Abstract
Furan is a heterocyclic organic compound formed during heat treatment for processing and preservation of various types of food. Rodent studies have previously shown that furan is a hepatocarcinogen. Those studies were conducted over a high dose range, which induced tumors at nearly 100% incidence at all doses. This ninety-day gavage study in mice was conducted to extend the dose to a lower range (0.0, 0.03, 0.12, 0.5, 2.0, and 8.0 mg/kg body weight [bw] per day) to identify a no-observed adverse effect level for hepatotoxicity and to characterize non-neoplastic effects, including those affecting clinical biochemistry, hematology, tissue morphology, and histopathology. The liver was the primary target organ with dose-dependent toxicity. Liver weights were increased at the 8.0 mg/kg bw dose in females only. Levels of the serum enzyme alanine transaminase, representative of liver damage, were increased three-fold at the highest dose. Histological changes in the liver were observed at 2.0 and 8.0 mg/kg bw in both sexes. Although clinical parameters were also altered for the kidney, these differences were not accompanied by histological changes. Based on these clinical biochemical and histological changes, a no-observed adverse effect level of 0.12 mg/kg bw per day of furan in mice is suggested.
Introduction
Worldwide, more than one thousand food samples have been analyzed for furan (Wegener and López-Sánchez 2010). Furan has been detected in cigarette smoke, wood smoke, and engine exhaust, as well as in heat-processed food such as canned fruits and vegetables (Bakhiya and Appel 2010; Becalski and Seaman 2005; Becalski et al. 2010; FDA 2004; Liu and Tsai 2010; Maga 1979). It is found in a wide range of food, including some thermally processed food, which includes levels as high as 170–5,000 ng/g (EFSA 2006; Liu and Tsai 2010; Maga 1979). Mean levels of furan ranged from 6 to 2,272 ng/g. The highest levels have been detected in coffee, baby food, cereal products, meat products, soups, and sauces (Bakhiya and Appel 2010; Becalski and Seaman 2005; Becalski et al. 2010; Jestoi et al. 2009; Liu and Tsai 2010). Thermally processed fruit juice contained furan at levels ranging from below 2.0 ng/g to 31 ng/g (Becalski et al. 2010; EFSA 2006; Maga 1979). Fruit juices contain high amounts of free sugars and ascorbic acid, both potential precursors of furan through thermal decomposition. In baby food, an average level of 25 ng/g was reported; this finding is of particular interest, as it can be the sole diet of infants (Arisseto et al. 2010; Jestoi et al. 2009; Lachenmeier et al. 2009; Owczarek-Fendor et al. 2010). The long-term effects of furan on the health of children is unknown, but the presence of furan in baby food is a concern because of the greater sensitivity of infants to carcinogens, as well as the larger amounts (relative to body weight) of certain food items (such as apple juice) that they consume (Bakhiya and Appel 2010; Becalski et al. 2010; Jestoi et al. 2009; Morehouse et al. 2008).
Furan can form from various precursors naturally present in food via complex steps, which might involve oxidation or the Maillard reaction (Becalski and Seaman 2005; Limacher et al. 2007; Limacher et al. 2008; Maerk et al. 2006; Perez Locas and Yaylayan 2004). The occurrence, analysis, and formation of furan were recently reviewed (Crews and Castle 2007). Several precursor classes have been suggested to release furan upon thermal treatment, such as ascorbic acid and related compounds, Maillard reaction systems (reducing sugars, specific amino acids), lipids comprising unsaturated fatty acids and the corresponding triglycerides, as well as carotenes and organic acids (Becalski and Seaman 2005; Fan 2005; Fan et al. 2008; Limacher et al. 2007; Maerk et al. 2006; Perez and Yaylayan 2004). The variety of precursors is not surprising, because furan is a rather stable reaction product that may be generated by the degradation and/or recombination of smaller fragments.
Currently, there are no studies linking furan exposure to adverse events in humans. Although it has long been known that furan is a constituent of food (Maga 1979), data on exposure through food are limited. Dietary exposure modeling for the U.S. population, conducted by the United States Food and Drug Administration’s Center for Food Safety and Applied Nutrition and based on measured concentrations of furan in numerous food items (Morehouse et al. 2008), predicts a mean consumption of 0.26 μg furan/kg bw/d for individuals over two years of age, 0.41 μg/kg bw/d for children between birth and one year of age, and 0.9 μg/kg bw/d for infants receiving formula as their primary source of nutrition. The widespread exposure to furan through the diet is a public health concern because of the demonstrated potential for hepatotoxicity and carcinogenicity observed in two rodent species. In particular, there was a high incidence of cholangiocarcinoma in male and female Fischer 344 rats at all doses tested in two-year NTP studies (1993), in which the lowest dose studied was 2.0 mg/kg bw/day. This finding is of concern, since cholangiocarcinomas also occur in humans and are associated with a very poor prognosis.
There appears to be a relatively small difference between human exposure levels and the doses in experimental animals that produce carcinogenic effects, presenting a potential risk to human health (Bakhiya and Appel 2010; Becalski et al. 2010; Heppner and Schlatter 2007; Jestoi et al. 2009). However, data on furan toxicity in rodents are insufficient to perform a risk assessment, since all doses administered previously to rats and mice were carcinogenic, and no no-observed adverse effect level (NOAEL) could be established in the National Toxicology Program (1993) study. Our earlier study on the Fischer rat (Gill et al. 2010) was designed to address this problem and establish a NOAEL in a subchronic study. Based on our findings, we proposed a NOAEL of 0.03 mg/kg bw in the rat model. In the present study, based on the OECD Guidance, we have examined the NOAEL for furan in a second animal model, the B6C3F1 mouse, using the same treatment protocol we used previously in the rat model.
