Abstract
Background:
Delayed neurological sequels (DNS) have been described after carbon monoxide (CO) poisoning. There is a need to find a new prognostic marker to guide the use of hyperbaric oxygen (HBO) therapy.
Aim:
To evaluate serum S-100β level in patients presenting with acute CO poisoning as an indicator of poisoning severity and predictor of DNS occurrence and HBO need in those patients.
Methods:
This prospective cohort study included patients with acute CO poisoning. On admission, carboxyhemoglobin (COHb) and S-100β levels were measured. Patients were followed up for 6 months for signs of DNS.
Results:
Out of 50 patients, 6 only developed DNS. The mean of S-100β levels was significantly higher in patients with severe poisoning and those with DNS. Receiver operating characteristic curve analysis revealed that S-100β had an area under the curve 0. 871; at a cutoff value ≥ 0.67 µg/L, its sensitivity and specificity were 100% and 77.3%, respectively. The sensitivity of S-100β was significantly higher than that of COHb, while its specificity and overall accuracy were significantly higher than those of HBO criteria.
Conclusion:
Serum S-100β level on admission could be a marker of poisoning severity and a predictor of CO-induced DNS development that guides the use of HBO therapy.
Introduction
Carbon monoxide (CO) is a colorless, tasteless, odorless, and nonirritating gas that is generated from incomplete combustion of organic material. 1,2 Statistics have revealed that CO poisoning is one of the chief causes of death worldwide. Annually, 50,000 cases with 1200 deaths were reported in the United States for non-fire CO exposures. 3
CO causes generalized and cerebral hypoxia by forming carboxyhemoglobin (COHb) as it has a greater affinity for hemoglobin (230–270 times) than oxygen. 2 Additionally, CO mediates direct cellular damage by interfering with mitochondrial enzyme activity and normal cellular respiratory function. 4,5
Presenting symptoms of acute CO poisoning are usually nonspecific and thus the patients may be misdiagnosed, resulting in significant morbidity and mortality. 6 Headache, dizziness, dyspnea, and confusion are the most common symptoms in mild CO exposure. However, severe CO exposures may result in coma, dysrhythmias, and death. 7,8
Following acute CO poisoning, some patients present with signs and symptoms of central nervous system (CNS) affection. The severity of these manifestations depends mainly on the duration of exposure to CO. 9 In the acute phase, patients may suffer from headache, dizziness, syncope, seizure, brain infarction, and loss of consciousness. 9,10 After recovery from the acute phase, delayed neurological sequels (DNS) develop in some patients. 9 –13
DNS may occur after a lucid interval (ranging from days to 6 weeks) after partial or complete recovery of the initial hypoxic stage of CO poisoning. The characteristic features of DNS are amnesia, mental disorientation, gait disturbance, urinary/fecal incontinence, depression, and Parkinsonism. Radiologic studies by magnetic resonance imaging and computed tomography have revealed that basal ganglia, hippocampus, and corpus callosum were affected in acute phase, 14 –17 while the deep white matter was more affected in the delayed phase. 18 –20
The CO-induced DNS manifestations may be permanent in 25% of cases. Therefore, prevention of this complication is a target goal in the treatment of CO poisoning. 21 –23
Brain hypoxia that ensues due to CO poisoning results in oxidative stress, necrosis, apoptosis, 24 lipid peroxidation, and reversible CNS demyelination. 25 Further damage to the CNS is produced by CO-activated neutrophils that produce reactive oxygen species. All these mechanisms result in inflammation and injury in the affected brain regions. 26 In patients with DNS, diffuse inflammations of the deep white matter and the periventricular area were detected. 15,27,28 The pathogenesis of CO-induced DNS is not precisely identified. Lesions in myelin base protein—which accounts for nearly one-third of myelin protein of the CNS—can explain the development of DNS. 10,29,30
Treatment of CO poisoning includes the administration of 100% normobaric oxygen to all patients presenting to the emergency department. Moreover, hyperbaric oxygen (HBO) therapy was recommended in patients with severe CO poisoning due to its beneficial effect in preventing inflammatory changes in the brain and lipid peroxidation, consequently reducing the DNS rates. 5,23,31
A meta-analysis showed that there was no significant difference in neurologic sequels in four out of six randomized controlled trials that investigated the use of HBO for the treatment of CO exposures. 32 The indications for HBO therapy in acute CO poisoning comprise various clinical manifestations and laboratory tests, including loss of consciousness, neurological deficits, significant metabolic acidosis, and COHb level above 25%. On the other hand, some adverse effects were associated with the use of HBO such as cataracts, reversible myopia, tracheobronchial symptoms, seizures as well as barotraumas to the middle ear, the cranial sinuses, or the lungs. 33 The development of more specific indications for HBO may lead to a better selection of high-risk patients who will ultimately benefit from HBO. Biochemical markers of neuronal injury have been proposed to be used as indicators of brain dysfunction and thus could help deciding the duration and the number of HBO sessions. Therefore, it is necessary to have an objective neurobiomarker for brain injury to support the decision of HBO therapy.
