Abstract
Herbacetin (HBN) is a glycosylated flavonoid, which possesses numerous pharmacological properties. Cyclophosphamide (CYC) is a chemotherapeutic drug that adversely affects the kidneys. The present investigation aimed to evaluate the curative potential of HBN against CYC-induced nephrotoxicity. Sprague Dawley rats (n = 48) were randomly divided into four groups: control (0.1% DMSO + food), CYC (150 mg/kg b.wt.), CYC+HBN (150 + 40 mg/kg b.wt.), and HBN (40mg/kg b.wt.). CYC treatment significantly decreased the activities of antioxidant enzymes such as catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione reductase (GSR) while elevating the concentration of reactive oxygen species (ROS) and malondialdehyde (MDA). Treatment with HBN significantly recovered the activity of CAT, SOD, GPx, and GSR while reducing the concentrations of ROS and MDA. Moreover, an increase in the level of renal functional markers, including Urea, creatinine, kidney injury molecule-1 (KIM-1), and neutrophil gelatinase-associated lipocalin (NGAL), and a decrease in creatinine clearance after CYC administration was recovered to control values by HBN treatment. Furthermore, HBN treatment normalized the increased levels of inflammatory markers such as nuclear factor kappa-B (NF-κB), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) after CYC administration. Besides, HBN administration increased the expression of anti-apoptotic markers (Bcl-2) while decreasing the apoptotic markers (Bax and Caspase-3). Furthermore, HBN decreased the activities of tricarboxylic acid (TCA) cycle enzymes (ICDH, αKGDH, SDH, and MDH) as well as renal mitochondrial respiratory-chain complexes (I-IV) and repolarized mitochondrial membrane potential (ΔΨm
Introduction
Cancer is a global menace that accounts for 7.6 million (13%) deaths worldwide, and this situation is predicted to further worsen by the end of 2030 as the deaths due to cancer may cross 13.1 million. 1 Undeniably, the discovery of new drugs for cancer therapy or even the improvement of available drugs may be helpful in dealing with such deathful crises all around the world. Cyclophosphamide (CYC) is used as one of the leading drugs for the medication of cancer with remarkable effects on the immune system. 2 It is prescribed to treat patients with various types of cancer, such as lymphoma, multiple myeloma, leukemia, ovarian cancer, breast cancer, small cell lung cancer, neuroblastoma, and sarcoma. 3 Furthermore, CYC is used to cure multiple sclerosis, lupus erythematosus, and rheumatoid arthritis. 4 Additionally, CYC is widely used in organ transplantation as well as to suppress the graft rejection due to its potent immunosuppressive properties. 2
Despite its wide clinical uses, CYC often leads to cardiotoxicity, hepatotoxicity, and nephrotoxicity.5–8 Nephrotoxicity is one of the main adverse effects of CYC-induced toxicity, as the kidneys are the main excretory organ of the human body and one of the major target organs for drug toxicity.9,10 The mechanism of CYC-induced toxicity involves its accumulation inside the cell that resultantly declines cellular antioxidant defense and increases the generation of reactive oxygen species (ROS), 11 which resultantly leads to oxidative stress (OS) and causes oxidative damage to cellular macromolecules such as lipids, proteins, and nucleic acids. Moreover, earlier reports have shown that CYC intoxication caused lipid peroxidation (LP), protein oxidation 12 as well as depolarization of mitochondrial membrane potential (MMP) in experimental animals. 13 OS may cause apoptotic cell death by activating mitochondria-dependent and mitochondria-independent apoptotic pathways. CYC exposure may cause tubular necrosis of renal epithelial cells, 5 tubular fibrosis, glomerular congestion, 14 and the release of inflammatory factors, and mediation of inflammatory response which ultimately causes renal dysfunction. 15
Flavonoids are well-known antioxidants that are present in a variety of plants and plant-based products. 16 Among such plant-based antioxidants, herbacetin (HBN; 3,4,5,7,8-pentahydroxyflavone) is a novel bioactive glycosylated flavonoid, 17 which was primarily isolated from a Rhodiola rosea L. and Ephedrae herba L. 18 with reported antidiabetic,19,20 anticancer, 21 antiviral, 22 antioxidant,21,23 and anti-inflammatory activities. 24 Furthermore, HBN has the potential to alleviate neurodegenerative diseases. 25 However, no study evaluated the role of HBN against CYC-induced nephrotoxicity. Thus, by considering the above-mentioned therapeutic effects of HBN, the present study was aimed to explore the antioxidant potential of HBN against CYC-induced nephrotoxicity by assessing antioxidant enzymes, lipid peroxidation (LP), renal functional markers, inflammation, apoptosis, tricarboxylic acid cycle enzymes, mitochondrial respiratory-chain complexes, mitochondrial membrane potential, and renal histology in Sprague Dawley rats.
