Abstract
Cyfluthrin is a pyrethroid insecticide and common household pesticide. The effect of cyfluthrin on Ca2+-related physiology in human osteosarcoma is unclear. This study investigated the effect of cyfluthrin on cytosolic-free Ca2+ concentrations ([Ca2+]i) and viability in MG63 human osteosarcoma cells. Cyfluthrin concentration-dependently induced [Ca2+]i rises. Cyfluthrin-induced Ca2+ entry was confirmed by the Mn2+-induced quench of fura-2 fluorescence. Cyfluthrin at concentrations of 10–100 μM induced [Ca2+]i rises. Ca2+ removal reduced the signal by approximately 50%. Cyfluthrin (100 μM) induced Mn2+ influx suggesting Ca2+ entry. Cyfluthrin-induced Ca2+ entry was inhibited 50% by protein kinase C (PKC) activator (phorbol 12-myristate 13-acetate) and inhibitor (GF109203X) and also by three inhibitors of store-operated Ca2+ channels: nifedipine, econazole, and SKF96365. In Ca2+-free medium, treatment with the endoplasmic reticulum Ca2+ pump inhibitor thapsigargin (TG) completely inhibited cyfluthrin-evoked [Ca2+]i rises. Conversely, treatment with cyfluthrin abolished TG-evoked [Ca2+]i rises. Inhibition of phospholipase C (PLC) with 1-[6-[((17β)-3-methoxyestra-1,3,5[10]-trien-17-yl)amino]hexyl]-1H-pyrrole-2,5-dion abolished cyfluthrin-induced [Ca2+]i rises. Cyfluthrin at 25–65 μM decreased cell viability, which was not reversed by pretreatment with the Ca2+ chelator 1,2-bis(2-aminophenoxy)ethane-
Introduction
Cyfluthrin belongs to a huge family of pesticides called pyrethroids 1 and is commonly used in household. 2 Its many forms of isomers are sold commercially. Most pyrethroids including cyfluthrin are considered to be highly toxic to animals and humans. 3 Therefore, it is important to explore the mechanisms underlying the effect of cyfluthrin on toxicologic research in cancer models. Regarding toxicity, cyfluthrin has been shown to have a genotoxic potential in human lymphocytes and rats 4 and several different cancer cell lines such as SH-SY5Y human neuroblastoma cells, HepG2 human hepatoma cells, and Caco-2 human colon cancer cells. 5 In terms of Ca2+ signaling, cyfluthrin has been reported to stimulate Ca2+ influx in neocortical neurons, 6 to alter N-type Ca(v)2.2 Ca2+ channel in neurons, 7 and to influence Ca2+- adenosine triphosphatase activity of rat brain synaptosomes and leukocyte membranes. 8 However, the effect of cyfluthrin on Ca2+ signaling and its related mechanism has not been explored in human osteosarcoma cells.
Ca2+ ions have a crucial role in cellular homeostasis. 9 An increase in intracellular free Ca2+ level ([Ca2+]i) can regulate many cellular responses, including proliferation, apoptosis, gene expression, protein folding, fertilization, channel gating, secretion, contraction, plasticity, enzyme activation, and so on. 10,11 Ca2+ homeostasis is also clinically relevant. Alzheimer’s disease, 12 cardiac disease, 13 and cancer 10,11 have been shown to involve abnormal cellular Ca2+ movement. In cell models, [Ca2+]i may rise as a result of Ca2+ influx from extracellular medium or be released from internal organelles like the endoplasmic reticulum. 10,11 Ca2+ could enter cells through various Ca2+ channels and receptors on the plasma membrane. 10,11 Literature shows that in MG63 human osteosarcoma cells, store-operated Ca2+ entry plays a key role in Ca2+ influx. 14 Phospholipase C (PLC)-related pathways play a pivotal role in Ca2+ release in many cell types. Activation of PLC leads to an increase in cytosolic levels of inositol trisphosphate (IP3) which stimulate Ca2+ release from the endoplasmic reticulum via the IP3 receptors. 15 Due to the importance of a Ca2+ signal, cells have different mechanisms to tightly control the signal. Elucidation of the pathways underlying a stimulus-induced Ca2+ signal is pivotal for understanding the effect of the stimulus on the physiology of the cell.