Numerous previous studies assessed many biomarkers (e.g. S-100β, glial fibrillary acidic protein, and neuron-specific enolase) for the prediction of DNS occurrence. 23,34 The S-100β protein is a calcium-binding protein found in high concentrations in astroglial cells. It regulates calcium-dependent signaling in neuronal differentiation, outgrowth, and apoptosis. It is considered as an indicator of glial activation and/or death in many disorders of the CNS. 35 The hypoxia and ischemia induced by acute CO poisoning activate gelatinase A and B. This results in degradation of the tight junctions of endothelial cells and basal lamina. Consequently, the blood–brain barrier is impaired, facilitating the passage of S-100β to peripheral blood. 36 –39
Several studies reported S-100β as a neurobiochemical marker of brain damage in traumatic brain injury, intracranial hemorrhage, stroke, and cardiac arrest. 36,40 Also, previous studies have evaluated S-100β level as an indicator of the severity of CNS affection in CO-poisoned patients. 34,36,41 –45 However, only a few studies 46,47 have investigated its utility as an indicator of HBO therapy that could decrease the incidence of DNS. Therefore, the current study aimed to evaluate serum S-100β level in patients presenting with acute CO poisoning as an indicator of poisoning severity as well as a predictor of DNS occurrence and the need of HBO therapy in those patients.
Patients and methods
Study design
This prospective cohort study was conducted on CO-poisoned patients admitted to Tanta University Poison Control Center (TUPCC) from the first of January 2018 to the end of March 2019. The study was approved by the Research Ethics Committee of Tanta Faculty of Medicine (approval number was 33208). Written informed consent was obtained from each patient and from relatives of unconscious patients. Confidentiality of the data was ensured by assigning a code number to each patient that was known only to the investigators.
Eligibility criteria
Inclusion criteria
All patients of both sexes above 18 years with acute CO poisoning were included in this study. Diagnosis of acute CO poisoning was based on the history of CO exposure as well as symptoms and signs of poisoning; confirmation was obtained by elevated COHb levels above 3% in nonsmokers and 10% in smokers. 34
Exclusion criteria
Patients were excluded from the study if they had the previous history of any disease that can cause serum S-100β elevation such as traumatic head injuries, status epileptics, and Parkinsonism. Additionally, patients with neuropsychiatric disorders, systemic comorbidities (e.g. heart, lung, liver, and kidney diseases), fire accidents, chronic CO poisoning, or co-ingestion of other drugs were excluded. Patients presenting after 6 h of CO exposure were also excluded.
Data collection
The data collection sheet consisted of three sections. The first section recorded the demographic characteristics (age and sex), as well as the medical and toxicological history of the patients (delay period between exposure and hospital arrival and duration of exposure).
The second section contained the findings of physical examination and the results of laboratory investigations.