Materials and Methods
Chemicals
Both CYC (Catalog No: 6055-19-2; Purity: 99%) and HBN (Catalog No: 527-95-7; Purity: 98%) were purchased from Merck, United States of America (USA).
Animals
Adult male Sprague-Dawley rats (170 ± 30 g) were kept in steel cages at standard temperature (22–25°C), humidity (45 ± 5%), and a12 h light/dark cycle in the animal house of the University of Agriculture, Faisalabad. Tap water and food were given ad libitum to experimental animals. All rats were treated in compliance with the European Union of animal care and experimentation (CEE Council 86/609) protocol. The experiment was approved by the Ethical Committee of University of Agriculture, Faisalabad (ETC No: 13701-04/ORIC).
Experimental design
Forty-eight adult male Sprague Dawley rats were divided into four groups, having twelve rats in each, and kept in different cages. Group-1 served as a control that received the vehicle only, and Group-2 received a single injection of CYC (150mgkg−1) intravenously on the first day of the trial, Group-3 was co-administered with CYC (150mgkg−1) and HBN (40mgkg−1) orally for the 7 days whereas rats of group-4 were administered with HBN (40mgkg−1) orally. The 150 mg/kg dose of CYC was used according to previous study conducted by Gunes et al., 26 while the 40 mg/kg dose of HBN was used as per a previous investigation. 27 After 7 days of treatment, rats were anesthetized with ketamine and xylazine mixture (60 and 5 mg/kg, respectively) via the intraperitoneal (i.p) route, dissected, and blood was collected from the Retro-orbital venous plexus. Furthermore, for the estimation of renal function markers (e.g., urea and creatinine), serum samples were obtained from the blood samples of rats and stored at 4°C, while creatinine clearance was determined from urine. Likewise, the right kidney was packed in zipper bags and stored at −80oC for biochemical analysis, while the left kidney was preserved in a 10% neutral formalin buffer solution for histopathological examination.
Isolation of kidney mitochondria
Mitochondria from the renal cells were separated by the method of Mingatto et al. 28 The kidney tissues were removed quickly and homogenized by using a medium labeled I (70 mM sucrose, 250 mM mannitol, 50 mMTris-HCl, 10 mM HEPES, 1 mM EDTA, 120 mMKCl and pH 7.4). Centrifugation of the homogenate was carried out for 5 min at 755×g and obtained homogenate was centrifuged again for 15 min at 13300×g. The medium labeled II (250 mM mannitol, 50 mM Tris-HCl, 70 mM sucrose, 10 mM HEPES and pH 7.4) was used to suspend the resulting pellets and then rinsed two times using the same buffer and centrifuged for 15 min at 13300 × g. The obtained pellets of mitochondria were suspended again in the same media and then used for further analysis.
