Abstract
Cutaneous side effects associated with sunitinib use are a major problem in patients receiving cancer treatment. The aim of this study was to investigate the protective effect of adenosine triphosphate (ATP) against possible skin damage resulting from sunitinib use in rats. Thirty Albino Winstar rats were divided into the following three groups: healthy controls (HCs, n = 10), sunitinib (SUN, n = 10), and sunitinib + ATP (SAT, n = 10). ATP was injected intraperitoneally at a dose of 2 mg/kg. One hour subsequent to the administration of ATP and 0.9% NaCl, the SAT and SUN groups were orally administered a dose of 25 mg/kg sunitinib to the stomach. Macroscopic evaluation of the skin indicated lower levels of skin damage in the SAT group than in the SUN group. As an indicator of oxidative stress, malondialdehyde (MDA), total oxidant status (TOS), and oxidative stress index (OSI) levels were significantly higher in the SUN group than in the HC group, while total glutathione (tGSH) and total antioxidant status (TAS) levels were significantly lower. However, MDA, TOS, and OSI levels were significantly lower in the SAT group than in the SUN group, while tGSH and TAS levels were significantly higher. Histopathological examination revealed keratin plugs with edema, vasopathology, and inflammatory cell infiltration in the SUN group. The SAT group showed less necrotic epithelium, keratin plugs, edema, and vasopathology than the SUN group. ATP can be effective in preventing skin damage caused by sunitinib use by reducing oxidative stress.
Introduction
Sunitinib is a tyrosine kinase inhibitor (TKI) anticancer drug used in the treatment of metastatic renal cell carcinoma, imatinib-resistant gastrointestinal stromal tumors, and pancreatic neuroendocrine tumors. 1 This group of drugs represents a new class of target-specific antineoplastic drugs. 2 Sunitinib suppresses endothelial cell proliferation and vascularization by inhibiting vascular endothelial growth factor (VEGF). 3 Preclinical and clinical studies have shown that sunitinib’s anticancer activity is associated with the inhibition of tumor cell proliferation and angiogenesis. 4 Although sunitinib and other TKIs are more specific and selective than classical chemotherapeutic drugs, their effects are known to be not limited to tumor cells. 5 Side effects tied to the use of these drugs include fatigue, asthenia, anorexia, diarrhea, abdominal pain, hypothyroidism, hypertension, myelosuppression, and oral mucositis, as well as cutaneous side effects. 2,5,6
Mucositis and cutaneous side effects may affect the quality of life, resulting in dose reduction, discontinuation, or a change in treatment. 1,7 Cutaneous side effects associated with sunitinib are thought to be due to VEGF receptor inhibition occurring in proangiogenic pathways. 8 Sunitinib toxicity has demonstrated to be primarily associated with mitochondrial disorders. Drugs that interfere with mitochondrial functions reportedly lead to depletion of adenosine triphosphate (ATP). 9 Bouitbir et al. showed that sunitinib increases the accumulation of mitochondrial reactive oxygen species (ROS) and reduces cellular ATP and reduced glutathione (GSH) stores. 10 A decrease in these high-energy phosphate concentrations has been documented to result in oxidative stress and membrane lipid peroxidation (LPO). 11 As is known, an increase in ROS production changes the balance between oxidant and antioxidant in favor of oxidants, causing oxidative damage. This phenomenon has been reported to lead to premature aging of the skin as well as pathological changes. 12
In the present study, we investigate the protective effect of ATP, an organic compound containing carbon (C), hydrogen (H), oxygen (O), nitrogen (N), and phosphate (P), 13 against skin damage in rats caused by sunitinib. ATP is continuously produced by the process of oxidative phosphorylation in mitochondria. 14 Often referred to as “molecular currency” for intracellular energy transfer, ATP functions as a chemical fuel by powering many organic life processes. 15 Khakh and Burnstock demonstrated that ATP also serves as a critical signaling molecule that allows intra- and intercellular communication. 16 Kumbasar et al. showed that ATP protects ovarian tissue from ischemia reperfusion injury by suppressing overproduction of xanthine oxidase and malondialdehyde (MDA). 17 Chiang et al. reported that ATP enhanced wound healing by increasing VEGF levels. 18 Thus, based on the literature, ATP may be hypothesized to be useful in the treatment of sunitinib-related skin damage. The aim of our study was to biochemically and histopathologically investigate the protective effect of ATP against possible skin damage due to sunitinib use in rats.