Since previous researches have shown that in MG63 cells, 16 [Ca2+]i rises can be evoked in response to the stimulation of external compounds such as niflumic acid, 17 amitriptyline, 18 and methoxsalen 19 . The goal of this study was to investigate the underlying pathways of this Ca2+ movement. To this end, fura-2 was applied as a Ca2+-sensitive dye to measure [Ca2+]i. The mechanisms of cyfluthrin-induced [Ca2+]i rises were explored. Furthermore, the cytotoxic effect of cyfluthrin and its relationship to Ca2+ were also examined.
Materials and methods
Chemicals
Cyfluthrin (Figure 1(a)) and other reagents were purchased from Sigma-Aldrich® (St Louis, Missouri, USA) unless otherwise indicated. The chemicals for cell culture were purchased from Gibco® (Gaithersburg, Maryland, USA). Aminopolycarboxylic acid/acetoxy methyl (fura-2/AM) and 1,2-bis(2-aminophenoxy)ethane-

Effect of cyfluthrin (a) on [Ca2+]i in fura-2-loaded MG63 cells. (b) Cyfluthrin was added at 25 s. The concentration of cyfluthrin was indicated. The experiments were performed in Ca2+-containing medium. (c) Effect of cyfluthrin on [Ca2+]i in the absence of extracellular Ca2+. Cyfluthrin was added at 25 s in Ca2+-free medium. (d) Concentration–response plots of cyfluthrin-induced [Ca2+]i rises in the presence or absence of extracellular Ca2+. The
Cell culture
MG63 human osteosarcoma cells purchased from Bioresource Collection and Research Center (Taiwan) were cultured in minimum essential medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U ml−1 penicillin, and 100 µg ml−1 streptomycin.
Solutions for [Ca2+]i measurements
Ca2+-containing medium (pH 7.4) had 140 mM NaCl, 5 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, and 5 mM glucose. Ca2+-free medium had similar chemicals as Ca2+-containing medium except that CaCl2 was substituted with 0.3 mM ethylene glycol tetraacetic acid (EGTA) and 2 mM MgCl2. Cyfluthrin was dissolved in ethanol as a 2 M stock solution. The other chemicals were dissolved in water, ethanol, or dimethyl sulfoxide. The concentration of organic solvents in the experimental solutions was below 0.1% and did not affect basal [Ca2+]i or viability.
[Ca2+]i measurements
Confluent cells grown on 6 cm dishes were trypsinized and made into a suspension in culture medium at a concentration of 106 cells per milliliter. Cell viability was determined by trypan blue exclusion (adding 0.2% trypan blue to 0.1 ml cell suspension). The viability was greater than 95% after the treatment. Cells were subsequently loaded with 2 μM fura-2/AM for 30 min at 25°C in the same medium. After loading, cells were washed with Ca2+-containing medium twice and were made into a suspension in Ca2+-containing medium at a concentration of 107 cells per milliliter. Fura-2 fluorescence measurements were performed in a water-jacketed cuvette (25°C) with continuous stirring; the cuvette contained 0.5 million cells suspended in 1 ml of medium. Fluorescence was monitored with a Shimadzu (Kyoto, Japan) RF-5301PC spectrofluorophotometer immediately after 0.1 ml cell suspension was added to 0.9 ml Ca2+-containing or Ca2+-free medium, by recording excitation signals at 340 and 380 nm and emission signal at 510 nm at 1-s intervals. For calibration of [Ca2+]i, after completion of the experiments, Triton X-100 (0.1%) and CaCl2 (5 mM) were added to the cuvette to obtain the maximal fura-2 fluorescence. The Ca2+ chelator EGTA (10 mM) was subsequently added to chelate Ca2+ in the cuvette to obtain the minimal fura-2 fluorescence. Control experiments showed that cells bathed in a cuvette had a viability of 95% after 20 min of fluorescence measurements. [Ca2+]i was calculated as previously described. 20