Materials and Methods
Test Compound and Dosing Solutions
Furan (Sigma-Aldrich Co., Oakville, ON, Canada) was mixed with Mazola corn oil to final concentrations of 0.03, 0.12, 0.5, 2.0, and 8.0 mg/mL. Each dose was prepared separately on a volume-to-weight (v:w) ratio. Chilled corn oil was weighed to the nearest milligram in a conical flask. Chilled furan solution was drawn up in a Hamilton syringe, measured to the nearest microliter, injected into the corn oil, and mixed using a magnetic stir bar. Dosing solutions were dispensed into brown glass vials and capped with plastic closures adapted with silicon septa. Dosing solutions were stored in a refrigerator at 4°C. Fresh solutions were prepared every fourteen days. Previous studies have demonstrated the stability of furan dosing formulations for at least fourteen days under these conditions (NTP 1993).
Animal Studies
Thirty-day-old male and female B6C3F1 mice (weighing between 12 and 18 g) were obtained from Charles River Laboratories Inc. (St.-Constant, QC, Canada) and acclimatized for a period of twelve days before treatment began. Animal handling and treatment procedures were conducted according to the Guidelines of the Canadian Council of Animal Care and were approved by the Health Canada Animal Care Committee (Ottawa, ON, Canada). Mice were identified by tattoo markings on the tail. Animals had ad libitum access to Purina Rodent Chow 5001 (Agribands Purina Canada, East Strathroy, ON, Canada) and city tap water. Male mice were housed singly, whereas females were housed in pairs. The temperature and humidity of the room were targeted for 18°C–26°C and 30–70%, respectively, and continuously monitored. Animals were observed for signs of illness twice daily during weekdays and once daily on weekends and holidays. Sentinel animals for health surveillance were housed in the same room as study animals and remained disease free throughout the study. Groups of sixteen female and sixteen male animals were gavaged with furan five days/week (as in the protocol of NTP 1993) over a ninety-day period such that they received a total dose of 0.0, 0.03, 0.12, 0.5, 2.0, or 8.0 mg/kg body weight (bw) per day. These animals were weighed daily on weekdays to calculate the adjusted dose per animal prior to gavage. Food consumption was measured on a weekly basis.
Organ Weights, Gross Necropsy, and Histopathology
At the end of the study, animals were killed by exsanguination via the abdominal aorta under isoflurane anaesthesia, and a complete gross examination was performed on each mouse. Liver, kidneys, heart, spleen, thymus, adrenals, brain, testes, prostate, seminal vesicles, epididymes, ovaries, and uterus were weighed upon removal. Paired organs such as the kidneys, testes, and ovaries were weighed separately. A subset for each sex (n = 5) was collected for histopathology analysis of liver, kidneys, spleen, thymus, uterus, ovaries, mammary glands, seminal vesicles, thyroid, lungs, stomach, pancreas, heart, adrenals, brain, testes, prostate, epididymes, colon, gall bladder, and small and large intestines. A second subset (n = 11) was collected for molecular and protein analysis, which is currently under investigation.
From the histopathology subsets, the testes were immersion fixed in Bouin’s solution. Other tissues were immersion fixed in 10% neutral buffered formalin. Representative sections from the left, median, right posterior, right anterior, and caudate lobes of the liver were identified for histological preparation. After fixation, the tissues were routinely embedded in paraffin and 5-μm sections were cut. Prepared slides were stained with Mayer’s hematoxylin and eosin (H&E) and examined by light microscopy. Liver sections from all dose groups were examined; however, for the kidney, only the 8.0 mg/kg bw–treated animals and control groups were examined. Photographs were taken on an Axiophot Zeiss microscope (Germany) with a digital camera linked to an image analysis system (Progress camera, ROCHE image analysis and archiving system).
Analysis of Serum Clinical Biochemistry
At the time of sacrifice under isoflurane anaesthesia, blood from the abdominal aorta was collected in SST Vacutainer tubes (Becton-Dickinson, Franklin Lakes, NJ) and allowed to clot at room temperature. Serum was separated by centrifugation at 3,000 × g and stored at −80°C until analysis. Clinical biochemistry was performed using a Beckman Synchron CX5 Clinical Biochemistry System (Beckman Instrument Canada Inc., Mississauga, ON, Canada). The parameters measured were serum amylase (AMY, U/L), albumin (g/L), globulin (g/L), albumin:globulin ratio, alkaline phosphatase (ALP, U/L), alanine transaminase (ALT, U/L), aspartate transaminase (AST, U/L), γ-glutamyl transpeptidase (U/L), bicarbonate (BIC, mmol/L), conjugated bilirubin (mmol/L), total bilirubin (mmol/L), calcium (mmol/L), chloride (mmol/L), cholesterol (mmol/L), creatinine (CRE, mmol/L), creatine kinase (U/L), glucose (mmol/L), lipase (U/L), magnesium (mmol/L), phosphorus (P, mmol/L), potassium (mmol/L), sodium (mmol/L), sodium:potassium ratio, total protein (g/L), triglycerides (mmol/L), uric acid (UA, mmol/L), and urea (mmol/L).
Hematology and Flow Cytometry
Blood was collected in EDTA tubes and analyzed using a Coulter Counter Model S-PLUS IV system (Coulter Electronics, Hialeah, FL). The following parameters were measured: red blood cell count, hematocrit, hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin concentration, red blood cell distribution width, platelet counts, mean platelet volume, and white blood cell counts including eosinophils, lymphocytes, monocytes, and neutrophils.
Statistical Analysis
Body weight, food consumption, clinical biochemistry, hematology, immunology, and organ weights are expressed as mean ± standard deviation. Statistical comparisons of control mice and furan-treated mice were performed by one-way analysis of variance followed by the Tukey multiple comparison test. Linear trend analysis was also performed. A value of p < .05 was considered statistically significant.
