Abstract
In this study, we aimed to show the effect of adenosine 5′-triphosphate (ATP) on sunitinib-induced cardiac injury in rats. The rats (
Introduction
Sunitinib malate (Su-11248; Sutent1; Pfizer Inc., New York City, New York, USA) is a multitargeted, oral, small molecule, tyrosine kinase inhibitor (TKI) and has antiangiogenic activities besides its anticancer effects. It is currently approved by the US Food and Drug Administration and by the European Commission for the treatment of several malignances including advanced renal cell carcinoma, 1 treatment of gastrointestinal stromal tumor after progression under imatinib therapy or for patients intolerant to imatinib, 2,3 and treatment of advanced stage pancreatic neuroendocrine tumors. 4 –6 In addition, the compound is currently under evaluation for the treatment of various solid tumors and efficacy was reported in colorectal cancer, non-small cell lung cancer, malign melanoma, chronic myeloid leukemia, and breast cancer. 7
Its mechanistic targets include various receptors for growth factors and cytokines including vascular endothelial growth factor receptor, platelet-derived growth factor receptor, the stem cell factor receptor, colony-stimulating factor 1 receptor, FMS-like tyrosine kinase-3, and rearranged during transfection receptor tyrosine kinase. 8 –11 Sunitinib targets the adenosine 5′-triphosphate (ATP) binding site of these receptors, and via inhibition, it prohibits cellular signaling and leads to interruption of angiogenesis and proliferation of tumor cells. As a consequence, tumor vascularization reduces and apoptosis of tumor cells is initiated. 9 But the inhibition of these wide range of kinases was considered to be the cause of undesired collateral toxic effects. 12 –15
An increasing number of cardiovascular toxicities (drug-induced cardiac injury) were reported with sunitinib. These serious adverse events such as the development of hypertension and angina pectoris might be resulted with acute ischemic myocardial damage, QT prolongation, symptomatic congestive heart failure, left ventricular systolic dysfunction, and sudden death. 10,11,16 –33 The underlying molecular mechanisms involved in the pathogenesis of these cardiotoxic effects are still unclear, but dependence of cardiomyocytes to aerobic metabolism and ATP was reported as an important factor for cardiotoxicity of sunitinib. 11,34,35 It was determined that being an ATP analog, sunitinib competitively inhibits the ATP binding domain of its target proteins. 36,37 The magnitude of the cardiotoxicity was usually reported to be dose dependent and cumulative. 38
The aim of this study was to explore the potential cardioprotective effects of ATP on sunitinib-induced cardiotoxicity as well as the effects of sunitinib on malondialdehyde (MDA), total glutathione (tGSH), tumor necrosis factor
Materials and methods
Animals
In this experiment, a total number of 30 male rats from albino Wistar species weighed between 265 g and 280 g were used. All of the rats were provided from Ataturk University Medical Experimental Practice and Research Center. The animals were sheltered at normal room temperature (22°C) in proper laboratory environment and were fed appropriately.
The protocols and procedures were made according to guidelines for experimental animal procedures and approved by the Atatürk University’s local animal ethics committee (Ethics Committee Number 2018/09 dated August 29, 2018).
Chemical materials
From the drugs used in the experiment, sunitinib (Sutent) was supplied by Pfizer, thiopental sodium (Pental Sodium) was supplied by I.E. ULAGAY (Istanbul, Turkey), and ATP was supplied by Zdorovye Narodu (Kharkiv, Ukraine).
Experiment groups
The rats that were planned to be used in our study were divided into three groups as sunitinib group (SG), sunitinib plus ATP group (SAG), and healthy group (HG).
Experiment procedure
ATP at a dose of 2 mg/kg (0.2 mL/kg) was injected intraperitoneally (ip) to the SAG group (
Biochemical analysis
Phosphate-buffered saline solution was used to rinse animal tissues prior to dissection. Tissue homogenization was made in the ice-cold phosphate buffers (50 Mm, pH: 7.4). After homogenization, samples were centrifuged at 5000 r/min for 20 min at 4°C, and in the result of this procedure, the supernatants were extracted for measurements of NF-
MDA analysis
Thiobarbituric acid and MDA formed a pink-colored complex. The measurement of the absorbance of this complex (a method by Okhawa et al.
