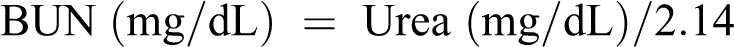Abstract
2,4-Dichlorophenoxyacetic acid (2,4-D) is an extensively used herbicide in the field of agriculture, its ever-escalating use induces toxicity, health effects, and environmental impact. Oxidative stress plays a key role in pathogenesis of 2,4-D-induced liver and kidney damage. Magnesium (Mg) is a highly effective antioxidant agent in restoring oxidative damage by directly influencing the metabolic and physiological processes. Therefore, the present study aimed to evaluate Mg role in ameliorating the oxidative damages provoked by 2,4-D in rat model. Male Wistar rats (180–220 g) were distributed into four groups and treated intragastrically for 4 weeks. Group 1: control, group 2: 2,4-D (150 mg/kg body weight/day), group 3: simultaneously treated with 2,4-D (150 mg/kg body weight/day) and Mg supplement (50 mg/kg body weight/day), and group 4: Mg supplement (50 mg/kg body weight/day). Under experimental conditions, plasma hepatic and renal biomarkers, tissue oxidative status, and antioxidant enzymes activities were investigated. Results demonstrated that 2,4-D intoxication caused hepatic and renal impairments as indicated by the significantly increased (p < 0.001) alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, urea, creatinine, and blood urea nitrogen levels. In addition, 2,4-D caused a significant enhancement (p < 0.001) in the level of malondialdehyde as well as reduction (p < 0.001) of the superoxide dismutase, catalase, and glutathione reductase activities in both hepatic and renal tissues. Mg treatment prevented and reversed the toxic variations induced by 2,4-D. In general, these outcomes suggest that Mg may have antioxidant potential and ameliorative effects against 2,4-D provoking hepatic and renal toxicity in rat model.
Introduction
2,4-Dichlorophenoxyacetic acid (2,4-D) is the most successful selective organic herbicide in phenoxy acids family 1,2 usually used in range land and most notably in agriculture due to its highest toxicity for controlling broadleaf weeds. 3 Unfortunately, this herbicide is not only specific for targeting weeds but also harmful for nontarget species. 4 There are many forms of 2,4-D including esters and alkyl amine salts which results in the production of harmful compounds including chlorophenols or dioxins. 5 These compounds are used as precursors in manufacturing agricultural chemicals, pharmaceuticals, biocides, and dyes. These derivatives get in our surroundings through industrial waste and pesticides 6,7 that may produce toxicological and eco-friendly problems due to its broad spectrum implementations. 8
Phenoxy herbicides stimulate the production of reactive oxygen species (ROS) which is associated with the production of free radicals and direct assault of these phenoxy acid radicals on biochemical perturbations in several sensitive metabolic pathways. 9 2,4-D influencing teratogenic, genotoxic, neurotoxic, immunosuppressive, cytotoxic, hepatotoxic, 10 and nephrotoxic effects have been well-documented. 11 Besides these adverse effects, it relates to an increased risk of non-Hodgkin’s lymphoma, 12 birth defects, 13 and deliberately considering as carcinogenic mediators of some mammalian species. 14,15
Phenoxy acids and its associated precursors are found in many soft tissues, particularly in hepatic and renal tissues. 16 In liver, 2,4-D acts as a toxicological negotiator by interacting with the hepatic cell membrane. As a result, structural deterioration and variation in enzyme effluence and metabolism of the cell components 17 involve in peroxidative damage of hepatic tissues. 10 Kidney is considered as the first targeted organ for toxicological accumulation analyses as its major role for xenobiotics detoxication, biotransformation, and elimination. After the uptake of entire xenobiotics, it is exposed through the portal circulation. 18 2,4-D is quickly eliminated through the kidneys and removed with no change in its form 19 by a kidney transport system. 20 This transport of 2,4-D may precede the delivery of its toxic compound into cells which afterward increases the susceptibility of oxidative stress 21 and the consequent induction of toxic effects in renal tissues. 22
