Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Adenosine, Adenosine Phosphate, Adenosine Triphosphate, Disodium Adenosine Phosphate, and Disodium Adenosine Triphosphate. These ingredients are reported to function in cosmetics as skin-conditioning agents – miscellaneous. The Panel considered the available data and concluded that the five adenosine ingredients reviewed in this report are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
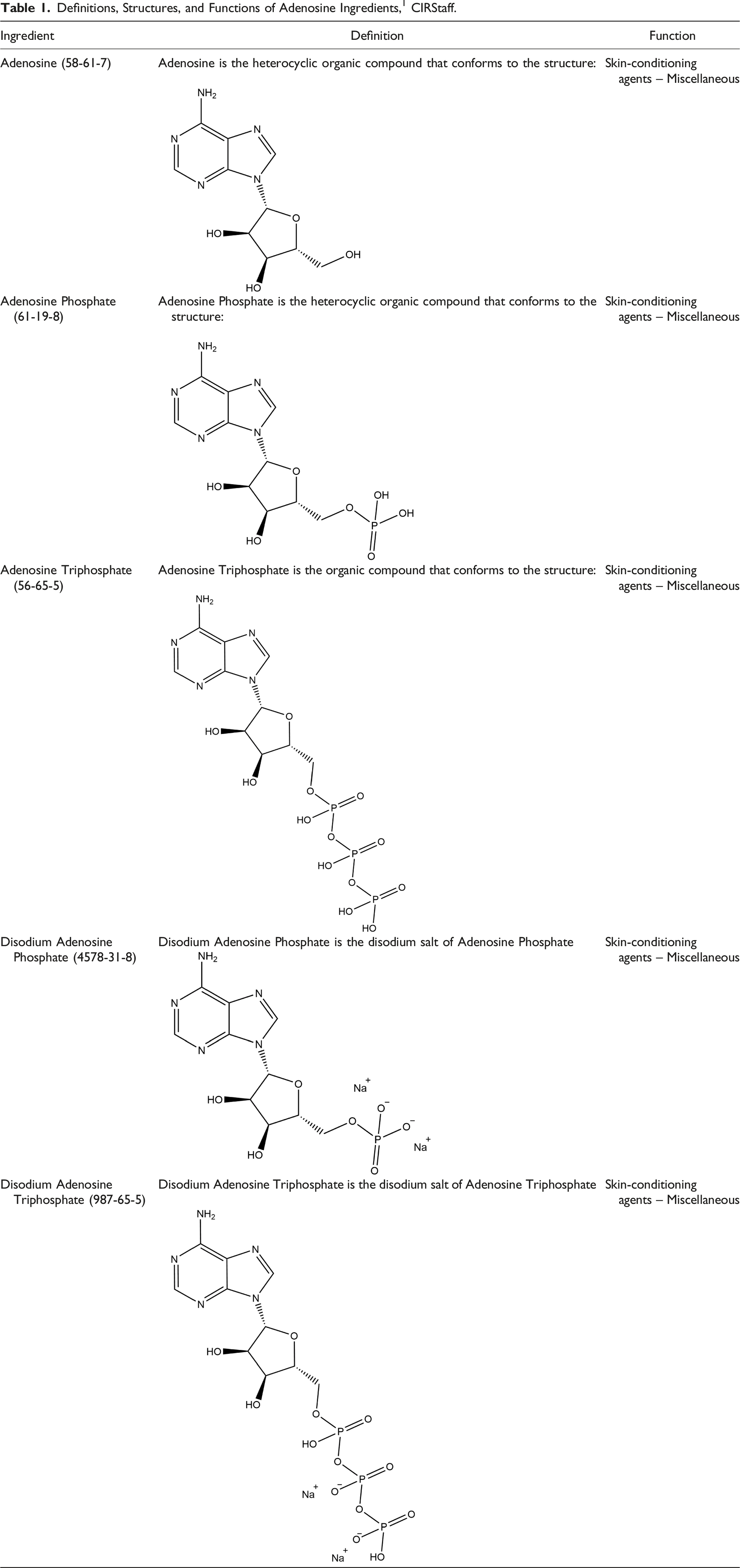
Definitions, Structures, and Functions of Adenosine Ingredients, 1 CIRStaff.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Some of the data included in this safety assessment were found on the European Chemicals Agency (ECHA) website. 2 These summaries are available on the ECHA website, and when deemed appropriate, information from these summaries have been included in this report.
