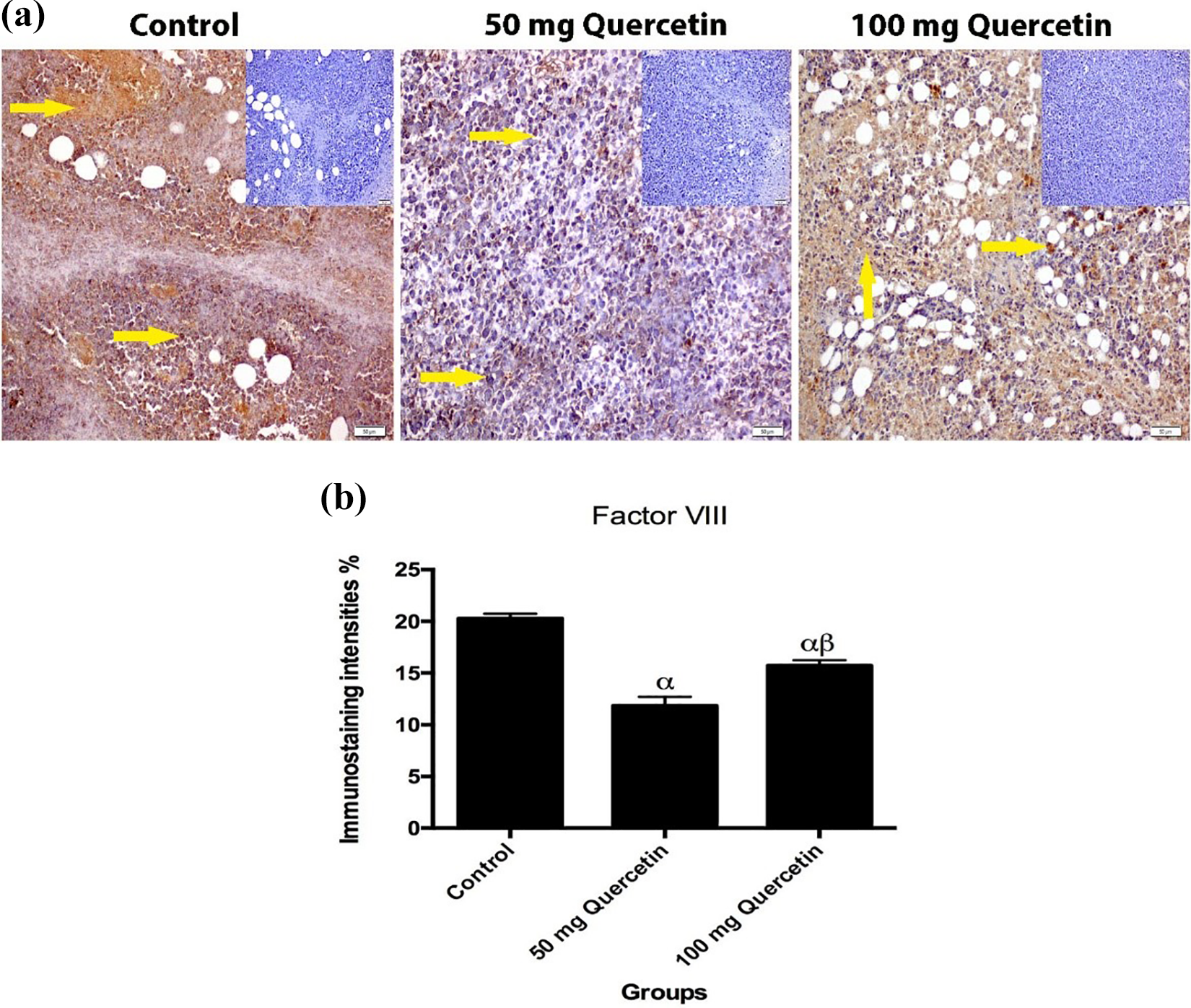
(a) Immune staining of factor VIII in solid tumors belonging to the control, 50 mg quercetin, and 100 mg quercetin experimental groups. Positive controls for each group were added to the immunoreactivity results of the factor VIII protein. Positive controls of each group can be seen in the corners of the photos in addition, the immunoreactive area is indicated by the yellow arrow (×20 magnification, bar = 50 µm). (b) Histopathological score data shown on histogram chart (expressed as mean ± SD). One-way ANOVA and Tukey post hoc multiple comparison test were applied (α, p < 0.05 control group; β, p < 0.05; 50 mg quercetin group). SD: standard deviation; ANOVA: analysis of variance.