The third section included the treatment received, the duration of hospital stay, and patients’ outcome on follow-up. Blood COHb level on admission was measured by using pulse CO-oximeter (Masimo SET Rainbow, Irvine, California, USA).
The severity of CO poisoning was graded as mild, moderate, and severe. Mild poisoning was characterized by COHb level above 10% without clinical manifestations of poisoning. In moderate poisoning, the COHb level was above 10%, but under 20–25%, with minor clinical manifestations (such as headache, lethargy, and fatigue). Severe cases presented with COHb level above 20–25% with disturbed consciousness level, or signs of cardiac ischemia, or both. 48
Treatment included administering 100% normobaric oxygen by a non-rebreather mask and/or providing HBO therapy. Indications for HBO therapy were unconsciousness on admission, seizures, persistence of neurological deficits after 4 h on normobaric oxygen treatment, metabolic acidosis, elevated COHb level >25%, and evidence of myocardial ischemia. 33,36 The decision to refer patients to HBO therapy was made by the treating physicians at TUPCC and not the authors.
Discharged patients were followed up for 6 months to observe the development of DNS manifestations such as gait disturbance, memory deficits, personality changes, dementia, learning difficulties, psychosis, depression, Parkinsonism, as well as disorders of behavior, attention, and concentration. 4,49,50 The symptoms of DNS were recorded in a simple questionnaire including leading questions.
Blood sample and biochemical investigations
On hospital admission and before receiving treatment, blood samples were collected from each patient for blood gas analysis and S-100β assay. The collection of blood samples took place within the first 6 h after exposure to CO as S-100β has a half-life of 6 h and is readily excreted in the urine. 51 Consequently, sampling was recommended to be within this time window. 52,53 Quantification of human S-100β was performed in accordance with the direction of the manufacturer by double-antibody sandwich enzyme-linked immunosorbent assay (ELISA) using human S1-00β ELISA kit (Shanghai Sunred Biological Technology Co., Ltd, Shanghai, China). The results were expressed as µg/L. Previous studies have reported that S-100β level of 0.10 µg/L is the reference value above which injury to neural tissue is detected. 54
Statistical analysis
The collected data were organized and statistically analyzed using SPSS software statistical computer package for windows, version 22. Categorical data were summarized as number and percentage. For quantitative data, the Shapiro–Wilk test for normality was performed. For normally distributed data, values were expressed as mean ± standard deviation. A comparison between the two groups was done using an independent-samples t test. One sample T test was used to compare S-100β level in studied patients to reference value for detection of injury to neural tissue (0.1 μg/L). Data that were not normally distributed were summarized as the median and interquartile range (expressed as 25th–75th percentiles). Comparison between two groups was done using the Mann–Whitney test and correlation was performed using Spearman’s rank-order correlation. Receiver operating characteristic (ROC) curves for predicting the occurrence of DNS were generated. Accuracy, sensitivity, and specificity were calculated for S-100β, COHb, and traditional criteria of HBO therapy and compared using Cochran’s Q test followed by post hoc Dunn’s test. Significance was adopted at p < 0.05 for interpretation of the test results. 55
Results
Table 1 summarizes the characteristics of the studied CO-poisoned patients. Their ages ranged from 18 to 63 years, with a median value of 23 years. Women outnumbered men (60% vs. 40%). The reported duration of exposure to CO ranged from 0.5 h to 3.5 h, with a median of 1 h. The duration that elapsed before admission to hospital and receiving medical care varied widely among the cases (0.5–6 h), with a median of 1.5 h. On presentation to TUPCC, the Glasgow coma scale (GCS) ranged from 10 to 15, with a median value of 15. Forty-four percent of patients presented with some degree of disturbed consciousness level. Hypotension was observed in 36% of patients on admission. Laboratory investigations—done on admission—revealed that 42% of patients had metabolic acidosis. In addition, COHb levels ranged between 4.5 g/dL and 54 g/dL, with a median of 16 g/dL. According to clinical manifestations and COHb level, 36% of cases had mild poisoning, while moderate and severe grades of poisoning were found in 32% of cases each. Serum S-100β levels on admission ranged from 0.23 µg/L to 0.87 µg/L, with an average of 0.54 ± 0.17 µg/L. The duration of hospital stay ranged from 3 h to 32 h, with a median of 9.3 h. Half the cases received HBO therapy. Follow-up for 6 months after discharge showed that DNS manifestations were reported by six patients (12%).