Results
In all study groups, the animals appeared healthy for the duration of the study. There appeared to be no changes in behavior, posture, or gait in either sex.
Body Weight, Organ Weights, and Food Consumption
Overall, body weights and weight gains were comparable for the control and treatment groups. Food consumption was also similar between groups (data not shown). At the highest dose (8.0 mg/kg bw), the absolute and relative liver weights of females increased significantly, by 14% (p < .01), but there were no changes in males (Table 1 ). No other organs showed significant changes in absolute weights.
Absolute liver weights (g) in male and female mice.
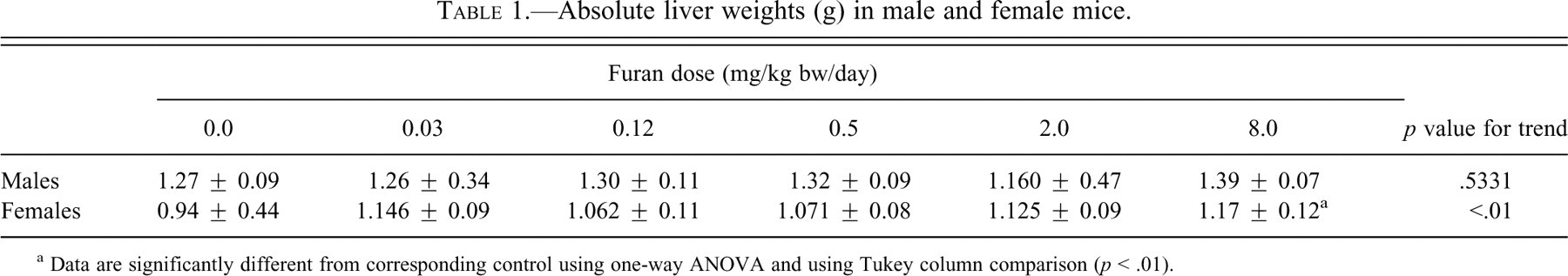
a Data are significantly different from corresponding control using one-way ANOVA and using Tukey column comparison (p < .01).
Clinical Biochemistry
Liver Markers
Levels of the enzyme activities of ALT, AST, and ALP in the serum can be indicative of liver injury. Alanine transaminase activity was significantly increased in males and females at 8.0 mg/kg bw (p < .01; Table 2 ). Activities of AST and ALP were not altered in either males or females (not shown).
Serum ALT (U/L) marker in male and female mice
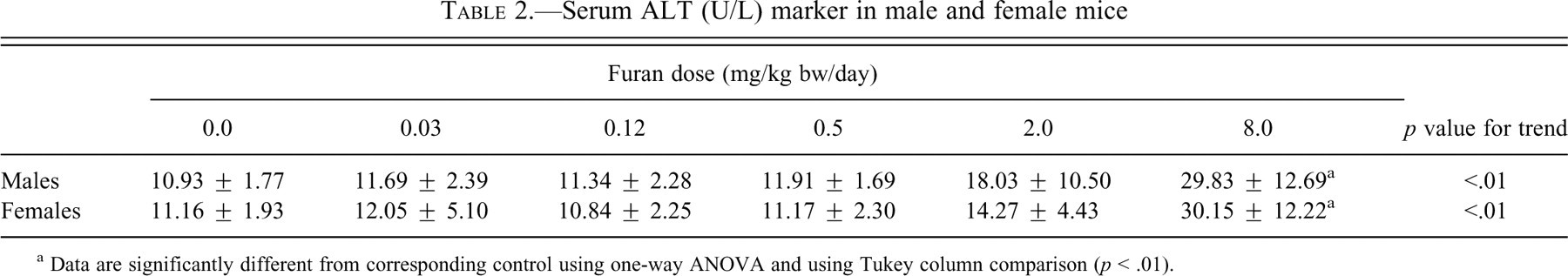
a Data are significantly different from corresponding control using one-way ANOVA and using Tukey column comparison (p < .01).
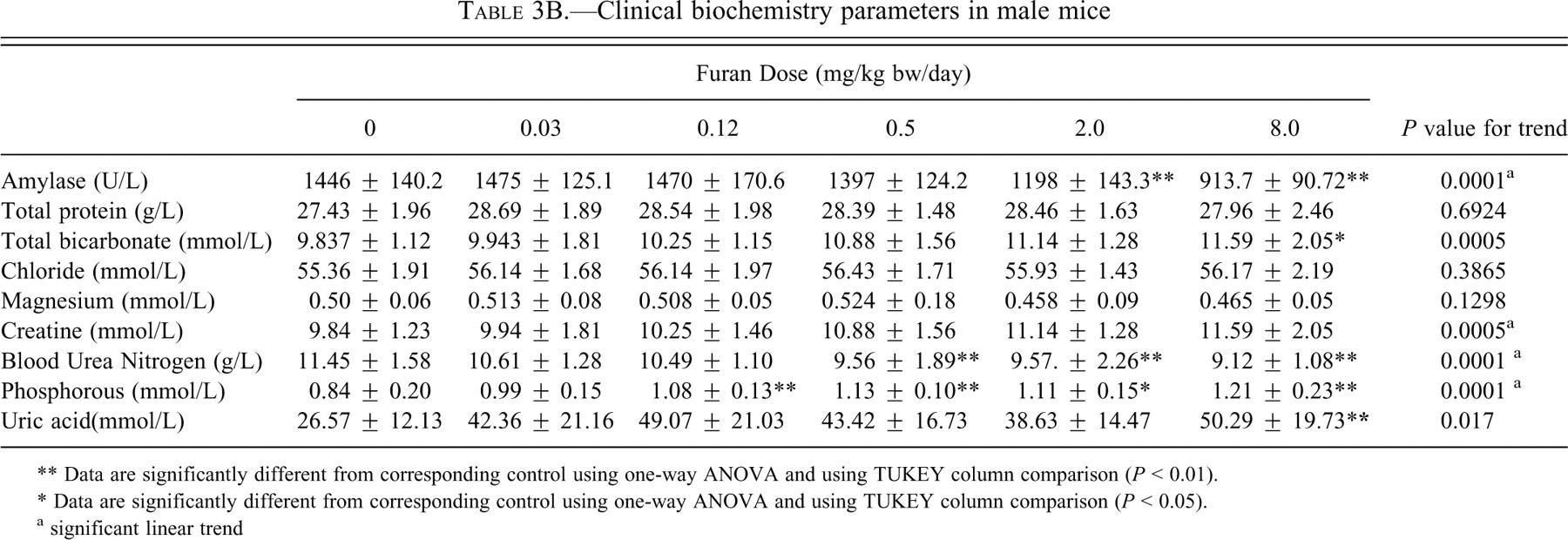
Serum AMY demonstrated a significant decreasing trend and was significantly decreased (p < .01) in males and females at 2.0 and 8.0 mg/kg bw (p < .01; Tables 3A and 3B).
