Abstract
The unfolded protein response (UPR) is one of a switch of autophagy and apoptosis, and the endoplasmic reticulum stress (ERS) which inducing UPR plays a role in the malformations caused by some genetic and environmental factors. Exposure to flutamide during pregnancy will also cause abnormalities in some male offspring reproductive organs such as cryptorchidism. In this study, after administered the pregnant mouse orally at a dose of 300 mg/kg body weight every day during gestational day (GD)12 to GD18, flutamide can not only caused hypospadias in the male mouse offspring but also damaged the morphology and function of their testis. And the expression of UPR-related genes and proteins, autophagy, apoptosis, and angiogenesis-related genes of the damaged/teratogenic testis and penis in the mice were investigated to determine the role of UPR in this model. It was found that flutamide activated maybe the Atg7-Atg3-Lc3 pathway through the UPR pathway, caused cells excessive autophagy and apoptosis, and inhibited the formation of penile and testicular blood vessels by activating UPR and affecting the messenger RNA level of vascular endothelial growth factor and hypoxia-inducible factor 1.
Introduction
Hypospadias is a congenital dysplasia in which the male urethra does not form a closed luminal structure, and ectopic opening in the ventral side of the penis often occurs with testicular abnormalities. 1 Flutamide, a nonsteroidal antiandrogen, is a water-insoluble environmental endocrine disruptors and commonly used in the field of reproductive toxicology to establish male animal reproductive organ damage/malformation models. 2 Some scholars established the rat hypospadias model by exposing flutamide during their mother’s pregnancy, and exposure to flutamide during pregnancy will also cause abnormalities in the other male reproductive organs such as cryptorchidism. 3
It is found that some molecular mechanisms of teratogenicity of xenobiotics, and some conservative signaling pathways, such as endoplasmic reticulum (ER) stress (ERS), induced unfolded protein responses (UPR) plays a role in the diseases caused by some genetic and environmental factors. 4 Some harmful factors such as toxic xenobiotics, hunger, metabolic disorders, and intracellular viral infection will lead to ERS. 5,6 In the case of ERS, the ER environment was destroyed, the misfolded proteins were accumulated, and the UPR occurred. 7
It is known that the UPR is composed of upstream specific stress sensors and downstream transcription factors that recode gene expression, and activating transcription factor 6 (ATF6), protein kinase (PKR)-like ER kinase, and inositol-requiring enzyme 1 (IRE1) are the main sensors, while glucose-regulated protein 78 (GRP78) binds the three ER protein cavity domains and anchors them on the membrane. 8,9 It was known that flutamide would induce oxidative stress (OS) in rat hepatocytes in vitro and lead to damage of them 10 ; the OS and ERS interaction can act as mediators that influence UPR signaling activity. 11
The UPR pathway is one of a switch of autophagy and apoptosis, and the activation and progression of autophagy require multiple steps to reach, involving many of the expression and regulation autophagy-related genes (ATG). 12 The appropriate autophagy can not only alleviate the damage caused by ERS to some extent but also promote cell survival; while excessive autophagy may initiate cell apoptosis. In the recent years, the role of autophagy and cell death induced by ERS in teratogenesis has attracted more and more attention. 13,14
In this article, we would developed a testicular and penile damage/teratogenic model of mouse administered with flutamide and then measured the expression of UPR key members Grp78, Perk, Atf6, Ire1, eukaryotic translation initiation factor 2 subunit alpha (eIF2α), activating transcription factor 4 (Atf4), X box-binding protein 1 (Xbp1), autophagy related genes (Atgs, unc-51-like kinase 1 (
Materials and methods
Animals and model construction
Institute of Cancer Research (ICR) mice aged about 8 weeks were purchased from Hunan SJA Laboratory Animal Co. Ltd (Certificate No.: SCXK Hunan 2016-0002, Hunan, China) and quarantined for 1 week on arrival at the institutional animal care facility prior to initiation of studies. All mice (including males for mating) were kept in an animal room maintained at constant temperature (21 ± 2°C), relative humidity of 55 ± 10%, and 12-h light/dark cycle (06:00–18:00). A week later, one male weighing 32–35 g mated with two virgin female weighing 29–32 g overnight, and pregnancy was confirmed by the presence of vaginal plugs the following morning as gestational day 0 (GD0). The female mice were allowed to cycle and ovulate naturally, and all mice had free access to food and distilled water. Use of the animals was approved by the Committee on Ethics of Biomedicine Research, Hunan Normal University in Changsha, China.
Forty pregnant mice were obtained and equally randomized to a flutamide group (
Organ measurement and sample collection for genes and proteins detection
Eight weeks later of birth, 91 offspring male mice were obtained in the treatment group and 99 in the control group. After being cervical dislocation, in each group the testis and penis of 12 male mice were taken out to freeze and store in the Trizol (Invitrogen, California, USA) and 6 in the radio-immunoprecipitation assay (RIPA) lysis buffer (Beyotime Biotechnology, Shanghai, China) immediately. And 40 male mice in each group were randomly selected for observing their overall appearance, measuring their body mass, and checking their urethral development. After they were sacrificed, some of them were photographed, and all their anal to genital distance (AGD), testicular volume, and penis length were measured with a vernier caliper.
Histopathological slice preparation
Twelve male pups were randomly selected from each group, and their penises and testes were took and fixed in formalin solution, dehydrated the tissue, embedded in paraffin and sectioned, stained by hematoxylin–eosin staining, and observed the pathological changes of the tissues under the light microscope (Olympus, Tokyo, Japan).
Total RNA extraction and real-time fluorescent quantitative PCR
Total RNA was extracted according to the protocol of the kit and dissolved in nuclease-free water, from which 1 µg total RNA was drawn and reverse transcribed to synthesize cDNA according to the reverse transcription kit (Promega, Wisconsin, USA).
The cDNA obtained was amplified with reverse transcription polymerase chain reaction (RT-PCR) on Applied Biosystems Inc. (ABI) ViiA 7 (Life Technology, USA) using ABI SYBR buffer (Life Technology, California, USA). Primers were designed with ABI Primer Express 3.0 according to ABI Primer Design Guidelines. All primers used in the present study were synthesized by Shanghai Invitrogen (China) using the sequences as listed in Table 1. The RT-PCR reaction system used is as follows: SYBR buffer 2.5 μl, cDNA 0.5 μl, upstream primer 0.3 μl, downstream primer 0.3 μl, Mili Q water 6.4 μl, under the reaction conditions: 95°C for 15 min, 40 cycles consisted of 95°C for 5 s, and 60°C for 1 min. 16,17
Primers for QRT-PCR.