Characteristics of studied CO-poisoned patients (n = 50).

GCS: Glasgow coma scale; COHb: carboxyhemoglobin; DNS: delayed neurological sequels; IQR: interquartile range; HBO: hyperbaric oxygen; SD: standard deviation.
The reported DNS manifestations included headache and vertigo (six cases, 12%), decreased concentration (four cases, 8%), impaired memory (four cases, 8%), impaired hearing (four cases, 8%) as well as disorders of mood (four cases, 8%) and sleep (four cases, 8%).
Figure 1 demonstrates that S-100β level was significantly higher (p < 0.001) in the studied patients compared to the reference value for the detection of injury to neural tissue (0.10 μg/L).

Comparing mean of S-100β as a cutoff for neural injury reported in other articles to the mean of the studied sample [S-100β level was significantly higher (p < 0.001) in the studied patients compared to the reference value for detection of injury to neural tissue (0.10 μg/L)].
Table 2 compares S-100β levels among different categories of clinical presentation and DNS in the studied patients. The mean S-100β levels were significantly higher in patients with disturbed consciousness (mean: 0.65 vs. 0.46 µg/L; p < 0.001), metabolic acidosis (mean: 0.66 vs. 0.47 µg/L; p < 0.001), hypotension (mean: 0.62 vs. 0.50 µg/L; p = 0.014), and DNS (mean: 0.73 vs. 0.52 µg/L; p < 0.001).
Serum S-100β levels in the studied patients according to clinical signs and DNS (n = 50).

DNS: delayed neurological sequels.
a Values are expressed as mean ± standard deviation.