Materials and methods
The animal subjects
A total of 30 male Albino Winstar rats weighing between 250 g and 267 g were used in the experiment. All rats were obtained from Ataturk University Medical Experimental Application and Research Center. The animals were housed and fed under suitable conditions at normal room temperature (22°C) in an appropriate laboratory environment. The protocols and procedures were approved by the local Animal Experimentation Ethics Committee (ethics committee number: 2019/115, dated: October 9, 2019).
Chemical compounds
Of the substances used in the experiment, sunitinib was obtained from Pfizer (Turkey), thiopental sodium from İ. E. Ulagay (Turkey), and ATP from Zdorove Naro (Ukraine).
Experimental groups
The rats were divided into healthy control (HC), sunitinib (SUN), and sunitinib + ATP (SAT) groups.
Test procedure
ATP was injected intraperitoneally (IP) into the SAT (n = 10) group at a dose of 2 mg/kg (0.2 mL/kg). 17 The SUN (n = 10) and HC (n = 10) groups were administered IP with an equivalent volume of normal saline (0.9% NaCl) solution. One hour following the administration of ATP and 0.9% NaCl, the SAT and SUN groups were orally administered sunitinib at a dose of 25 mg/kg to the stomach. This procedure was repeated once a day over the course of 4 weeks. At the end of this period, the skin tissues of all animals were evaluated macroscopically. The animals were then euthanized with high-dose thiopental sodium (50 mg/kg) anesthesia. Samples were taken from the skin tissue immediately after the animals were euthanized. MDA, total glutathione (tGSH), total oxidant status (TOS), and total antioxidant status (TAS) levels were measured, and oxidative stress index (OSI) was calculated using some of the skin tissues. Some of the skin samples underwent histopathological examination.
Biochemical analysis
Preparation of the samples
Prior to dissection, all tissues were rinsed with phosphate-buffered saline solution. The tissues were homogenized in ice-cold phosphate buffers (50 mM, pH 7.4), appropriate for the variable to be measured. The tissue homogenates were centrifuged at 5000 r/min for 20 min at 4°C, and the supernatants were extracted to analyze tGSH, MDA, TOS, and TAS. Total tissue results were expressed by dividing by grams of protein. All spectrophotometric measurements were performed using a microplate reader (BioTek, Winooski, Vermont, USA).
MDA analysis
MDA measurements were based on the method used by Ohkawa et al. involving spectrophotometrical measurement of the absorbance of the pink-colored complex formed by thiobarbituric acid and MDA. The tissue homogenate sample (25 μL) was added to a solution containing 25 µL of 80 g/L sodium dodecyl sulfate and 1 mL of mixture solution (200 g/L acetic acid + 1.5 mL of 8 g/L 2-thiobarbiturate). 19 The mixture was incubated at 95°C for 1 h. Upon cooling, 1 mL of n-butanol: pyridine (15:1) was added. The mixture was vortexed for 1 min, then centrifuged for 10 min at 4000 r/min. The absorbance of the supernatant was measured at 532 nm. The standard curve was obtained using 1,1,3,3-tetramethoxypropane.
tGSH analysis
tGSH analysis was performed using the method described by Sedlak and Lindsay. 5,5′-Dithiobis [2-nitrobenzoic acid] (DTNB) disulfite is chromogenic in the medium and easily reduced by sulfhydryl groups. 20 The yellow color produced during reduction is measured using spectrophotometry at 412 nm. A cocktail solution consisting of 5.85 mL of 100-mM Na-phosphate buffer, 2.8 mL of 1-mM DTNB, 3.75 mL of 1-mM NADPH, and 80 µL of 625 U/L glutathione reductase was prepared. Before measurement, 0.1-mL metaphosphoric acid was added to 0.1-mL tissue homogenate and centrifuged for 2 min at 2000 r/min for deproteinization. Then, 0.15 mL of the cocktail solution was added to 50 µL of supernatant. The standard curve was obtained using glutathione disulfide.