Manganese ions (Mn2+) and Ca2+ use the same movement pathways in cells, thus Mn2+ influx implicates Ca2+ entry. MnCl2 (50 μM) was added to cell suspension in the cuvette 30 s before fura-2 fluorescence recording began to determine cyfluthrin-induced Ca2+ influx. Data were recorded at excitation signal at 360 nm (Ca2+-insensitive) and emission signal at 510 nm at 1-s intervals as described previously. 21
Cell viability assays
The assessment of cell viability was based on the rationale that cells can cleave tetrazolium salts by dehydrogenases. Increases in the intensity of color correlated with the live cell number. Assays were conducted according to the manufacturer’s instructions (Roche Molecular Biochemical, Indianapolis, Indiana, USA). Cells were seeded in 96-well plates at a concentration of 104 cells per well in culture medium for 24 h in the presence of cyfluthrin. The cell viability detecting tetrazolium dye 4-[3-[4-lodophenyl]-2-4(4-nitrophenyl)-2H-5-tetrazolio-1,3-benzene disulfonate] (WST-1; 10 µl pure solution) was added to samples after cyfluthrin incubation, and cells were incubated for 30 min in a humidified atmosphere. In experiments using BAPTA/AM to chelate cytosolic Ca2+, cells were incubated with 5 μM BAPTA/AM for 1 h before incubation with cyfluthrin. The cells were washed once with Ca2+-containing medium and incubated with/without cyfluthrin for 24 h. The absorbance of samples (A450) was determined using an enzyme-linked immunosorbent assay reader. Absolute optical density was normalized to the absorbance of untreated cells in each plate and expressed as a percentage of the control.
Statistics
Data are shown as mean standard deviation of three independent experiments. Data were analyzed by one-way analysis of variances using the Statistical Analysis System (SAS®, SAS Institute Inc., Cary, North Carolina, USA). Multiple comparisons between group means were performed by post hoc analysis using the Tukey’s honestly significantly difference procedure. A
Results
Cyfluthrin-induced [Ca2+]i movement in MG63 cells
The basal [Ca2+]i level was 52 ± 1 nM (Figure 1(b)). Cyfluthrin evoked [Ca2+]i rises in a concentration-dependent manner at concentrations between μM 10 and 100 μM in Ca2+-containing medium. At a concentration of 100 μM, cyfluthrin evoked [Ca2+]i rises that attained a net increase of 51 ± 2 nM. In the absence of extracellular Ca2+, 10–100 μM cyfluthrin induced concentration-dependent [Ca2+]i rises (Figure 1(c)). The Ca2+ response saturated at 100 μM cyfluthrin because 200 μM cyfluthrin did not cause greater responses (Figure 1(b) and (c)). The concentration–response relationships of cyfluthrin-caused [Ca2+]i rises were depicted in Figure 1(d). The half-maximal effective concentration value was 50 ± 4 μM in Ca2+-containing medium or 48 ± 2 μM in Ca2+-free medium, respectively, by fitting to a Hill equation. Ca2+ removal reduced the Ca2+ signal by approximately 50%.
Cyfluthrin-induced Mn2+ influx suggests Ca2+ entry in MG63 cells
Mn2+ and Ca2+ share the same routes of movement in cells but the former quenches fura-2 fluorescence at all excitation wavelengths, 21 while fura-2 binding to Ca2+ amplifies the fura-2 fluorescence except at the Ca2+-insensitive excitation wavelength of 360 nm. Figure 2 shows that 100 μM cyfluthrin evoked an instant decrease in the 360 nm excitation signal that reached a maximum value of 51 ± 1 arbitrary units at 75 s. The Mn2+ quenching results imply that Ca2+ entry contributed to cyfluthrin-induced Ca2+ signal.