bIndependent-samples t test.
c Significant at p < 0.05.
Table 3 compares COHb levels among different categories of clinical presentation and DNS in the studied patients. The median levels of COHb showed similar significant differences (being higher in patients with disturbed consciousness, metabolic acidosis, and hypotension). However, the median levels of COHb did not differ significantly between patients with DNS and those without (median: 17 vs. 15 g/dL; p = 0.827).
COHb levels in the studied patients according to clinical signs and DNS (n = 50).
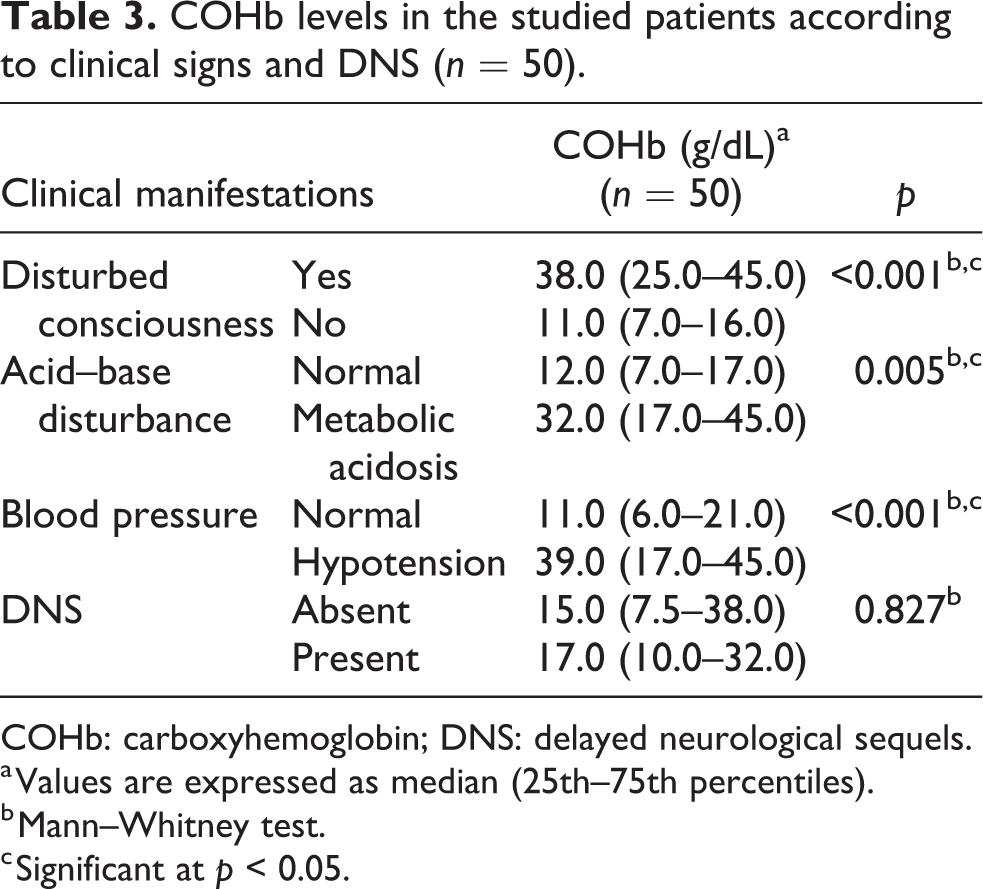
COHb: carboxyhemoglobin; DNS: delayed neurological sequels.
a Values are expressed as median (25th–75th percentiles).
b Mann–Whitney test.
c Significant at p < 0.05.
Table 4 shows the correlation between each of S-100β and COHb levels on one side and other relevant patients’ variables. A moderate, negative, significant correlation was found between GCS and each of S-100β (r = −0.640, p < 0.001) and COHb (r = −0.736, p < 0.001). Weak/mild correlations (r = 0.3 or less) were found between each of the duration of exposure, pre-hospitalization period, and hospital stay with S-100β and COHb levels.
Spearman’s correlation between S-100β and other relevant variables (n = 50).

COHb: carboxyhemoglobin; rs: correlation coefficient of Spearman’s rank-order correlation; GCS: Glasgow coma scale.
a Significant at p < 0.05.
