Abstract
The present study investigated the reproductive toxicity of furan in an Extended One-Generation Reproductive Toxicity Study in rats. Sprague Dawley F0 weaning rats (30 per sex per group) were exposed to furan orally at 0, 1, 2.5, 5, and 10 mg kg−1 for 10 weeks (males) and 2 weeks (females) and then mated. Results of F0 indicated that in the furan-treated groups (5 mg kg−1 and 10 mg kg−1), body weight (bw) gain decreased during prebreed and gestational period while increased during lactation periods. F0 animals prebreeding exposure resulted in head tilt and foot splay at 10 mg kg−1. Number of live pups at birth were decreased (p < 0.001) at 10 mg kg−1. At postnatal day (PND) 70, a significant (p = 0.03) decrease in testosterone levels of male rats and estrogen levels of female rats (p = 0.05) was observed in 10 mg kg−1 furan-treated group in F1 generation. Luteinizing hormone, follicle-stimulating hormone, and progesterone levels were also reduced, but their reduction was not statistically significant in all groups. In higher dose furan group (10 mg kg−1), testicular and ovarian weights were reduced in F1 generation at PND 70, with decreased daily sperm production (p = 0.01) and disturbed estrous cyclicity (p < 0.01). Some histopathological changes were also observed in testis and ovaries in groups whose parents were previously exposed to 10 mg kg−1 bw of furan group. Based on the above results, it is suggested that exposure to food-based contaminant furan induced remarkable changes in the F0 (parental stage) and F1 (offspring, pubertal, and adult stage) generations of Sprague Dawley rats.
Introduction
Furan (C4H4O) is a colorless volatile organic compound that is used in several industries and is also found in various common food products that undergo excessive heat treatment, such as canned and jarred foods, coffee, sauces, soups, baby foods, infant formula, and beverages. 1 –5 Furan is also present in the environment, as it is the main constituent of cigarette smoke, wood smoke, and exhaust gases from engines. 6 Because of the toxic nature of furan and its excessive daily intake in humans through diet and air, it has become a matter of concern. The US Food and Drug Administration has reported the presence of furan in food items in various surveys. 1,7 The European Food Safety Authority also reported that among adults, prepared coffee is the main source of furan. 8 The International Agency for Research on Cancer has classified furan as a possible human carcinogen (group 2B). 9 Previous observations reported that furan caused various types of cancer and has an adverse effect on the biological system of humans and animals. 9 –12 Furan has also been known as hepatoxic and carcinogenic in the rats and mice approved by National Toxicology Program. 13
Furan needs to be removed from consumer products because of its negative effects on human health. 6 Various toxicants are dangerous to cell membranes since they cause increase in lipid peroxidation, production of reactive oxygen species (ROS), and DNA damage in the cells. 5,11,14 Furan can easily pass through biological membranes due to its low polarity and acts as an endocrine disruptor as it has been reported previously that exposure to different concentrations of furan resulted in the disturbance of the reproductive neuroendocrine system in rats. 5 It has been previously reported that furan is cytotoxic, genotoxic, and an apoptotic inducer. 15,16 Previous literature on furan reported that its exposure causes reproductive impairment by disturbing reproductive organ growth; spermatogenesis; testosterone (T), luteinizing hormone (LH), and follicle-stimulating hormone (FSH) secretion; and reproductive performances. 5,6,16 It has been previously reported that hazardous environmental toxicants can bring variations in the signaling pathways of cells and defensive and protective systems of the antioxidant levels of the cells by affecting physiological conditions. 12,17 Furan caused significant changes in histological structure of cells, malondialdehyde levels, antioxidant enzyme activities, and caused DNA damage in lymphocytes and sperm cells by producing ROS. 6,11,12 Adverse effect of furan has been tested for a variety of reproductive parameters, and prominent effects were reported in the reproductive systems of males and females. 11,18 –20 However, a study was conducted on Taiwanese boys whose mothers used contaminated (with polychlorinated biphenyls (PCBs) and furan) rice oil that resulted in smaller size of penis in comparison with age-matched control boys. 21 Similarly, Guo et al. reported that sperms of prenatally exposed young men with PCBs and furan have abnormal sperm morphology, reduced sperm motility, and also reduced ability of sperm to enter hamster oocytes. 22 Tyla et al. observed the reproductive toxicity of food toxicant acrylamide in the next two generation of Fisher (F344) rats. 23 Furan was also reported to occur in the morning urine samples of humans in different concentrations. 24
Previously, many studies revealed the reproductive toxicity of furan in adult rats. Thus far, no information is available in the literature about the toxic effects of furan in the next generation. There is a considerable debate whether furan is a toxicant in the next generation. The current study was designed with the aim to study the Extended One-Generation Reproductive Toxicity Study (EOGRTS) across different life stages (parental, offspring, and adults). The main reason for this EOGRTS was to assess the possible effects of furan on F0 (parental) reproduction and F1 (first generation) offspring growth and development with calculations of endocrine function following exposure during early period of development. This EOGRTS design was performed in compliance with Environmental Protection Agency (EPA; Organisation for Economic Co-operation and Development (OECD) #443) testing guidelines and under EPA Good Laboratory Practice regulations. 25 –29 In view of these concerns, we performed a frequent oral dose-based EOGRTS in Sprague Dawley rats to find out the potential toxic effects of furan on reproductive performance of rats in F0 (parental) and F1 (first generation).
