Abstract
The current study was aimed to investigate the ameliorative effect of lycopene against gentamicin-induced testicular toxicity in adult rat testes. Pretreatment with lycopene (4 mg/kg/day) significantly prevented the decrease in the absolute testes weight and relative testes weight and the reduction in sperm count, motility, viability, and daily sperm production in gentamicin (100 mg/kg/day)-treated rats. Gentamicin significantly decreased the level of serum testosterone and testicular lactate dehydrogenase-X and G6PDH activities but a marked increase was observed upon pretreatment with lycopene. Testicular caspase-3 and -9 activities were significantly increased but lycopene showed significant protection from gentamicin-induced apoptosis. Oxidative stress was induced by gentamicin treatment as evidenced by increased hydrogen peroxide level and lipid peroxidation and decreased the antioxidant enzymes superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase activities and glutathione content. These alterations were effectively prevented by lycopene pretreatment. Histopathological examination showed loss of spermatogenesis and morphological abnormalities of the testis after treatment with gentamycin. These abnormalities were effectively normalized by pretreatment with lycopene. In conclusion, gentamicin decreases rat testes weight and inhibits spermatogenesis. It induces oxidative stress and apoptosis by possible mitochondrial dysfunction. These data provide insight into the mode of action of gentamicin-induced testicular toxicity and the beneficial role provided by lycopene to restore the suppressed spermatogenesis.
Introduction
Aminoglycosides including gentamicin have been successfully used for decades in the treatment of gram-negative bacterial infections 1 and currently regain popularity because of widespread resistance to other antibiotic classes. 2 However, nephrotoxicity is an associated limitation for the clinical use of gentamicin. 3 Moreover, gentamicin produces testicular toxicity, thereby limiting the therapeutic window. 4
Previous studies have demonstrated that gentamicin (100 mg/kg for 10 days) can impair sperm motility, reduce reproductive organ weights, and cause apoptosis in the rat testes, eventually resulting in testicular failure. 5 –7 Oxidative stress and generation of reactive oxygen species (ROS) have been implicated in the pathophysiology of gentamicin toxicity. 8 Oxidative stress plays a critical role in the pathogenesis of reproductive disorders, the etiology of defective sperm function and male infertility. 9 Therefore, oxidative stress has been reported in the renal and testicular toxicity of gentamicin and several antioxidants have been used to protect against gentamicin-induced renal toxicity. 10,11
Lycopene, a naturally occurring carotenoid, has attracted particular attention in recent years because it is a highly efficient antioxidant, possessing a singlet-oxygen and free-radical scavenging capacity. 12 Thus, lycopene can reduce oxidative stress by scavenging free radicals and preventing damage of the essential components in cells such as lipids, proteins, and DNA. 13 It has also many biochemical functions, some of which include antihyperlipidemic, anti-inflammatory, and antiapoptotic effects 14 and chemopreventive agent against certain types of cancers such as prostate cancer. 15 In addition, lycopene serves as a promising intervention to testicular toxicity associated with oxidative stress. 16,17 Therefore, the current study was aimed to investigate the ameliorative effect of lycopene against gentamicin-induced testicular toxicity in adult rat testes.
Materials and methods
Reagents
Gentamicin (CAS-No: 1405410, 100% pure) and lycopene (CAS-No: 502658 purity ≥ 98%) were purchased from Sigma-Aldrich Chemical Company (St. Louis, Missouri, USA). Other reagents were of analytical grade.
Animals
Adult male Wistar rats (80–90 days old) weighing 170 ± 10 g were obtained from our animal facility (Faculty of Pharmacy, King Abdulaziz University, Jeddah, Saudi Arabia), housed in clean polypropylene cages and maintained on a 12 h light/dark cycle and a temperature of 20–25°C with ad libitum access to standard food and water. For 7 days before the experiment, rats were handled daily for 5 min to acclimatize them to human contact and minimize their physiological responses to handling for subsequent protocols. 18 All animal treatment described was conducted according to Research Ethics Committee of King Abdulaziz University, Jeddah, Saudi Arabia.
