Abstract
A broad definition of preconditioning is “the preparation for a subsequent action.” Mounting evidence demonstrates that novel remote preconditioning paradigms, in which protective stimuli experienced locally can capacitate systemic tolerance and enhanced cell viability upon exposure to ensuing cellular insults, have been largely successful in the field of cardiovascular ischemia/reperfusion injury. To ensure successful protective preconditioning, some models (including the uterus) have been demonstrated to activate the unfolded protein response (UPR), which is a cellular stress response controlled at the level of the endoplasmic reticulum. However, in the context of remote preconditioning, activation of these intracellular molecular pathways must result in the extracellular transmission of adaptive signals to remote targets. In our recently published manuscript, we have described the activation of the UPR in the pregnant uterine myocyte to be associated with increased uterine myocyte quiescence and normal gestational length. We hypothesize that ubiquitous uterine gestational stresses experienced in every pregnancy, which have been demonstrated in other systems to activate the UPR, may induce a robust paracrine dissemination of a uterine secretome, for example, glucose-regulated protein 78, with preconditioning-like properties. Furthermore, we speculate that the gestational stress-induced uterine secretome acts to promote both local and systemic tolerance to the ensuing gestational insults, allowing for the maintenance of uterine quiescence. In this context, preterm labor may be the result of a pregnant uterus experiencing a stress it cannot accommodate or when it is unable to host an appropriate UPR resulting in insufficient preconditioning and a diminished local and systemic capacity to tolerate pregnancy-dependent increases in normal gestational stress. This is highly attractive from a clinical viewpoint as we ultimately aim to identify local and systemic adaptations that may serve as preconditioning stimuli for use as a strategy to restore appropriate preconditioning profiles to prolong uterine quiescence in pregnancy.
Introduction
The length of a term human pregnancy is between 37 and 42 weeks of gestation. Preterm birth, defined as parturition prior to 37 weeks of gestation, is the number one cause of mortality in children younger than 5 years of age. 1 Each year, approximately 15 million babies are born prematurely worldwide and this number continues to rise. 2,3 Unfortunately, the mechanisms responsible for facilitating uterine quiescence, regulation of gestation length, and the triggers that lead to the onset of term and preterm labor largely remain unresolved. Hence, to date the development of tocolytic intervention therapies for the prevention of premature labor have been unsuccessful. Administration of exogenous progesterone, however, has been found to be successful in delaying the onset of premature labor in a subset of women previously identified as at high risk for preterm birth. However, women in their first pregnancy, those unresponsive to exogenous progesterone treatment, and those not identified as high risk continue to undergo preterm birth due to unknown etiologies. 4,5 Recently, our laboratory has established that unfolded protein response (UPR)-dependent activation of nonapoptotic caspase-3 during pregnancy, specifically within the uterine smooth muscle cell, plays a critical role in the maintenance of uterine quiescence across gestation. 6 Caspase-3 activation is normally associated with apoptotic cell death; however, in the context of the pregnant uterus, it remains non-apoptotic and preserves uterine quiescence by targeting and disabling the contractile architecture of the myometrium, for example, connexin 43, α-actin, and γ-actin. 6,7 Subsequently, we speculate that the gestationally induced UPR, seen to activate local non-apoptotic caspase-3, may also promote the paracrine dissemination of a uterine secretome. Similarly, a variety of studies have demonstrated the ability of cellular stress, including endoplasmic reticulum (ER) stress, oxidative stress, hypoxia, and inflammation, to augment UPR-dependent cellular secretions. 8,9 Interestingly, in the field of ischemia/reperfusion injury, the purposeful induction of cellular stress in one tissue provided protective effects in a secondary remote tissue upon exposure to a subsequent cellular stress. 10 Therefore, we propose that applying a similar remote preconditioning paradigm, targeting the hormetic response to gestationally regulated myometrial stressors, will ensure an appropriate uterine UPR and promote tolerance to gestational stress, ultimately facilitating uterine quiescence and an appropriate gestational length. Accordingly, identifying preconditioning-like strategies that evoke these protective effects against subsequent uterotonic stresses will be key in devising novel tocolytic interventions for the prevention of preterm birth.