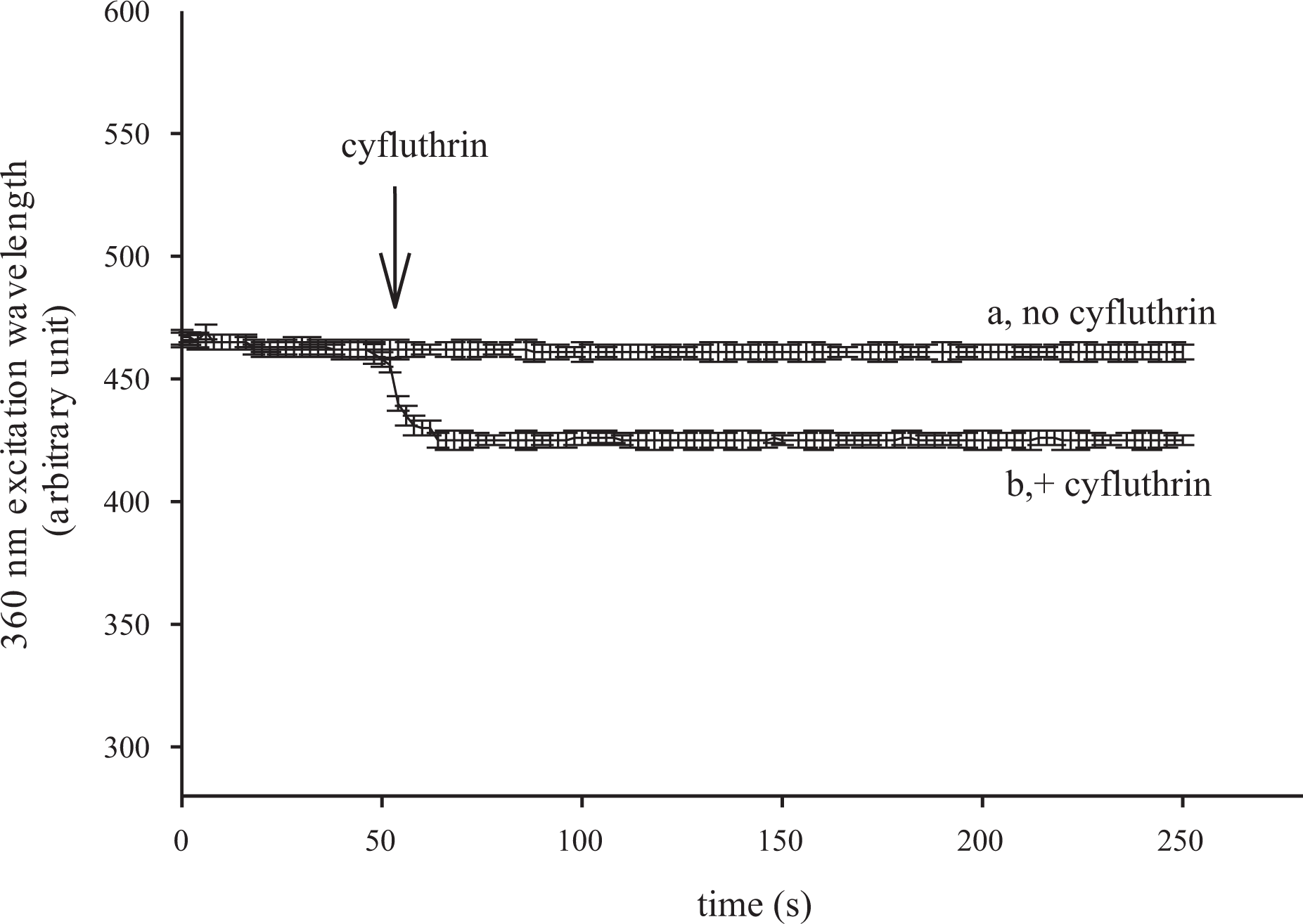
Effect of cyfluthrin on Ca2+ influx by measuring Mn2+ quenching of fura-2 fluorescence. Experiments were performed in Ca2+-containing medium. MnCl2 (50 μM) was added to cells 1 min before fluorescence measurements. The
Regulation of cyfluthrin-induced [Ca2+]i rises in MG63 cells
In Ca2+-containing medium, three store-operated Ca2+ entry inhibitors: nifedipine (1 μM), econazole (0.5 μM), and SKF96365 (5 μM); phorbol 12-myristate 13-acetate (1 nM; a protein kinase C (PKC) activator); and GF109203X (2 μM; a PKC inhibitor) were added 1 min before cyfluthrin (100 μM). All the chemicals inhibited cyfluthrin-induced [Ca2+]i rises by approximately 50% (Figure 3).