Dietary supplement intake has been gradually escalating during the last decades, in spite of diagnosis nutrient deficiency. 23 In the human body, magnesium (Mg) is considered as the fourth most abundant positive charge ion. 24 It is essential for optimal metabolic activities such as biomolecules synthesis and stability, mitochondrial function, and acts as a cofactor for more than 300 enzymes, mainly in energy-dependent activities in the form of Mg-adenosine triphosphate (ATP) complex. 25 Mg is a cofactor of enzymes which are involved in the synthesis of an essential cellular antioxidant, glutathione reductase (GSH), responsible for maintaining the cell’s redox state. 26 Mg insufficiency is associated with oxidative stress, lipid peroxidation, 27,28 and diminishes glutathione peroxidase (GPX) activity. 29 Conversely, it is well-established that Mg provides protective effects against oxidative stress 30 and lipid peroxidation. 31 It has been suggested that Mg by increasing the levels of reduced GSH 32 and the activity of superoxide dismutase (SOD) 33 reduces the free-radical-mediated peroxidative damages. 34 From the literature review, it is confirmed that Mg treatment plays a beneficial role in the management of pesticide intoxication in both human and animal. 35 –37
Despite the above-mentioned reports regarding the role of Mg in restoring oxidative damage provoked by various pesticides, no experimental data exist against 2,4-D herbicide toxicity. Thus, the present study was planned to assess the ameliorative action of Mg supplementation against 2,4-D-induced hepatic and renal toxicity in the rat model.
Materials and methods
Chemicals
All analytical grade chemicals were purchased from the local suppliers of Pakistan. 2,4-D (Cat # 2094D; CAS No. 94-75-7) was acquired from Research Organics, Inc./Sigma-Aldrich Corporation (Cleveland, Ohio, USA) and Mg sulphate (MgSO4) was obtained from Merck (D-6100; E. Merck, Darmstadt, Germany).
Animals
Forty healthy male albino Wistar rats weighing (180–220 g) were obtained from the International Center for Chemical and Biological Sciences, animal care facility, University of Karachi, Pakistan. The animals were maintained at surrounding temperature (23 ± 4°C) and a 12-h light/12-h dark cycle in polyethylene cages with the saw-dust covered floor. Rats were given ad libitum access to standard pellet chow (a mixture of wheat, alfalfa, soybean, vitamins, and minerals) and water. Before initializing experiment, animals were acclimatized to laboratory condition for 1 week. The current research for the handling and caution of laboratory animals was carried out in agreement with the worldwide acknowledged guiding principle in animal research (Health research, extension act of 1985).
Study design
After the acclimatization period, 40 rats were randomly distributed into four treated groups (n = 10) and provided the following treatment daily for 4 weeks. Group 1 (control): control group, no treatment. Group 2 (2,4-D): 2,4-D group, intragastrically treated with 2,4-D (150 mg/kg body weight/day) dissolved in deionized water.
38
Group 3 (2,4-D + Mg): 2,4-D and Mg-treated group, concurrent intragastrically received 2,4-D (150 mg/kg body weight/day) and Mg supplement (50 mg/kg body weight/day) dissolved in deionized water. Group 4 (Mg): Mg group, treated only with Mg supplement (50 mg/kg body weight/day) dissolved in deionized water by intragastric route.
39
Animals from all experimental groups were monitored regularly with normal activity and weighed individually during the experiment. % Change in body weight of individual rat was determined using this formula 40
Blood and sample collection
Twenty-four hours after last dose, the rats were weighed, anesthetized, and euthanized by cervical dislocation. Blood samples were collected into tubes coated with heparin and centrifuged at 2200×g for 10 min to separate plasma and refrigerated at −80°C until hepatic and renal biomarkers assessment. Liver and kidneys were obtained by surgical operation, washed in chilled saline (0.9% w/v) to eliminate blood contamination, dried, and weighed individually. The liver and kidney weight (organ weight/gram of body weight) of each animal was noted.
Liver and kidney homogenate preparation
Liver tissues (1:10 w/v) were minced and homogenized in chilled potassium chloride buffer (1.17%) and centrifuged (800 × g for 5 min at 4°C) to clear the nuclear fragments. It was again processed to centrifuge (10,500 × g for 20 min at 4°C).
Renal tissues at the ratio of 1:10 (w/v) were homogenized in 100 mM of potassium chloride buffer (pH 7.0) using a Ultra-Turrax T-25 polytron homogenator (IKA-Labortechnik, Germany). All homogenates were then allowed to centrifuge at 600 × g for 1 h at 4°C.