Materials and methods
Animals
Chemicals and animals
Furan (Catalog no.185922, 99% purity) was purchased from Sigma–Aldrich, St Louis, Missouri, USA. Sprague Dawley rats (postnatal day (PND) 35) were collected from the Animal House Facility of Department of Animal Sciences, Quaid-i-Azam University, Islamabad, Pakistan. The rats were housed in steel cages. Prior to the start of the experiment, standard colony conditions were maintained accordingly. 6,20,30
Ethical considerations
Animal handling and subsequent euthanization were done according to the guidelines provided by the “Ethics Committee” of the Department of Animal Sciences, Faculty of Biological Sciences, on care and use of animals for scientific research (Ref No.As-2018/431). Animal handling and euthanization protocols used currently were according to the European Union guidelines for human use of animals for experimental purposes.
Study design
By following OECD 443, an EOGRTS was performed and additional analyses at the age of puberty was also performed. The animals were scattered among five dose groups for better assessment of dose–response relations. Male rats were exposed to furan 70 days prior to mating and female rats were exposed 14 days prior to mating to cover at least one full spermatogenic cycle or two estrogenic cycles. The first control group received a daily oral dose of corn oil (50 μL), while second, third, fourth, and fifth group received furan by gavage administration (1, 2.5, 5, and 10 mg kg−1 in 50 μL corn oil, respectively) for 70 days (male rats) and 14 days (female rats). Dose time was chosen according to the standard protocol 443 of the OECD (2011). Different doses of furan were selected to assess the dose-dependent EOGRTS exposure response. Furan doses used in this experiment were selected on the basis of doses used in previous studies on furan and acrylamide with some slight modifications in concentration 6,16,20,31,32 and OECD guideline #408 (doses can be used according to the relevant substance if doses have not been previously reported). In male rats, dose exposure continued during mating, and in female dams, dose exposure continued during pregnancy and lactation. Animals were housed in couples for mating, avoiding sibling mating. Pregnant females were single housed. Presence of sperm in vaginal smear or in the dams’ vaginal plug was considered as gestational day (GD) 0 or day 0 of pregnancy. On GD 0, 6, 13, and 20, mated females were weighed. Each female was shifted to a shoe-box cage with bedding on GD 18. On the beginning of GD 20, females were monitored twice daily, for verification of littering. Male rats were monitored during prebreed and mating period and female rats during prebreed, gestation, and lactation period with a complete physical examination. Also, the number of living and dead pups, their weights, sex ratio, anogenital distance (AGD), nipple retention (NR), and survival rate were recorded. Litter size was not uniform. During the lactation period, the mortality, the time to vaginal opening, and preputial separation in pups were recorded. F1 females were housed individually, while F1 males had to be housed in small groups of five to six animals due to space limits. All the reproductive parameters of F1 males and females were documented. At around 10 weeks of age, F1 animals were marked for reproductive tests (hormonal profile, body weight (bw), and histopathology).
Formulas for indices:
Blood and tissue collection
At PND 70, male rats (n = 10 per group) and female rats (n = 10 per group) were weighed and blood samples were collected using cardiac puncture for hormonal analysis, while testicular, epididymis, prostrate, seminal vesicles, uterus, and ovarian organs were sampled, weighed, and processed for histological and sperm parameter.
Body weight gain
On PND 70, animals were weighed and mean bw gain was attained.
Determination of AGD, NR, and puberty onset
On PND 1, number of male and female pups were totaled, and the weight of each pup was recorded. The pups were examined for deformities, and in all pups, the AGD was examined in an ocular stereomicroscope. All the pups were weighed continuously on PND 0, PND 4, PND 7, and PND 14. On PND 14, male pups were inspected for number of NRs. Puberty in male and female pups was examined through the appearance of external signs of puberty onset. From PND 34, the vaginal opening (female) and preputial separation (male) of the pups (n = 10 per group) was noticed every day. The day that a complete preputial separation and vaginal opening was seen was considered as the day of puberty onset for that animal, and the average day of puberty onset for each group was attained and described.