Experimental protocol
The animals were randomly divided into four groups consisting of six animals each. Group I (control) received vehicle. Group II (lycopene group) received lycopene (4 mg/kg/day suspended in corn oil by oral gavage). Group III (gentamicin group) treated with gentamicin (100 mg/kg/day by intraperitoneal) in a single injection. Group IV (lycopene-gentamicin group) treated with lycopene and gentamicin by the same schedule. Groups II and IV received lycopene from day 1 till day 15 (total period of the experiment). Groups III and IV received gentamicin for 10 consecutive days, starting from the sixth day of the experiment till day 15. The doses of gentamicin 3,4 and lycopene 19 –21 in this study were selected on the basis of the previous studies. The experiment was continued for 15 consecutive days.
Necropsy
Twenty-four hours after the last dose, blood samples were collected from the retro-orbital sinus under anesthesia with sodium thiopental (20 mg/kg body weight). Samples were centrifuged and supernatant serum was separated from the clot as soon as possible and stored at −20°C until testosterone assay. Animals were euthanized and the testes were excised immediately, cleaned from adhering fat and connective tissues and the weights were recorded in grams. The cauda epididymides from each animal were used for sperm count, motility, and viability. Half of the right testis from each rat was homogenized in ice-cold phosphate buffer (pH 7.0) using a glass-teflon homogenizer. The homogenate was centrifuged at 10,000 ×
Sperm count and motility
Cauda epididymides were dissected out, immediately minced in 5 ml of physiological saline and then incubated at 37°C for 30 min to allow spermatozoa to leave the epididymal tubules. The percentage of motile sperms was recorded using a phase contrast microscope at a magnification of 400×. Total sperm number was determined by using a Neubauer hemocytometer as previously described. 22 To determine sperm motility, 100 sperms each were observed in three different fields, and classified into motile and nonmotile sperms, and the motility was expressed as percentage incidence.
Sperm viability
A 20 µl of sperm suspension was mixed with an equal volume of 0.05% eosin-Y. After 2 min incubation at room temperature, slides were viewed under the microscope using 400× magnification. Dead sperms appear pink and live sperms were not stained. 23 Two hundred sperms were counted for each sample and viability percentage was calculated. Sperm viability was defined as the percentage of intact cells. 24
Daily sperm production
DSP was determined in adult rats as previously described. 25 Half of the right testis was decapsulated and homogenized in 50 ml of ice-cold 0.9% sodium chloride solution containing 0.01% Triton X-100 using a Polytron homogenizer. 26 The homogenate was allowed to settle for 1 min and then was gently mixed, and a 10 ml aliquot was transferred to a glass vial and stored on ice. After thorough mixing of each sample, the number of sperm heads (step 19 spermatid head) in four chambers of Neubauer type hemocytometer was counted under a light microscope with 40× objective. To calculate DSP, the number of spermatids at stage 19 was divided by 6.1, which is the number of days of the seminiferous cycle in which these spermatids are present in the seminiferous epithelium.
Serum testosterone
Testosterone was measured by use of the Pathozyme Testosterone ELISA kit. Briefly, standards, specimens, and controls were dispensed into appropriate wells, followed by testosterone HRP reagent and antitestosterone reagent, before mixing thoroughly and incubating at 37°C for 90 min. Wells were then rinsed with deionized water and substrate solution was dispensed into each well, gently mixed, and incubated for 20 min. The reaction was stopped with the “stop reagent” and the absorbance recorded at 450 nm. 27 The intensity of the color formed is proportional to the amount of enzyme present and is inversely related to the amount of unlabeled testosterone in the sample.
Lactate dehydrogenase-X and glucose-6-phosphate dehydrogenase activities
Lactate dehydrogenase-X (LDH-X) activity was measured using α-ketovaleric acid as the substrate. 28 Glucose-6-phosphate dehydrogenase (G6PDH) activity was assayed by following the rate of reduction of NADP. 29 The assay mixture contained 0.1 ml of 3.8 mM of NADP, 0.1 ml of 63 mM MgCl2, 0.1 ml of 33 mM glucose-6-phosphate, and 0.5 ml of distilled water. Solutions were mixed, incubated for 5 min at 30°C, and the reaction was started by adding the enzyme sample. The rate of increase in absorbance was recorded for 5 min at 2340 nm.