Chemistry
Definition and Structure
The definitions and structures of the ingredients included in this review are provided in Table 1. All of these ingredients share Adenosine (CAS No. 58-61-7; Molecular Weight = 267.25 g/mol; log Kow = −1.05; Figure 1) as the core structure. Adenosine.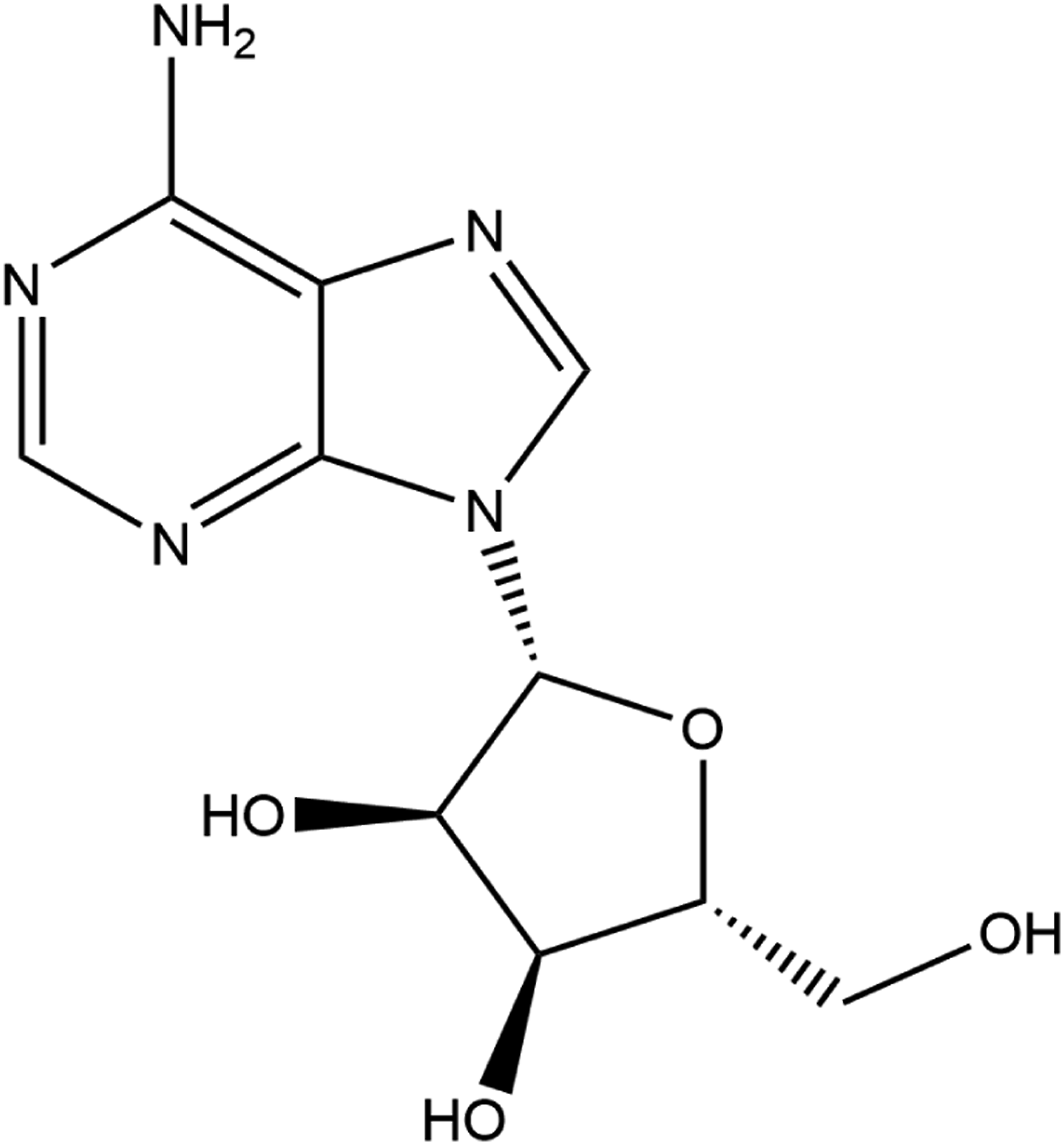
Adenosine Triphosphate (ATP; CAS No. 56-65-5; Molecular Weight = 507.18 g/mol; log Kow = −3.61) is composed of a purine nucleoside esterified with a triphosphate. 3 Adenosine Triphosphate is a ubiquitous organophosphate that connects anabolism and catabolism, but also fuels processes such as motile contraction, phosphorylations, and active transport. 4 Both Adenosine and Adenosine Phosphate (AMP; CAS No. 61-19-8; Molecular Weight = 507.18 g/mol; log Kow = −3.1) are formed when Adenosine Triphosphate is consumed in metabolic processes. Adenosine, a ribonucleoside comprising adenine and ribose, exerts pleiotropic functions throughout the body, primarily by interaction with G-protein coupled receptors. 5 Adenosine Phosphate is an ester of phosphoric acid and Adenosine. Like Adenosine Triphosphate, Adenosine Phosphate plays an important role in many cellular metabolic processes, and is an intermediate in the synthesis of nucleic acids.
Chemical Properties
Chemical Properties of the Adenosine Ingredients in This Safety Assessment.

Method of Manufacture
These methods are general to the production of Adenosine and Adenosine Triphosphate. No methods specific to cosmetic ingredient manufacture were found in the literature or submitted as unpublished data.
Adenosine
The main methods of manufacturing Adenosine include chemical synthesis, RNA degradation, and microbial fermentation.
6
Adenosine Triphosphate
Adenosine Triphosphate may be produced by microbial phosphorylation of Adenosine Phosphate. 7
Impurities
Adenosine
A supplier reported that Adenosine has a purity of ≥ 99%. 8
Adenosine Triphosphate
A supplier reported the following impurities for Adenosine Triphosphate: < 2% Adenosine Phosphate, < 2% Adenosine diphosphate, and < .01% methanol. 9
Disodium Adenosine Triphosphate
Disodium Adenosine Triphosphate used in a trade name mixture sold to the cosmetic industry was reported to be free from carcinogens, mutagens, and reproductive toxicants (CMR) as defined by European Union (EU) chemical regulations, and from phthalates, pesticides, and glycol ether. 10 Additional details were not provided.