Determination of estrous cyclicity
For the measurement of estrous cyclicity of the female rats, the vaginal smear from PND 60 to PND 70 was collected and observed under a microscope for 10 consecutive days. The uniformity of the estrous cycle was attained and described.
Daily sperm production (DSP)
Frozen testicular tissues were thawed at room temperature; parenchyma was weighed after removing tunica albuginea. Testicular homogenate was prepared by solution (5 mL) containing sodium chloride (0.9%) and Triton X-100 (0.5%) using a rotor-stator homogenizer (IKA-Werke, Staufen, Germany) for 30 s. After that, the homogenate was diluted five times, and the sample was taken on Neubauer chamber and 19th-stage spermatids were counted under a light microscope at 40× magnification. At least three readings were taken for average number of spermatids calculations. To obtain the number of spermatids, these values were used and divided by 6.3 (number of days the spermatids remain in seminiferous epithelium) to determine DSP.
Histopathology
Testis and ovaries collected from the experiment were fixed in 10% formalin for 48 h. Next to fixation, dehydration was carried out at room temperature in the ascending grades of alcohol, and tissues were shifted to xylene. After clearing, tissues were embedded in paraffin wax, and 5- to 7-µm-thick sections were cut using microtome (Thermo, Shandon Finesse 325, Cambridge, UK). Sections were transferred to albumenized slides that were preheated at 37°C. For complete stretching of tissues and removal of the bubbles from glass slides, slides were placed overnight in incubator at 58 C. Then, tissues were rehydrated in descending grades of alcohol, stained with hematoxylin–eosin stain, and covered with cover slip. Prepared slides were observed under Leica microscope (New York City, New York, USA) equipped with digital camera (Canon, Japan).
Hormonal analysis
Plasma levels of T were determined by enzyme-linked immunosorbent assay (ELISA) kit (Cat # BC-1115, Bio Check Inc., California, USA) based on the principle of competitive binding between T in the specimen and T-horseradish peroxidase (HRP) conjugate for a constant amount of rabbit anti-T. The FSH (RD-FSH-Ra, Reddot Biotech Inc., Kelowna, British Columbia Canada) ELISA is principally based on the competitive inhibition binding reaction between biotin-labeled FSH and FSH in the plasma test samples, with the FSH-specific antibody. LH (RD-LH-Ra, Reddot Biotech Inc. Kelowna, British Columbia, Canada) ELISA is principally based on the competitive inhibition binding reaction between biotin-labeled LH and LH in the plasma test samples, with the LH-specific antibody. Estrogen (ELISA) kits (Cat # ES-180S) are based on the principle of competitive binding between estrogen in the test specimen and estrogen-HRP conjugate for a constant amount of rabbit anti-estradiol. Progesterone concentration was quantitatively determined in plasma through ELISA kit (Cat # PG-129S), which was based on the principle of competitive binding between progesterone in the test specimen and progesterone-HRP conjugate for a constant amount of rabbit anti-progesterone. The ELISA kits were validated by verifying the slope of the curve obtained by serial dilutions (0%, 20%, 40%, 60%, and 80%) of the sample with ELISA buffer and the curve created with kit standards, T (slope (m) = 0.0697, r 2 = 0.988), FSH (m = 0.039, r 2 = 0.974), LH (m = 0.0241, r 2 = 0.96), estrogen (m = 0.066, r 2 = 0.966), and progesterone (m = 0.021, r 2 = 0.9824) indicating the positive linear relationship.
Statistical analysis
The results are stated as mean ± SD. Statistical analysis was done using lme4 33 and easyanova 34 package of R 3.2.5. 35 Using Shapiro–Wilks’s, Levene’s, and Tukey’s 1 − df test, we tested the statement of normality, homogeneity of variances, and additivity of the model. The effects of different treatments were analyzed by analysis of variance ea1 command of R with completely randomized design, followed by post hoc Tukey’s honest significant difference (HSD), while the data of indices, F0 head tilt, foot spray, parental F0 reproductive parameters (no. (%) of plug/sperm-positive females, no. (%) of pregnant females, no. of live litters on PND 0, no. of live litters on PND 4, etc.), and percentages were compared using χ 2 test. The values of p < 0.05 were considered statistically significant.