Biochemical markers
The activity of catalase (CAT) was estimated by using the method of Aebi. 29 Superoxide dismutase (SOD) activity was determined according to the methodology of Sun et al. 30 The glutathione peroxidase (GPx) activity was measured by using the protocol of Lawrence and Burk, 31 whereas for the estimation of glutathione reductase (GSR) activity, the method of Carlberg and Mannervik, 32 was used. The concentration of reactive oxygen species (ROS) was measured by the methodology of Hayashi et al., 33 while the malondialdehyde (MDA) level was determined according to the method of Placer et al. 34
Renal function markers
The kits provided by abcam were used to assess the levels of urea (Cat No. ab83362), creatinine (Cat No. ab65340) and creatinine clearance (Cat No. ab65340) with the help of enzyme-linked immunosorbent assay (ELISA) plate reader (Mark™ microplate absorbance reader1681130). Urinary kidney injury molecule-1 (KIM-1) and neutrophil gelatinase-associated lipocalin (NGAL) were determined according to the manufacturer’s guidelines by using KIM-1 Quantikine ELISA kits and NGAL Quantikine ELISA Kit (R & D Systems China Co. Ltd., Changning, China).
Inflammatory markers
The levels of inflammatory markers such as nuclear factor kappa-B (NF-κB), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), and activities of inducible nitric oxide synthase (iNOS) as well as cyclooxygenase-2 (COX-2) were ascertained with ELISA kit as per the manufacturer’s guidance, BioTek, Winooski, VT, USA.
Ribonucleic acid (RNA) extraction and real-time quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
Primers sequences for the r

RT-qPCR: reverse transcription-polymerase chain reaction; 3β-HSD: 3β-hydroxysteroid dehydrogenase; 17β-HSD: 17β-hydroxysteroid dehydrogenase; StAR: steroidogenic acute regulatory protein.
Tricarboxylic acid (TCA) cycle enzymes
The activity of isocitrate dehydrogenase (ICDH) was measured using the Bernt and Bergmeyer method. 37 Succinate dehydrogenase (SDH) was tested using the Slater and Borner method. 38 Malate dehydrogenase (MDH) activity was measured by the method of Mehler et al. 39 Alpha-ketoglutarate dehydrogenase (α-KGDH) activity was evaluated via following the process of Reed and Mukherjee. 40
Renal mitochondria respiratory-chain complexes
Mitochondrial respiratory-chain complex assay kits (Suzhou-Comin Biotechnology Ltd. China) were used to assess respiratory chain complexes like complex
Mitochondrial Membrane Potential (ΔΨm)
The ΔΨm changes have been evaluated by measuring RH-123 fluorescence quenching under the following conditions: 0.15 mg rat liver mitochondria were added to 0.5 mL buffer (250 mM sucrose, 10 mM HEPES, 100 AM K-EGTA, 2 mM MgCl2, 4 mM KH2PO4, pH 7.4) containing an ADP regenerating system (10 mM glucose and 2.5 U hexokinase). Before rhodamine (50 nM) addition, samples were incubated with 33 nM cyclosporin A, 1 Ag/ml rotenone, and 0.1 mM ADP. Finally, mitochondria were energized by 20 mM succinate in the presence or absence of oligomycin (0.2 AM) to detect membrane potential changes. 41
Histology
The kidneys were fixed in 10% formalin solution for 48 h. Then, specimens were dehydrated in ascending grades of alcohol and embedded in paraffin wax. 4–5 μm thick slices were cut by using microtome and finally stained with hematoxylin-eosin (dissolved in 70% alcohol). Finally, these slides were observed under a light microscope (Nikon, 187842, Japan) at 400X and microphotography was performed by Leica LB microscope connected to Olympus Optical Co. LTD, Japan. Image
Statistical analysis
One-way analysis of variance (ANOVA), was used to analyze the variations in data by using Minitab 19.2.0 Software and CFX Maestro™ was used for the analysis of the results of qRT-PCR. The level of significance was set at p < 0.05. Values are expressed as Mean ± SE (Standard Error) in the tables.
Results
Effect of CYC and HBN on biochemical markers
Mean ± SEM of rats (n = 12/group) biochemical markers in the renal tissues of control, cyclophosphamide treated, cotreated, and herbacetin groups.

Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others.
CYC: cyclophosphamide; HBN: herbacetin.