Caspase-3 and -9
Enzymatic activities of caspase-3 and -9 were assayed as per the manufacturer’s protocol supplied in the caspase colorimetric assay kit (Enzo Life Sciences, New York, USA). Briefly, an aliquot of enzyme source (100 µg protein/50 µl) was added to 50 µl of reaction buffer containing 200 µM of chromogen (Ac-DEVD-pNA/Ac-LEHD-pNA for caspase-3 and -9, respectively), kept at 37°C for 2 h and terminated by adding stop buffer. The change in the absorbance due to the release of p-nitroanilide (pNA) was measured at 405 nm using a microtiter plate reader. Enzyme activity was defined as produced μmol pNA/min/mg protein and expressed as U/mg protein.
Oxidative status
Hydrogen peroxide production
Hydrogen peroxide (H2O2) generation was assayed by the method of Pick and Keisari. 30 Briefly, the incubation mixture contained 1.641 ml phosphate buffer (50 mM, pH7.6), 54 µl horseradish peroxidase (8.5 units/ml), 30 µl of 0.28 nM phenol red, 165 µl of 5.5 nM dextrose, and 600 µl of enzyme source, incubated at 35°C for 30 min. The reaction was terminated by the addition of 60 µl of 10 N sodium hydroxide. The absorbance was read at 610 nm against a reagent blank on a spectrophotometer. For standard curve, known amounts of H2O2 and all the above reagents except enzyme source were incubated for 30 min at 35°C, followed by the addition of 60 µl of 10 N sodium hydroxide, and reading of optical density at 610 nm. The quantity of H2O2 produced was expressed as nmol of H2O2 generated/min/mg protein at 35°C.
Lipid peroxidation
Malondialdehyde (MDA), formed as an end product of the peroxidation of lipids, served as an index of the intensity of oxidative stress. MDA reacts with thiobarbituric acid to generate a colored product that can be measured optically at 532 nm. A breakdown product of lipid peroxidation (LPO), thiobarbituric acid reactive substance was measured by the method of Buege and Aust.
31
Briefly, the stock solution contained equal volumes of trichloroacetic acid 15% (w/v) in 0.25 N HCl and 2-thiobarbituric acid 0.37% (w/v) in 0.25 N HCl. One volume of the test sample (sperm suspension) and two volumes of stock reagent were mixed in a screw-capped centrifuge tube, vortexed and heated for 15 min on a boiling water bath. After cooling on ice, the precipitate was removed by centrifugation at 1000 ×
Antioxidant status
Superoxide dismutase
Superoxide dismutase (SOD) was assayed by the method of Marklund and Marklund. 32 Briefly, the assay mixture contained 2.4 ml of 50 mM Tris–HCl buffer containing 1 mM EDTA (pH 7.6), 300 µl of 0.2 mM pyrogallol, and 300 µl enzyme source. The decrease in absorbance was measured immediately at 420 nm against blank at 10 s intervals for 3 min on a spectrophotometer. The activity of enzyme was expressed in nmol pyrogallol oxidized/min/mg protein.
Catalase
Catalase (CAT) was assayed as previously mentioned. 33 Briefly, the assay mixture contained 2.40 ml of phosphate buffer (50 mM, pH 7.0), 10 µl of 19 mM H2O2, and 50 µl enzyme source (sperm suspension). The decrease in absorbance was measured immediately at 240 nm against blank at 10 s intervals for 3 min on a spectrophotometer. The activity of enzyme was expressed in µmol of H2O2 consumed/min/mg protein.
Glutathione peroxidase
Glutathione peroxidase (GPx) was assayed by the method of Paglia and Valentine. 34 Briefly, the assay mixture contained 1.59 ml of phosphate buffer (100 mM, pH 7.6), 100 µl of 10 mM EDTA, 100 µl of sodium azide, 50 µl of glutathione reductase (GR), 100 µl of reduced glutathione (GSH), 100 µl of 200 mM NADPH, 10 µl of H2O2, and 10 µl enzyme source. The oxidation of NADPH was measured immediately at 340 nm against blank at 10 s intervals for 3 min on a spectrophotometer. The activity of enzyme was expressed in nmol of NADPH oxidized/min/mg protein.