Clinical biochemistry parameters in female mice.
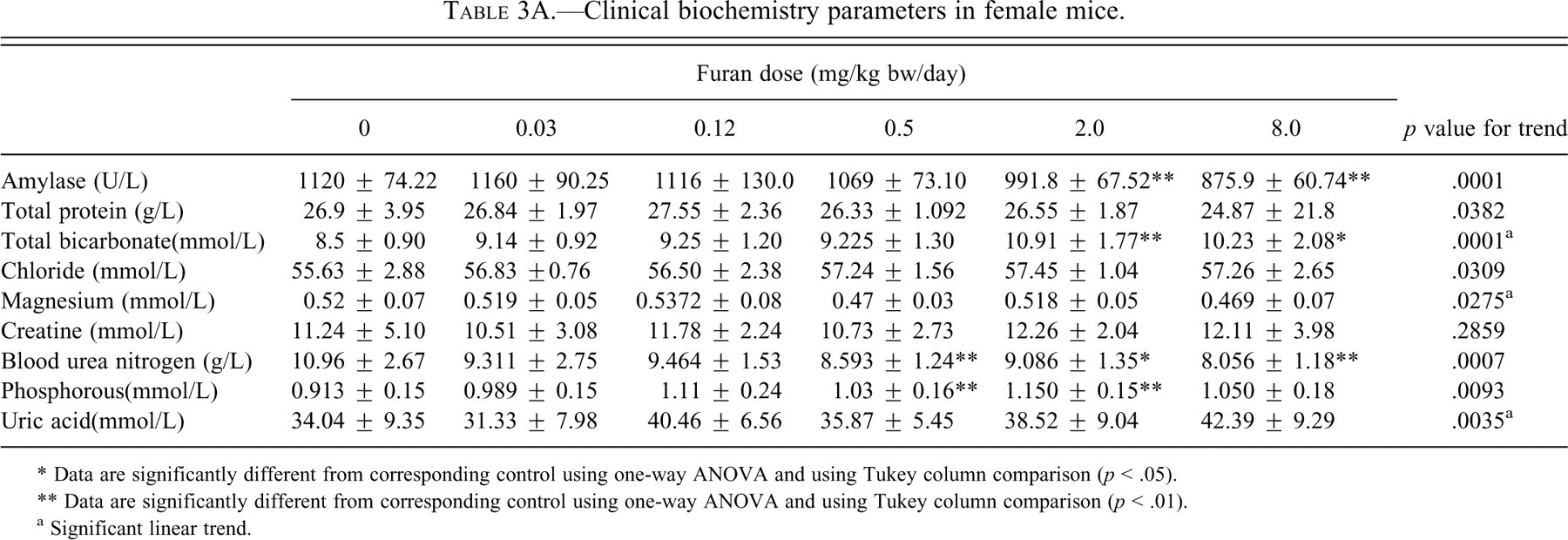
* Data are significantly different from corresponding control using one-way ANOVA and using Tukey column comparison (p < .05).
** Data are significantly different from corresponding control using one-way ANOVA and using Tukey column comparison (p < .01).
a Significant linear trend.
Clinical biochemistry parameters in male mice
** Data are significantly different from corresponding control using one-way ANOVA and using TUKEY column comparison (P < 0.01).
* Data are significantly different from corresponding control using one-way ANOVA and using TUKEY column comparison (P < 0.05).
a significant linear trend
No significant changes were observed in total protein. Chloride levels were not affected in either sex. Magnesium showed a significant decreasing trend in females, whereas there were no changes in males.
Renal Function
Renal function was also assessed using several serum biomarkers indicative of renal damage (Tables 3A and 3B). Some of these biomarkers showed changes, including BUN, CRE, P, BIC, and UA. Blood urea nitrogen was significantly (p < .01) decreased simliarily in both sexes starting with the 0.5 mg/kg bw group of furan. Creatinine was not affected in females, whereas in males it showed an increased linear trend. Phosphorus levels were significantly increased linearly starting at a dose of 0.12 mg/kg bw (p < .01) in males, but no clear trend is evident in females. Magnesium levels were not affected in males, whereas they showed a significant decreasing trend in females. Uric acid levels were significantly increased, particularly in males, where they rose by 92% at the highest dose (p < .01) compared with an increase of only 21% in females. Despite the changes in serum markers for kidney damage, there were no significant changes in either absolute or relative kidney weights and no histological changes were observed.
Hematology Parameters
In males and females, there were no significant changes in hematological parameters including erythrocyte and leucocyte counts, hemolysis, icterus, and hemoglobin levels (data not shown). No changes in platelets were observed.