Effect of CYC and HBN on renal functional markers
Mean ± SEM of rats (n = 12/group) renal functional markers in the control, cyclophosphamide treated, cotreated, and herbacetin groups.

Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others.
CYC: cyclophosphamide; HBN: herbacetin.
Effect of CYC and HBN on inflammatory markers
Mean ± SEM of rats (n = 12/group) inflammatory markers in the renal tissues of control, cyclophosphamide treated, cotreated, and herbacetin groups.

Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others.
CYC: cyclophosphamide; HBN: herbacetin.
Effect of CYC and HBN on pro-or-anti-apoptotic markers
CYC administration substantially (p < 0.05) reduced the gene expression of the anti-apoptotic-marker (Bcl-2), whereas the gene expressions of apoptotic-markers (Caspase-3 and Bax) were considerably (p < 0.05) increased in the CYC-treated group as compared to the control group. However, co-treatment of HBN with CYC substantially (p < 0.05) reversed the gene expressions of anti-apoptotic and apoptotic markers as compared to the CYC administered group. However, HBN alone treatment displayed standard gene expressions of anti-apoptotic and apoptotic markers with no remarkable changes, which were comparable to the control group (Figure 1). Effect of cyclophosphamide and herbacetin on the renal pro (Bax, and Caspase-3)- or-anti-apoptotic (Bcl-2) markers. Values are expressed as Mean ± SEM (12 rats per group). Significant differences displayed at p < 0.05.
Effect of CYC and HBN on tricarboxylic acid cycle enzymes
Mean ± SEM of rats (n = 12/group) tricarboxylic acid cycle enzymes in the renal tissues of control, cyclophosphamide treated, cotreated, and herbacetin groups.

Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others.
CYC: cyclophosphamide; HBN: herbacetin.
Effect of CYC and HBN on renal mitochondrial respiratory chain complexes
Mean ± SEM of rats (n = 12/group) renal mitochondrial respiratory chain complexes and ΔΨm in the control, cyclophosphamide treated, cotreated, and herbacetin groups.

Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others.
CYC: cyclophosphamide; HBN: herbacetin.
Effect of CYC and HBN on ΔΨm
Rats treated with CYC showed a significant (p < 0.05) depolarization of ΔΨm as compared to the control group. Nonetheless, the co-treatment of HBN with CYC restored the loss of ΔΨm as compared to the CYC-exposed rats. However, HBN alone treated group showed normal ΔΨm as in the control group (Table 6).
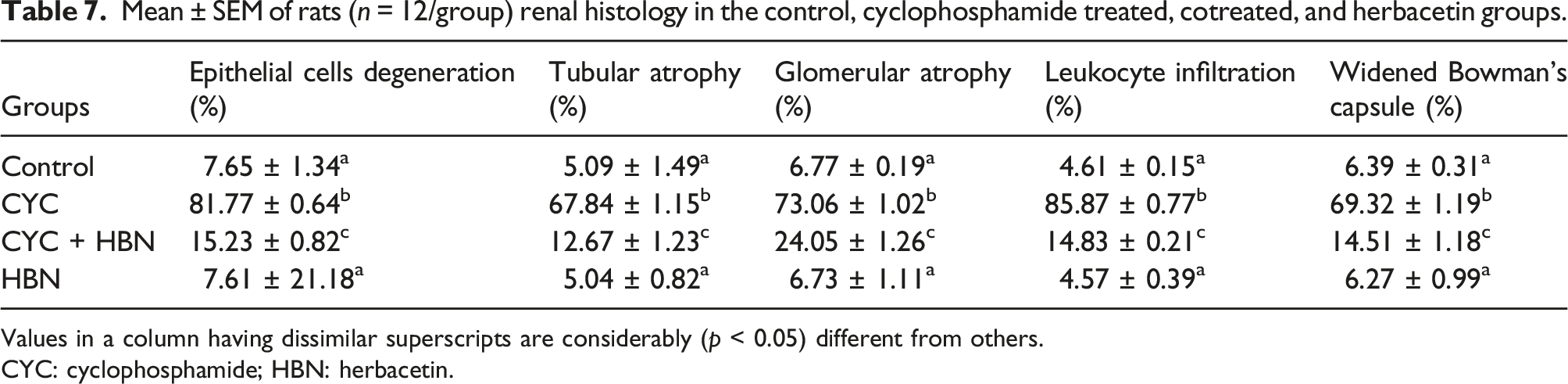
Effect of CYC and HBN on renal histology
Figure 2 and Table 7 presents the effects of CYC and HBN on the histological profile. CYC treatment-induced degeneration in epithelial cells and leukocyte infiltration, as well as leading to tubular and glomerular atrophy, widened Bowman’s capsule and thickened the basement membrane of Bowman’s capsule as compared to the control group, indicating the CYC-induced nephrotoxicity. Nevertheless, HBN co-administration with CYC reduced the renal damage with improved glomerular degradation, inflammatory cell infiltration, and normal renal tissues as compared to the CYC-induced group. However, normal