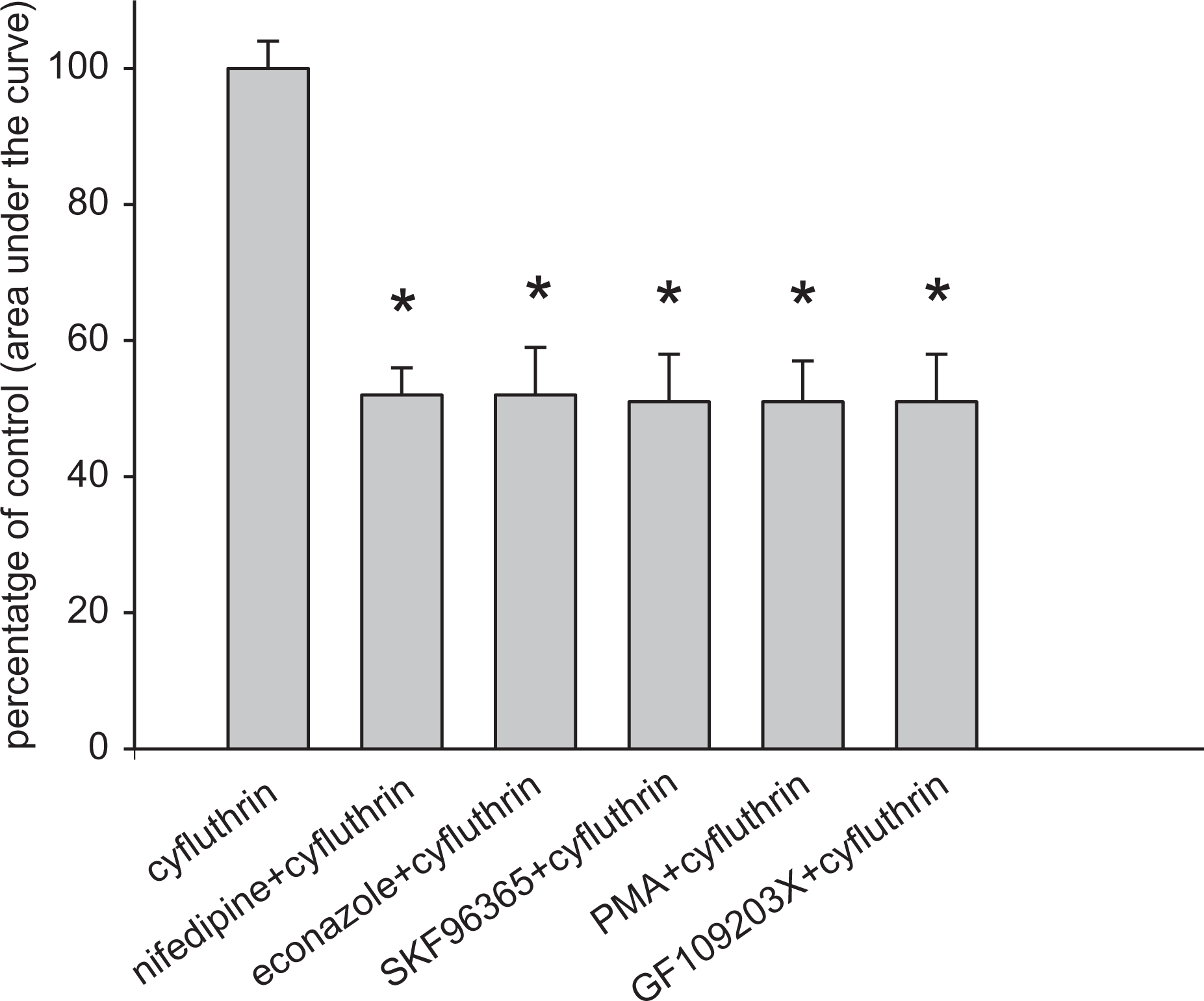
Effect of Ca2+ channel modulators on cyfluthrin-induced [Ca2+]i rises. In blocker- or modulator-treated groups, the reagent was added 1 min before cyfluthrin (100 μM). The concentration was 1 μM for nifedipine, 0.5 μM for econazole, 5 μM for SKF96365, 10 nM for PMA, and 2 μM for GF109203X. Data are expressed as the percentage of control (first column, 100%) that is the area under the curve (25–200 s) of 100 μM cyfluthrin-induced [Ca2+]i rises in Ca2+-containing medium and are mean ± SD of three independent experiments. *
The role of the endoplasmic reticulum in cyfluthrin-evoked Ca2+ release in MG63 cells
In MG63 cells, the endoplasmic reticulum was thought to play a key role in releasing store-operated Ca2+. 17 –19 To exclude the contribution of Ca2+ entry, the experiments were performed in Ca2+-free medium. Figure 4(a) shows that addition of 1 μM thapsigargin (TG), an endoplasmic reticulum Ca2+ pump inhibitor, 22 evoked [Ca2+]i rises of 22 ± 2 nM. Subsequently added 100 μM cyfluthrin did not induce [Ca2+]i rises. Conversely, Figure 4(b) shows that the addition of TG failed to release more Ca2+ after cyfluthrin-induced [Ca2+]i rises. It appears that the endoplasmic reticulum played a pivotal role in cyfluthrin-evoked Ca2+ release from organelles.