Measurements of TOS and TAS
TOS and TAS levels for tissue homogenates were determined using a novel automated measurement method and commercially available kits (Rel Assay Diagnostics, Turkey), both developed by Erel. 21,22 The method used to measure TOS is based on the bleaching of the characteristic color of the more stable 2,2′-azino-bis [3-ethylbenzothiazoline-6-sulfonic acid] radical cation by antioxidants, measured at 660 nm. The results are expressed as nmol hydrogen peroxide equivalent/L. To measure TAS levels, the oxidants present in the sample oxidized the ferrous ion-o-dianisidine complex to ferric ion. The oxidation reaction was enhanced by glycerol molecules, which are abundantly present in the reaction medium. The ferric ion produced a colored complex with xylenol orange in an acidic medium. The color intensity, spectrophotometrically measured at 530 nm, was related to the total amount of oxidant molecules present in the sample. The results are expressed as μmol Trolox equivalent/L. The percentage ratio of TOS to TAS was used to determine the OSI, calculated as TOS divided by TAS × 100.
Histopathological examination
All of the tissue samples were first identified in a 10% formaldehyde solution for light microscopic assessment. Following the identification process, tissue samples were washed under tap water in cassettes for 24 h. Samples were then treated with conventional grade alcohol (70%, 80%, 90%, and 100%) to remove the water within tissues. Tissues were then passed through xylol and embedded in paraffin. Four-to-five micron sections were cut from the paraffin blocks and stained with hematoxylin–eosin. Photographs were then taken using the Olympus DP2-SAL Firmware Program assessment (Olympus® Inc., Tokyo, Japan). Histopathological assessment was carried out blindly by a pathologist. Histopathological data, including keratin plugs, collagen fiber disorganization, epithelium necrosis, polymorphonuclear cell infiltration, and edema, were analyzed by the same pathologist and quantified between 0 and 3 (0: not observed, 3: intensive).
Statistical analyses
Statistical analyses were performed using SPSS version 22.0 software (IBM Corp., Armonk, New York, USA). Data are expressed as mean ± standard deviation. In the macroscopic, biochemical, and histopathological analyses, significant differences between groups were determined using Kruskal–Wallis analysis of variance, a nonparametric test. For dual comparison, the Mann–Whitney U test was performed to evaluate significance levels, with a value of p < 0.05 accepted as statistically significant.
Results
Macroscopic evaluation of the skin
Skin tissues of all animals were evaluated macroscopically for erythema, dermatitis-like redness, desquamation, and hyperkeratosis. Each criterion was graded on a scale of 0 to 3. While the highest score was found in the group receiving only sunitinib, a decrease in the severity of skin toxicity was observed in the ATP + sunitinib group. The differences between all groups were statistically significant (p < 0.001) (Table 1).
The results of macroscopic evaluation of the skin.

HC: healthy control group; SUN: sunitinip group; ATP: adenosine triphosphate; SAT: sunitinib + ATP group.
a p-Value of Kruskal–Wallis test.
b p = 0.002, Statistically significantly different when compared with control group.
c p = 0.004, Statistically significantly different when compared with SAT group.
d p = 0.003, Statistically significantly different when compared with SAT group.
Biochemical results
tGSH and MDA analysis
The results of the biochemical analyses of the 30 rats assigned to the three groups are summarized in Table 2. A significant increase in MDA as a marker of oxidative stress (p = 0.004) was observed in the SUN group compared with the control group. In the SAT group, which received ATP treatment prior to the administration of sunitinib, MDA levels were significantly lower than those of the SUN group (p = 0.004). While there was no statistically significant difference between tGSH values in the SAT and control groups (p = 0.109), tGSH levels were significantly higher in the SAT group than SUN group (p = 0.004).