The obtained clear supernatant of both homogenates was kept at −80°C for biochemical assays. 41
Plasma renal biomarkers assessment
Plasma samples were used for renal functional assessment. Urea was measured according to Oxime method as described by Mather and Roland 42 and creatinine by Jeff’s method as described by Spierto et al. 43 Values of plasma urea and creatinine were expressed as mg/dL. Blood urea nitrogen (BUN) values were calculated using mentioned formula
Plasma hepatic biomarkers assessment
Potential biomarkers of liver function, including alkaline phosphatase (ALP), alanine aminotransferase (ALT), and aspartate aminotransferase (AST), were estimated using the diagnostic assay kit (LABKIT, Chemelex, S.A., Canovelles (Barcelona), Spain) as defined by the manufacturer. Results were represented as U/L.
Lipid peroxidation
The malondialdehyde (MDA), a lipid peroxidation indicator was determined according to the earlier described method of Okhawa et al. 44 Thiobarbituric acid reacting substances as a byproduct of lipid oxidative degradation were identified by measuring the absorbance at 530 nm via Shimadzu UV-1900 spectrophotometer. Results were represented as µmol/g of tissue.
Superoxide dismutase
SOD activity in liver and kidney homogenate was assayed by the method of Kono. 45 % Inhibition of the rate of formazan dye, nitroblue tetrazolium-diformazan in the experimental sample was noted per minutes on spectrophotometer (UV-1900; Shimadzu, Japan) at 560 nm. The activity was measured in terms of % inhibition and represented as U/mg of protein.
Catalase
Catalase (CAT) activity in tissues was assessed by the method of Sinha. 46 In brief, dichromate/acetic acid reagent boiled for 10 min in the presence of hydrogen peroxide (H2O2) at 100°C. The absorbance of chromic acetate produced in the reaction mixture was recorded by UV-1900 Shimadzu spectrophotometer at 570 nm. The calculated enzyme activity was expressed as U/mg of protein.
Glutathione reductase
GSH activity was determined by Carlberg and Mannervik. 47 The assay system was consisted of sample, potassium phosphate buffer, reduced nicotinamide adenine dinucleotide phosphate (NADPH), and oxidized glutathione. The individual reaction mixture was mixed gently and noted the absorbance at 340 nm for 5 min on kinetic spectrophotometer (PRIM 500) at 25°C. The enzyme activity was measured by following the molar coefficient for NADPH of 6.22 μmol−1 cm−1. Calculated enzyme activity was expressed as µg/g of tissue.
Statistical analysis
Results were expressed as mean value ± SD (n = 10). Experimental data were analyzed by one-way analysis of variance using IBM (Anmork, New York, USA) statistical package for social sciences, version 19. Tukey’s test was used for multiple comparisons and differences were accepted significant at different levels (p < 0.001, p < 0.01, and p < 0.05).
Results
% Change in body weight and absolute and relative organs weight of control and treated groups
Table 1 presents the % change in body weight and absolute and relative liver and kidney weight of investigational rats. A considerable decrease in % body weight change was found in 2,4-D exposed group than that in the control group (p < 0.001). Likewise, a remarkable decrease in % body weight change was also observed in the 2,4-D + Mg group when compared with the control group (p < 0.001). However, treatment provided with Mg supplement to the 2,4-D exposed group increased the percentage of weight change when compared with 2,4-D alone (p < 0.001).
Change in body weight and absolute and relative organs weight of rats of four treated groups.a

2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; ANOVA: analysis of variance.
a2,4-D: rats intoxicated with 2,4-D; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D; Mg: rats treated with Mg. Values are expressed as mean (±standard deviation); n = 10 rats per group (one-way ANOVA Tukey’s post hoc test).
bp < 0.001 as compared with the control group.
cp < 0.001 as compared with the 2,4-D group.
dp < 0.01 as compared with the 2,4-D group.
Further, the 2,4-D + Mg exposed group showed a significant recovery in absolute liver weight when compared with 2,4-D intoxicated rats (p < 0.01). While no significant changes were observed in absolute kidney weight.