41
) was used for measurement of MDA. The homogenate sample (25 µL) was added to a solution containing 25 µL of 80 g/L sodium dodecyl sulfate and 1 mL mixture solution (200 g/L acetic acid + 1.5 mL of 8 g/L 2-thiobarbiturate). Firstly, we incubated at 95°C for 1 h. After cooling, 1 mL of
tGSH analysis
We made
TNF-α and NF-κ B analysis
We measured tissue homogenate NF-
Tp-I analyses
Tp-I levels in plasma obtained from animals were measured using VIDAS Tp-I Ultra Kit, enzyme-linked fluorescent assay technique. All process of the test was performed automatically with VIDAS machine by prepared test reactants in the kit. The sample was transferred to well containing anticardiac Tp-I antibodies marked with alkaline phosphatase conjugate. Adherence of the antigen to Tp-I attached to the inner wall of solid-phase connector and conjugate was achieved by pulling and releasing the sample conjugate mixture into solid-phase connector. The unbound ingredient was removed by washing. Conjugate enzyme catalysis hydrolyzes of substrate known as 4-methylumbelliferone phosphate to 4-methylumbelliferone, a product that’s fluorescent measured at 450 nm. Density of the fluorescent is proportional to the antigen concentration in the sample.
Histopathological examination
We used 10% formaldehyde solution to fix all of the tissue samples for microscopic examination. After this procedure, samples were washed under tap water in cassettes for 24 h. Conventional grade of alcohol (70%, 80%, 90%, and 100%) was used to remove the remaining water within tissues. After passing through xylol, tissues were embedded in paraffin. We cut 4- to-5-µm sections from the paraffin blocks and hematoxylin–eosin staining was made. Their photos were taken using the Olympus DP2-SAL firmware. A pathologist who was blinded for the study groups made histopathological examinations.
Statistical analysis
The results obtained from experiments were stated as “mean value ± standard error” (
Results
Inflammatory marker results
MDA, tGSH, TNF-
Effect of adenosine triphosphate on sunitinib-induced cardiac injury in rats.a
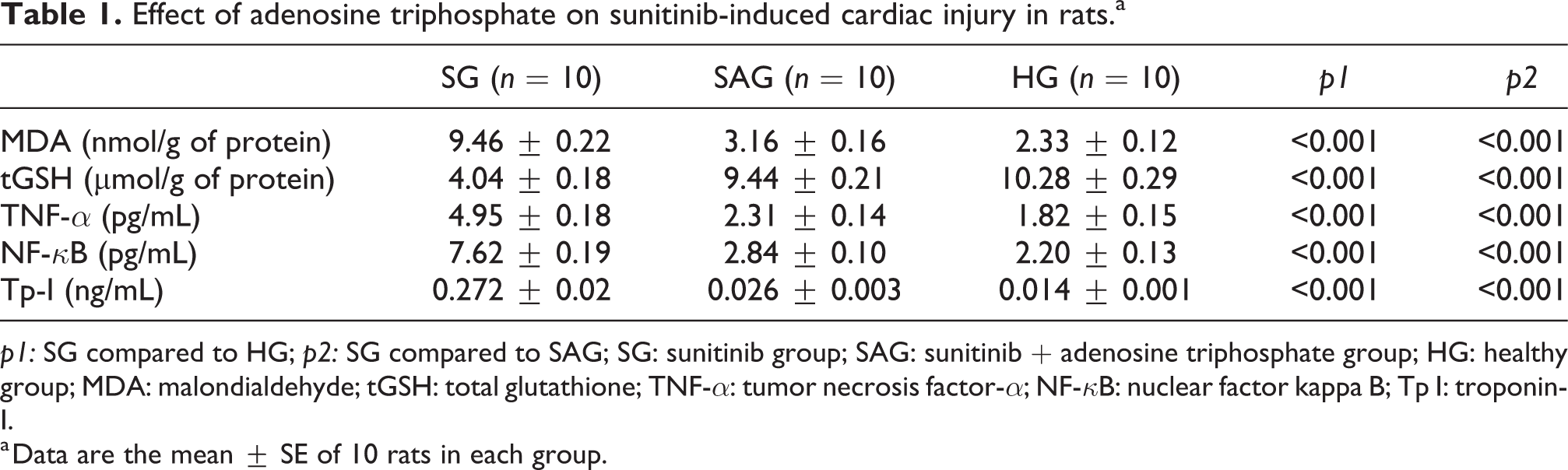
a Data are the mean ± SE of 10 rats in each group.