Preconditioning
Preconditioning refers to the phenomenon where sublethal insults can paradoxically induce tolerance to subsequent stressors. The process of preconditioning biological systems against pathophysiological events can be observed in multiple forms. Ischemia/reperfusion preconditioning in cardiovascular tissue has become an increasingly active area of research over the last 3 decades. In the event of acute ischemia due to a myocardial infarction, persistent hypoxia induces both cellular necrosis and apoptosis in the area of infarct, and apoptosis in the bordering tissue zone. 11 ∓13 Murry and colleagues demonstrated the application of brief ischemic events prior to a prolonged myocardial infarction reduced the infarct size by approximately 25% compared to control animals that did not receive ischemic preconditioning. 14 Subsequent studies validating Murry’s initial findings have further linked cardioprotective effects to mitochondrial stabilization via G-protein–coupled receptor activation and canonical downstream events, release of circulating humoral factors, and neurogenic activation of protein kinase C. 15 ∓20 Preconditioning paradigms utilizing similar principles have been demonstrated to limit damage from other lethal hypoxic events, such as stroke or liver failure. 21,22 Recent studies have also demonstrated the act of ischemia conditioning in an isolated tissue can capacitate further cytoprotection in a secondary remote tissue. 10,23 Intriguingly, this phenomenon of remote preconditioning does not appear to be isolated to the myocardium and has been demonstrated to show protective effects in additional parenchymal tissues including but not limited to the kidneys, lungs, and ovaries. 24 While ongoing studies continue to examine the molecular mechanisms responsible for ischemia/reperfusion preconditioning effects in the various tissue paradigms, it is clear that low-dose stress facilitates preinduction of cellular readiness, which has a paradoxical protective effect against further cellular damage. Based on these studies, we hypothesize that similar remote preconditioning-like paradigms occur across gestation in a normal pregnancy and that identifying these mechanisms/events may serve as a tool for reducing the risk of preterm birth.
Preterm Birth
In addition to fetal death, secondary effects of preterm birth which primarily result from immature organ development include increased risk of neurologic and developmental disabilities, such as cerebral palsy, hearing, and vision impairments, as well as respiratory complications. 25 ∓28 Recent studies have also demonstrated direct correlations between preterm birth (PTB) and latent diseases such as asthma, insulin resistance, and hypertension. 29 ∓31 The major discernable risk factors for preterm labor include low socioeconomic status, advanced age, tobacco use, high stress, and race. 32 ∓36 Independently, women who have reduced cervical length, multiple fetuses, infection, or previously undergone spontaneous preterm birth are also at an increased risk of early onset of labor. 37 Currently, the prophylactic treatment of intramuscular or vaginal progesterone to women with a high risk of preterm birth has proven effective in reducing but not eliminating the risk of preterm delivery. 4,5,38 In a multicenter, randomized, double-blind, placebo-controlled trial performed by Hassan and colleagues, midtrimester vaginal progesterone treatment to women, with a sonographic short cervix, reduced preterm delivery prior to 33-week gestations by 45%. 4 Though progesterone intervention is promising for women with established high risk of preterm birth, tocolytic (anticontractile) agents are the only treatments available to delay birth in women who present with preterm labor symptoms. Nifedipine, a calcium channel blocker, and Indomethacin, a cyclooxygenase-2 inhibitor, however, are only effective in impeding the immediate processes of active labor for 24 to 48 hours. 39 ∓41 While this does provide a short time period in which antenatal corticosteroids and magnesium sulfate can be administered to promote fetal lung maturation and provide neuroprotection, respectively, this is not a long-term solution for the prevention of preterm birth. 42,43 Consequently, we propose understanding how the uterine UPR mediates both local and systemic adaptations to gestationally induced stress will aid in the development of effective prophylactic tocolytic therapies.