The results of biochemical evaluations of tissue samples among groups.
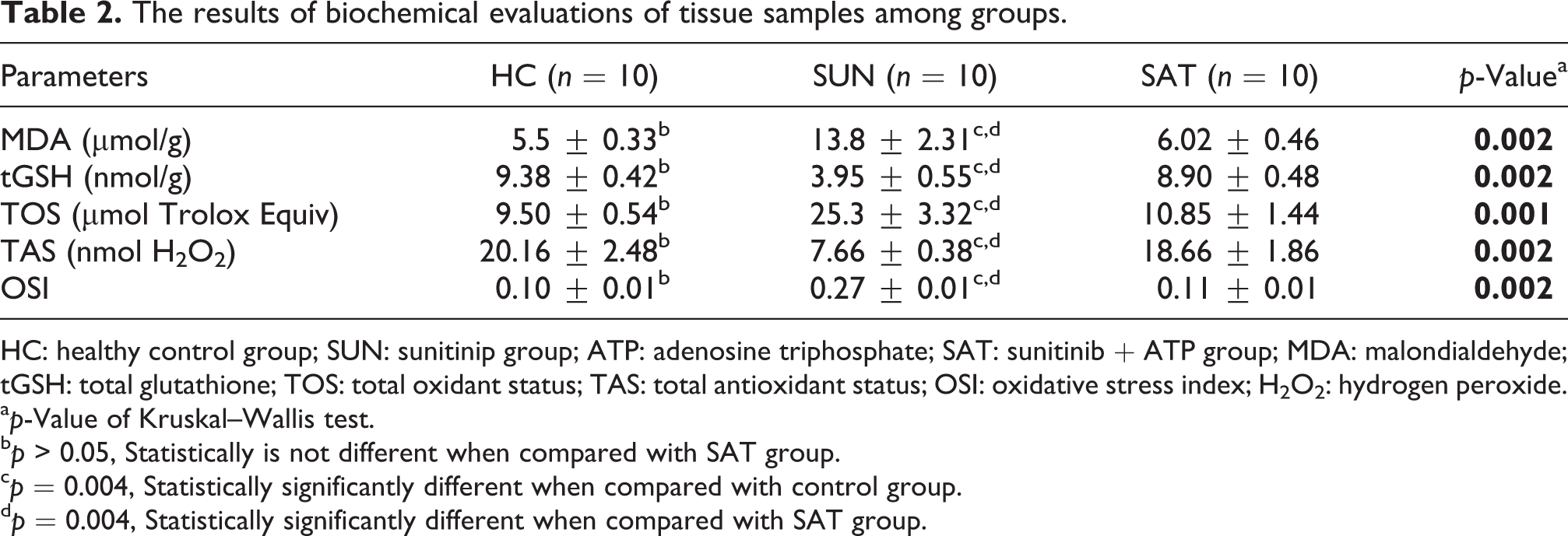
HC: healthy control group; SUN: sunitinip group; ATP: adenosine triphosphate; SAT: sunitinib + ATP group; MDA: malondialdehyde; tGSH: total glutathione; TOS: total oxidant status; TAS: total antioxidant status; OSI: oxidative stress index; H2O2: hydrogen peroxide.
a p-Value of Kruskal–Wallis test.
b p > 0.05, Statistically is not different when compared with SAT group.
c p = 0.004, Statistically significantly different when compared with control group.
d p = 0.004, Statistically significantly different when compared with SAT group.
TAS, TOS, and OSI levels
Compared with the control group, the TAS levels of the SUN group were significantly lower (p = 0.004), while those of the SAT group were significantly higher (p = 0.004). TOS and OSI (p = 0.004) levels increased only in the sunitinib group, while skin TOS and OSI levels were lower in the SAT group (p = 0.004). There were no statistically significant differences between the SAT and HC groups with respect to TOS (p = 0.06), TAS (p = 0.224), and OSI (p = 0.224) values (Table 2).