Edem: ER degradation enhancing α mannosidase like protein.
QRT-PCR: quantitative reverse transcription polymerase chain reaction; Grp78: glucose-regulated protein 78; Perk: protein kinase (PKR)-like ER kinase; eIF2α: eukaryotic translation initiation factor 2 subunit alpha; Atf4 and 6: activating transcription factor 4 and 6; Ire1: inositol-requiring enzyme 1; Xpb1: X box-binding protein 1; Bcl-xl: B-cell lymphoma-extra-large; Ask1: apoptosis signal-regulating kinase 1; Jnk1, 2, 3: c-Jun N-terminal kinase 1, 2, 3; Caspase 3, 9, 12: cysteinyl aspartate-specific proteinase 3, 9, 12; Chop: C/EBP-homologous protein; Ulk1: unc-51-like kinase 1; Lc3b: light chain 3b; Atg3, 7: autophagy-related genes 3, 7; Hif-1α: hypoxia-inducible factor 1; Vegf: vascular endothelial growth factor.
The relative mRNA expression levels were analyzed using the 2–ΔΔCt method, and the
Western blotting
The penises and testes harvested from the control and flutamide groups were homogenized and centrifuged at 10,000 g for 10 min at 4°C to clear the protein lysate. Then the protein concentrations were quantitated using a modified bicinchoninic acid protocol, and the detection kit was purchased from Beyotime Biotechnology, Shanghai, China. Total protein was boiled for 5 min in sodium dodecyl sulfate loading buffer containing dithiothreitol and then loaded about 25 µg of total protein per lane in an SDS-polyacrylamide gel electrophoresis gel. Following electrophoresis, the separated proteins were blotted onto a polyvinylidene difluoride membrane, which was blocked overnight with 5% skimmed milk powder in Tris-buffered saline buffer with tween 20 (TBST) at 4°C. Blots were incubated overnight at 4°C with
Statistical analysis
Data were expressed as
Results
Hypospadias, changes of genital phenotype, and histology of offspring males induced by flutamide during their maternal pregnancy
All the male pups which randomly selected for anatomy and measurement of reproductive organs had no changes of the urethra, genital phenotype, and histology in the control group, while those in the flutamide group were with hypospadias, and the incidence rate was 100% (Figure 1(a)). The body mass of the control group (39.67 ± 3.23 g) was more than that of the flutamide (37.21 ± 2.89 g;