Histological Changes in the Liver
Liver sections from animals in the histopathology subset (n = 5) per dose group consisted of the left, median, right, and caudate lobes. These sections were stained with H&E and examined by light microscopy.
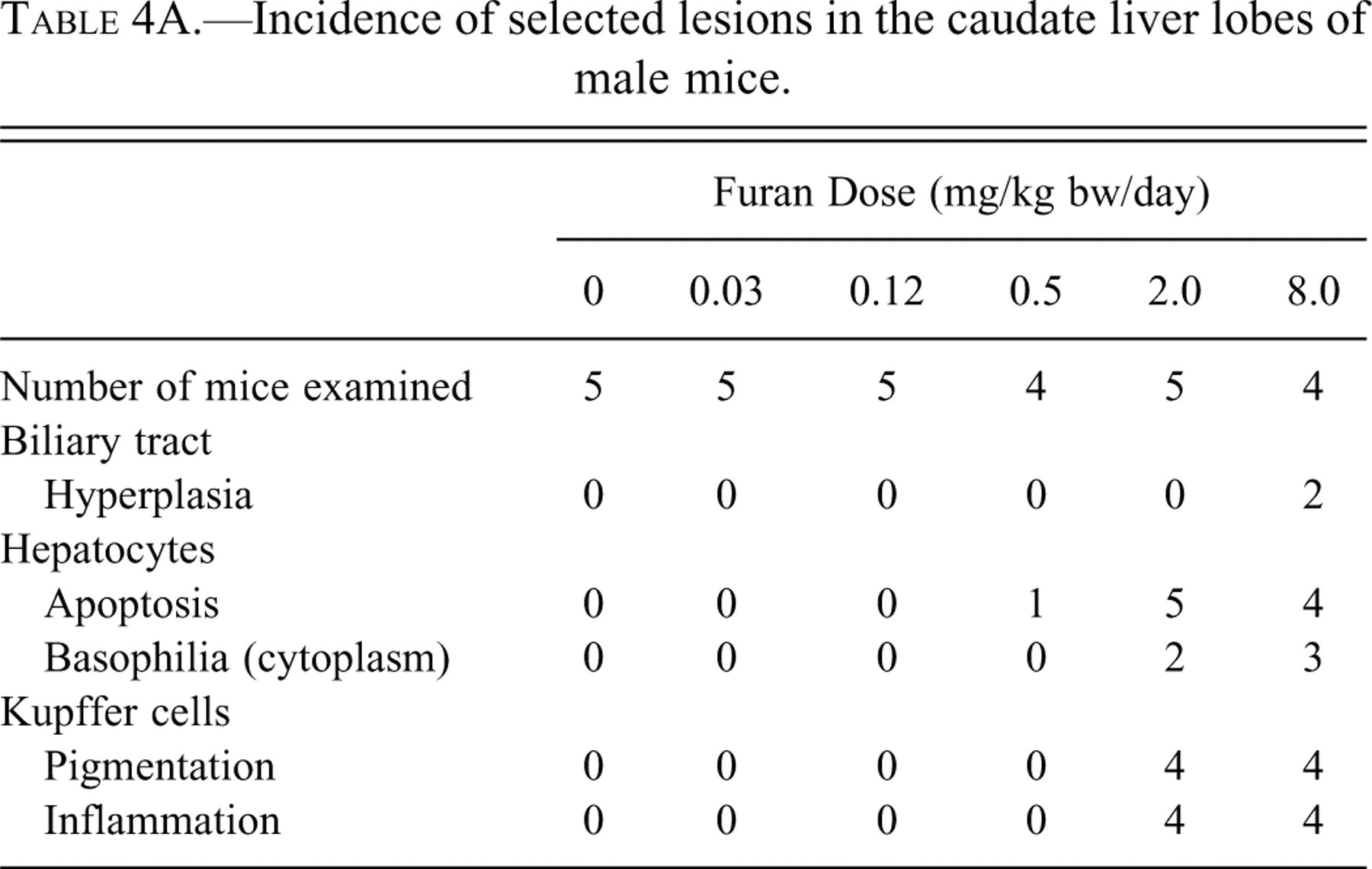
Microscopic lesions were detected in the caudate lobes of one male at 0.5 mg/kg bw and in both males and females in the 2.0 and 8.0 mg/kg bw groups. These lesions were restricted to the subcapsular region. Apoptotic hepatocytes, Kupffer cells with yellow/green pigment in the cytoplasm, oval cell hyperplasia, and inflammatory infiltrate were all observed (Figure 1 ). These changes were graded mild to focally moderate, and animals in the 8.0 mg/kg bw group were slightly more affected than those dosed with 2.0 mg/kg bw of furan. In addition, two females at 8.0 mg/kg bw and one group of males at 8.0 mg/kg bw had a mild, focal biliary tract hyperplasia (Tables 4A and 4B). In some mice, there was a border one to three cells thick consisting of mildly enlarged hepatocytes with slightly basophilic cytoplasm and prominent nucleoli between these subcapsular lesions and the deeper parenchyma. This border was detected in two males and two females in the 2.0 mg/kg bw group and three males and three females dosed with 8.0 mg/kg bw of furan.
Incidence of selected lesions in the caudate liver lobes of male mice.