Unfolded Protein Response in Human Disease
In all mammalian cells, including the uterine myocyte, the UPR originates from the intracellular compartment termed ER. The ER is a perinuclear organelle composed of 2 distinct subdomains, the smooth and rough ER. Smooth ER functions include lipid synthesis, drug metabolism, and particularly the management of intracellular calcium stores, which is important for multiple physiological functions (eg, intracellular signaling and muscle contraction). 44 ∓47 In contrast, the primary function of the rough ER is to coordinate synthesis, folding, and packaging of secretory proteins to the Golgi apparatus. 48 With the onset of various proteotoxic cellular stressors, an accumulation of unfolded proteins in the ER leads to the intrinsic activation of the molecular cascade termed the UPR (Figure 1). Upon proteotoxic activation, the immediate goal of the UPR is to restore ER/cellular homeostasis and to avoid cell death. 49,50 It is clear, however, that the dysregulation of the UPR can inhibit critical cellular functions in response to stress (eg, Alzheimer, atherosclerosis, type 2 diabetes, Creutzfeldt-Jakob, and lysosomal storage diseases) or facilitate the adaptation to pathophysiological stressors, as seen in different models of cancer. 51 ∓55 Although numerous modalities of the tumor environment have been demonstrated to induce the UPR (eg, nutrient limitation and low pH), hypoxia is thought to be the primary activator. 49,56 Specifically, activation of UPR-associated proteins (Figure 1) has been identified in the presumptive ischemic regions of tumors and are induced in cultured cancer cells subjected to hypoxia in vitro. 54,55 Utilizing various knockdown techniques, it has been demonstrated that cancer cells or tumors deficient in glucose-regulated protein 78 (GRP78), X-box binding protein 1 (XBP1), or protein kinase RNA-like ER kinase (PERK) have increased sensitivity to apoptotic stimuli. 57 ∓61 In contrast, tumor-dependent increases in the secretion of GRP78 into the circulation of patients with cancer has been directly correlated to tumor resistance and decreased patient survival. 62 Furthermore, it has been demonstrated that activation and secretion of adaptive UPR signals renders the tumor insensitive to apoptotic stimuli, thereby permitting the tumor to survive in the face of chemotherapeutic agents. 58,63 In the pregnant uterus, we have identified a similar local UPR-mediated resistance to apoptotic cell death, despite elevated levels of caspase-3 activation, and further speculate that the dissemination of the UPR uterine secretome acts to increase systemic tolerance to ensuing gestational stress.
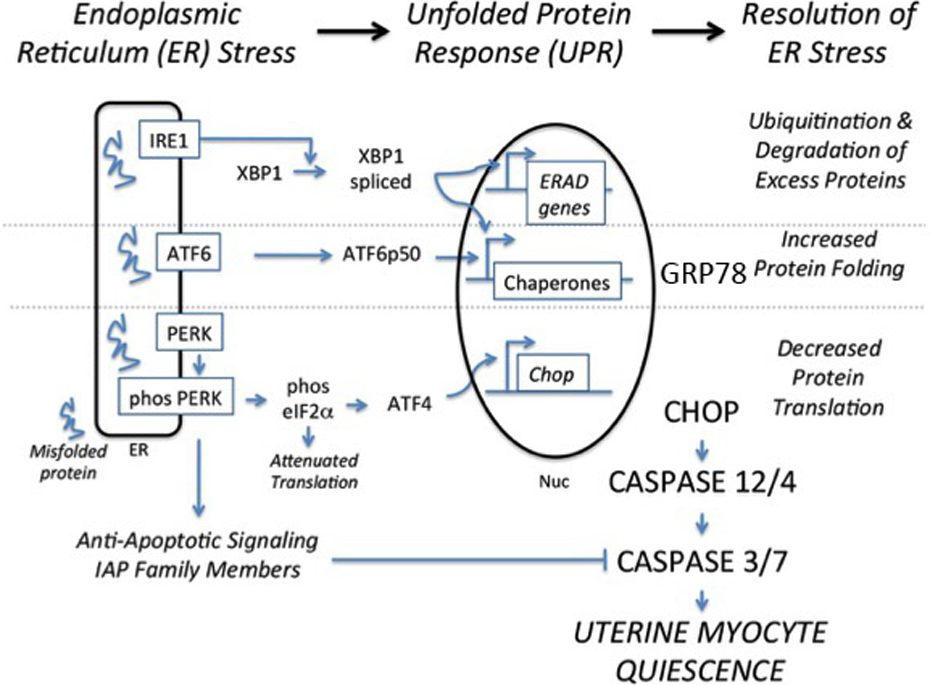
The ERSR-UPR signaling cascade. ATF6 indicates activating transcription factor 6; BiP, binding immunoglobulin protein; CHOP, CCAAT-enhancer-binding protein homologous protein; ERSR, endoplasmic reticulum stress responses; eIF2α, eukaryotic initiation factor 2 alpha; IRE1, serine/threonine-protein kinase/endoribonuclease inositol-requiring enzyme 1; PERK, protein kinase RNA-like endoplasmic reticulum kinase; UPR, unfolded protein response; XBP1, X-box binding protein 1. Adapted from Tsang et al. 49
Preconditioning the ER UPR
Although the UPR has the capacity to activate apoptotic processes, the initial signaling response via PERK, inositol-requiring enzyme 1 alpha (IRE1α), and activating transcription factor 6 (ATF6) functions to aid in the restoration of cellular homeostasis, promoting the avoidance of apoptotic consequence. Accordingly, UPR-activated genes increase chaperone protein expression, for example GRP78, attenuate protein translation, and increase ER-associated degradation of unfolded proteins. 64 ∓66 Both in vivo and in vitro studies have proven that direct and remote preconditioning with minor insults of ER stress stimulate a protective adaptive UPR, promoting tolerance, and antiapoptotic signaling, fostering resistance to various cellular insults. 67 ∓71 Utilizing loss of function mutant alleles in various UPR genes, Mao and Crowder demonstrated the necessity of functioning IRE1α, XBP1, and ATF6 for appropriate UPR-mediated preconditioning against lethal hypoxic insults. 