Morphologic comparison of the mouse overall, penis and testis between the control group (L) and the Fluentamide group (R). (a): The male mice of the Fluentamide group was with “vaginal opening” in the external genitalia (R, arrow pointed), and their AGD was 11.03 ± 1.75 mm; while those of the control group without that, and the AGD was 17.68 ± 1.43 mm, which was significantly greater than that of the flutamide group (
Histopathological sections showed that the urethra of the flutamide group did not form a closed structure, which was connected to the skin, and there were loose sponges with disordered layers and unevenly distributed blood vessels, which full of many red blood cells in the lumen (Figure 2(b)). In the testicular section, the sperm cells were less than the control group, many of the spermatogenic cells were dissociated and autolyzed, and some of the Sertoli cells felled off (Figure 2(d)).

Comparison of the penis and the testis histological morphology between the control group and the flutamide group. (a) and (b): In the control group, the penile cross-section of the male offspring mice was complete, while there was an obvious fissure in the flutamide group (pointed by the arrow in Figure 2(b)). (c) and (d): In the control group, the spermatogenic cells, sertoli cells, interstitial cells, and interstitial cells were arranged neatly in the testes of the male offspring. A large number of spermatogenic cells in the flutamide group fell off the basement membrane, and most of the cells were arranged disorderly.
Effect of flutamide on UPR pathway and other related genes in offspring male mice
The relative mRNA abundance of

(a). The relative mRNA abundance of UPR key genes in the penises of the control group and the flutamide group (
The expression abundance of UPR key proteins Perk, p-Elf2α, Atf6, Grp78, and Ire1 in the penis and testis of the flutamide group was higher than that of the control group (
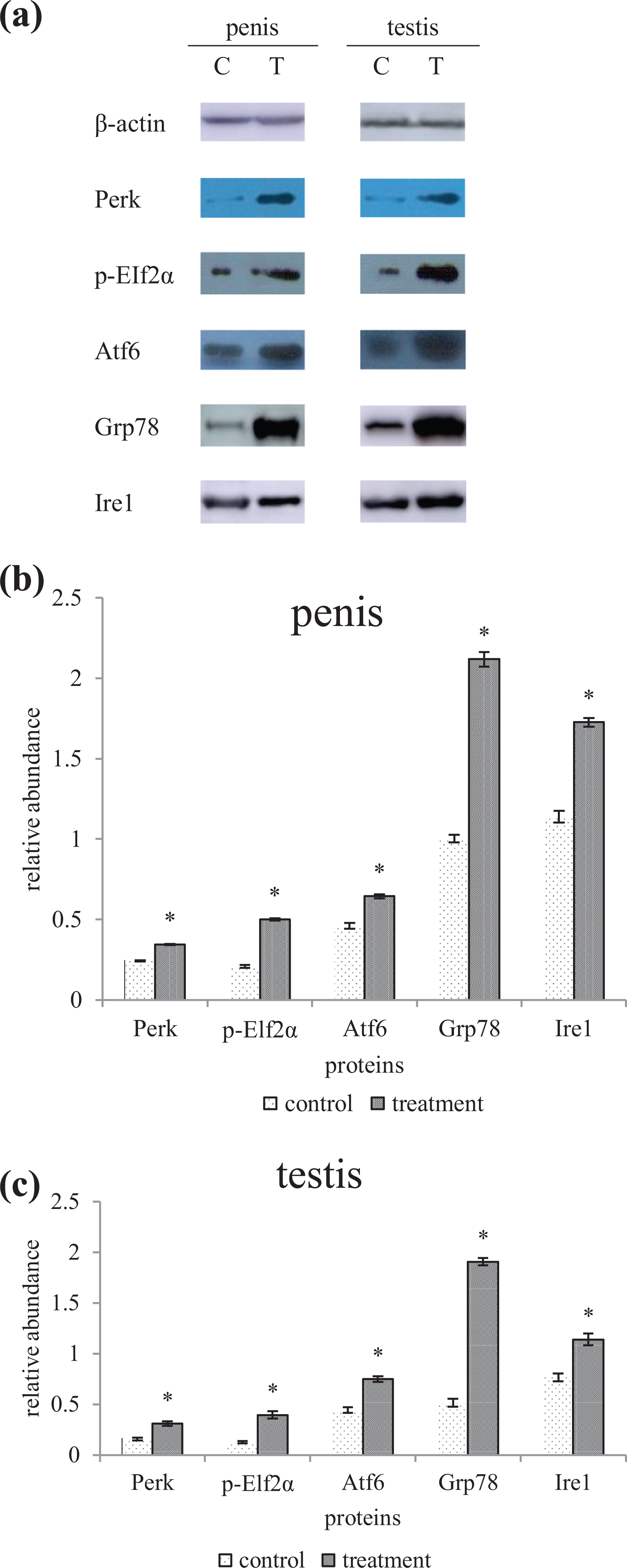
(a). Influence of flutamide on Perk, p-EIf2α, Atf6, Grp78, and Ire1 of the offspring male penises and testes 8 weeks after birth. All the expression level of five proteins in the flutamide group was higher than that of the control. (b) Expression of Perk, p-EIf2α, Atf6, Grp78, and Ire1 of the offspring male penises 8 weeks after birth. (
The relative mRNA abundance of