Histopathological findings
The results of the data quantified in pathological analyses are summarized in Table 3. The results for the SAT group were similar to those of the control group in terms of a number of parameters. The most adverse pathological alterations were detected in the SUN group. Histological examination of skin tissues obtained from the control group revealed normal keratinized stratified squamous epithelium, a normal dermis layer with well-organized collagen fibers, hair follicles surrounded by sebaceous glands, and normal dermal blood vessels (Figure 1(a)). In the sunitinib group, sections of the skin showed extensive keratin plugs on the surface of the epithelium, thinning of stratified squamous epithelium, and some necrotic keratinocytes. Skin taken from the SUN group also exhibited polymorphonuclear cell infiltration within the dermis, hair follicle rupture, decreased sebaceous gland activity surrounding hair follicles, disorganization and irregularity of collagen fibers of the dermis, superficial dermal edema, and mild congestion in blood vessels (Figure 1(b) and (c)). In the SAT group, stratified squamous epithelium thickness similar to that the control group, diminished keratin plugs, mostly normal hair follicles, increased sebaceous gland activity, normal organization of collagen fibers in the dermis, and normal blood vessels were observed. Polymorphonuclear cell infiltration and edema were not detected (Figure 1(d)).
The results of histopathological analysis.

HC: Healthy control group; SUN: sunitinip group; ATP: adenosine triphosphate; SAT: sunitinib + ATP group.
a p-Value of Kruskal–Wallis test.
b p = 0.002, Statistically significantly different when compared with control group.
c p = 0.003, Statistically significantly different when compared with SAT group.
d p = 0.004, Statistically significantly different when compared with SAT group.

(a) Hematoxylin–eosin staining in skin tissue in the control group;
Discussion
In this study, we investigated the effect of ATP in the prevention of skin damage caused by sunitinib, which until now constituted a gap in the literature, and determined that ATP was significantly effective in preventing the increase of sunitinib-induced oxidative stress. Furthermore, through macroscopic and histopathological evaluations of skins samples, we found that ATP reduces skin damage associated with the use of sunitinib.
Novel anticancer angiogenesis inhibitors offer a well-tolerated safety profile, but they also have many side effects. Signal pathways and/or receptors inhibited by these new drugs are physiologically expressed in the skin and/or hair follicles, creating the potential for skin toxicity. 23 Cutaneous side effects include hand–foot skin reaction (HFSR), stomatitis, facial edema, yellowing of the facial skin, whitening of the hair, subungual splinter hemorrhage, erythematous rashes on the trunk, edema of lower extremities, facial erythema, alopecia, facial acneiform rash, and scalp dysesthesia. 5,24 Toxicities of the oral cavity, such as mucositis and stomatitis, are among the main unavoidable side effects associated with cancer treatment. 25 HFSR is the most common cutaneous side effect. A literature review found that HFSR was observed in 36% of patients treated with sunitinib; in that study, the mean onset time for patients receiving sunitinib treatment was 32.4 days. 8 In the present study, effects of skin toxicity such as erythema, dermatitis-like rashes, desquamation, and hyperkeratosis were observed on the epidermis following continuous intragastric administration of 25 mg/kg sunitinib for 1 month. The incidence of skin toxicity in the sunitinib group was 100%, its severity ranging from mild to severe. The administration of ATP in conjunction with sunitinib reduced the incidence and severity of skin toxicity. Sunitinib is a small molecule that inhibits multiple receptor tyrosine kinases (RTKs), which are involved in tumor growth, pathological angiogenesis, and metastatic progression of cancer. It also inhibits the VEGF receptor as well as other RTKs such as PDGFR, EGFR, KIT, RET, Flt3, and RAS and affects cellular energy homeostasis by disrupting mitochondrial function through the inhibition of AMPK signaling. This disruption leads to opening of the mitochondrial permeability transition pore, transmembrane potential (ΔΨ) dissipation, and swollen mitochondria, disrupts cristae, and greatly reduces intracellular ATP, while inducing the release of cytochrome c at low concentrations and apoptosis. 9 In the literature, sunitinib has been shown to increase the accumulation of mitochondrial ROS and decrease stores of cellular ATP and GSH. 10 The decrease in these high-energy phosphate concentrations has been documented to result in oxidative stress and membrane LPO. 11
Lipid molecules are among the biomolecules most affected by oxidative stress. MDA, the product of oxidative stress during polyunsaturated fatty acid peroxidation, 26,27 is known to cause the degradation of DNA and protein structures due to its mutagenic properties. 28 Excessive ROS production or impaired detoxification of aggressive molecules may induce oxidative stress, identified as a critical factor in the pathogenesis of chronic, nonhealing wounds. MDA levels have been shown to be higher in chronic ulcers than in acute wounds, and this molecule is a product of excess ROS-induced LPO in skin wounds. 12,29 In our study, MDA levels were significantly increased in the SUN group; however, there was a significant decrease in the tissue MDA levels in the SAT group. This result demonstrated the effect of ATP on preventing oxidative stress.