72 It has also been demonstrated that conditioning renal epithelial cells with minor insults of chemically induced ER stress significantly reduced oxidative injury in response subsequent oxidative stress. 69 Along with providing cytoprotection against hypoxic and oxidative injury, UPR preconditioning has also been shown to diminish downstream ER stress-mediated inflammatory signaling. In the scope of pregnancy, reduced inflammation, as a downstream consequence of preconditioning, is highly significant as many events initiated by the inflammatory signaling molecule nuclear factor kappa B result in the establishment of uterine contractility. 73 One study in particular demonstrated the preactivation of adaptive UPR signaling pathways within the mouse eye blunted tumor necrosis factor alpha (TNFα)-mediated retinal endothelial inflammation and apoptosis. 74 Similarly, we speculate that gestational stresses experienced in every normal pregnancy act to directly precondition the pregnant uterine myocyte UPR, capacitating local tolerance to subsequent endogenous and exogenous uterotonic stresses, thereby promoting uterine quiescence in a nonapoptotic caspase-3–dependent manner. We also suspect, however, that these stressors may indirectly promote systemic pro-quiescent adaptations through remote secretome-mediated preconditioning. In the context of the kidney, remote preconditioning, via systemically administered pretreatments of tunicamycin or thapsigargin, ameliorated the consequences of a chemically induced form of glomerulonephritis in rats. 75 In addition, remote systemic ER stress preconditioning has been shown to suppress translation of UPR apoptotic effector proteins ATF4 and CCAAT-enhancer binding homologous protein, inhibiting tunicamycin (Tm)-mediated apoptosis in splenic macrophages, renal tubule cells, and hepatocytes, preventing hepatosteatosis and renal dysfunction. 76 Based on these data, it is important to note for future studies that the UPR is a dynamic signaling network capable of exhibiting cell type- and stimulus-specific responses. Subsequently, a wide range of gestational stressors stemming from the uterine myocyte may promote remote preconditioning in systemic tissues such as the vasculature or immune system. Although there is no direct evidence for this, there are a multitude of studies dissecting the effects of pregnancy on hemodynamics, cardiac function, and postpregnancy cardiovascular outcomes. For example, during pregnancy, there is a significant decrease in mean arterial pressure, which is typically attributed to a reduction in peripheral vascular resistance, mediated by both systemic and uterine vascular remodeling. 77,78 Based on the evidence of extracellular GRP78 modulating inflammation and biochemical stressors, it is easy to postulate that the accommodation of systemic pregnancy-dependent hemodynamic changes such as these may, in part, be due to uterine UPR remote conditioning. Additional changes seen in vascular function include an approximate 40% increase in blood volume and subsequent increases in total cardiac output. 78 Interestingly, there is evidence to suggest these adaptations in maternal blood volume are regulated by fetal/placental endocrine feedback systems. 79 In addition to normal physiological cardiac adaptations during pregnancy, studies have also found pregnancy-dependent attenuation of pathophysiological cardiac function, which may be in part the result of remote UPR preconditioning experienced explicitly during pregnancy. 80 Specifically, in a mouse model of left ventricular (LV) pressure overload, pregnancy was found to mitigate pathological LV remodeling, pulmonary congestion, and transverse aortic constriction-dependent gene expression. 80 Finally, it has been suggested that the hemodynamic changes experienced during pregnancy may reduce a women’s risk of cardiovascular disease later in life. If proven to be true, this phenomenon perfectly corroborates the idea that pregnancy-dependent uterine UPR positively influences maternal systemic vascular function (Figure 2).