Effect of TG on cyfluthrin-induced Ca2+ release. ((a) and (b)) TG (1 μM) and cyfluthrin (100 μM) were added at time points indicated. Experiments were performed in Ca2+-free medium. Data are mean ± SD of three independent experiments. TG: thapsigargin; Ca2+: calcium ion; SD: standard deviation.
PLC played a role in cyfluthrin-evoked [Ca2+]i rises in MG63 cells
PLC-dependent activation of IP3 receptors results in the release of Ca2+ from the endoplasmic reticulum. 11 Thus, the role of this pathway in the cyfluthrin-induced release of Ca2+ was examined. 1-[6-[((17β)-3-Methoxyestra-1,3,5[10]-trien-17-yl)amino]hexyl]-1H-pyrrole-2,5-dion (U73122) 23 is a commonly used PLC inhibitor that is applied to examine if the activation of PLC was necessary for cyfluthrin-induced Ca2+ release. First, Figure 5(a) shows that adenosine triphosphate (ATP; 5 μM) was found to evoke [Ca2+]i rises of 23 ± 2 nM. ATP was chosen in our experiments because it is a well-established PLC-dependent stimulus of [Ca2+]i rises in most known cells. 12 U73122 was used to test its inhibitory effect on ATP-induced [Ca2+]i rises. Figure 5(a) and (b) shows that incubation with U73122 (2 μM) did not alter basal [Ca2+]i but completely inhibited ATP-induced [Ca2+]i rises. This suggests that U73122 effectively suppressed PLC activity. Subsequently, the data show that incubation with 2 μM U73122 did not alter basal [Ca2+]i but abolished 100 μM cyfluthrin-induced [Ca2+]i rises. Although U73122 is generally deemed as a selective inhibitor of PLC, the possibility that U73122 acted by PLC-independent action needs to be excluded. Thus, U73343 was used as a negative control. U73343 is structurally very similar to U73122 while it lacks inhibitory effect on PLC. Our results show that U73343 (2 μM) failed to alter ATP-induced [Ca2+]i rises (Figure 5(b)). Therefore, this most likely suggests that U73122 selectively inhibited PLC activity in cyfluthrin-induced [Ca2+]i rises.