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
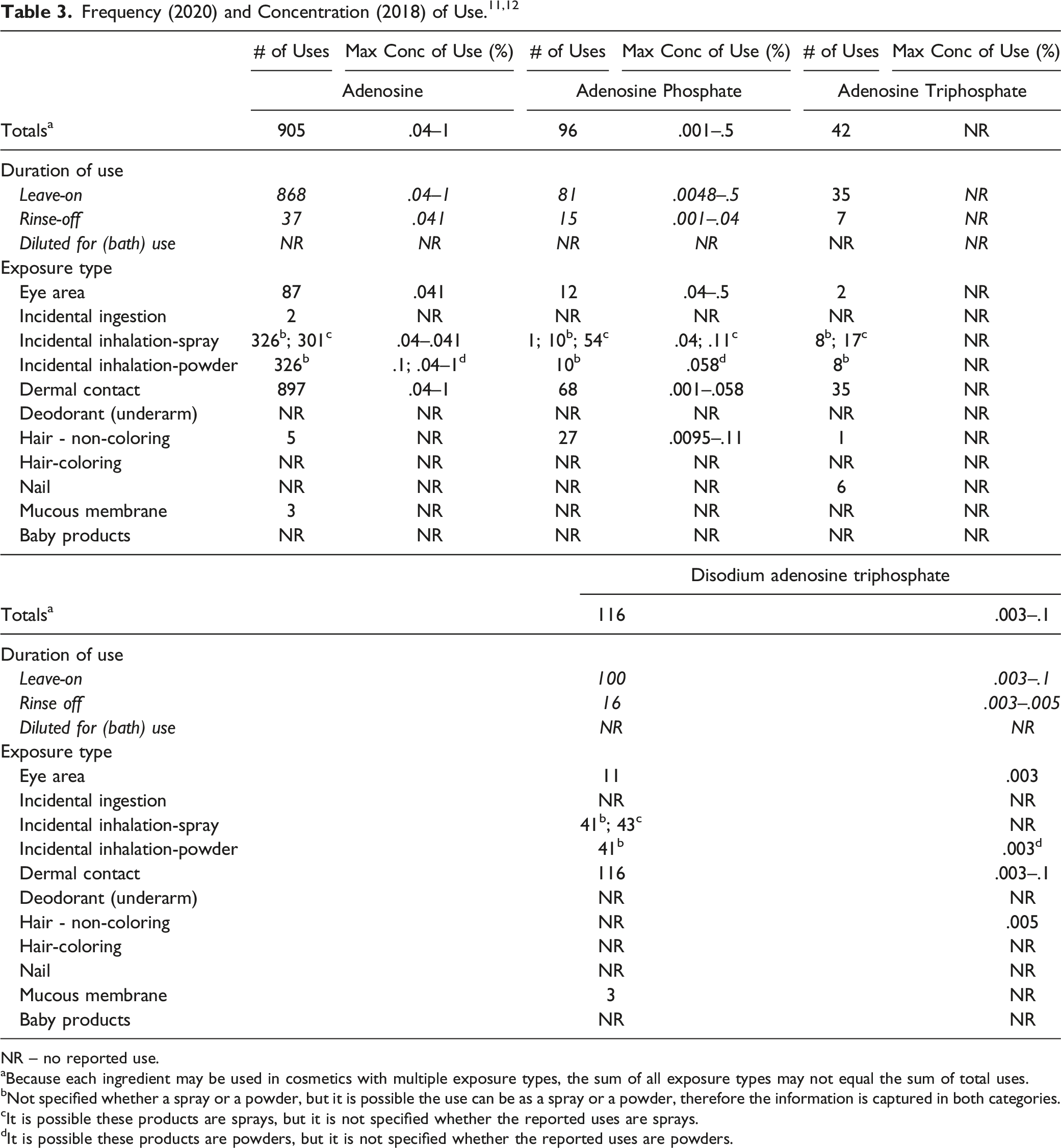
NR – no reported use.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
cIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
dIt is possible these products are powders, but it is not specified whether the reported uses are powders.
These ingredients have been reported to be used around the eyes (e.g., at up to .5% Adenosine Phosphate in mascara). In addition, Adenosine could result in incidental ingestion as it is used in lipstick and dentifrices (concentrations of use not reported). Some of the adenosine ingredients are used in cosmetic sprays and could possibly be inhaled; for example, Adenosine is reported to be used at .041% in spray moisturizing formulations, and Adenosine Phosphate is used in aerosol hair spray formulations at up to .04%. In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared with pump sprays.13,14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., would not enter the lungs) to any appreciable amount.15,16 Adenosine is reported to be used in face powders at concentrations up to .1% and could be incidentally inhaled. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.17-19
All of the adenosine ingredients named in this report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 20
Non-Cosmetic
Adenosine
According to the US FDA, Adenosine is used for the treatment of paroxysmal supraventricular tachycardia and approved for use in nuclear stress testing in patients who cannot exercise adequately. 21 Adenosine is typically given intravenously at a dose of .84 mg/kg. 22 In 2013, the FDA issued a warning informing health care professionals of the rare but serious risk of heart attack with the use of Adenosine-containing drugs in nuclear stress testing. Health care professionals are advised to avoid using this ingredient in patients with signs or symptoms of unstable angina or cardiovascular instability. In addition, Adenosine is used to treat surgical and nerve pain, and pulmonary hypertension.23,24
Adenosine Phosphate and Adenosine Triphosphate
According to 21 CFR 216.24, all drug products containing Adenosine Phosphate or Adenosine Triphosphate were withdrawn or removed from the market because the product or product components were found to be neither safe nor effective for its intended use as a vasodilator and anti-inflammatory. Adenosine Phosphate is used in the therapeutic treatment of herpes, post-herpetic neuralgia, photosensitivity, and porphyria cutanea tarda.25-27 Adenosine Triphosphate has been previously reported to treat acute kidney failure, high blood pressure, cystic fibrosis, and lung cancer.28,29 Adenosine Triphosphate has also been reported to be used as an ergogenic aid used by athletes. 30
Toxicokinetic Studies
Dermal Penetration
In vitro
Adenosine
In a dermal penetration study, human skin samples (500 μm thick) were mounted in stainless steel doubly jacketed diffusion cells. 31 The acceptor solution consisted of phosphate buffered saline and the test substance consisted of Adenosine (1.5 or 3%) in propionic acid, (.5%) in hexanoic acid, or (1.5%) in a binary vehicle of propionic and hexanoic acid. A volume of 450 μL of the test substance was pipetted into the donor reservoir. Perfusate samples were collected after 25 or 30 min, and analyzed. The observed optimal permeability coefficients (Kp) of Adenosine from the binary vehicle, propionic acid solution, and hexanoic acid solution were .0004, .00012, and .00016 cm/min, respectively.