In the case of oxidative stress, free oxidant radicals appear in the cell. 30 These molecules cause changes in the cell, such as protein damage and deterioration of the DNA structure, triggering a process that results in apoptosis and tissue damage. 31 Glutathione, an endogenous tripeptide, is present in two different forms, GSH and oxidized glutathione (GSSG), 32 and plays an important role in removing these radicals. When the number of free oxidant radicals increase in a cell, the reduction of GSSG to GSH is catalyzed by the glutathione reductase enzyme to reduce these radicals. While the GSH/GSSG ratio is greater than 100 in the resting state, this ratio can decrease to between 1 and 10 under situations of oxidative stress. 33 The skin utilizes a number of antioxidant substances, including GSH, to maintain oxidative balance. 12 In the present study, significantly lower levels of glutathione were detected in the tissues of the SUN group compared to those of the HC and SAT groups. High levels of glutathione in the SAT group were also evidence of the antioxidant effect of ATP.
Under physiological conditions, the oxidant/antioxidant balance is maintained by the predominance of antioxidants. Disruption of this balance causes tissue damage called oxidative stress. Therefore, the oxidant/antioxidant balance is used to assess whether tissue damage has occurred. 34 There are a number of markers for evaluating oxidative stress and antioxidant status, as well as different methods of measuring them. However, individual measurement of these markers is both time-consuming and costly. For this reason, in recent years, TOS and TAS have been measured. 21,22 The OSI, calculated as the ratio of TOS to TAS, is used as an indicator of oxidative stress. 21,22,35 Previous studies have suggested that OSI may be a more accurate indicator of oxidative status than single TOS or TAS levels. 36,37 Various chemotherapy agents, such as cisplatin in particular, by leading to excessive oxidant production in the body or weakening of the antioxidant mechanism, have been shown to cause an increase in TOS and OSI and a decrease in TAS. 38 In addition, these parameters have been used as an oxidative stress parameter in various inflammatory skin diseases such as psoriasis and dermatitis. 39,40 The current study also evaluated the overall effects of sunitinib use on oxidative and antioxidant status in skin tissues proceeding from this perspective. In the group treated with only sunitinib, TOS and OSI levels increased dramatically when compared to the group treated with the SAT and HC groups, whereas TAS levels in the SUN group were clearly reduced compared to the other groups. These results indicate that exposure to sunitinib causes an increase in oxidative stress in skin tissues and that high oxidative stress can be reduced by treatment with ATP.
Data on the histopathology of HFSR induced by multikinase inhibitors (MKIs) such as sunitinib are currently limited to case series. In MKI-associated hand–foot syndrome, there are histological signs of keratinocyte damage in the epidermis and dermis with dilated blood vessels, a mild to moderate perivascular lymphohistiocytic infiltrate, and occasionally syringometaplasia of the eccrine sweat glands. 41 Lacouture et al. 24 reported that the level of epidermal necrosis varies according to the time interval between the onset of MKI and skin biopsy. In biopsies of skin lesions taken less than 1 month after MKI induction, keratinocyte necrosis, including stratum spinosum and stratum granulosum, was at lower levels, whereas in biopsies taken 1 month later the stratum corneum had begun to exhibit changes. In addition, biopsies of lesions obtained at later dates contained a hyperkeratotic phase characterized by hyperkeratosis, papillomatosis, and parakeratosis. In the present study, the skin tissues of rats were examined following 1 month of sunitinib administration. In the sunitinib group, sections of the skin showed large-scale keratin plugs at the top of the epithelium, thinning of stratified squamous epithelium, and some necrotic keratinocytes. Polymorphonuclear cell infiltration within the dermis, hair follicle rupture, increased sebaceous gland surrounding hair follicles, disorganization and irregularity of collagen fibers of the dermis, superficial dermal edema, and mild congestion in blood vessels were also observed in the SUN group. However, in the group treated with ATP, histopathological findings were similar to those of the HC group.