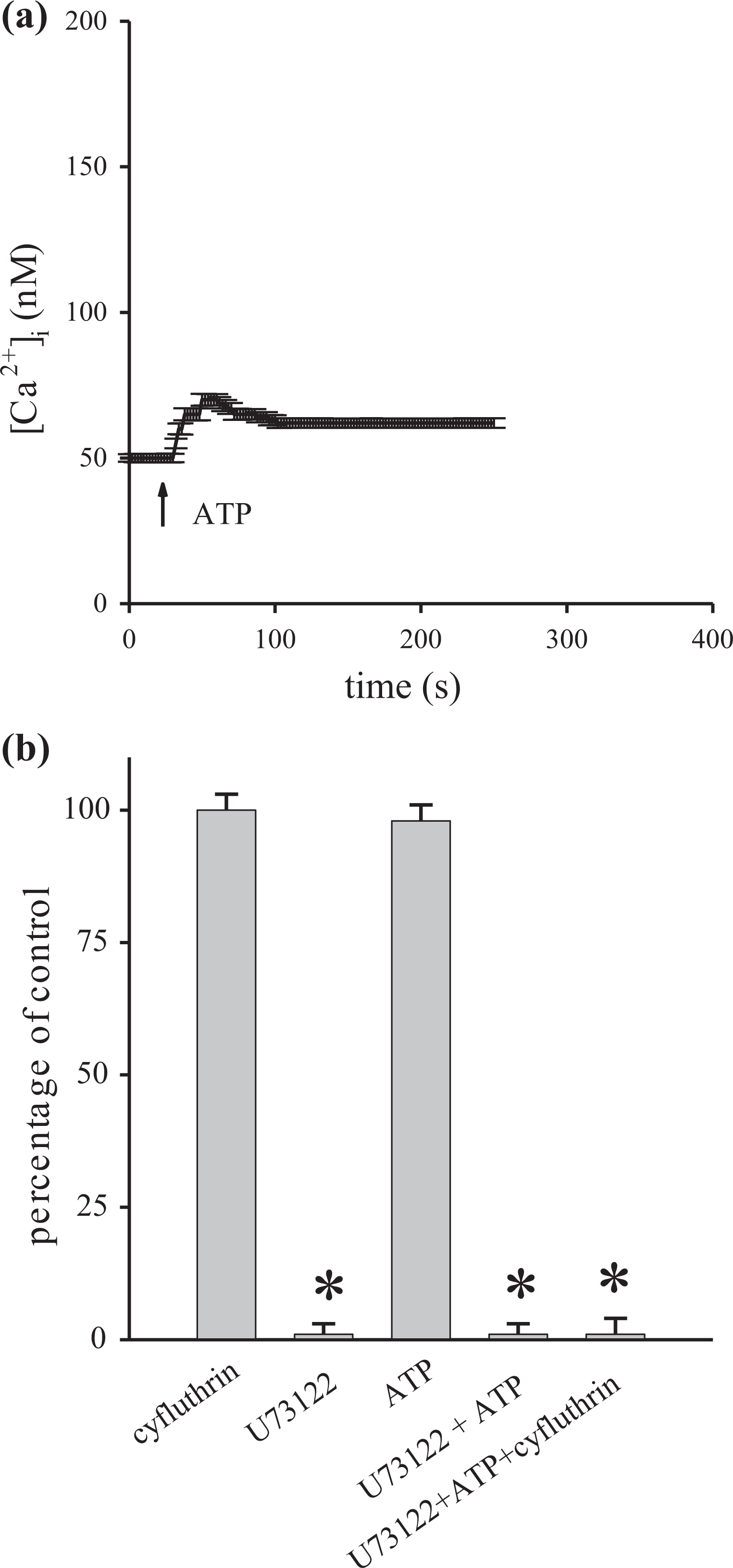
Effect of U73122 on cyfluthrin-induced Ca2+ release. Experiments were performed in Ca2+-free medium. Experiments were performed in Ca2+-free medium. (a) ATP (5 μM) was added at 25 s. (b) First column is 100 μM cyfluthrin-induced [Ca2+]i rises. Second column shows that 2 μM U73122 did not alter basal [Ca2+]i. Third column shows ATP-induced [Ca2+]i rises. Fourth column shows that U73122 pretreatment for 30 s abolished ATP-induced [Ca2+]i rises (*
Cyfluthrin decreased cell viability in MG63 cells
Cyfluthrin (0–65 μM) was added to cells for 24 h, and tetrazolium assay was performed. Cyfluthrin caused a decrease in viability in a concentration-dependent manner between 25 μM and 65 μM (Figure 6). Because cyfluthrin significantly induced [Ca2+]i rises, the next question was whether cyfluthrin-caused cytotoxicity was induced by preceding [Ca2+]i rises. The intracellular Ca2+ chelator BAPTA/AM (5 μM) 24 was applied to prevent [Ca2+]i rises during cyfluthrin incubation. At a concentration of 100 μM, cyfluthrin did not evoke [Ca2+]i rises in BAPTA/AM-treated cells (not shown). Figure 6 shows that 5 μM BAPTA/AM incubation did not change the viability of control cell. In the presence of 25–65 μM cyfluthrin, BAPTA/AM loading did not reverse cyfluthrin-induced cell death. Thus, the findings implicate that cyfluthrin-evoked cytotoxicity was not caused by [Ca2+]i rises.