Results
Results of parental F0 weight gain after furan exposure
The significant decrease in bw of prebreed male (p = 0.02) 5 mg kg−1 and (p = 0.001) 10 mg kg−1 and prebreed female (p = 0.1) 5 mg kg−1 and (p = 0.03) 10 mg kg−1 was observed in the furan-treated groups when compared with the control (nontreated) group. Similarly, the F0 females showed significant decrease in bw during gestational period in highest dose (p = 0.05) 10 mg kg−1 furan-treated groups (Table 1). However, the bw of F0 females significantly increased during lactation period in 5 mg kg−1 (p = 0.05) and 10 mg kg−1 (p = 0.02) furan-treated groups when compared to the control group (Table 1).
Effects of oral furan (0, 1, 2.5, 5, and 10 mg kg−1) exposure on F0 parental body weight gains.a

SEM: standard error of the mean.
a p and F values in the rows from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SEM.
bSignificant at p < 0.05 versus control.
cSignificant at p < 0.001 versus control.
dSignificant at p < 0.01 versus control.
Results of parental F0 head tilt and foot splay after furan exposure
After the observation of physical parameters, a significant difference was observed in head tilt and foot splay of F0 males of furan-treated groups when compared with control group. Similarly, in F0 females, only significant difference was also observed in foot splay (Table 2).
Mean (±SD) results of effects of parental F0 head tilt and foot spray after furan exposure orally (0, 1, 2.5, 5, and 10 mg kg−1).a

a These data are presented as the number (%) of the 30 animals per specified dose group per sex, which exhibited the finding(s) at least once from weaning to demise. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other-treated groups using χ 2 test.
Results of parental F0 reproductive parameters after furan exposure
The reproductive parameters of F0 parental rats (to delivered F1 offspring) are presented in Table 3. The percentage of plug/sperm-positive females (p = 0.97), age of pregnant females (66.6% and 56.6%, respectively), and gestational length (p = 0.87) were nonsignificantly changed among all furan-treated groups as compared to control group (Table 3). Additionally, the number of live pups/litters was reduced significantly in 10 mg kg−1 groups when compared with the control group (p = 0.001, F = 4.81; Table 3).
Results of effects of parental F0 reproductive parameters after furan exposure orally (0, 1, 2.5, 5, and 10 mg kg−1).a

PND: postnatal day; SD: standard deviation.
a p and F values in the rows of gestational length and no. of live pups/litter (PND 0) from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Data of number (%) and index were compared using χ 2 test. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SD.
bSignificant at p < 0.05 versus control.
cSignificant at p < 0.001 versus control.
Results of F1 generation results during lactation period
In F1 generation, total number of pups born and total number of live pups on PND 0, PND 4, PND 7, PND 14, PND 21, and PND 28 were significantly (p < 0.001) reduced in 10 mg kg−1 furan-treated group. There was no difference observed in sex ratio and survival of F1 pups during lactation period. The litter size of F0 high-dose dams was reduced significantly (p < 0.001) during the lactation periods. Similarly, the bw of all the pups on PND 4 was reduced significantly (p < 0.001) in 10 mg kg−1 furan-treated group (Figures 1 -3). AGD was nonsignificantly affected by treatment in both male and female pups. Males usually have zero nipples, but sometimes a few nipples could be observed. The exposure of parents to furan did not affect the NR in F1 males (p = 0.07, F = 2.28; Table 4).

Mean body weights of F1 litters from PND 1 to PND 70. Data are presented as mean ± SD. ANOVA with complete randomized designs followed by Tukey’s post hoc shows a pairwise comparison of control with parently furan (0, 1, 2.5, 5, and 10 mg kg−1) exposure groups in F1 generation. ANOVA: analysis of variance; PND: postnatal day.

Male body weights of F1 litters from PND 1 to PND 70. Data are presented as mean ± SD. ANOVA with complete randomized designs followed by Tukey’s post hoc shows a pairwise comparison of control with parently furan (0, 1, 2.5, 5, and 10 mg kg−1) exposure groups in F1 generation. ANOVA: analysis of variance; PND: postnatal day; SD: standard deviation.

Female body weights of F1 litters from PND 1 to PND 70. Data are presented as mean ± SD. ANOVA with complete randomized designs followed by Tukey’s post hoc shows a pairwise comparison of control with parently furan (0, 1, 2.5, 5, and 10 mg kg−1) exposure groups in F1 generation. ANOVA: analysis of variance; PND: postnatal day; SD: standard deviation.
Results of F1 litters parameters during lactation.a

NR: nipple retention; PND: postnatal day; AGD: anogenital distance; SD: standard deviation.
a p and F values in the rows from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SD.
bSignificant at p < 0.05 versus control.
cSignificant at p < 0.001 versus control.