Glutathione reductase
The activity of GR was assayed by the method of Carlberg and Mannervik. 35 Briefly, the assay mixture contained 1.75 ml of phosphate buffer (100 mM, pH 7.6), 100 μl of 200 mM NADPH, 100 μl of 10 mM EDTA, 50 μl of 20 mM oxidized GSH, and 50 μl enzyme source. The oxidation of NADPH was measured immediately at 340 nm against blank at 10 s intervals for 3 min on a spectrophotometer. The activity of enzyme was expressed in nmol of NADPH oxidized/min/mg protein.
Total reduced glutathione
Total reduced GSH was determined by the method of Moren et al. 36 Briefly, 1.0 ml of cell extract was precipitated with 1.0 ml of 10% trichloroacetic acid. The precipitate was removed by centrifugation. To an aliquot of the supernatant, 4 ml of phosphate solution and 0.5 ml of 5,5′-dithiobis-2-nitrobenzoic acid reagent were added. The color developed was read at 412 nm against a reagent blank. The amount of GSH was expressed as μg/mg protein.
Histopathological examination of the testes
Autopsy samples were taken from the testes of euthanized rats and fixed in 10% formalin solution for 10 h at least, then washed in tap water for 12 h. Serial alcohols (methyl, ethyl, and absolute) were used for dehydration of the tissue samples. Tissue specimens were cleared in xylene and embedded in paraffin. The paraffin blocks were sectioned at 3 µm thickness by sledge microtome. The obtained tissue sections were collected on the glass slides and stained by hematoxylin and eosin stain for histopathological examination by the light microscope.
Statistical analysis
Differences between obtained values (mean ± standard deviation,
Results
Testes weight and sperm parameters
The body weight of animals did not show any significant change at the end of gentamicin treatment (data not shown). Table 1 displays the changes in testes weight and sperm parameters. Treatment of male rats with gentamicin caused significant decrease (
Effect of gentamicin and lycopene on testes weight and sperm parameters.a

SD: standard deviation; ANOVA: analysis of variance.
aData are expressed as mean ± SD (
Serum testosterone
Serum testosterone was significantly (17.16%,

Serum testosterone level in gentamicin-treated rats in the absence (gentamicin) and presence of lycopene (gentamicin + lycopene). Group I: control, group II: lycopene, group III: gentamicin, group IV: lycopene + gentamicin. Data are expressed as mean ± SD (