Figure 2 demonstrates the ROC curve analysis of serum S-100β level as a predictor of DNS. The area under the curve (AUC) was 0. 871 (95.0% confidence interval = 0.746–0.949), p < 0.001. At a cutoff value ≥ 0.67 µg/L, serum S-100β level had a sensitivity of 100% (was able to predict all cases that had DNS) and a specificity of 77.3% (identified correctly 77.3% of cases that did not develop DNS).
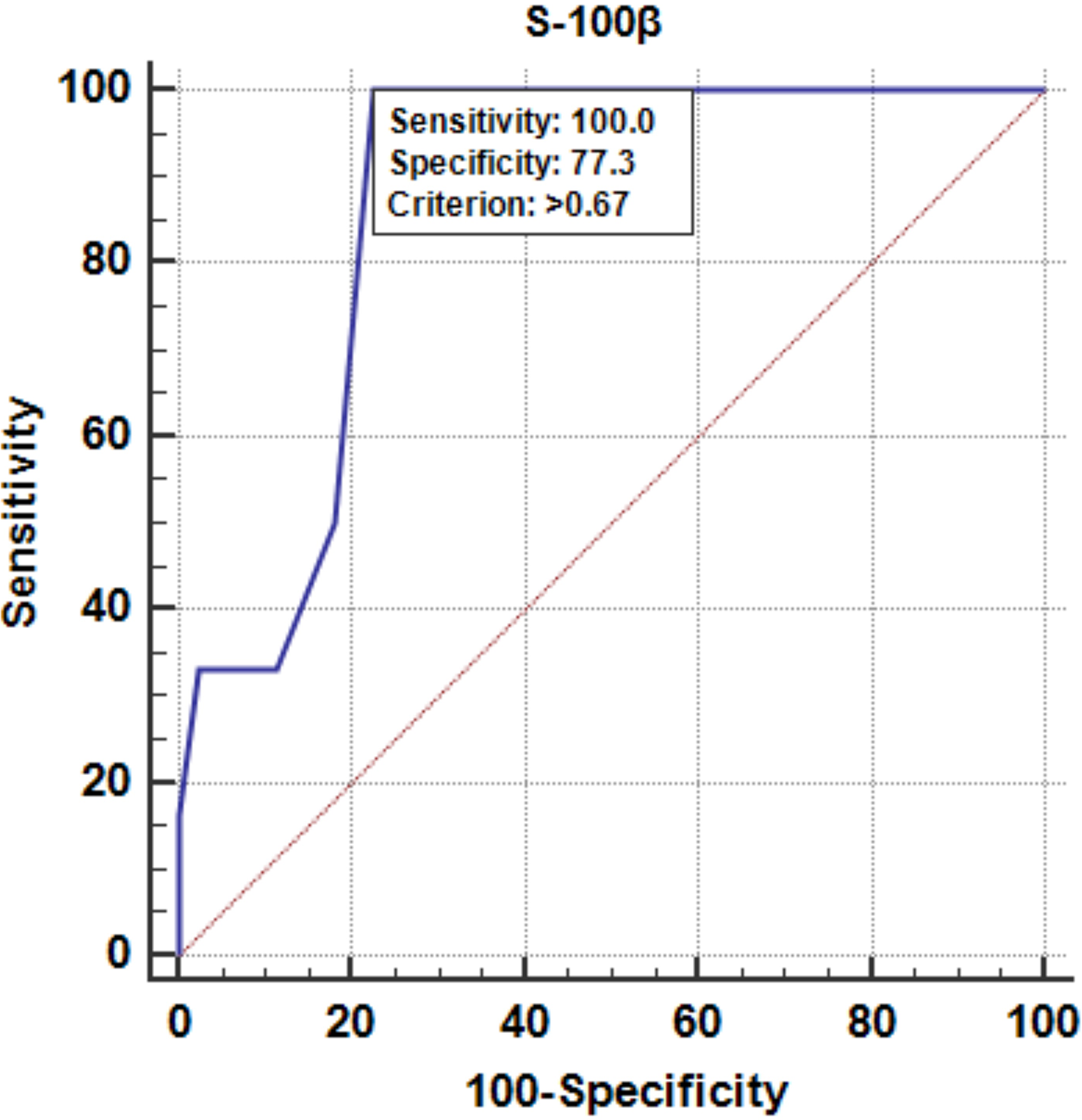
Receiver operating characteristic curve analysis of serum S-100β level as a predictor of delayed neurological sequels. Area under the curve = 0.871. Confidence interval: 0.746–0.949, p < 0.001*, sensitivity was 100.0% (was able to predict all cases that had delayed neurological sequels) and specificity was 77.3% (identified correctly 77.3% of cases that did not develop delayed neurological sequels) at a cutoff value ≥0.67 μg/L.
Table 5 compares the sensitivity, specificity, and accuracy of COHb level alone, indications for HBO therapy, and S-100β level as predictors of DNS occurrence. The S-100β level had the highest sensitivity, specificity, and accuracy compared with COHb alone or the combined indications of HBO therapy. The sensitivity of S-100β was significantly higher than that of COHb (p = 0.043) but insignificantly higher than that of HBO indications (p = 0.662). The specificity and overall accuracy of S-100β level were significantly higher than those of HBO indications (p = 0.002 and 0.001) and insignificantly higher than those of COHb level (p = 1.000 and 0.231).
Accuracy, sensitivity, and specificity of HBO indications, COHb level alone, and S-100β level for prediction of DNS (total n = 50).