Results of F1 generation at PND 70
From PND 30 onward, all the male and female pups were analyzed daily for preputial skin separation and vaginal opening. The day when the preputial skin was separated and the vaginal opening appeared was consider as day 1 of puberty. The analysis of external signs of puberty onset showed that parental furan exposure did not affect the pup’s puberty (Table 5). On PND 70, male and female rats were dissected. Results of male rats indicate that the testicular weight was significantly (p = 0.03) decreased in the high-dose treated group (10 mg kg−1), while there were no significant change observed in weight of prostate, seminal vesicle, and epididymis in dose-dependent manner compared with control group. The DSP was also significantly decreased in 5 mg kg−1 (p = 0.04) and 10 mg kg−1 (p = 0.01) treated group. Similarly, in female rats at PND 70, no significant change was observed in uterus weight, while a significant (p = 0.02) decrease in paired ovary weight was observed in 10 mg kg−1 furan-treated groups. The estrous cycle length of female rats was also significantly (p < 0.01) increased in 5 mg kg−1 and 10 mg kg−1 treated group compared to control group (Table 5).
Results of F1generation at PND 70.a
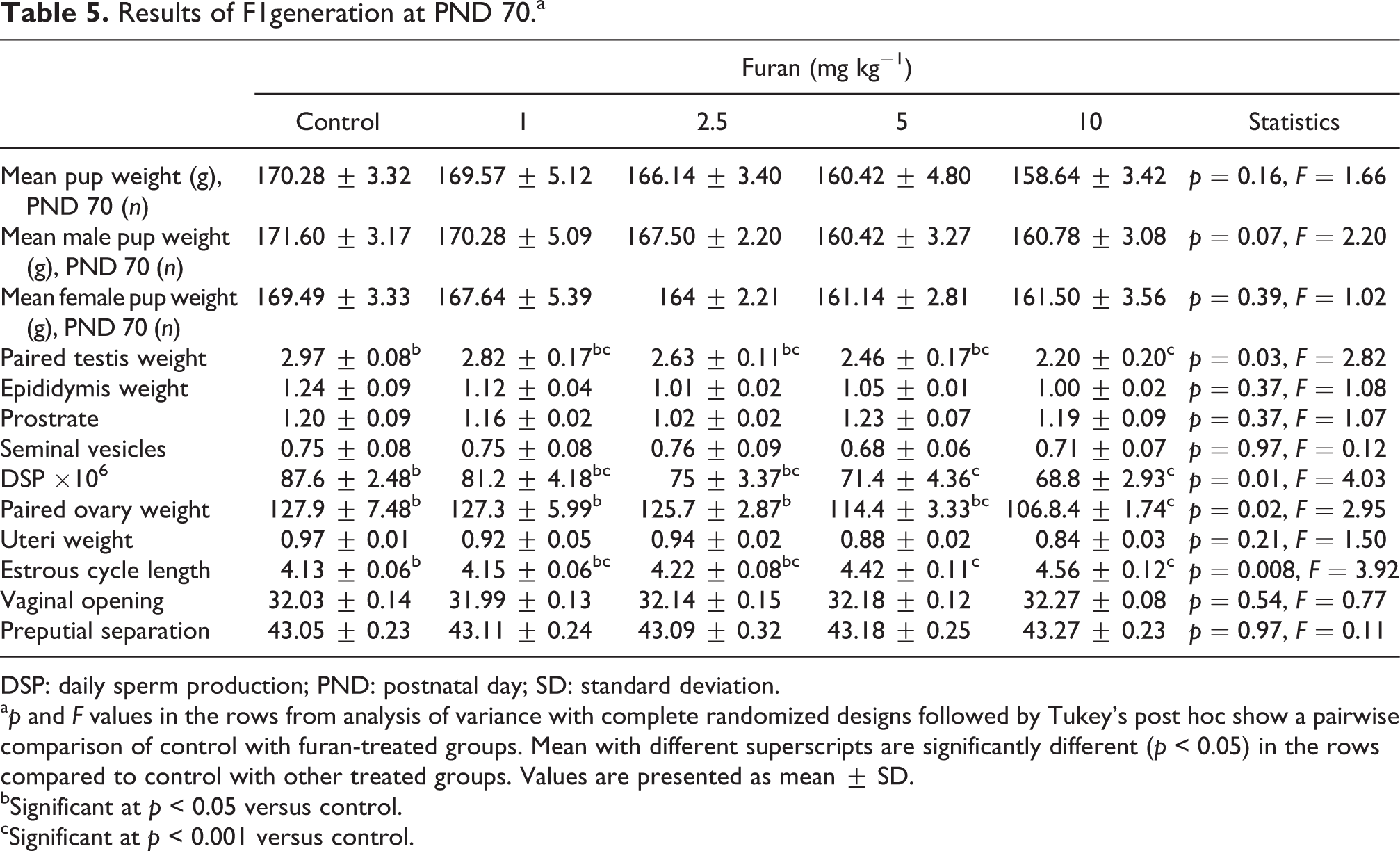
DSP: daily sperm production; PND: postnatal day; SD: standard deviation.
a p and F values in the rows from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SD.
bSignificant at p < 0.05 versus control.
cSignificant at p < 0.001 versus control.