Testicular LDH-X and G6PDH activities
The activities of LDH-X and G6PDH enzymes revealed a significant decrease (26.27% and 30.19%, respectively,

Testicular LDH-X and G6PDH activities in gentamicin-treated rats in the absence (gentamicin) and presence of lycopene (gentamicin + lycopene). Group I: control, group II: lycopene, group III: gentamicin, group IV: lycopene + gentamicin. Data are expressed as mean ± SD (
Caspase-3 and -9
Caspase-3 and -9 activities were significantly (
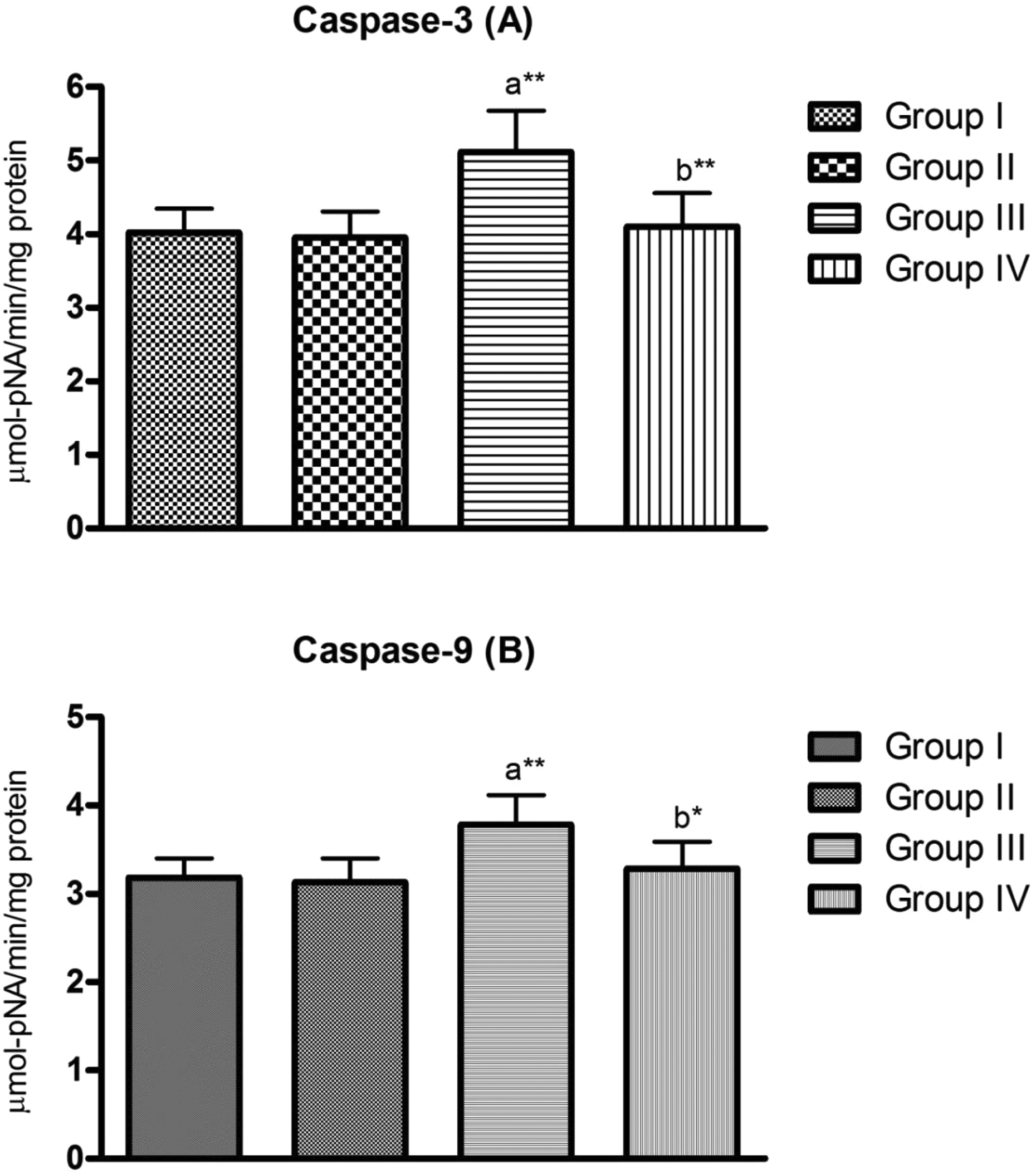
Testicular caspase-3 and -9 activities in gentamicin-treated rats in the absence (gentamicin) and presence of lycopene (gentamicin + lycopene). Group I: control, group II: lycopene, group III: gentamicin, group IV: lycopene + gentamicin. Data are expressed as mean ± SD (
Oxidative and antioxidant status
Markers of oxidative stress and antioxidant status in testicular tissue are presented in Table 2. H2O2 level and LPO were significantly (
Effect of gentamicin and lycopene on oxidative and antioxidant status.a

H2O2 production: hydrogen peroxide, nmol of H2O2 generated/min/mg protein; LPO: lipid peroxidation, µmol of MDA equivalent formed/min/mg protein; SOD: superoxide dismutase, nmol pyrogallol oxidized/min/mg protein; CAT: catalase, µmol of hydrogen peroxide consumed/min/mg protein; GPx: glutathione peroxidase, nmol of NADPH oxidized/min/mg protein; GR: glutathione reductase, nmol of NADPH oxidized/min/mg protein; GSH: total reduced glutathione, µg/mg protein; SD: standard deviation; ANOVA: analysis of variance.
aData are expressed as mean ± SD (
Gentamicin treatment (group III) significantly decreased (
Histopathological examination
Normal testicular architecture with an orderly arrangement of germinal cells and Sertoli cells was observed in the testes from control group (group I, Figure 4(a)). Animals treated with lycopene (group II, Figure 4(b)) showed normal testicular histology and normal spermatogenesis. Gentamicin treatment at a dose of 100 mg/kg (group III, Figure 4(c)) resulted in atrophy, degeneration, and loss of spermatogenesis in some of the seminiferous tubules. However, the findings in the lycopene-pretreated rats showed normal histological structure in the seminiferous tubules and the intestinal tissues with complete spermatogenesis series in the lumen(group IV, Figure 4(d)).