The aim of the treatment is to employ preventive measures to reduce the risk of cutaneous toxicities such as HFSR and stomatitis developing secondary to the use of MKI such as sunitinib. MKI-induced HFSR is managed in two ways, by preventative or restrictive measures. There are few randomized clinical trials concerning the management of HFSR in the literature, and recommendations have been largely developed by expert panels with reference to case reports, case series, clinical experience, and algorithms. Various topical and systemic prophylactic treatments have been reported in the literatüre. 42 –44 In a large randomized clinical trial on prophylactic effects, Ren et al. reported that, in HFSR associated with sorafenib, urea-based creams reduce symptoms and delay toxicity. However, urea-based creams had no effect on sorafenib dose reduction, discontinuation, or termination rate. 43 Despite the relatively high incidence of MKI-induced skin toxicity, evidence supporting preventive measures is very limited.
Most processes requiring energy, including mitotic and biosynthetic activities of cells during wound healing, occur directly or indirectly via hydrolysis of ATP. ATP is used during wound healing in various metabolic processes, including protein and lipid biosynthesis, growth factor production, signal transduction, and mitosis. Low blood flow and low supply of oxygen in the wound area cause ischemic and potentially hypoxic conditions that may impair the balance of energy production and energy use in cells. This imbalance, by leading to the depletion of high-energy phosphate ATP, may reduce the cellular energy supply and potentially interfere with the healing process. 45 Encapsulation of ATP in small unilamellar lipid vesicles (ATP-vesicles) has been shown to accelerate healing of the skin at full thickness in both nondiabetic and diabetic rabbits. 18,46 The introduction of ATP-vesicles into ischemic ear wounds in rabbits initiated early angiogenesis in the wound bed and resulted in faster wound closure than in untreated rabbits, as well as improved development of granular tissue and reepithelialization. 46 These findings indicate that the intracellular distribution of ATP increases wound healing in normal and diabetic wounds and present a potential option for the treatment of chronic nonhealing wounds. Although the mechanisms by which intracellular ATP delivery improves wound healing are not fully understood, it is clear that the presence of ATP is an important aspect of the healing process.
There are several limitations to the current study. First of all, patients with various tumors clinically present with dermal toxicity caused by MKIs as a result of the use of these drugs. To further assess the effects of MKIs and ATP on skin toxicity, a skin toxicity model for mice with given types of tumors should be established. Secondly, the number of rats used in this study was kept low, mainly due to animal rights concerns. A further limitation was the lack of data in the literature with which to compare our results regarding skin toxicity caused by the use of TKI and the use of antioxidant treatment to prevent such toxicity.
Conclusion
We found that ATP is effective in reducing oxidative stress caused by sunitinib use and that it protects against sunitinib-induced skin damage at macroscopic and histopathological levels. The results presented herein demonstrate that antioxidant therapies may be effective in the treatment of cutaneous toxicities where oxidative stress plays a prominent pathogenic role. We expect this study to provide further insight that may be of benefit to clinical trials concerned with improving skin damage secondary to the use of sunitinib. Compared to other cutaneous toxicities, severe HFSR and stomatitis are early symptoms requiring therapy modifications to alleviate symptoms, whereas other cutaneous toxicities do not affect the course of treatment. Therefore, the development of agents to reduce such side effects will improve both the quality of life and the success rate of treating patients with advanced disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