Results of F1 generation histopathology of testis and ovary at PND 70
On PND 70, parental exposure to different concentrations of furan exhibited some obvious changes in the testis histology of F1 generation. The lumen diameter was increased in higher dose treated groups. Changes were also found in tubular diameter and epithelial height but were not statistically different in the furan-exposed groups when compared with the control group (Table 6; Figure 4). Histological analysis of ovary revealed the presence of different types of ovarian follicles, that is, primordial follicle, primary follicle, secondary follicle, antral follicle, atretic follicle, cystic follicle, and corpus luteum (Table 6). Many minor differences were observed in follicles of female ovary. However, the number of cystic follicles was prominently observed in 10 mg kg−1 treated group (Table 6; Figure 5).
Results of F1 generation histopathology at PND 70.a

POF: preovulatory follicle; PND: postnatal day; SD: standard deviation.
a p and F values in the rows from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SD.
bSignificant at p < 0.001 versus control.
cSignificant at p < 0.05 versus control.

Photomicrograph (×40) of cross section of seminiferous tubules of male rats in F1 generation, with both parents receiving doses of furan: (a) Control group shows regularly arranged tubules with thick epithelium lumen filled with spermatids; (b) 1 mg kg−1 group shows normal epithelial height, lumen filled with mature spermatozoa; (c) 2.5 mg kg−1 group shows normal morphology, lumen filled with immature spermatozoa; (d) 5 mg kg−1 group shows increased lumen diameter and interstitial spaces; (e) 10 mg kg−1 group shows thin degenerated epithelium with empty lumen and interstitial spaces. L: lumen; E: epithelium; IS: interstitial spaces.

Photomicrograph (40×) showing different stages of folliculogenesis in ovarian tissues of rats in F1 generation, with both parents receiving doses of furan: (a) control group shows all the stages of folliculogenesis; (b) 1 mg kg−1 group shows no significant effect on number of follicles as compared to control group; (c) 2.5 mg kg−1 group represents no significant difference except a slight decrease in number of ovarian follicles; (d) 5 mg kg−1 group shows reduction in number of ovarian follicles; (e) 10 mg kg−1 group indicates large number of Cys Fs in ovary. Pr F: primordial follicle; PF: primary follicle; SF: secondary follicle; AF: antral follicle; Atr F: atretic follicle; Cys F: cystic follicle; CL: corpus luteum.
Results of F1 generation hormonal analysis at PND 70
There were no statistically significant difference observed in FSH and LH concentration in 10 mg kg−1 furan-treated male groups compared with the control group, while the plasma T level was significantly (p = 0.03) reduced in 10 mg kg−1 treated group (Table 7). Similarly, in female rats, plasma concentration of estrogen was reduced significantly (p = 0.05), while the levels of progesterone, LH, and FSH were not statistically different in 10 mg kg−1 treated groups when compared with control animals (Table 7).
Results of F1 generation hormonal profile at PND 70.a

PND: postnatal day; LH: luteinizing hormone; FSH: follicle-stimulating hormone; SD: standard deviation.
a p and F values in the rows from analysis of variance with complete randomized designs followed by Tukey’s post hoc show a pairwise comparison of control with furan-treated groups. Mean with different superscripts are significantly different (p < 0.05) in the rows compared to control with other treated groups. Values are presented as mean ± SD.
bSignificant at p < 0.05 versus control.
cSignificant at p < 0.001 versus control.
Discussion
After fertilization, complex physiological and endocrine mechanisms are involved, which control the embryonic development. Any alteration in these mechanisms can cause abnormal growth and functional changes in the life of an adult organism. It has been previously observed that, when parents were exposed to environmental toxicants, it may cause developmental, behavioral, and hormonal changes in the body of offspring. 23,30,36 In the present study, the potential effects of furan on rats were evaluated in an EOGRTS and demonstrated that long-term exposure of furan to parents resulted in parental F0 toxicity and F1 generation toxicity in both sexes at higher doses (5 mg kg−1 and 10 mg kg−1). Normal development of the male and female reproductive systems depends upon the proper functioning of testis, ovaries, pituitary, and hypothalamus. The hormonal secretions from pituitary and gonads are necessary for the development of reproductive systems.