Histopathological results
According to the conducted microscopic evaluations of cardiac muscle tissue, the structure and morphology of cardiac muscle fibers and blood vessels were normal in the control group (Figure 1(a) and (b)). In the sunitinib group, microscopic examinations showed obvious cardiac muscle tissue damage. Dilated myofibers and locally atrophic myofibers were outstanding across the tissue. Congested blood vessels were observed at the same time (Figure 1(c)). In rats treated with ATP prior to sunitinib, marked amelioration in these findings and normal blood vessels were evidenced (Figure 1(d)).

Hematoxylin–eosin staining in cardiac muscle tissue. (a) Healthy group: →: muscle nuclei; *: blood vessel; >: muscle fiber ×200. (b) Healthy group: →: muscle nuclei; *: normal blood vessel; >: muscle fiber ×200. (c) Sunitinib group: →: muscle nuclei; *: congested blood vessel; >: dilated and atrophic muscle fibers ×100. (d) Sunitinib + adenosine triphosphate group: →: muscle nuclei; *: normal blood vessel; >: muscle fibers ×200.
Discussion
Targeted therapies including TKIs that inhibit molecular pathways have changed the course of many different solid tumors. Sunitinib is one of the most commonly used TKIs and is approved for the treatment of many different cancer types, and research studies are ongoing with sunitinib in various malignancies. 1,2,5 But like other TKIs, sunitinib may lead to drug-induced cardiac toxicity, an undesired adverse effect of this group of drugs. Primary pharmacological effects were defined to be the most important underlying reason of cardiotoxicity because of overlapping of regulatory signaling pathways of tumor growth and survival with cardiac function permanence mechanisms. The most frequent manifestation of cardiotoxicity was reported to be the decrease in left ventricular ejection fraction. 43,44
Over the past few years, many different underlying mechanisms of sunitinib-induced cardiotoxicity were established in various in vivo and in vitro studies. These mechanisms include deterioration of the structure and functions of mitochondria, activation of apoptosis, and depletion of ATP. 33,45,46 Besides determination of these mechanisms, the extent of cardiotoxicity, the effect of cardiotoxicity on prognosis and survival of the patients, and potential cardioprotective agents that could prevent the heart from damage were also defined.
In this present study, we explored the potential cardioprotective effects of ATP on sunitinib-induced cardiotoxicity and also analyzed the effects of sunitinib on MDA, tGSH, TNF-
In this study, according to the histopathological analysis of cardiac muscle tissues, the structure and morphology of cardiac muscle fibers and blood vessels were normal in HG (Figure 1(a) and (b)). We determined obvious cardiac muscle tissue damage in SG including dilated myofibers, locally atrophic myofibers, and congested blood vessels (Figure 1(c)). In this study, the pathological specimens of the patients who experienced heart failure due to sunitinib treatment were evaluated. The microscopic findings were determined as hypertrophy in cardiac myocytes and abnormalities in mitochondria of these cells such as swelling, membrane whorls, and effaced cristae. 19 The authors also have reported similar findings in tissues obtained from mice after sunitinib administration. In another study, an increase in the collagen deposits was determined in hearts of rats treated with sunitinib. 28 Also, pro-inflammatory findings were reported in the rats that isolated heart perfused with sunitinib in the same study.
When we evaluated the samples of the rats treated with ATP prior to sunitinib, evident improvement with normal blood vessel features was observed (Figure 1(d)). Thus, we found that cardiotoxic effects of sunitinib were blocked by ATP. Although this finding could be considered to be a new protective way, it is not clear if ATP administration with sunitinib will reduce antitumor efficacy of sunitinib. Various agents evaluated for cardioprotection failed to demonstrate a clinical benefit because of reduction in antitumor efficacy of the primary agent. 48 Contrary to these results, attenuation of sunitinib-induced cardiotoxicity was determined with the A3 adenosine receptor selective agonist IB-MECA without influencing the antitumor efficacy of sunitinib. 49
Conclusion
TKIs and some other biological agents inhibiting kinases maintained an important clinical benefit for cancer patients with added cardiac toxicity. As being a member of TKI class, cardiotoxicity is also a problem of sunitinib treatment. Our results suggest that sunitinib has inflammatory effects on cardiac myocytes, and MDA, TNF-
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study conformed to the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments and was approved by the ethics committee at the University of Seville. The manuscript does not contain clinical studies or patient data.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