UPR preconditioning maintains uterine myocyte quiescence. Gestational stresses precondition the uterine myocyte UPR throughout gestation to maintain the tocolytic potential of caspase-3/7 activation while avoiding its apoptotic consequences. UPR indicates unfolded protein response.
Extracellular GRP78: Candidate Molecule for Remote UPR Preconditioning During Pregnancy
Trafficking of GRP78 to Non-ER Sites
To propagate systemic preconditioning events, we propose local uterine myocyte UPR stressors stimulate the transmission of a uterine secretome throughout the course of gestation. Components of the UPR, such as GRP78, have been found to not only be expressed in the ER but also on the extracellular surface of many cell types and in the extracellular space. 81,82 GRP78 has also been found in the serum, synovial fluid, saliva, and oviductal fluid. 82 –85 The mechanism whereby components of the UPR signaling cascade traffic from the ER to the cell surface and the extracellular space is not fully resolved. It has been confirmed, however, that extracellular GRP78 does not arise from apoptotic cell death-mediated protein leakage during ER stress, as GRP78 release into the extracellular space precedes any evidence of apoptotic cell death. 86 Moreover, in cells exposed to Brefeldin A, which inhibits ER to Golgi protein transport, extracellular levels of GRP78 were significantly decreased while they intracellularly continued to increase, suggesting that extracellular GRP78 is actively trafficked. 86,87 More recently, the extracellular trafficking of UPR proteins was confirmed by the finding that protease-activated receptor 4 was identified as a partner for GRP78 that was necessary for its translocation to the cell surface during periods of ER stress in normal and cancer cells, underscoring the regulated nature of GRP78 translocation to the cell surface. 88 However, the GRP78 amino acid sequence contains the classical ER retention signal (KDEL) in its C-terminal. The KDEL sequence should dictate that GRP78 is a lumen bound, ER resident protein, which cannot traffic to the surface of cell or be secreted extracellularly. It is thought that an oversaturation of the specific KDEL receptors in the ER during periods of ER stress may allow for GRP78 to escape the KDEL retention system and accumulate in the plasma membrane and the extracellular space. 87,86,89,90 The severity of ER stress needed to facilitate GRP78 secretion into the extracellular space, however, remains ambiguous, and further studies are required to characterize cell-type and stimulus-specific thresholds. We speculate that during remote preconditioning events secreted components of the uterine UPR have the capacity to transmit and propagate the tocolytic-preconditioned phenotype to adjacent naive cells. Thus, as previously mentioned, we propose that a uterine myocyte-derived UPR secretome, in the circulation of pregnant women, may play a similar role in systemic remote preconditioning of the vasculature and immune response during pregnancy.
Extracellular Cell-Free GRP78 Is Anti-Inflammatory
One mechanism whereby the UPR secretome may modulate systemic conditioning is through the propagation of anti-inflammatory signaling. Extracellular GRP78 specifically has been demonstrated in multiple studies, both in vitro and in vivo, to play an anti-inflammatory immunomodulatory role.