Endocrine regulation involves the secretion of gonadotropin-releasing hormone from hypothalamus that further stimulates the secretion of LH and FSH from anterior pituitary, all these hormones are necessary for the normal functions of reproduction (spermatogenesis and folliculogenesis). 37 –39 It has been previously observed that exposure to some endocrine disrupting chemicals (EDCs) can lead to disturbance of the several normal body functions. In rodents during the development period, exposure to EDCs at higher concentrations disturbed the reproductive functions in adulthood. 40 Similarly, human exposure to EDCs brings changes in reproductive functions, and genital abnormalities are evident in last half century. 41,42 Exposure to environmental toxicants, for example, acrylamide, bisphenol, tetrabromobisphenol A, and imidacloprid, brought variations in growth and endocrine functions and thus affected F1 and F2 generations. 23,30,36,43
It has been previously reported that exposure of furan caused reproductive toxicity in several animal studies. 5,16 Guo et al. reported that young men prenatally exposed to dioxins and furan have a high incidence of abnormal sperm morphology, motility, and reduced ability to penetrate hamster ovary. 22 Furan was also investigated to induce chromosomal aberrations and sister chromatid exchanges in Chinese hamster ovary cells and Chinese hamster V79-derived cell line. 13,44 Previously, it has been observed that different concentrations of furan induced toxic effects on the endocrine systems of animals and humans. 5,12,16
In current investigation, a significant change in F0 parental weight gain of males (prebreed period) and female (prebreed period, gestation, and lactation) was observed after exposure with high doses of 5 mg kg−1 and 10 mg kg−1 of furan. Similar findings previously reported that reduction in weight gain of animals was observed when exposed to environmental toxicants such as acrylamide, furan, and bisphenol. 6,23,30 The toxicity of furan at parental stage (F0) in current study is similar to the previous findings. In animals, EDCs has been widely studied, and different physical parameters have been estimated and standardized to confirm the toxicological effects of EDCs on development of different organs by physical examination. Study of these parameters reveals the state of development of organs within the body. Subchronic furan and acrylamide exposure resulted in reduced seminal vesicle and testicular weight, along with reduced serum level of T, LH, FSH, and prolactin. 6,19,45 The bw is the most sensitive indicator of adverse effect of xenobiotics. It is notable that observing the bw is important for interpretation of reproductive effects of toxins. 46 The results of the current study showed the decrease in prebreed bw gain of F0 (parents). Explanation of the reduced bw may be due to growth and protein deficiencies because of malnutrition during development. 47,48 It may also be due to excessive breakdown of tissue and plasma protein. 49 In current study, weight gain in females during gestation period was decreased, while increase in bw gain was noticed during lactating period. This might be due to increased food consumption during gestation and lactation period. Metabolism of females increases during gestation and lactation period, and females gain additional calories during this period due to fast metabolism. 50 In our study, reduction in gestational weight gain may be due to the reason that lactation makes greater demand of mother’s body than pregnancy does. 43 Another possible reason is low number of implantation sites during pregnancy.
In this current study, head tilt and foot splay were also observed in F0 parents after exposure with various doses of furan. This is related to the neurotoxic effects of furan. Similar results were obtained previously in which acrylamide exposure caused head tilt and foot splay in high treatment groups, and results were related to the degeneration of brain monoaminergic system. 32 Spindle fiber proteins present in peripheral axons play an important role in anterograde axonal transport. It was reported that acrylamide interfered with these proteins and caused hind limb weakness like foot splay, reduce grip strength, and so on. 51 The effects observed in present study must be related to neurotoxic and reproductive toxic nature of furan.