AgNOR results
An example of AgNOR staining cells is shown in Figure 4. The TAA/NA ratio and average AgNOR number were determined in the quercetin (50 and 100 mg/kg) groups and in the tumor control group (Table 3). There were statistically significant differences between the three groups for TAA/NA ratio (x 2 = 84.321, p = 0.000) and average number of AgNOR (x 2 = 212.320, p = 0.000; Table 4). TAA/NA ratio (between quercetin 50 mg/kg and positive control (Z = −2.502, p = 0,003), quercetin 100 mg/kg and positive control (Z = −3.611, p = 0.001), quercetin 50 mg/kg, and quercetin 100 mg/kg (Z = −5.428, p = 0.001)) was significant in the double comparison of the groups (Table 5).

Examples of AgNOR staining cells in experimental groups: (a) positive control group, (b) 50 mg/ kg group, and (c) 100 mg/kg group. AgNOR: argyrophilic-nucleolar organizer region.
TAA/NA and the mean AgNOR number values of positive controls, quercetin 50 mg/kg, and quercetin 100 mg/kg groups.

TAA/NA: total AgNOR area/nuclear area; AgNOR: argyrophilic nucleolar organizer region.
Comparison of three groups for the mean AgNOR number and TAA/NA ratio.
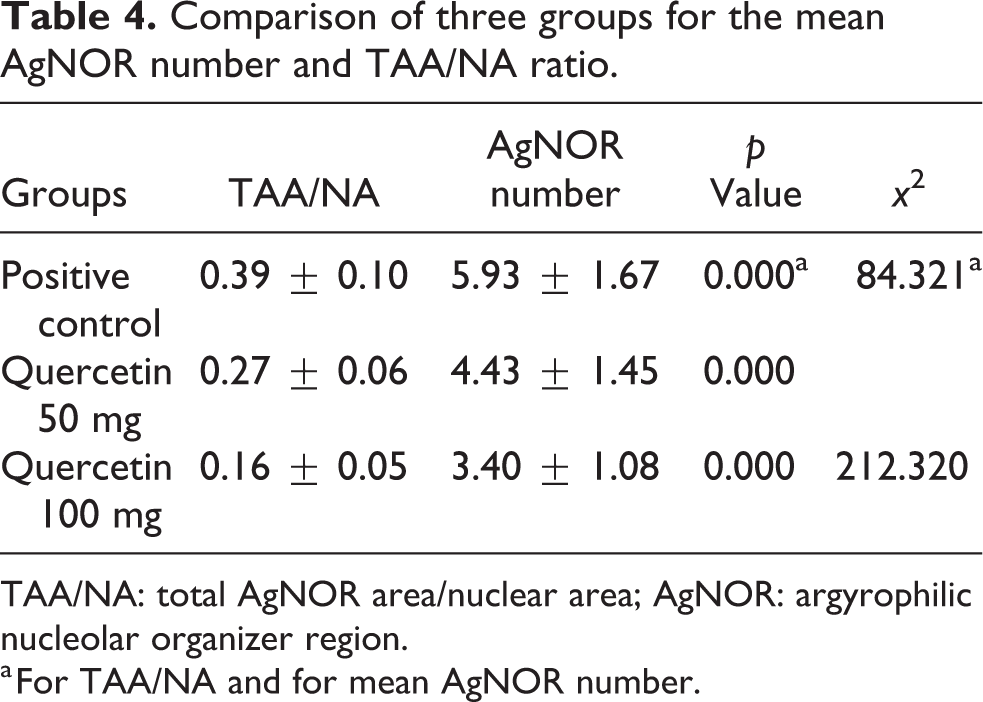
TAA/NA: total AgNOR area/nuclear area; AgNOR: argyrophilic nucleolar organizer region.
a For TAA/NA and for mean AgNOR number.