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
Oral
Adenosine Phosphate
Male and female Wistar rats (number of animals not stated) were given a single dose of 10 mg/kg [14C]Adenosine Phosphate dissolved in 9% aqueous sodium chloride via gavage. 32 The specific activity of the [14C]Adenosine Phosphate was reported to be 46 mCi/mmol. Within 72 h of administration, 28% of the administered activity was excreted in the urine and 6% was recovered in the feces. Plasma levels of Adenosine Phosphate were maximal approximately 30 min after oral administration. Adenosine Phosphate was considered to be rapidly absorbed by the intestinal mucosa and quickly distributed 2 h after absorption; only 20% of the maximal concentration remained in the plasma.
Human
Oral
Adenosine Triphosphate
Eight volunteers were given single doses of 5000 mg Adenosine Triphosphate or placebo via an ingested pellet targeted at release in the proximal or distal small intestine, or via a naso-duodenal tube. 30 Blood Adenosine Triphosphate and metabolite concentrations were monitored by high performance liquid chromatography (HPLC) 4.5 h (naso-duodenal tube) or 7 h (pellets) post-administration. Adenosine Triphosphate concentrations in the blood did not increase after supplementation of Adenosine Triphosphate via pellets or naso-duodenal tube. Concentrations of uric acid were significantly increased compared to placebo by approximately 50% after administration via proximal-release pellets and naso-duodenal tube, but not after administration via distal-release pellets. The mean time to peak uric acid concentration was shorter for naso-duodenal tube administration (75 to 195 min) as compared to the pellet administration (150–390 min).
Toxicological Studies
Acute Toxicity Studies
Oral
Adenosine
An LD50 of >2000 mg/kg bw was established for mice given Adenosine orally. 33 No other details regarding this study were provided.
An acute oral toxicity study on Adenosine was performed on female Wistar rats (3 rats/group) according to Organization for Economic Cooperation and Development (OECD) Test Guideline (TG) 423. 2 In both groups, the test substance (Adenosine in methylcellulose) was given at a dose of 2000 mg/kg bw. Animals were observed for 14 d following treatment and killed on day 15. All rats survived treatment and no treatment-related clinical symptoms were observed. Necropsy revealed pale kidneys in two animals of group 1 and all animals of group 2. The LD50 was reported to be >2000 mg/kg bw.
Adenosine Triphosphate
The oral LD50 of Adenosine Triphosphate was reported to be >2000 mg/kg in rats. 34 No other details regarding this study were provided. In a different study, groups of 5 male anesthetized New Zealand White rabbits were given 2 or 20 mg/kg Adenosine Triphosphate via a gastric cannula. 35 The test substance did not have an effect on diastolic aortic pressure, heart rate, central venous pressure, iliac venous blood flow, lung resistance, or the arterial partial pressure of oxygen (PaO2).
Disodium Adenosine Triphosphate
An oral LD50 of >2000 mg/kg was reported for both mice and rats treated with Disodium Adenosine Triphosphate. 36 No other details regarding these studies were provided.
Short-Term Toxicity Studies
Oral
Adenosine and Adenosine Triphosphate
New Zealand White rabbits were given doses of either 3 mg/kg/d (n = 4) or 20 mg/kg/d (n = 12) Adenosine Triphosphate mixed with cellulose, or 20 mg/kg/d adenosine hemisulfate salt (n = 4) for 14 d. 35 Adenosine Triphosphate and adenosine hemisulfate, dissolved in saline, were administered daily via gastric cannula. Control rabbits received a corresponding amount of saline. No modification of electrocardiogram morphology or heart rate was detected in treated animals compared to controls. Central venous and arterial pressures were comparable in all groups. After treatment with 3 and 20 mg/kg/d, increases of 30 and 50% in the intervillous vein blood flow (IVBF) were observed, respectively. The left ventricular work index (LVWI) was significantly increased by 10% in animals given 20 mg/kg/d Adenosine Triphosphate. In addition, treatment with the higher dose level led to a 12.5% decrease of the spontaneous respiratory frequency. A 26% reduction of lung resistance was noted in all Adenosine Triphosphate-treated groups. Increases of 22 and 23% of PaO2 were observed in rabbits treated with 3 mg/kg/d and 20 mg/kg/d Adenosine Triphosphate, respectively. Similar results were noticed in rabbits treated with adenosine hemisulfate; however, lung resistance and PaO2 levels remained unchanged.
Developmental and Reproductive Toxicity Studies
Intraperitoneal
Adenosine
Adenosine (50, 100, and 150 mg/kg) was administered intraperitoneally to mice and rats once a day for 5 d. 33 Decreased spermatogenesis and increased numbers of abnormal sperm were noted. No other details regarding this study were provided.
Genotoxicity
In Vitro
Adenosine
The potential mutagenicity of Adenosine (up to 333 μg/plate, vehicle not stated), was evaluated in
The genotoxicity of Adenosine was also evaluated in a Chinese hamster ovary cell/hypoxanthine-guanine phosphoribosyl-transferase (CHO/HGPRT) assay. 38 Adenosine in DMSO was not genotoxic when tested at up to 2000 μg/mL, with and without metabolic activation.
Carcinogenicity Studies
No data regarding the carcinogenicity of these ingredients were found in the published literature, and unpublished data were not submitted.
Other Relevant Studies
Cytotoxicity
Adenosine
The cytotoxic effect of Adenosine in Swiss albino mouse embryo fibroblasts (3T3 and 3T6) and immortalized cervical cancer (HeLa) cells, cultured with and without adenosine deaminase, was studied. 39 [14C]Adenosine (.2–2.5 μCi) was diluted with unlabeled Adenosine (to 10−5–10−3 M) in .3 mL of a solution containing serum-free medium, 50 mM phosphate buffer, and 10% serum. Both calf and horse serum were used; however, horse serum did not contain adenosine deaminase. Cells were exposed to Adenosine at concentrations of 0, .002, .005, .01, .02, .20, 1.0, and 2.0 mM, and cultures were observed over a period of 1 week. When Adenosine was added to cell cultures in a medium containing horse serum, it was found to be toxic at low concentrations. In 10% calf serum, there was no effect on cell growth at low or moderate Adenosine concentrations, while in medium containing 10% horse serum, there was definite inhibition of growth at a concentration of .005 mM and a killing of cells at .02 mM. Cell inhibition in calf serum was observed when Adenosine was used at concentrations of 1.0 mM and higher. When the same experiment was performed with horse serum with the addition of 1 mM uridine to the cell culture medium, toxic effects were not observed at any concentration up to .2 mM.