In current study, reduction in number of pregnant F0 female was observed in higher dose treated groups. The possible reason for reduced pregnancy may be that furan generates ROS in sperm cells and causes sperm DNA damage. 12 Another reason is mounting behavior/libido failure. 23 Furan treatment might affect the rat’s limbs grip, disturbing mounting behavior, so that they cannot penetrate sperm into the uterus of females resulting in reduced pregnancy. Previously, it is reported that human exposure to furan revealed that furan affected lymphocytes and sperm cells by producing ROS. 12
AGD development is related to the reproductive development. The distance between anus and genitalia of an organism reveals the degree of development of the reproductive system of the animal. 52 In current study, there was no significant difference observed in the male and female AGD and male NR in the furan-exposed group as compared to the control group. In the last 20 years, during critical period of sexual differentiation, the assessment of NR and AGD has been considered as important marker of altered androgen exposure. 53,54 These parameters are used to study the hazardous effects of toxins on reproductive toxicity in adulthood. 54,55 According to OECD protocols, these parameters are considered mandatory for testing of newly synthesized chemicals. 56
In our study, we observed the reduced fertility rate/number of pups in F1 generation in high-dose treatment group (10 mg kg−1). It is well known that the total number of live pups and litter size reduction is correlated with the reduction in implantation sites. 57 Similarly, chemical exposure suppresses the LH surge or delays preovulatory LH surge, which is correlated with delayed or blocked ovulation. 57,58 This continuous suppression of LH and delayed ovulation is associated with the significant reduction in litter size and number of implantation sites that affect number of live pups, fertility, and mating process. Although the AGD in males and females were not significantly affected, a mild decrease was observed. Reduced weight in pups was observed only in PND 0 and PND 4 when the pups depend on their mother for milk. This may be due to the furan exposure to the mother may cause detrimental effects in term of milk quality and quantity and may have potential toxic effects on the growth retardation of the offspring. No postnatal mortality was observed in F1 generation. Aforementioned data on the F0 rats are directly related with reported studies on acrylamide and furan reproductive toxicity. 5,6,16 However, one of the main purposes for EOGRTS is to provide the awareness about chemical toxicity in parents, offspring, and adulthood life stage.
It was noticed that puberty onset in males and females in F1 generation was not affected significantly in the current study. Similarly, no difference was observed in the bw gain on PND 70, but testicular and prostate weight were reduced dose dependently. Previous study has stated that prostrate and testicular weights depend on sex hormones as prostrate response to androgen antagonists is well established 59 and testicular dependency on androgen has also been shown. 60 Similarly, in this study, paired ovarian weight was also reduced in F1 generation females dose dependently. It means that exposure to furan during sensitive development period of life badly effects the accessory organ weight. Similarly, in an early study, decrease in ovarian weight was also recorded. 61
The hormonal concentration, that is, T, LH, and FSH in males on PND 70, is slightly affected in F1 generation. In current finding, T and LH levels were reduced in male rats because of the furan toxicity on reproductive system. DSP was also reduced in our current study because of low T concentration. T is essential for maintaining all functions of accessory sex glands, 16 and the lack of T disrupts spermatogenesis. 62 Similarly, female’s estrogen level was reduced in high treatment group, but progesterone, LH, and FSH concentrations in adult animals were not affected. In addition, estrous cycle was disturbed with increase in estrous cycle length of F1 animals at higher doses. Our findings are supported by previous studies in which premenarcheal exposure of women with 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) was associated with longer menstrual cycles. 63 In mammalian reproduction, FSH is main hormone; it play a vital role in gonadal development, maturation at puberty, and gamete production during the fertile phase of life. 64 Estrogen and FSH concentration is important for female secondary sexual characteristics, development, and proliferation of the ovarian follicles, and it plays a significantly important role in women’s health. 64,65 Significant changes in ovarian and testicular architecture were seen in F1 generation as examined by histological analysis. Spermatogenesis and folliculogenesis are dependent on endocrine regulation of sex hormones. 37,38,66 Results from the present study confirmed that parental furan exposure generated alterations in the hypothalamic pituitary testicular and ovarian axis that altered folliculogenesis and spermatogenesis in F1 generation. The parental furan exposure disturbed the levels of T in male rats and estrogen level in female rats in F1 generation. The testicular and ovarian weights were reduced in F1 generation at PND 70 with decreased DSP and disturbed estrous cyclicity in groups treated with high dose (10 mg kg−1) of furan. Histopathological changes were also observed in testis and ovaries of F1 generation, whose parents were previously exposed with higher (10 mg kg−1) doses of furan. Our results are in line with previous findings on furan exposure and highlight that parental exposure to furan can cause adverse effects on the F1 generation.
Conclusion
Our study demonstrates for the first time that extended one-generation exposure of rats to furan at different concentrations impairs their hypothalamic pituitary testicular and ovarian axis, which over long-term exposure might affect generations. As we predict an adverse effect of the food-based toxicant furan to mammals, long-term and generation studies that include the detection of altered folliculogenesis and spermatogenesis are warranted. The toxicological profile of furan presented in this study supports the identification of furan as an endocrine disruptor of concern for human health. To enable environmental risk assessment, more comprehensive molecular-based studies are recommended to further clarify the health hazardous effects of furan in living organisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was entirely funded by the Reproductive Physiology Laboratory, Department of Animal Sciences, Quaid-i-Azam University, Islamabad, Pakistan.