Double comparison of all groups.
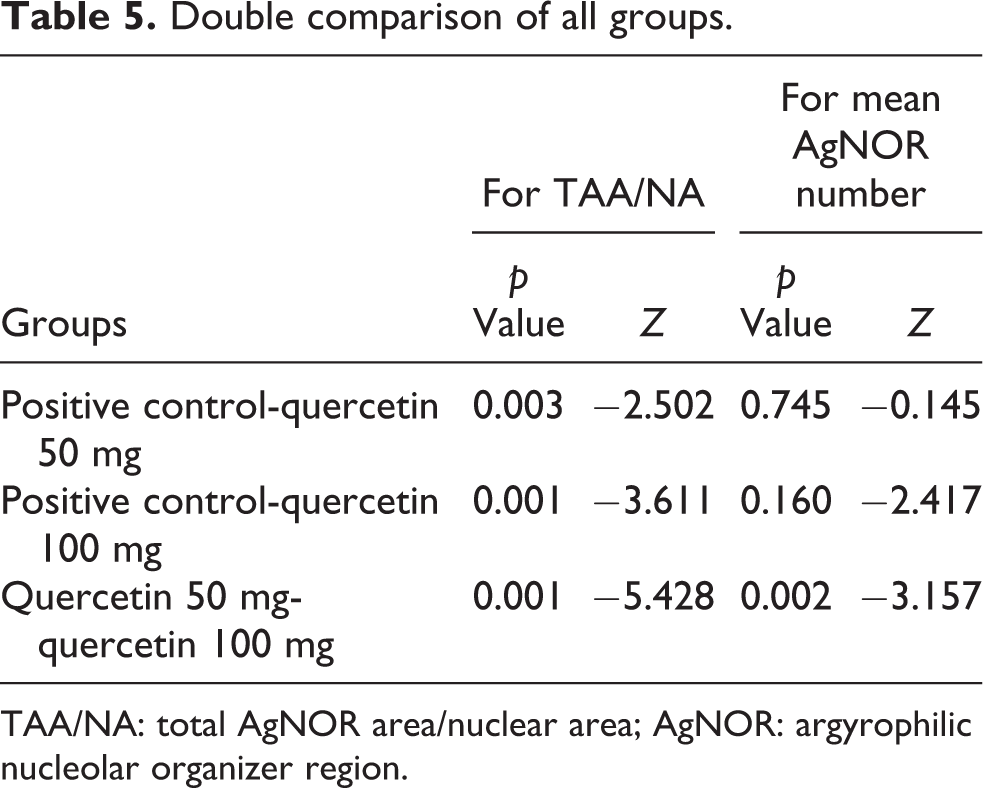
TAA/NA: total AgNOR area/nuclear area; AgNOR: argyrophilic nucleolar organizer region.