Representative illustrations of histological morphology of rat testes. Testes from (a) control and (b) lycopene groups of rats exhibit normal testicular histology and normal spermatogenesis (S). Testes of rats treated with gentamicin 100 mg/kg/day (c) display atrophy, degeneration, and loss of spermatogenesis (T). There was improvement in histopathological morphology of testes of animals pretreated with lycopene before gentamicin (d) treatment (I). The findings in the lycopene-pretreated rats showed normal histological structure in the seminiferous tubules and the intestinal tissues with complete spermatogenesis series in the lumen. Cross sections of testes were stained with H&E (×40). H&E: hematoxylin and eosin.
Discussion
The current study was conducted to investigate the ameliorative effect of lycopene against gentamicin-induced testicular toxicity in adult rat testes. Treatment of rats with gentamicin for 10 days had no effect on body weight but the absolute and relative testes weight decreased. The absence of gentamicin effect on body weight may be explained by the absence of general toxicity. 37 The weight of the testes is basically dependent on the mass of the differentiated spermatogenic cells and spermatozoa. 38,39 The decrease in the absolute weight of testes in this study may be due to low serum testosterone level, inhibition of spermatogenesis, and decreased sperm production, as reported by Aly and Khafagy. 40 Lycopene pretreatment protected these animals from the decrease in the testes weight. The decrease in sperm count is correlated with decrease in testosterone level and increase in ROS as evident from suppressed antioxidant enzyme activities which coincide with that obtained by Aitken and Sawyer. 41 The decrease in sperm count in gentamicin-treated rats may be due to the lower concentration of testosterone as the sperm production in the testis and maturation in epididymis is under the control of testosterone. 42 Motility and viability were closely correlated. Typically, viable (based on staining pattern) sperm were motile while nonviable sperm were immotile. Further, the decreased sperm motility and viability may be due to free-radical attack, which results in disturbed sperm motility. 43 Impaired sperm motility may result in infertility due to the failure of sperm to reach the site of fertilization as well as their ability to penetrate zona pellucida. 44 In addition to decrease in sperm count and motility, the effect of gentamicin on sperm parameters was further demonstrated by decrease in DSP manifested as decrease in step 19 spermatids/stage VII. 45 The reduction in DSP of the rats treated with gentamicin was consistent with the reduction in concentration of serum testosterone. These alterations were effectively prevented by lycopene pretreatment.
Testosterone is important for normal sperm production. In the absence of testosterone, germ cells are unable to progress and result in failure of spermatogenesis and infertility. 46,47 The decreased serum testosterone in the gentamicin-treated rats may be due to Leydig cell impairment caused by ROS generation. 48 Pretreatment with lycopene could normalize serum testosterone level by reducing ROS generation.
The present findings also show decreased activities of LDH-X and G6PDH in gentamicin-treated rats, which are indicative of testicular toxicity. LDH-X enzyme is a marker for normal spermatozoa metabolism. 49 This could be one of the contributory factors leading to reduced sperm concentration and sperm motility in gentamicin-treated animals. 50 G6PDH provides reducing equivalents for the hydroxylation of steroids. 51 G6PDH is also directly associated with GSH metabolism and lowered activity of this enzyme provokes increased oxidative stress to the tissue and may lead to cell death. 52 Lycopene could, however, effectively suppress the gentamicin-induced alterations in the activities of these enzymes.