Tumor Cell Proliferation
Adenosine
The effects of Adenosine on DNA synthesis and cell growth in human (HT-29, T84, HRT-18, Colo320HSR) and mouse (MCA-38) colorectal carcinoma cell lines were studied. 40 Cells were seeded in 24-well plates at 20,000 cells/well. Adenosine was added at final concentrations of 1 μCi/mL, 1 μM, with methyl-[3H]thymidine. Plates were incubated for 36–48 h. DNA synthesis and cell proliferation were stimulated in all cell lines tested, with a half maximal effective concentration (EC50) of 2.8–30 μM, and a maximum stimulation being reached at 10-100 μM. Effects were similar among mouse and human cell lines.
Effect on Histamine Release
Adenosine Phosphate and Adenosine Triphosphate
Thirty-nine patients with various dermatoses were used in a study evaluating histamine release from human cutaneous mast cells following intracutaneous injection with the polycondensation product of
The effects of intradermal injections of Adenosine Phosphate and Adenosine Triphosphate, as compared to intradermal injections of histamine, were evaluated. 42 The backs of subjects were injected with 50 μL isosmotic phosphate buffered saline containing Adenosine Triphosphate, Adenosine Phosphate, histamine, compound 48/80, or phosphate-buffered saline alone. Injections were carried out in 2.5-min intervals. The area of erythema induced by the injection was delineated at 30 seconds and after 4.5 min. Solutions that were extremely acidic were neutralized with sodium hydroxide prior to injection. Injection of Adenosine Triphosphate resulted in immediate erythematous reaction of the surrounding skin. This reaction faded after 1 min, and was replaced by slightly darker erythema that lasted for up to 2 h. The extent of these reactions was dose-dependent. No wheals were formed after injection with Adenosine Phosphate or phosphate-buffered saline. Adenosine Triphosphate produced wheals in 5 out of 7 subjects injected with 180 nmol, and in all subjects at higher doses, in a dose-dependent manner. Wheals that resulted from 1080 nmol Adenosine Triphosphate were approximately equal to wheals due to histamine (1.63 nmol). Injections of Adenosine Triphosphate at high doses produced sensations of persistent pain which was not observed with injection of saline or histamine.
In order to evaluate the role of histamine and prostaglandins in the inflammatory response to Adenosine Triphosphate, the study was also performed with the addition of pre-treatment with either diphenhydramine, cimetidine, indomethacin, or doxantrazole. Erythema and wheal responses were significantly suppressed with the addition of diphenhydramine pre-treatment. Indomethacin, doxantrazole, and cimetidine did not alter the Adenosine Triphosphate reaction.
Dermal Irritation and Sensitization
Irritation
In vitro
Adenosine
An in vitro skin irritation study was performed using reconstructed human epidermis according to OECD TG 439. 2 Ten mg of Adenosine (in powder form; concentration not provided) were applied to the epidermal surface. (The epidermal surface was first moistened with 5 μL deionized water to improve contact between the powder and the epidermis). Phosphate buffered saline and sodium dodecyl sulfate (5%) were used as the negative and positive controls, respectively. The test substance did not significantly reduce cell viability compared to the negative control. The test substance was predicted to be non-irritating to the skin.
Animal
Adenosine
According to a risk profile from the Norwegian Food Safety Authority (NFSA), Adenosine was non-irritating to animal skin in multiple conventional tests. 33 No other details regarding these studies were provided.
Human
Adenosine
A 48-h patch test was performed on 10 subjects. 43 Each subject received an occlusive patch with 15 μL of a cosmetic ingredient containing .2% Adenosine on the inside upper arm. Skin reactions were evaluated 1, 24, and 48 h after patch removal. One hour after patch removal, slight erythema was observed on one volunteer. However, after 24 and 48 h, no skin reaction was observed in any subject.
Sensitization
Animal
Adenosine
According to a risk profile from the NFSA, Adenosine was non-sensitizing in a Magnusson and Kligman maximization study. 33 No other details regarding this study were provided.
Disodium Adenosine Triphosphate
A Magnusson-Kligman test was performed on Pirbright white guinea pigs (number of animals not stated). 44 The test substance was a trade name mixture containing 15% mannitol and 15% Disodium Adenosine Triphosphate. A .5% aqueous dilution of the test substance (i.e., .075% mannitol and .075% Disodium Adenosine Triphosphate) was used for the intracutaneous induction, and a 10% aqueous dilution of the test substance (i.e., 1.5% mannitol, 1.5% Disodium Adenosine Triphosphate) was used for the epicutaneous induction and challenge. No signs of irritation or skin reactions indicative of an immune response were observed.
Human
Adenosine
A human repeated insult patch test (HRIPT) was completed in 205 subjects using a test material containing .2% Adenosine. 43 Each of the subjects received .2 mL of the test substance on the upper back area under a semi-occlusive patch. After a 24-h exposure period, the patches were removed and sites were evaluated. A series of 9 test patches were applied followed by a 2-wk non-treatment period. Challenge patches were applied to previously unexposed sites and allowed to remain in skin contact for 24 h. Challenge sites were scored at 24 and 72 h post patching. No signs of sensitization were observed.
Disodium Adenosine Triphosphate
An HRIPT was completed on 50 volunteers using a trade name material consisting of 15% mannitol and 15% Disodium Adenosine Triphosphate. 44 A 10% aqueous dilution of the trade name material (i.e., 1.5% mannitol, 1.5% Disodium Adenosine Triphosphate) was applied to the backs of subjects under an occlusive patch for a total of 9 applications within a 3-wk period. A challenge patch was applied 2 wk later to the previously exposed area, as well as an unexposed area. Readings were taken 24, 48, and 96 h after patch removal. No skin reactions were noted in any volunteers.