In vitro
Upon UPR activation, GRP78 levels increase in the stressed cell and translocate to the surface with high amounts being released into the culture media. 81,86 Cells that secrete GRP78 into their extracellular environment were found to gain the ability to promote a prosurvival and antiapoptotic phenotype displaying resistance to antiangiogenic chemotherapeutic agents such as Bortezomib. 81 Cell-free GRP78 has been demonstrated to confer an anti-inflammatory prosurvival phenotype by binding to target cell surface receptors such as Cripto-1, which allows for the attenuation of transforming growth factor β tumor suppressor functions. 91 Extracellular GRP78 has also been demonstrated to block p53 action and thereby inhibit its proapoptotic targets, BOK and NOXA. 81 Taken together, these data suggest that GRP78 secreted into the extracellular space has the capacity to confer an anti-inflammatory, prosurvival phenotype.
In vivo
Elevated extracellular cell-free GRP78 is found in saliva, serum, oviductal fluid, and the synovial fluid of patients with rheumatoid arthritis. 82 –85 It was determined in this context that GRP78 also performed anti-inflammatory and immunomodulatory functions. When exposed to cell-free GRP78, human peripheral blood mononuclear cells (PBMCs) displayed a dose-dependent increase in the anti-inflammatory cytokines TNFα and interleukin (IL)-10 secretion. In stimulated PBMCs, the presence of extracellular GRP78 lowered the levels of IL-1β and increased the levels of IL-1 receptor antagonist. The concentration of soluble tumor necrosis factor receptor 2 (TNFR2) levels, which act to suppress the pro-inflammatory activities of TNFα, also increased confirming the role of extracellular GRP78 as a propagator of an anti-inflammatory signaling cascade. 83 It has become apparent that extracellular GRP78 has remarkable anti-inflammatory and immunomodulatory properties. In animal models of collagen-induced arthritis, prophylactic administration of recombinant GRP78 1 week before the initiation collagen immunization was sufficient to prevent the induction of collagen-induced arthritis. 92 Administration of GRP78 at the onset of collagen-induced arthritis was also successful in suppressing the development of arthritis. 93 Further, suppression of collagen-induced arthritis was achieved by parenteral (gavage) administration of lentiviral vectors expressing GRP78. A single dose of exogenous GRP78 was sufficient to induce permanent remission of inflammation in collagen-induced arthritis, suggesting not only does GRP78 mediate anti-inflammatory actions but it is also capable of driving resolution of inflammation, likely through immune cell differentiation. 93 The effects of serum GRP78 have been found to be at least partially dependent of IL-4, as the suppressive effects of GRP78 are abrogated in the IL-4−/− mouse. These analyses suggest that administration of exogenous extracellular GRP78 can allow for increased resistance to and active resolution of inflammatory challenges. In a similar manner, in our recent Proceedings of the National Academy of Sciences (PNAS) paper, we administered 4-phenyl butyric acid, a chemical chaperone that mimics GRP78 action, and found we could reverse ER stress-induced preterm birth. 6
Glucose-Regulated Protein 78-Mediated Transmission of Biochemical Stress
In addition to possessing anti-inflammatory properties, extracellular GRP78 has also been demonstrated to play a critical role in the transmission of biochemical stress signals from one cell to another, which may also allow for a remote systemic preconditioning phenotype to be propagated across gestation. Specifically, this form of paracrine and potential endocrine signaling allows for amplification and expansion of a local tissue response to a systemic alarm or danger signal. The GRP78-mediated transmission of ER stress has been observed when conditioned media isolated from stressed tumor cells were exposed to naive macrophages, resulted in UPR induction and a concomitant transcriptional activation and secretion of pro-inflammatory cytokines. 94 In validation of the in vitro data, when mice were intraperitoneally injected with conditioned media isolated from stressed tumor cells versus unstressed control media, an activated ER stress response was recapitulated only in the liver of the stress-conditioned media-injected mouse. Cell–cell interactions mediated by soluble secreted factors have also been defined in the extracellular matrix of tumors, where this phenomenon has been implicated in modulating tumor cell progression. 8 Cancer-associated fibroblasts have been demonstrated to secrete factors that promote the multipotent mesenchymal stem cells to differentiate into cancer-associated fibroblast contributing further to tumor development. Size fractionation and mass spectrometry analysis identified conclusively that GRP78 was the factor promoting the transition from mesenchymal stem cell to cancer-associated fibroblast. 95 These data clearly indicate an exciting potential mechanism whereby the ER stressed pregnant uterus or even the fetus gives rise to a uterine secretome, which is secreted into the circulation to provide a systemic alert or update to circulating immune cells, other somatic cell types, or remote organs of the need to adapt, accommodate, or protect themselves against a possible stress events. We speculate that transmission of uterine stress signals in the form of a secretome acts in a remote preconditioning-like fashion during pregnancy, allowing for improved resistance to and increased tolerance of the normal gestational stresses every pregnancy experiences, promoting the maintenance of uterine quiescence.