Effect of cyfluthrin on cell viability. Cells were treated with 0–60 μM cyfluthrin for 24 h, and the cell viability assay was performed. Data are mean ± SD of three separate experiments. Each treatment had six replicates (wells). Data are expressed as a percentage of control response that is the increase in cell numbers in cyfluthrin-free groups. Control had 10,555 ± 112 cells per well before experiments, and had 13,125 ± 211 cells per well after incubation for 24 h. *
Discussion
Ca2+ signaling plays a key role in numerous cellular responses. Cyfluthrin has been shown to trigger Ca2+ entry in neocortical neurons, 6 to alter N-type Ca2+ channel in neurons, 7 and to alter Ca2+-ATP pump activity of rat brain synaptosomes and leukocyte membranes. 8 However, the effect of cyfluthrin on human osteosarcoma cells is unclear. The present study shows that cyfluthrin induced [Ca2+]i rises in MG63 cells by depleting Ca2+ stores and inducing Ca2+ entry. Removal of extracellular Ca2+ inhibited 50% of cyfluthrin-induced Ca2+ entry throughout the measurement of 200 s, suggesting that cyfluthrin induced both Ca2+ entry and Ca2+ release. The Mn2+ quenching results also implicate that Ca2+ entry contributed to cyfluthrin-induced Ca2+ signal.
The pathway of cyfluthrin-induced Ca2+ entry was examined. In MG63 cells, store-operated Ca2+ channels have been shown to play a crucial role in [Ca2+]i rises triggered by different compounds such as niflumic acid, amitriptyline, and methoxsalen. 17 –19 Although there are so far no selective inhibitors for this Ca2+ entry, nifedipine, econazole, and SKF96365 were commonly applied for this purpose. 25 –27 Our results suggest that all of these three chemicals inhibited cyfluthrin-induced [Ca2+]i rises. Therefore cyfluthrin-induced Ca2+ entry appears to involve store-operated Ca2+ pathway.
The changes in the activity of protein kinases are well established to be able to alter Ca2+ signaling. 28,29 Our data show that cyfluthrin-evoked [Ca2+]i rises were inhibited by 50% by enhancing or inhibiting PKC activity. It appears that cyfluthrin can only trigger full Ca2+ responses when PKC activity is at a normal status. The relationship between PKC and Ca2+ movement has been reported. For example, Na+-Ca2+ exchanger 3 was reported to be differentially regulated by PKC and protein kinase A. 30 Calretinin was shown to participate in regulating steroidogenesis by PLC-Ca2+-PKC pathway in Leydig cells. 31
Regarding the Ca2+ stores involved in cyfluthrin-evoked Ca2+ release, the TG-sensitive endoplasmic reticulum store seemed to be the dominant one. TG was originally purified from the Mediterranean plant
Cell viability could be altered in a Ca2+-dependent or -independent manner. Previous studies have shown that rapid disruption of cellular integrity of zinc-treated astroglia was regulated by Ca2+-dependent mechanisms. 34 In contrast, sensitization to autoimmune hepatitis in Ca2+-independent phospholipase A2-null mice led to duodenal villous atrophy with apoptosis. 35 Our data show that cyfluthrin induced Ca2+-independent cell death in a concentration-dependent manner in MG63 cells. Literature shows that the plasma level of cyfluthrin in human patients may reach 10 μM. 36,37 In patients with liver or kidney dysfunction or healthy persons taking higher doses this level may go much higher. Our findings show that at a concentration of 45 μM cyfluthrin evoked death of 50% of MG63 cells. Thus, the clinical relevance of our data may not be excluded.
In sum, our data show that cyfluthrin evoked [Ca2+]i rises via inducing Ca2+ influx through a PKC-sensitive store-operated Ca2+ entry and also via Ca2+ release from the endoplasmic reticulum in a PLC-dependent fashion. In the physiological level, cyfluthrin also induced Ca2+-dissociated cell death. Thus, caution should be exercised when using cyfluthrin for other in vitro research in MG63 human osteosarcoma cells.
Footnotes
Author contribution
W-ZL and Y-CL contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by VGHKS107-149 to Y-CL.