Phototoxicity/Photosensitization
Human
Disodium Adenosine Triphosphate
A phototoxicity study was conducted with a trade name mixture consisting of 15% mannitol and 15% Disodium Adenosine Triphosphate in 10 volunteers. 44 A 10% aqueous solution of the trade name mixture (i.e., 1.5% mannitol, 1.5% Disodium Adenosine Triphosphate; .2 mL) was applied under an occlusive patch to two different areas of the forearm, one irradiated and one non-irradiated. After a 24-h exposure, one site was irradiated with long-wave ultraviolet light (UVA; 320–400 nm) for 15 min; the other test site served as a non-irradiated control. Skin reactions were scored immediately after light exposure as well as 24 and 48 h later. No reactions were noted on either the irradiated or non-irradiated test site in any subject.
A photosensitization test was completed on 34 subjects with a trade name mixture consisting of 15% mannitol and 15% Disodium Adenosine Triphosphate . 44 For 3 wk, six 24-h induction patches were applied containing a 2% aqueous solution (i.e., .3% mannitol, .3% Disodium Adenosine Triphosphate) of the trade name mixture. Applications were performed in duplicate; one site was subsequently irradiated with UV light (260–400 nm) for 15 min each session. After 2 wk, a challenge patch was applied at virgin sites with and without irradiation. At the challenge phase, no skin reactions were exhibited at either the irradiated site or the non-irradiated site.
Ocular Irritation Studies
In Vitro
Adenosine
According to a risk profile from the NFSA, Adenosine was predicted to be slightly irritating to the eyes in an in vitro hen’s egg test-chorioallantoic membrane (HET-CAM) assay. 33 No other details were provided for this study.
Animal
Adenosine
A Draize assay was performed on 3 Japanese White rabbits according to OECD TG 405. 2 The test substance, 100 mg undiluted Adenosine, was instilled into the left eye of each animal. The eyes, which were not rinsed, were observed for 21 d. The test substance was considered to be non-irritating to the eye.
Clinical Studies
Effects of Inhalation
Adenosine
The effect of inhaled Adenosine was studied in eight asthmatic subjects. 45 Before administration of Adenosine, two baseline blood samples were taken, and five baseline measurements of specific airway conductance (SGaw) were made. Volunteers then inhaled a single sample of Adenosine, ranging from .6 to 6.7 mg/mL. The test material was nebulized from a volume of 4 mL in disposable nebulizers driven by compressed air at 8 L/min. Approximately .5 mL of the test solution left the nebulizer as an aerosol each minute; 12.5% of this entered the lungs with a mass median particle diameter of 4.5 microns. After inhalation, SGaw and blood sample measurements were taken at 1, 3, 5, 10, 15, 20, 25, and 30 min. Significant falls in SGaw from a mean baseline of .124 ± .024 to .046 ± .008 and .066 ± .012 s/cm/water, were observed at 3 and 30 min, respectively. Inhalation did not produce significant changes in levels of histamine, neutrophil chemotactic factor, or cyclic adenosine phosphate in the blood.
Adenosine Phosphate and Adenosine Triphosphate
The effects of aerosolized Adenosine Triphosphate and Adenosine Phosphate on dyspnea and airway caliber were studied. 46 The perception of dyspnea quantified by a modified Borg Scale of Perceived Exertion and other symptoms was determined in 10 nonsmokers and 10 patients with asthma. Each subject attended the laboratory on three occasions. The first visit included a screening, recording of medical history, lung function assessment, and skin-prick testing of common aeroallergens. On the second and third visit, subjects were administered either Adenosine Triphosphate or Adenosine Phosphate, in aerosolized form. Before, immediately after, and 30 min after the challenge, spirometry was performed, the Borg score was determined, and symptoms other than dyspnea were recorded. In order to determine the Borg scale, subjects were asked to determine the degree of breathlessness they were experiencing on a scale of 0–10. For the inhalation challenge tests, Adenosine Triphosphate (.125–512 mg/mL) and Adenosine Phosphate (.048–400 mg/mL) were dissolved in a normal saline solution and administered via a breath-activated dosimeter with an output of 10 μL per inhalation. Participants wore a nose clip and inhaled 5 breaths of the normal saline solution, followed by sequential doubling concentrations of either Adenosine Triphosphate or Adenosine Phosphate. Subjects who were healthy nonsmokers did experience dyspnea when given Adenosine Triphosphate or Adenosine Phosphate. All patients with asthma experienced dyspnea when given Adenosine Triphosphate, and 90% of patients with asthma experienced dyspnea when given Adenosine Phosphate. The geometric mean provocative dose (PD20) in responsive subjects was 26.9 mg/mL and 39.6 mg/mL for Adenosine Triphosphate and Adenosine Phosphate, respectively. In patients with asthma, the perception of dyspnea assessed by the Borg score increased from .1 to 3.3 and .2 to 2.5 after Adenosine Triphosphate and Adenosine Phosphate, respectively. Eighty percent of subjects coughed after the Adenosine Triphosphate challenge, whereas 40% of subjects coughed after the Adenosine Phosphate challenge. Throat irritation was noted after the Adenosine Triphosphate and Adenosine Phosphate challenge in 70% and 35% of subjects, respectively.