HBO: hyperbaric oxygen; COHb: carboxyhemoglobin; ROC: receiver operating characteristic; DNS: delayed neurological sequels.
ap value from Cochran’s Q test.
b p value from post hoc Dunn’s test; p1: comparison between COHb level and HBO indications; p2: comparison between COHb level and S-100β level; p3: comparison between HBO indications and S-100β level.
c Significant at p < 0.05.
Discussion
Although HBO therapy is indicated in cases of acute CO poisoning and is expected to decrease the rate of DNS, some patients suffer from these complications even after receiving multiple sessions of HBO. We hypothesized that the criteria used as indicators for HBO therapy cannot effectively predict the occurrence of such complications and, consequently, HBO is not prescribed for some patients who may benefit from such therapy. The present study aimed to evaluate serum S-100β level in patients presenting with acute CO poisoning as an indicator of poisoning severity and predictor of DNS occurrence and HBO need in those patients.
The results of the present study showed that the sex distribution and the median of both the patients’s age and the duration of exposure to CO were more or less in agreement with findings in the majority of studies, both in Egypt and across the world. 36,41,43,56
Half the cases in the current study received HBO. Previous studies have shown that HBO exerts its beneficial effect in cases with CO poisoning through several mechanisms, including accelerating the rate of CO elimination from hemoglobin, improving the mitochondrial oxidative metabolism, inhibiting both lipid peroxidation and leukocytic adherence to injured microvessels as well as attenuating immune-mediated DNS. 57
Follow-up of our patients for 6 months after discharge showed that DNS manifestations were reported by six patients (12%), which is nearly similar to the rates (14–16.5%) reported by previous studies. 46,47,58,59 However, both higher rates (24.1–26%) 21,60 and lower rates of DNS (9.8%) 23 were also found in other studies. These discrepancies in the incidence of DNS among the studies may be due to various factors such as the age of the studied patients, the duration of exposure to CO, the delay before receiving medical care, and the concentration of the gas.
Determination of COHb levels is traditionally used to diagnose CO poisoning in suspected cases and to determine the severity of poisoning. However, COHb levels were found to be a poor marker of clinical presentation and outcome in those patients. 56,61 –64 This could be attributed to hypoxia-independent inflammation of the brain. 26,61 In addition, the elimination half-life for CO is 50–70 min on 100% oxygen. 65,66 Consequently, COHb levels fall quickly after removal from the site of exposure, and most likely are decreased considerably by the time the measurement is done after reaching the health-care facility. Moreover, even low or moderate COHb levels can trigger pathophysiologic processes that result in brain damage and morbidity. Hence, new markers are needed to determine the clinical severity and the prognosis in cases of CO poisoning.
In the current study, serum S-100β level on admission ranged from 0.23 µg/L to 0.87 µg/L, with an average of 0.54 ± 0.17 µg/L, and this level was significantly higher than the reference value for detection of injury to neural tissue (0.1 μg/L). This result is in accordance with previous studies 41,43 that reported an elevation of serum S-100β levels in patients with acute CO poisoning. However, Brvar et al. 36 found that serum S-100β level was elevated only in patients who experienced disturbed consciousness following CO exposure, while those with intact consciousness had normal serum levels.
Our results revealed that both S-100β and COHb levels correlated with clinical manifestations (disturbed consciousness, hypotension, and metabolic acidosis). However, only S-100β level was found to be significantly elevated on admission in cases that developed DNS later.