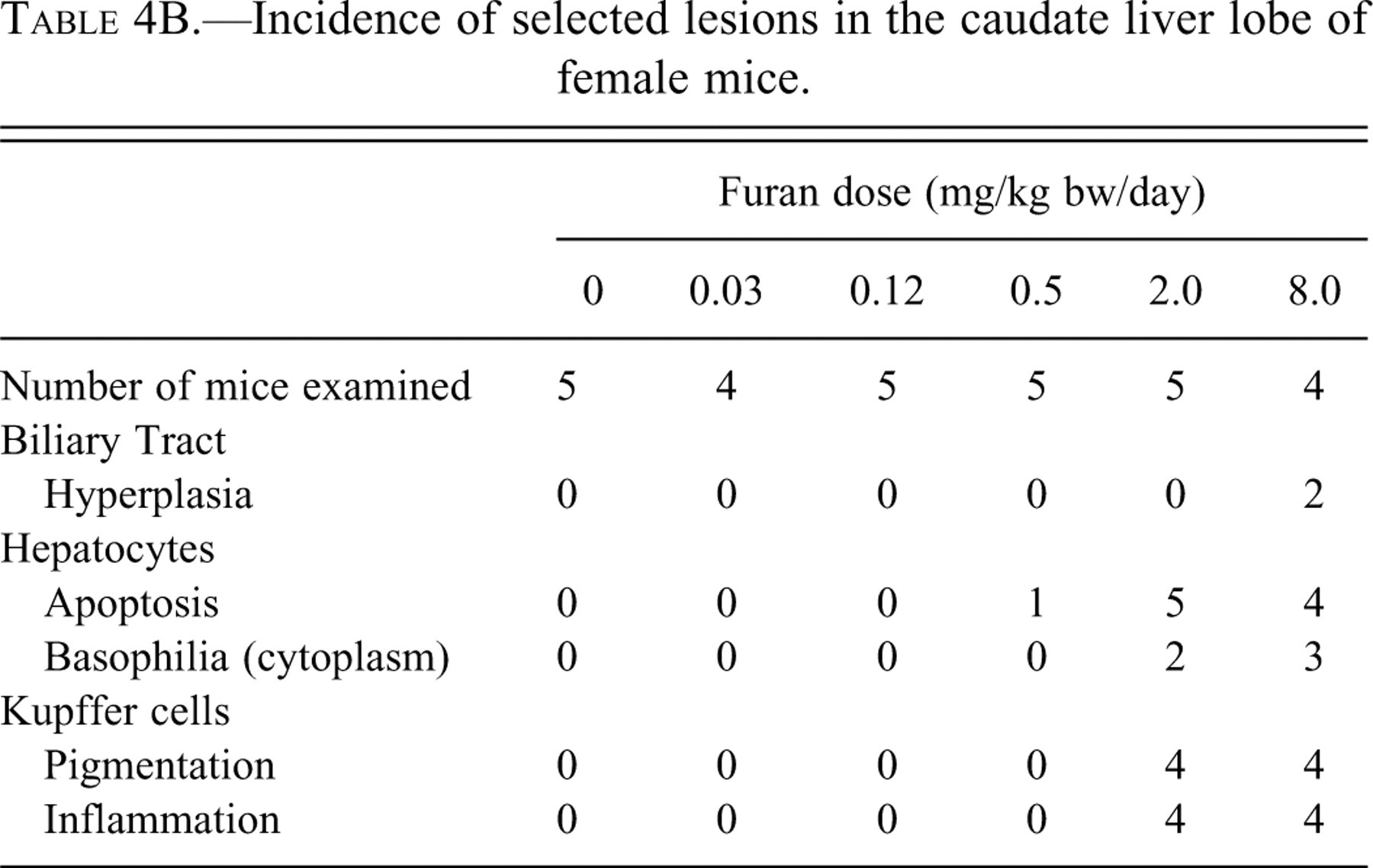
Incidence of selected lesions in the caudate liver lobe of female mice.
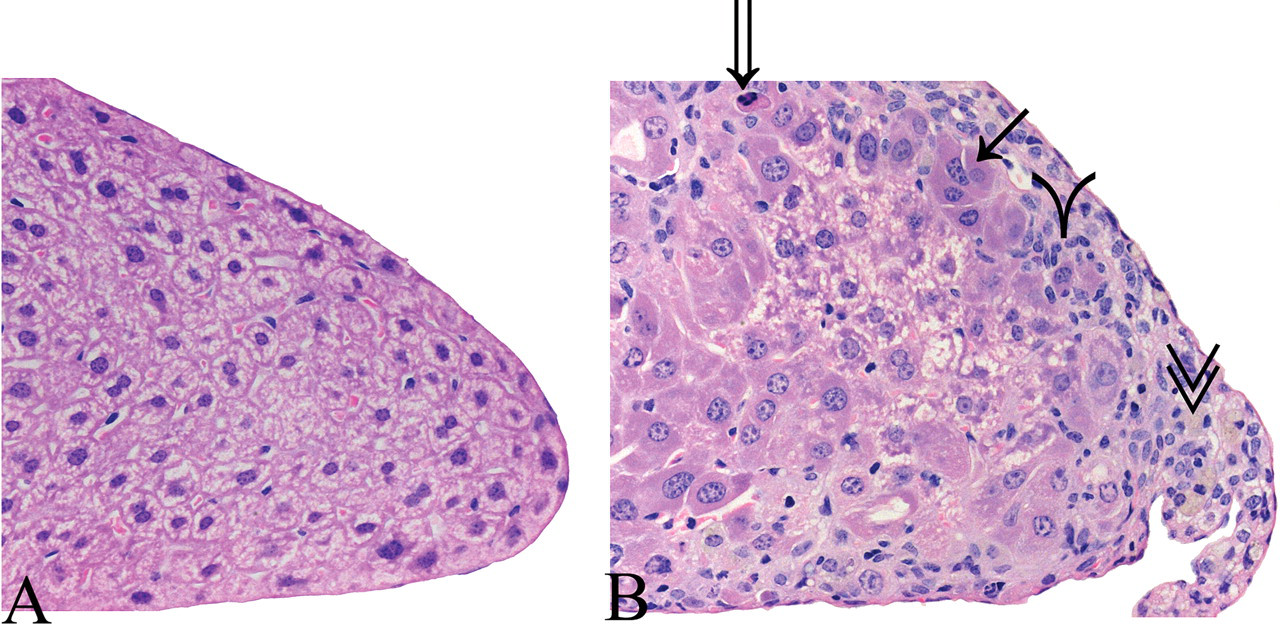
Photomicrographs of the furan liver fixed in 10% neutral buffered formalin and stained with hematoxylin and eosin. Image A (40×): margin of caudate lobe from male control mouse. Image B (40×): liver of male from 8.0 mg/kg bw dose group showing margin of caudate lobe with subcapsular lesions, Kupffer cell with pigment in cytoplasm (double arrowheads); apoptotic hepatocyte (open arrow); a mix of inflammatory and oval cells (single arrowhead); and hepatocytes with basophilic cytoplasm between subcapsular lesion and normal parenchyma (solid arrow).