A different study was performed to evaluate whether inhaled Adenosine Triphosphate or Adenosine Phosphate produces a tussive response, and whether chronic cough patients are hypersensitive to these ingredients compared to healthy volunteers. 47 All participants received two cumulative cough challenges, one with Adenosine Triphosphate and one with Adenosine Phosphate. Saline (.9%) was used as the solvent for both Adenosine Phosphate and Adenosine Triphosphate. The two challenges were administered on two different days, at least 48 h apart. Each volunteer started with a saline inhalation, followed by Adenosine Triphosphate or Adenosine Phosphate delivered in increasing concentrations on a half-log scale from .1 to 300 mM. The number of coughs produced in the first 15 seconds after inhalation were counted. The challenge was terminated once the volunteer coughed at least fivetimes (C5), or the maximum concentration was inhaled. Two out of 19 healthy patients coughed with Adenosine Phosphate, none reaching C5. Eighteen out of 20 volunteers coughed after administration of Adenosine Triphosphate, with 15 reaching C5. Eight out of 20 chronic cough patients coughed with Adenosine Phosphate, two reaching C5. Eighteen of 19 chronic cough patients reached C5 after inhalation of Adenosine Triphosphate. The C5 in chronic cough patients was predominately distributed between 1 mM and 100 mM, as all patients who reached C5, did so by a concentration of 100 mM.
Adenosine Triphosphate in Resistance-Trained Athletes
Adenosine Triphosphate
The effect of Adenosine Triphosphate in resistance-trained athletes was investigated in a 12-wk, 3-phase randomized, double-blind, and placebo- and diet-controlled intervention. 48 Phase 1 consisted of a periodized resistance-training program. Phase 2 was a 2-wk overreaching cycle in which volume and frequency were increased, followed by a 2-wk taper (Phase 3). Biochemical parameters were evaluated on weeks 0, 4, 8, and 12 to assess the chronic effects of the test substance. Twenty-four males were randomly assigned to receive either 400 mg/d of Adenosine Triphosphate or maltodextrin (placebo), consumed orally via a two-piece gelatin capsule 30 minutes prior to resistance training sessions. On non-training days, participants were instructed to consume one dose on an empty stomach prior to breakfast. No statistically or clinically significant changes in blood chemistry or hematology were observed. No adverse effects were reported in this study.
Summary
The safety of Adenosine, Adenosine Phosphate, Adenosine Triphosphate, Disodium Adenosine Phosphate, and Disodium Adenosine Triphosphate as used in cosmetics is reviewed in this safety assessment. According to the
According to 2020 VCRP survey data, Adenosine, Adenosine Phosphate, Adenosine Triphosphate, and Disodium Adenosine Triphosphate are reported to be used in 905, 96, 42, and 116 formulations, respectively. The results of the concentration of use survey conducted by the Council indicate that Adenosine has the highest concentration of use; it is used at up to 1% in body and hand products. Disodium Adenosine Phosphate is not reported to be in use.
The penetration ability of Adenosine in different vehicles was evaluated in human skin. The observed optimal Kps of Adenosine from a binary vehicle (propionic and hexanoic acid), propionic acid solution, and hexanoic acid solution were .004, .012, and .016 cm/min, respectively.
Wistar rats were given 10 mg/kg [14C]Adenosine Phosphate dissolved in 9% aqueous sodium chloride via gavage. Within 72 h of administration, 28% of the injected activity was excreted in the urine and 6% was recovered in the feces. Eight volunteers were given singles doses of 5000 mg Adenosine Triphosphate or placebo via an ingested pellet targeted at release in the proximal or distal small intestine, or via a naso-duodenal tube. Concentrations of uric acid were significantly increased compared to placebo after administration via proximal-release pellets and naso-duodenal tube, but not after administration via distal-release pellets.
No treatment-related symptoms were observed when Wistar rats were orally given 2000 mg/kg bw Adenosine in methylcellulose, however, pale kidneys were observed in all females in one group given 2000 mg/kg Adenosine. In a different study, the reported oral LD50 of Adenosine in mice was >2000 mg/kg. The acute oral LD50 of Adenosine Triphosphate was reported to be >2000 mg/kg in rats. No changes in diastolic aortic pressure, heart rate, central venous pressure, IVBF, lung resistance, or PaO2 were observed in New Zealand White rabbits given a single dose of up to 20 mg/kg Adenosine Triphosphate orally. An oral LD50 of >2000 mg/kg was reported for both mice and rats for Disodium Adenosine Triphosphate in two different studies.
In a short-term toxicity study, New Zealand White rabbits were given doses of either 3 mg/kg/d or 20 mg/kg/d Adenosine Triphosphate mixed with cellulose, or 20 mg/kg/d adenosine hemisulfate salt, for 14 d. Administrations occurred via gastric cannula. The LVWI was significantly increased by 10% in animals given 20 mg/kg/d Adenosine Triphosphate. In addition, treatment with the highest dose level led to a 12.5% decrease of the spontaneous respiratory frequency. A 26% reduction of lung resistance was noted in all Adenosine Triphosphate -treated groups. Increases of 22 and 23% of PaO2 were observed in rabbits treated with 3 mg/kg/d and 20 mg/kg/d Adenosine Triphosphate, respectively.
In a reproductive study, Adenosine (50, 100, and 150 mg/kg) administered intraperitoneally in mice and rats for 5 d caused decreased spermatogenesis and an increased number of abnormal sperm.
Adenosine was non-genotoxic in Ames assays performed with and without metabolic activation on
The cytotoxic effects of Adenosine in Swiss albino mouse embryo 3T3 and 3T6 and in HeLa cells cultured with and without adenosine deaminase were studied. Cells were exposed to Adenosine at concentrations of 0, .002, .005, .01, .02, .20, 1.0, and 2.0 mM. When Adenosine was added to cell cultures in a medium containing horse serum (does not contain adenosine deaminase), it was found to be toxic at low concentrations. Cell inhibition in calf serum was observed when Adenosine was used at concentrations of 1.0 mM and higher.
The effect of Adenosine on DNA synthesis and cell growth in human HT-29, T84, HRT-18, and Colo320HSR and mouse MCA-38 cell lines was studied. Adenosine was added with methyl-[3H]thymidine (final concentrations, 1 μCi/mL, 1 μM). DNA synthesis and cell proliferation were stimulated in all cell lines tested, with an EC50 of 2.8–30 μM, and a maximum stimulation was reached at 10–100 μM.