histological features were observed in the HBN alone treated group as in the control group. (A) Control group; displaying normal histological structure (H& E; 400X), (B) CYC treated group; showing dilatation, vacuolation, degeneration and widened Bowman’s capsule necrosis in the kidney tissues (C) CYC + HBN treated group showing improved histoarchitecture with reduced degenerative alterations in renal epithelium and renal tubules (D) HBN treated group showing normal renal histoarchitecture. CYC: Cyclophosphamide; HBN: Herbacetin. Mean ± SEM of rats (n = 12/group) renal histology in the control, cyclophosphamide treated, cotreated, and herbacetin groups. Values in a column having dissimilar superscripts are considerably (p < 0.05) different from others. CYC: cyclophosphamide; HBN: herbacetin.
Discussion
CYC is a primary antineoplastic and immunosuppressive agent that adversely affects different organs, 42 particularly kidneys. 43 During the course of excretion through the urine, CYC metabolites cause nephrotoxicity. 44 There are two reported cytotoxic metabolites of CYC; phosphoramide mustard and acrolein. Phosphoramide mustard is believed to have an antineoplastic activity, while acrolein, a highly reactive metabolite with a short biological half-life, that is responsible for CYC-induced renal injury.45,46 However, HBN treatment shielded the renal tissues against adverse effects of CYC and lessened oxidative stress (OS). According to Wei et al., 47 HBN has significant potential against OS–induced toxicities due to its antioxidant, anti-inflammatory, and anti-cancer properties. Therefore, it is assumed that HBN might play a vital role as a pharmacologic agent for averting the toxic effects of CYC.
CYC administration significantly reduced the activities of CAT, SOD, GPx, and GSR while increasing the concentration of ROS and MDA levels. Imbalance in ROS and antioxidant defense systems leads to OS, which ultimately results in oxidative damage.48,49 An increase in free radicals causes overproduction of MDA, which is an indicator of lipid peroxidation. 50 LP is commonly known as a marker of OS and antioxidant status. 51 SOD, CAT, and GPx are the main enzymes participating in ROS elimination. SOD converts O−2 into hydrogen peroxide, which is the primary stage antioxidant pathway. CAT and GPx catalyze the conversion of hydrogen peroxide into water and oxygen. 52 GPx degrades hydrogen peroxide into water and oxygen by converting reduced glutathione (GSH) into glutathione disulfide (GSSG). 53 On the other hand, GSH functions as an electron donor in these reactions. The concentration of GSH is retained by GSR. 54 The increased production of ROS and OS is known to play a significant role in CYC-induced renal toxicity.55,56 Furthermore, the reaction of acrolein with GPx leads to the low level of intracellular glutathione, which causes OS. 55 Our findings suggested that HBN alleviated OS and increased the antioxidant enzymes activities such as CAT, SOD, GPx, and GSR, while decreased the concentration of ROS and MDA levels. According to Cho et al., 57 the presence 8-OH group in the structure of HBN may attribute to its antioxidant activity.