There is a wide agreement that male reproductive organs are particularly susceptible to the detrimental effects of ROS and LPO, which lead to impaired fertility. 53 There is evidence that ROS plays a role in the induction of both death receptor and mitochondrial apoptotic pathways. 54 Particularly, spermatozoa are highly susceptible to ROS damage due to the high content of polyunsaturated fatty acids in their plasma membrane. 55 Literature reveals as a consequence of ROS generation in cells, mitochondrial dysfunction should occur. 56
Apoptosis is an active process by which cells commit suicide. 57 Caspases are a group of cysteine proteases whose activation cascade plays an important role in following induction of apoptosis. 58 The increase in the activity of caspase 9 initiates an irreversible path to cell death via cleavage and activation of the downstream effector, caspase-3. 57,59 Caspase-3, which is the main final executor of apoptosis, is responsible for the cleavage of key cellular proteins, leading to apoptosis. 60 The increased activities of caspase-9 and caspase-3 attest that caspase cascades are involved in gentamicin-induced spermatogenic cell apoptosis and that the mitochondrial pathway plays an important role in gentamicin-induced reproductive system injuries in male rats. The results of this study have been shown to coincide with the increase in the level of H2O2. 61,62 Increased generation of ROS could not only be a cause but also the consequence of mitochondrial dysfunction. 63 Lycopene pretreatment, however, effectively suppressed the activation of caspase-3 and -9.
In the present study, administration of gentamicin increased the levels of H2O2 and LPO and concomitantly decreased the activities of SOD, CAT, GPx, and GR as well as GSH level in the rat testis. SOD is considered the first line of defense against deleterious effects of oxyradicals in the cell by catalyzing the dismutation of superoxide radicals to H2O2 and molecular oxygen. The reduction in the activity of CAT may reflect an inability to eliminate H2O2 produced by gentamicin due to enzyme inactivation caused by excess ROS production in the rat testis. 64 The reduced activity of GPx can be explained on the basis of the observed lack of substrate GSH. The antioxidant enzymes CAT and GPx protect SOD against inactivation by H2O2. Reciprocally, the SOD protects CAT and GPx against inhibition by superoxide anion. Treatment with gentamicin also reduced GR activity. GR is involved in the supplementation of GSH to spermatogenic cells. 65 The reduction in the activities of antioxidant enzymes and the increase in H2O2 and LPO could reflect an adverse effect of gentamicin on the antioxidant system in the testis. 66 ROS cause damage to sperm and other cytoplasmic organelle membrane structures through peroxidation of phospholipids, proteins, and nucleotides, thereby altering sperm motility. 67 In addition to their direct effect on cellular constituents, ROS produce oxidative stress by decreasing enzymatic defenses. 68
GSH plays an important role in the cellular antioxidant defenses and most importantly to remove H2O2 and organic peroxides. 69 It can act as a nonenzymatic antioxidant by direct interaction of SH group with ROS, or it can be involved in the enzymatic detoxification reaction for ROS, as a cofactor or coenzyme. 70 Pretreatment with lycopene protected the rat testis against LPO induced by gentamicin by increasing antioxidant enzymes and decreasing ROS generation.
The significant decline in testes weights and sperm parameters was accompanied by histopathological changes in testis as displayed by alterations of spermatogenesis with a complete loss of all the stages of germ cell maturation, atrophy, and degenerative aspects of seminiferous tubules. Pretreatment of rats with lycopene resulted in normal histological structure in the seminiferous tubules and the intestinal tissues with complete spermatogenesis series in the lumen.
Lycopene is the most potent free-radical scavenger among the carotenoids. 71,72 It has high lipid solubility and so can pass through the blood–testis barrier, 73 suggesting it may be a potentially useful treatment for testicular toxicity associated with oxidative stress. The protective effects of lycopene were correlated with a decrease in ROS production and subsequent inhibition of the mitochondrial apoptotic pathway.
In conclusion, gentamicin decreased rat testes weight, inhibited spermatogenesis, decreased testosterone level, and induced apoptosis accompanied by abnormal histologic findings. It induced oxidative stress and apoptosis by possible mechanism of mitochondrial dysfunction. The histopathological studies confirmed the cytoprotection rendered by lycopene. These data provide insight into the ameliorative effect of lycopene against gentamicin-induced testicular toxicity and its beneficial role to counteract oxidative stress and apoptosis in rat testis and to restore the suppressed spermatogenesis.
Footnotes
Acknowledgment
The authors gratefully acknowledge the technical and financial support by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, Saudi Arabia, under Grant Number D-051-166-1439.