The effects of intradermal injections of Adenosine Phosphate and Adenosine Triphosphate compared to intradermal injections of histamine were evaluated. The backs of volunteers were injected with 50 μL isosmotic phosphate buffered saline containing Adenosine Triphosphate, Adenosine Phosphate, histamine, compound 48/80, or saline. Adenosine Triphosphate produced wheals in 5 out of 7 subjects injected with 180 nmol, and in all subjects at higher doses, in a dose-dependent manner. Wheals that resulted from 1080 nmol Adenosine Triphosphate were approximately equal to wheals due to histamine (1.63 nmol). Injections of Adenosine Triphosphate at high doses produced sensations of persistent pain which was not observed with injection of saline or histamine.
Adenosine (10 mg) was considered to be non-irritating in an in vitro skin irritation study performed using reconstructed human epidermis, according to OECD TG 439. According to a risk profile from the NFSA, Adenosine was non-irritating to animal skin in multiple conventional tests. According to the same risk profile, Adenosine was considered to be non-sensitizing to the skin of guinea pigs. A 48-h patch test performed using .2% Adenosine on 10 subjects yielded negative results. Negative results were also observed in an HRIPT performed on 205 subjects using the same test substance. A trade name material consisting of 15% mannitol and 15% Disodium Adenosine Triphosphate was used in different aqueous dilutions in a Magnusson-Kligman maximization test (.075% mannitol and .075% Disodium Adenosine Triphosphate (intracutaneous induction); 1.5% mannitol, 1.5% Disodium Adenosine Triphosphate (epicutaneous induction and challenge)) and HRIPT (1.5% mannitol, 1.5% Disodium Adenosine Triphosphate). No signs of sensitization were observed in either study.
A phototoxicity and photosensitization study was performed with a trade name mixture consisting of 15% mannitol and 15% Disodium Adenosine Triphosphate. The test substances were applied at 10% (i.e., 1.5% mannitol, 1.5% Disodium Adenosine Triphosphate and 2% (i.e., .3% mannitol, .3% Disodium Adenosine Triphosphate) aqueous dilutions in the phototoxicity and photosensitization studies, respectively. No skin reactions were noted in either study.
Adenosine was predicted to be slightly irritating to the eyes in a HET-CAM assay. It was considered to be non-irritating to rabbit eyes in a different study.
The effect of inhaled Adenosine (.6 to 6.7 mg/mL) was studied in 8 asthmatic subjects. Significant falls in SGaw from a mean baseline of .124 ± .024 to .046 ± .008 and .066 ± .012 s/cm/water were observed at 3 and 30 min, respectively. Inhalation did not produce significant changes in levels of histamine, neutrophil chemotactic factor, or cyclic adenosine phosphate in the blood.
The effects of aerosolized Adenosine Triphosphate and Adenosine Phosphate on dyspnea and airway caliber were studied. The PD20 was 26.9 mg/mL and 39.6 mg/mL for Adenosine Triphosphate and Adenosine Phosphate, respectively, in responsive subjects. The perception of dyspnea assessed by the Borg score increased from .1 to 3.3 and .2 to 2.5 after Adenosine Triphosphate and Adenosine Phosphate, respectively, in patients with asthma. In a different study, 2 out of 19 healthy patients coughed after inhalation of Adenosine Phosphate, none reaching C5. Two out of 18 volunteers coughed after administration of Adenosine Triphosphate, with 15 reaching C5. Eight out of 20 chronic cough patients coughed with Adenosine Phosphate, 2 reaching C5. Eighteen of 19 chronic cough patients reached C5 after inhalation of Adenosine Triphosphate.
Thirty-nine patients with various dermatoses were used in a study evaluating histamine release from human cutaneous mast cells following intracutaneous injection with 48/80 (1 mg/mL water), Adenosine Triphosphate (60 mg/mL water), adenosine diphosphate (30 mg/mL water), or Adenosine Phosphate (37 mg/mL water). In addition, 3 concentrations of histamine dihydrochloride were also injected (1, 3, and 10 μg/mL), and used to compare the responses elicited from the test substance. Injection of Adenosine Triphosphate in the skin caused a response similar to that of histamine, but only at high concentrations. Adenosine Triphosphate released histamine at concentrations >1 mg/mL, while 48/80 stimulated histamine release in skin at concentrations >1 μg/mL.
The effect of orally ingested Adenosine Triphosphate (400 mg/d) in resistance-trained athletes was evaluated over a 12-wk period. No significant changes in blood chemistry or hematology were observed, and no adverse effects were reported.
Discussion
The Adenosine ingredients reviewed in this document are naturally-occurring, ubiquitous chemicals. Because the noted, safe use of these ingredients in therapeutics results in significantly greater systemic exposures than could be possible from cosmetic use, the concern for systemic toxicity is mitigated. The Panel determined that the available human sensitization data on Disodium Adenosine Triphosphate and Adenosine can be used as read-across sources to mitigate any concerns for the other ingredients lacking these data. The safety of the ingredients included in this report was further supported by low concentrations of use, sufficient impurities data, negative animal oral toxicity assays, and human clinical studies. In addition, The Panel noted the effects of Adenosine administered via a nebulizer in asthmatic patients and determined that these effects would not be pertinent to cosmetic exposure, as delivery of Adenosine via cosmetic products would result in a much lower exposure than that of a nebulizer.
The Panel discussed the issue of incidental inhalation exposure from powders and hair sprays. The Council survey results indicate that Adenosine is being used in face powders at concentrations up to .1%. In addition, Adenosine is used in spray moisturizing products at up to .041%, and Adenosine Phosphate is used at up to .04% in hair sprays. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Adenosine, Adenosine Phosphate, Adenosine Triphosphate, Disodium Adenosine Phosphate, and Disodium Adenosine Triphosphate 1 are safe in cosmetics in the present practices of use and concentrations described in the safety assessment.
Footnotes
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