CYC administration caused considerable elevation in the levels of urea, creatinine, KIM-1, and NGAL while decreasing the creatinine clearance. Elevated plasma levels of urea and creatinine reflect the glomerular filtration rate (GFR). 58 Decreased creatinine clearance indicates chronic kidney disease or severe kidney damage, 59 whereas KIM-1 and NGAL are the biomarkers of acute kidney injury (AKI). 60 KIM-1 is a transmembrane protein that is a universally accepted marker for the initial diagnosis of AKI. 61 Under normal physiological conditions, urine does not contain KIM-1, but its presence in the urine indicates proximal tubular injury. 62 Likewise, NGAL is a cytosolic protein: its significant amount in the blood, urine, renal, and proximal-distal tubules is detected only after renal injury and ischemia. 63 Hence, increased levels of these markers indicate renal damage. However, HBN treatment significantly mitigated all these toxic changes in the levels of renal markers by its free radical scavenging potential.
NF-κB is a transcription factor in several tissues that control the immune response and multiple inflammatory disorders. NF-κB plays a pivotal role in activating pro-inflammatory cytokines, including TNF-α, IL-1β, COX-2, IL-6, and iNOS.64,65 NF-κB gets activated in CYC-treated tissues due to OS, which as a result causes tissue damage through the production of pro-inflammatory cytokines including IL-1β, TNF-α, and IL-6. 56 Furthermore, iNOS and COX-2 enzymes are two major inflammatory markers that control the inflammatory response by producing nitric oxide and prostaglandin E2, respectively. 66 Nitric oxide over-expression renders the cell susceptible to ROS by lowering the activity of intracellular glutathione. 67 Thus, a drug that inhibits the expression of COX-2 and iNOS may have tremendous potential for treating chronic inflammation. 68 In the present research, it was observed that HBN significantly suppressed the levels of NF- κB, IL-6, TNF-α, and IL-1β as well as the activities of COX-2 and iNOS in CYC-treated rats, which may be attributed to its ring structure as supported by the previous investigation, which stated that the anti-inflammatory effects of flavones are due to the methoxylation of the 5- or 7-hydroxyl groups on the A-ring or non-methoxylation of the 3′-hydroxyl groups on the B-ring. 69
Apoptosis is also known as a mechanism of programmed cell death. 70 The entire process is categorized into intrinsic and extrinsic pathways. 71 A class of proteins belonging to the Bcl-2 family regulates the overall factors involved in this pathway. This family of proteins is either classified into pro-apoptotic (Bax) and anti-apoptotic (Bcl-2) proteins. Bax triggers the discharge of cytochrome c into the cytosol via the mitochondrial inter-membrane permeability, while Bcl-2 inhibits the liberation of cytochrome c. Thus, Bcl-2 plays a critical role in the event of apoptosis by regulating the induction and prevention of cytochrome c discharge. 72 Reduction in Bcl-2 and elevation in Bax adversely alter the mitochondrial inter-membrane permeability, resulting in a rise in the liberation of cytochrome c within the cytosol. 73 This augmented cytochrome c in cytosol ultimately activates the expression of Caspase-3. 74 Caspase-3 is a highly activated cysteine protease in the caspase family, which performs an essential part in both apoptotic pathways, including extrinsic or intrinsic pathways. Therefore, Caspase-3 is considered as a reliable bio-indicator for cellular apoptosis. 75 Outcomes of the investigation showed that CYC exposure lowered the gene expression of Bcl-2 while boosting the gene expression of Bax and Caspase-3. Nevertheless, co-administration of HBN downregulated the gene expression of Bax and Caspase-3 and renovated the gene expression of Bcl-2. Our outcomes verify the assumption that the tissue-shielding impact of HBN on apoptosis might be, at least, mediated by regulating the Bcl-2/Bax ratio.