In the left lobe of the liver (Tables 5A and 5B), subcapsular hepatocyte apoptosis and/or Kupffer cells with pigment and/or inflammatory infiltrate were also detected in four of four male mice and three of four female mice dosed with 8.0 mg/kg bw of furan. These same changes were observed in males (three of five mice) and females (two of five mice) dosed with 2.0 mg/kg bw of furan.
Incidence of selected subcapsular lesions in the other liver lobes of male mice.
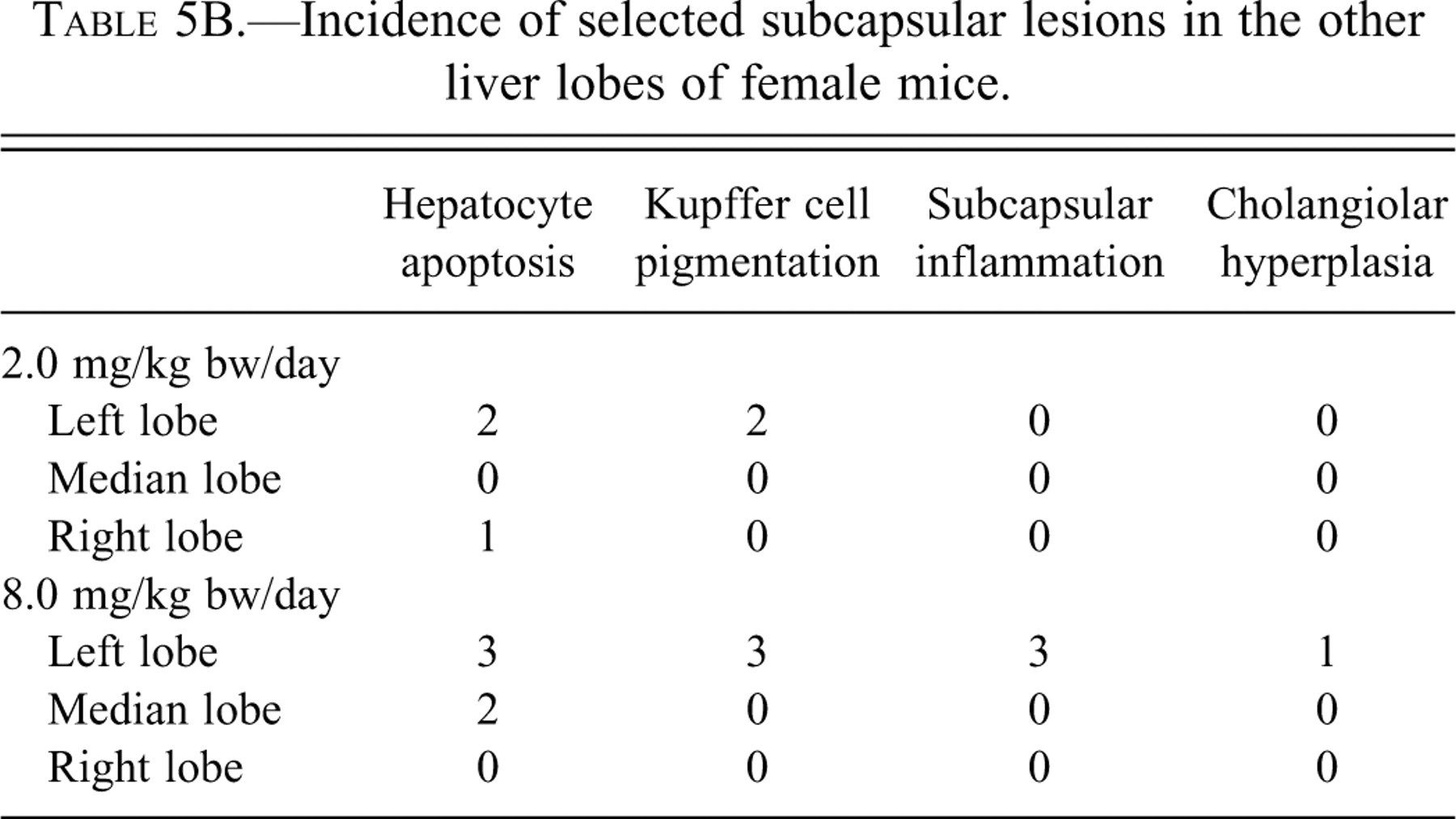
Incidence of selected subcapsular lesions in the other liver lobes of female mice.
In the right lobe (Tables 5A and 5B), subcapsular apoptotic hepatocytes were also rarely detected, in one animal administered 2.0 mg/kg bw of furan in both males and females. In the median lobe, this was the finding in two females administered 8.0 mg/kg bw of furan. Also, one female mouse at the dose of 8.0 mg/kg bw of furan had very mild, focal biliary tract hyperplasia.