(a). The relative mRNA abundance of ATGs in the penises of the control group and the flutamide group (
The relative mRNA abundance of

(a). The relative mRNA abundance of apoptosis-related genes in the penises of the control group and the flutamide group (
The relative mRNA abundance of

(a). The relative mRNA abundance of angiogenic genes in the penises of the control group and the flutamide group (
Discussion
It had found that flutamide would induce hypospadias of rats by subcutaneous injection or by oral, 18 –20 but the flutamide dose and incidence of hypospadias are not consistent. 21 In this study, we found after administered the pregnant mouse orally at a dose of 300 mg/kg body weight every day during GD12–GD18, flutamide can not only cause hypospadias in the male mouse offspring but also damage the morphology and function of their testis.
Persistent OS and protein misfolding trigger the apoptotic cascade and play a major role in the pathogenesis of a variety of human diseases. 11 Misfolded proteins induce reactive oxygen species, and OS interferes with the ER redox state, which disrupts proper disulfide bond formation and protein folding. 22 It was reported some birth defects were related to UPR regulation, for example, it was found that exposure of mouse embryos to short-term gestational hypoxia could induce the most common types of heart defects, which was due to the fact that UPR mediated a significant reduction in fibroblast growth factor (FGF) signaling in cardiac progenitor cells in the second heart region. 23
ERS is important in some environmental endocrine disruptors, such as bisphenol A (BPA) and diethylstilbestrol, induced damage in rat testes.
24
The expression level of key UPR genes and proteins in the penile and testis of the flutamide group was higher than those in the control group; we speculated that the three UPR branches of Perk-eIF2α-Atf4, Atf6-Grp78-Xbp1, and Ire1-Xbp1 may be activated. In the penile and testis of the flutamide group, the
Perk activation and Elf2α phosphorylation can selectively upregulate autophagy genes such as Atg7 and Lc3, thereby activating autophagy,
27
and excessive autophagy was known to cause apoptosis.
28
The expression of
It is reported that exposure to 4-nonylphenol dose-dependently induces the formation of autophagosomes in Sertoli cells and increases the expression of the mRNA expression of Atg3, Atg5, Atg7, and Atg12 in testis,
30
and Atg7 could be selectively upregulated by PERK and its downstream substrate eIF2a in the UPR pathway.
31
In this study,
It is known that antiandrogen such as flutamide may have the ability to inhibit HIF-1,
32
the transcriptional activity of HIF-1 was decreased, and secretion of VEGF was increased by BPA and benzyl butyl phthalate under hypoxia in Michigan Cancer Foundation (MCF)-7 cells.
33
In addition, accumulating evidence suggested that UPR played a vital role in regulating angiogenesis, in part by coordinating the secretion of VEGF.
34
In this study, we found in the treatment group, fewer blood vessels on testicular surface than in control group and unevenly distributed blood vessels in the penis, and
In summary, the male offspring mice exposed to flutamide at a dose of 300 mg/kg · d during GD12–GD18 could cause penile and testicular deformity/damage. The reason may be that flutamide activated the Atg7-Atg3-Lc3 pathway through the UPR pathway, caused cells excessive autophagy and apoptosis, and inhibited the formation of penile and testicular blood vessels by activating UPR and affecting
Supplemental Material
Supplemental Material, Supplementary_material_(6) - Role of unfolded protein response in genital malformation/damage of male mice induced by flutamide
Supplemental Material, Supplementary_material_(6) for Role of unfolded protein response in genital malformation/damage of male mice induced by flutamide by H Yu, K Wen, X Zhou, Y Zhang, Z Yan, H Fu, J Zhu and Y Zhu in Human & Experimental Toxicology
Footnotes
Author’s contribution
Haiming Yu and Kexin Wen have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Project of Hunan Provincial Department of Education [17A131].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