Mitochondria are the most important subcellular organelles that produce energy and are susceptible to OS. 76 Mitochondrial enzymes (ICDH, αKGDH, SDH, and MDH) catalyze the oxidation of substrate by the TCA cycle producing reducing-equivalents. These reducing equivalents are channeled through the respiratory chain for adenosine triphosphates (ATP) production through oxidative phosphorylation. 77 Constant energy supply is considered an important necessity for the proper functioning of tissues and a reduction in the activity of TCA cycle enzymes shows the defect in pyruvate aerobic oxidation that could lead to low ATP production. 78 The present study revealed that CYC reduced the TCA cycle enzyme activities. A decrease in the activities of TCA cycle enzymes could be due to CYC-induced membrane destabilization. However, in the present study, co-treatment of HBN with CYC reversed the enzyme’s activity. Decreased production of ROS might be responsible for the restoration of enzymatic activities. Outcomes of the study are further in compliance with the findings of Ijaz et al., 79 in which morin, a close relative of HBN, significantly reversed the activities of mitochondrial enzymes potentially due to its antioxidant activity.
The administration of CYC induced a significant decline in the activity of mitochondrial complexes (I–IV) of the electron transport chain (ETC). ETC is considered as the key site of ROS generation under physiological conditions. 80 Previous data revealed that the accumulation of intracellular ROS is the main reason for the dysregulation of mitochondrial ETC. 81 Once the transmission of the mitochondrial ETC is blocked, the synthesis of ATP is affected and this condition is induced due to mitochondrial damage. 82 NADH dehydrogenase, a flavin-linked dehydrogenase, transmits electrons from NADH to Coenzyme Q. The decrease in its activity may be due to the reduction of reducing equivalents that are used to form glutathione to prevent oxidative damage in mitochondrial components. 83 Mitochondrial dysfunction is an important factor in renal dysfunction. 84 Our results are consistent with the results of Sudharsan et al., 85 who revealed that CYC treatment decreased the level of mitochondrial complexes. However, the co-administration of HBN restored the levels of ETC complexes due to its ameliorative effects.
In the current study, CYC administration caused the ΔΨm collapse. Increased ROS production can disturb the various mitochondrial functions, detected by ΔΨm collapse, mitochondria swelling, and dehydrogenase activity reduction. 86 The ΔΨm is essential for mitochondrial movement, 87 and it has been observed that anterograde movement of the mitochondrial membrane is beneficial in its proper functioning. 88 ΔΨm also plays a key role in maintaining mitochondrial homeostasis. 89 It is reported that the inhibition of ETC decreases proton efflux across the inner membrane of mitochondria and that is generally linked with the depolarization of ΔΨm. 90 Our finding presented that HBN has the potential to reverse CYC-induced ΔΨm loss by increasing the ETC complexes' activities.
CYC treatment-induced degeneration of epithelial cells, leukocyte infiltration, as well as tubular and glomeruli atrophy, widened Bowman’s space and thickened the basement membrane of Bowman’s capsule in the renal proximal tubules when compared with the control group, indicating the CYC-induced nephrotoxicity. Our results are consistent with the study of Caglayan et al., 55 who revealed that CYC treatment showed degenerative changes in tubule epithelium, tubular dilation, glomerular hyperemia, and kidney tissue inter-tubular vessels. However, HBN cotreatment with CYC substantially restored the above-mentioned histopathological damages to renal tissues, which might be due to its antioxidant, anti-apoptotic and anti-inflammatory effects.
Conclusion
In conclusion, outcomes of the present study showed that HBN treatment potentially alleviated the CYC-instigated adverse effects on antioxidant enzymes, urea, creatinine, creatinine clearance, respiratory chain complexes, TCA cycle enzymes, and ΔΨm. Moreover, HBN regulated renal functions by decreasing the overgeneration of ROS and MDA levels. Furthermore, HBN treatment restored the levels of inflammatory, anti-or-pro-apoptotic markers, besides the benchmark histological profile. Hence, HBN ameliorated CYC-induced nephrotoxicity may be due to its antioxidant, anti-inflammatory, and anti-apoptotic properties.
Footnotes
Author contributions
MUI and SM designed the experiment. MUI, SM and HA performed the experiments. RB and HN helped in data interpretation and HA wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