We found that the mean S-100β level was significantly higher in patients with disturbed consciousness and it correlated negatively and significantly with GCS. This finding is in line with previous studies that reported higher serum levels of S-100β in CO-poisoned patients with disturbed consciousness. 34,36,41,43,46 However, Rasmussen et al. 44 reported the lack of significant increase in serum S-100β level in CO-poisoned patients with a GCS less than 13. However, their study was conducted on a small sample size, 20 patients, which may impact their results. In addition, other factors may cause this contradictory result, such as the sampling time, as serum S-100β level is recommended to be measured within the first 6 h after neural insult. 52,53
In agreement with our results, Park et al. 47 found a nonsignificant increase in COHb and a significant increase in serum S-100β protein levels in patients who developed DNS. Moreover, Pepe et al., 21 Moon and Kim, 60 Kudo et al., 58 and Kitamoto et al. 59 found a lack of correlation between COHb level and DNS. Also, Di et al. 67 and Hassan et al. 46 reported that serum S-100β levels were significantly higher in patients with DNS. Kim et al. 23 found that serum S-100β protein level was independently associated with DNS. However, Ide et al. 45 and Rasmussen et al. 44 argued that serum measurements of S-100β protein could not be used for predicting CO-induced DNS as the blood–brain barrier is not compromised. They suggested that measuring S-100β level in the cerebrospinal fluid is more accurate. Nevertheless, the affection of blood–brain barrier in cases of acute CO poisoning has been demonstrated by Yang and Rosenberg 39 as mentioned before.
Previous studies have observed that elevated S-100β levels were decreased by HBO therapy. 42,43 Therefore, elevated S-100β level has been suggested to be used as a potential additional criterion for HBO in CO-poisoned patients with disturbed consciousness. 68 The present study has evaluated serum S-100β level as a predictor of DNS. The AUC was 0. 871; at a cutoff value ≥ 0.67 µg/L, serum S-100β level had a sensitivity of 100% and a specificity of 77.3%. We compared the sensitivity, specificity, and accuracy of serum S-100β levels against the traditional criteria of HBO and COHb level as predictors of DNS.
A few previous studies have examined the utility of S-100β as a predictor of DNS development and their results indicated a very good to excellent discriminatory power, but with varying cutoff values among the different studies along with their associated sensitivities and specificities. Park et al. 47 reported that a serum S-100β level above 0.165 µg/L independently predicted the development of DNS after acute CO poisoning, with 90% sensitivity and 87% specificity.
Knapik et al. 69 assessed the utility of plasma S-100β in predicting death or permanent neurological damage on admission to the intensive care unit in patients with various neurological disorders. All patients with plasma S-100β levels over 0.532 μg/L on ICU admission developed a permanent neurological deficit or died. They found S-100β to be a good marker (AUC = 0.82) for the prediction of permanent neurological deficit. However, the sensitivity for S-100β at a cutoff value of 0.27 µg/L was only 68%, while the specificity was 89%.
In addition, Hassan et al. 46 conducted an ROC curve analysis to examine serum S-100β as a predictor of DNS and found that S-100β is an excellent predictor (AUC = 0.990), with 87.5% sensitivity and 100% specificity at a cutoff value >18.9 Pg/L. Kim et al. 23 found S-100β to have an AUC of 0.85 with 90% sensitivity and 84.8% specificity at a cutoff value above 0.103 µg/L.
The current study went further than the previous aforementioned studies by comparing the discriminatory power of S-100β level as a predictor of DNS occurrence against the already established criteria of HBO therapy. We found that S-100β had significantly higher sensitivity than that of COHb and significantly higher specificity and overall accuracy than those of HBO indications. These results suggest that using serum S-100β level as a criterion for HBO therapy—rather than depending on the traditional criteria of HBO—can significantly decrease the rate of DNS after acute CO poisoning.
In conclusion, serum S-100β level on admission, in CO-poisoned patients, could be a measure of poisoning severity and a predictor of DNS development. Prediction of DNS can help selecting the patients who will benefit most from HBO therapy.
The present study bears some points of strength, being a prospective study, one of the few studies that examined the predictive ability of S-100β for DNS occurrence and the first study to compare this marker to traditional indications of HBO therapy. However, the study was also subject to some limitations. The sample size was relatively small. Also, serum S-100β protein levels could be impacted by its extracranial sources in fat, skin, and muscle; thus, false-positive results may be obtained by immunoassay in some patients. 70 The rate of DNS was based on manifestations reported by the patients during the follow-up period and was not confirmed by specialized neuropsychiatric examination.
Footnotes
Acknowledgment
The authors thank the staff members of Tanta University Poison Control Center for their support and help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