An increased incidence of randomly distributed apoptotic hepatocytes in the parenchyma, as opposed to immediately subcapsular, subjectively rated as occasional was detected in three of the females in the 8.0 mg/kg bw group. This increase occurred most consistently in the median lobe and was not detected in males of the same dose group. This incidence was rated as rare in control animals.
Histological Changes in Other Organs
Histological sections of other tissues in the control group and the 8.0 mg/kg bw group were formalin-fixed and stained with H&E to be examined by light microscopy. No changes were observed in the lungs, spleen, thymus, stomach, adrenal, testes, or ovaries.
Conclusion and Discussion
The results of long-term studies in rodents show that the liver is the main target of both furan toxicity and carcinogenicity. In the National Toxicology Program (1993) bioassay in B6C3F1 mice and Fisher 344 rats, multiple non-neoplastic lesions were detected in all treated animals, in addition to benign and malignant tumors. In this subchronic study with B6C3F1 mice, the liver was also the target organ. The changes in the biochemical serum markers indicative of liver damage were supported by gross and histological changes in the liver. These changes are comparable to those reported by other researchers (Elmore and Sirica 1991, 1992, 1993; Maronpot et al. 1989; Maronpot et al. 1991; Moser et al. 2009; NTP 1993; Sirica 1996; Wilson et al. 1992). In the current mouse study, serum ALT levels were notably elevated at 2.0 mg/kg bw and showed a three-fold increase over controls in the 8.0 mg/kg bw group of both sexes. Similar types of changes in ALT levels were observed by Moser et al. (2009), whose dose groups were the same as those in the current study. Alanine transaminase is released following reversible hepatocellular injury or necrosis. The other serum liver-related markers, AST and ALP, were not significantly altered. In our previous study in Fischer 344 rats, all three serum biomarkers were altered, but ALT levels were decreased. Liver weights showed no significant increase at any dose in males but were significantly (p < .01) increased at the highest dose in females. Sex differences in effects on liver weight were also observed in previous studies with furan.
Histological changes were first observed in the 0.5 mg/kg bw dose group and increased in severity at 8.0 mg/kg bw in both males and females, with minimal effect in one out of five mice examined. These lesions were detected in the caudate lobes in the 0.5 mg/kg bw dose group and were restricted to the subcapsular region. Moser et al. (2009) observed that two of ten mice dosed with 0.5 mg furan/kg bw had evidence of hepatic cytotoxicity and that higher doses had a dose-related increased severity and/or incidence of hepatic cytotoxicity. These changes were similar in rats, but much more severe. Hepatic lesions in mice of the high-dose group (8.0 mg/kg bw of furan) were comparable to the lesions in rats dosed with 0.5 mg/kg bw. In rats, the first histological changes were evident at a dose of 0.12 mg/kg bw, and similar changes were seen in mice at 0.5 mg/kg bw (Gill et al. 2010). Apoptotic hepatocytes, Kupffer cells with yellow/green pigment in the cytoplasm, and inflammatory infiltrate were all observed. The changes observed included subcapsular hepatocyte necrosis and inflammation localized on the dorsal margin along visceral surfaces of the liver. As in the rat, the caudate lobes were most severely affected, but changes were also seen in the left and right lobes. In a few animals, lesions were also evident in the margin of the right lateral lobe. A possible explanation for the differential effects of furan on different liver lobes is that they are caused by “streaming” in the portal venous circulation to the liver. A higher rate of blood flow to the caudate lobes via the portal vein relative to flows to the median and left lateral lobes would expose the caudate lobes to higher concentrations of furan (Metzger and Schywalsky 1992). Another possible explanation could be that the physical proximity of these lobes to the stomach-gastrointestinal tract, associated with leaking of furan into the intraperitoneal space, could lead to higher exposure in these lobes. In addition, there could also be intrahepatic lobe differences in the metabolism of furan (Elmore and Sirica 1991).
Serum biochemical biomarkers related to renal damage were significantly altered to a much greater extent in males than females. These biomarkers included BUN, UA, BIC, and CRE. Changes were also seen in some electrolytes: P was significantly increased in males of the four highest dose groups starting at 0.12 mg/kg bw, and no clear trends were observed in females. The magnesium level was not affected in males, whereas it showed a significant decreasing trend in females. These changes in electrolyte levels could be the result of multiple metabolic changes including gastrointestinal and renal dysfunction. Although the increases in P, Ca, and potassium are suggestive of renal injury, the major markers for renal injury including BUN and CRE were not altered and no histological changes or differences in organ weights among groups were observed in the kidney.
Some of the other clinical biochemistry parameters were also altered. Serum AMY was decreased significantly in both sexes, but the changes were more pronounced in males. The major contributor to serum AMY is the pancreas, suggesting that the pancreas may be affected by furan. However, the other marker of pancreatic injury, lipase, was not affected in either sex. Amylase has been shown to be decreased in animals with liver disease and infections (Dere et al. 2007).
In conclusion, the results obtained demonstrate that the mouse is more resistant than the Fischer 344 rat to the oral administration of furan. The study does demonstrate a dose-dependent increase in furan-induced cytotoxicity of the liver in both male and female mice. The liver was the only organ that showed an increase in absolute weight. The histological changes in mice were similar to those observed in the rat liver, except that the changes occurred at doses >2.0 mg/kg bw and these changes were much less severe. These changes were first observed in the caudate lobe and were followed by changes at higher doses in the left lateral lobe in both sexes. Changes in clinical biochemistry, including P and BUN, and histology were observed at a dose of >0.5 mg/kg bw in males and females. Hence, based on these clinical biochemical and histological changes, a NOAEL of 0.12 mg/kg bw per day of furan in mice is suggested.
Footnotes
Acknowledgments
We thank Kevin Little, Julie Snider, Don Demers, Dominque Patry, Wendy Lezama, and Martha Navarro for their help during the animal phase of the study.