Discussion
Cancer is a common case in people and is increasing day by day. 27 It is known that chemotherapeutic agents used in cancer treatments have high toxicity and side effects. Chemotherapies naturally kill normal and neoplastic cells. Although reactive oxygen species play a role in cancer cell death mechanisms, they generally affect the human body negatively. 28 Therefore, in addition to these treatments, phytochemicals obtained from plants have been reported to have no side effects. Flavonoids are polyphenolic substances derived from plants that play a role in various pharmacological activities. They have antiviral, antimicrobial, anti-inflammatory, and antiallergic potential that can affect different cell types in both animal and human experiment models. Many inhibit a large molecular target range in the micromolar concentration range by downregulating or suppressing a large number of inflammatory pathways and functions. 9 –11,29,30 Phenolic components obtained from the products of plants play an important role in the prevention of cancer. Epidemiological studies show that diets containing polyphenolic compounds are effective in decreasing cancer rates. 31 One of those described as flavanol derived from nature is considered quercetin. Quercetin exhibits biphasic behavior in basophils at nanomolar doses, and therefore, its effect on cells involved in allergic inflammation has been reported in studies. Like many other molecules that share a flavone ring, quercetin affects immunity by acting mainly on leukocytes and targeting many intracellular signal kinases and phosphatase, enzymes, and membrane proteins that are often very important for a cellular-specific function. 32 Herbal medicines lead cancer cells to apoptosis by blocking the nuclear factor kappa B (NF-κB) pathway, increasing CD95L signaling, and increasing CD95L expression, tyrosine kinase, angiogenesis, and cell inhibition. 33 Many pharmacological properties of quercetin have been extensively investigated and have been reported to block the progression of various human tumors, such as oral cancer and breast cancer. 7 In a study on quercetin and its doses, it has been reported that quercetin reduces angiogenesis. 34 Whether the antioxidants investigated in experimental studies have antiangiogenic effects on tumor vascularization is important in determining the anticarcinogenic activity of the agent. 32,33 In the literature, there are articles reporting that the angiogenesis is decreased in the tumor groups as a result of factor VIII staining in studies with plants with antioxidant properties (Cornus mas L, curcumin, resveratrol, Viburnum opulus, quercetin). 8 –11,35 –37 Yang et al. showed that AuNPs-Qu-5 inhibits angiogenesis and metastasis of quercetin in breast cancer cells tested by targeting the EGFR/ vascular endothelial growth factor receptor 2 (VEGFR-2) signal pathway. 5 In our study, the expression of FVIII in solid tumor masses was evaluated and the decrease in FVIII expression was found statistically significant in the groups given quercetin (p < 0.05). Some transcriptionally active proteins and rDNA form NOR on chromosomes, and these active proteins are mostly argyrophilic. Transcribed rDNA, which binds to silver, silver-stained NORs, and AgNOR-related proteins, is identified as region-dependent proteins (AgNORs) that regulate argyrophilic nucleus. As a technique, AgNOR staining is not specific for a particular protein. Various types of proteins associated with the silver-binding ribosome can form AgNORs. The determination of the activities of these proteins is used as a marker of the proliferation and metabolic activity of the cells. 21,22 In these studies, it was found appropriate to use the average AgNOR number and TAA/NA ratio in routine cytopathology to detect the proliferation activity of tumor cells. We aimed to show whether quercetin has an antitumor effect on the amount of AgNOR protein. As far as we know, this study is the first research on the determination of quercetin EAC AgNOR quantities. When the three groups were compared in this study, a statistically significant difference was found between the groups in terms of the average number of AgNOR. When groups were compared in pairs, there was a statistically significant difference between the control and quercetin (50 mg/kg) and TAA/NA (100 mg/kg) ratio. According to AgNOR stabbing results, it can be said that 100 mg/kg quercetin dose is more reliable in cancer treatment. Evaluation of AgNOR points using a light microscope is subjective and cannot be reproduced. Also, single AgNOR points can be stacked together or overlapped. The different size of each silver point is not taken into account only when counting AgNOR. In cancer cells, in addition to gene expression, more reliable information about the proliferative and metabolic activity of cells can be determined by calculating the use of the NOR region, the nucleus area. It is important to identify new biomarkers to differentiate benign and malignant tumors 38 –41 Also, choosing the most reliable therapeutic strategy for cancer treatment is crucial to increase the success rate of treatment.
Conclusion
Additional research should be conducted to further investigate the effectiveness of antioxidant plants and fruits used in cancer treatments. According to the results we obtained in our study, quercetin is an important antioxidant that can be used for the treatment of cancer development in immunohistochemistry and AgNOR staining results. Research in the literature has shown that the results of the TAA/NA ratio can be used as a reliable biomarker on the therapeutic success rate and selection of a reliable dose for cancer treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