Potential Fetal Implications
Until now, we have focused primarily on the maternal implications of remote preconditioning. However, as the fetal unit interacts with the maternal circulation, we speculate that there is the potential for the uterine secretome to affect the fetal compartment. Although there is evidence that the fetal compartment likely induces gestational stressors (such as hypoxic challenges, infection-mediated events, and stretch stimuli), which may have the capacity to trigger the uterine UPR secretome, there is no direct evidence that the maternal UPR affects fetal function. It is clear that the fetal UPR is responsible for modulating multiple cellular processes necessary for development, as various attempts to create whole-cell animal knockouts for multiple components of the UPR, including GRP78, IRE1, ATF6, and XBP1, have resulted in embryonic lethality. 96 –99 Further, it has been demonstrated that fetal UPR function is altered under pathophysiological conditions at the level of the placenta and fetal membranes. In fetal membranes, Lappas and colleagues revealed infection-mediated increases in the UPR increased prolabor proteins such as pro-inflammatory cytokines and matrix metalloproteinases. 100 Similarly, in cases of early-onset preeclampsia, it is thought ER stress-dependent increases in the UPR result in small placental phenotypes and subsequently intrauterine growth restriction. 101 Thus, as the fetal UPR is important in both physiological and pathophysiological states of fetal development, it is not hard to imagine that components of the maternal uterine UPR secretome can influence fetal UPR function, especially in fetal tissues that come in direct contact with the maternal circulation. In fact, we suspect maternal–fetal UPR signaling may be an important feedback tool in which both maternal and fetal stresses propagate an adaptive response. In support of this hypothesis, 1 study by Wright and colleagues revealed maternal stress significantly affected cytokine responses in fetal cord blood mononuclear cells. 102 Again, while no direct evidence exists for maternal to fetal trafficking of uterine UPR components, it has been demonstrated that fetal exosomal cargo can be passed to the maternal side of uterine tissues in a murine pregnancy model. 103 Currently, we are examining in vivo mouse models that will better allow us answer this question.
Potential Clinical Interventions
Many other disease states are dependent on the dysregulation of the UPR. Subsequently, a more recent effort has been made to create therapeutic interventions targeting different components of the UPR response, which may also be effective as potential therapeutic preconditioning agents. Many of the current interventions targeting the UPR are recognized as chemotherapeutic agents and are designed to enhance tumor sensitivity to apoptotic stimuli. For example, Bortezomib, a proteasome inhibitor, successfully blocks the phosphorylation of PERK and eukaryotic initiation factor 2 alpha and leads to an increase in the accumulation of unfolded proteins and eventual caspase-mediated apoptosis in human pancreatic cancer cells. Other UPR targeting chemotherapeutic agents include MKC-3946 (small-molecule IRE1α endoribonuclease inhibitor) and EGF-SubA (a fusion protein that selectively cleaves GRP78), both have similarly been shown to reduce the apoptotic threshold in various cancers. 104,105 In addition to traditional cancer therapies currently being utilized in the clinic, more preliminary studies have proven to be effective in mimicking the beneficial prosurvival anti-inflammatory effects of the UPR, utilizing recombinant technologies. As previously mentioned, studies examining the anti-inflammatory effects of cell-free synthetic GRP78 demonstrated a dose-dependent increase in the secretion of anti-inflammatory cytokines IL-10 and TNFR2 in cultured PBMCs. 83 Another promising agent, currently approved by the federal drug administration for the treatment of pregnant women with intrahepatic cholestasis, is tauroursodeoxycholic acid (TUDCA), an endogenously produced tertiary bile salt that acts as an ER chaperone. 106 Intrahepatic cholestasis, a late-onset liver disorder in pregnancy, is typically characterized by pruritis, increased maternal serum bile acids, and adverse fetal outcomes. 106 Repeated studies examining the efficacy of TUDCA treatment in intrahepatic cholestasis pregnancies have demonstrated significant decreases in maternal serum bile acids and alleviation of disease symptoms. 107,108 More recently, the use of TUDCA as a potential treatment for preterm birth has also been examined. 109 Specifically, in an ex vivo model of infection-mediated preterm birth, TUDCA was seen to significantly ameliorate prolabor mediators in lipopolysaccharide (LPS)-treated human term fetal membranes and myometrial explants. 109 Currently, however, no clinical trials have been performed directly analyzing the efficacy of TUDCA as a tocolytic therapy. Overall, it is clear that much work is needed to determine the effects of these various agents in context of the pregnant uterus. Each reagent does, however, have the potential to act as a therapeutic preconditioning agent, aimed to reduce systemic and local uterine inflammation, manage myometrial stress levels, and promote the maintenance of uterine quiescence.
Conclusion
Cervical length measurement and a previous preterm birth are currently the strongest predictors for a subsequent preterm birth; however, nulliparous women with no obstetrical history remain at a heightened risk. With limited treatments such as progesterone for prior preterm birth or shortened cervix, cervical cerclage for shortened cervix or cervical insufficiency, and possibly cervical pessary, the ability to accurately predict and prevent preterm labor remains one of the most critical challenges facing modern obstetrics. 4,110,111 With such a large subset of women unidentifiable or unresponsive to the currently available treatments, 2 major questions remain, (1) what cellular mechanisms lead to the initiation of premature uterine contractility? and (2) what therapies can be developed to predict and inhibit spontaneous preterm labor in all women? Previous studies from our laboratory have demonstrated that the uterine UPR plays a major role in modulating myometrial quiescence and that exogenous progesterone, the most successful of preventative treatments, and progesterone receptor inhibitors modify the local uterine UPR drastically in favor of quiescence or labor, respectively. Thus, it remains critical that we gain a better understanding of the exact cellular UPR-dependent processes necessary for facilitating quiescence. We predict gestationally regulated, stress-preconditioning of the myometrium and the resulting extracellular uterine secretome-dependent remote systemic preconditioning are important for the maintenance of a normal term pregnancy. Furthermore, the dysregulation of balance between uterine adaptation and maternal stress may play a role in the etiology of pregnancy-dependent complications, such as intrauterine growth retardation and tobacco smoke-induced uteroplacental hypoxia. In this context, we speculate the severity of stress evoked by these conditions and other uterine stressors correlated with preterm birth (eg, twin pregnancy or preeclampsia) may exceed the prophylactic threshold of the endogenous uterine UPR, resulting in a maladaptive prolabor phenotype. Subsequently, there are multiple pharmaceutical agents, currently used for the treatment of UPR-dependent diseases, that could be repurposed for preconditioning therapies similar to those currently being utilized in the field of cardiovascular ischemia/reperfusion. TUDCA, in particular, has already been shown to alleviate extreme UPR stress in other pregnancy-related diseases and may be a promising agent for the restoration of a preconditioned-like uterine profile. 109 Overall, this hypothesis represents a paradigm shift in how the UPR controls cellular homeostasis in a paracrine and endocrine manner during pregnancy and how dysfunctional regulation of this system may lead to deleterious pregnancy outcomes. Gaining a greater understanding of the mechanisms associated with the onset of preterm birth will ultimately allow for enhanced preterm birth diagnostics, novel tocolytic drug design, and more accurate preventative intervention protocols.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the March of Dimes #21 FY2012-152 and the NICHD 1R01HD06511.