2,4-D exposed rats also displayed a considerable increase in relative liver and kidney weight in comparison with control rats (p < 0.001). However, supplementation with Mg restored both the kidney weight and the relative weight of the 2,4-D + Mg exposed group when compared with the 2,4-D intoxicated group (p < 0.001). On the other hand, a slight reduction in relative kidney weight of the Mg exposed group was observed as compared with the control group (p < 0.001).
Renal function biomarkers of control and treated groups
Table 2 presents the levels of renal biomarkers including plasma urea, creatinine, and BUN of experimental rats. The 2,4-D treatment caused a considerable enhancement in the levels of urea, creatinine, and BUN in comparison with the control group (p < 0.001, p < 0.01). In contrast, the 2,4-D + Mg group showed considerably less elevated levels of urea (p < 0.05) when compared with 2,4-D exposed. Likewise, treatment provided with Mg supplement to the 2,4-D exposed group also showed decreased levels of creatinine when compared to the control and 2,4-D groups (p < 0.001).
Plasma level of kidney indices of rats of four treated groups.a

2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; BUN: blood urea nitrogen; ANOVA: analysis of variance.
a2,4-D: rats intoxicated with 2,4-D; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D; Mg: rats treated with Mg.
Values are expressed as mean (±standard deviation); n = 10 rats per group (one-way ANOVA Tukey’s post hoc test).
bp < 0.001 as compared with the control group.
cp < 0.001 as compared with the 2,4-D group.
dp < 0.05 as compared with the 2,4-D group.
ep < 0.01 as compared with the control group
Hepatic function biomarkers of control and treated groups
Table 3 presents the hepatoprotective effects of Mg supplement on 2,4-D intoxicated rats. In the 2,4-D and 2,4-D + Mg groups, plasma ALP, AST, and ALT levels were remarkably increased when compared with control rats (p < 0.001). However, in the 2,4-D + Mg group reduced levels of ALP, AST, and ALT were observed when compared to the 2,4-D alone group (p < 0.001).
Hepatic enzymes level of rats of four treated groups.a

2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ANOVA: analysis of variance.
a2,4-D: rats intoxicated with 2,4-D; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D; Mg: rats treated with Mg. Values are expressed as mean (±standard deviation); n = 10 rats per group (one-way ANOVA Tukey’s post hoc test).
bp < 0.001 as compared with the control group.
cp < 0.001 as compared with the 2,4-D group.
Hepatic oxidative stress biomarkers of control and treated groups
Hepatic oxidative stress markers (MDA, SOD, CAT, and GSH) of 2,4-D exposed rats treated with or without Mg supplement are illustrated in Figures 1(a) to 4(a). Levels of MDA in the hepatic tissue of rats intoxicated with 2,4-D were found to be considerably (p < 0.001) increased when compared to the control rats. Likewise, in the 2,4-D + Mg group, elevated MDA levels were also noticed than the control (p < 0.01) group. However, in the 2,4-D + Mg group, a remarkable decrease (p < 0.01) in levels of MDA was observed when compared to 2,4-D exposed rats. In addition, a remarkable decline in MDA levels was also observed in the alone Mg group when compared with the control group (p < 0.01) (Figure 1(a)). On the other hand, activity of SOD was remarkably reduced in the 2,4-D exposed group compared with that of control (p < 0.001). However, Mg treatment provided to 2,4-D intoxicated rats showed a remarkable recovery in hepatic activity of SOD when compared with the 2,4-D group (p < 0.001) (Figure 2(a)). In a similar trend, CAT activity was found to be significantly decreased in the 2,4-D exposed group when compared to that the control group (p < 0.001). While, treatment provided with Mg supplement to the 2,4-D group showed remarkable improvement in CAT activity when compared to that of the 2,4-D group (p < 0.05) (Figure 3(a)). GSH activity was reduced significantly as a result of 2,4-D administration than the control group (p < 0.001). But the noticeable elevation of GSH activity was observed in the alone Mg group when compared to control (p < 0.05). However, Mg supplement significantly restored decreased GSH activity of the 2,4-D + Mg group when compared to 2,4-D alone (p < 0.001) (Figure 4(a)).

(a) Hepatic and (b) renal MDA level of rats of four treated groups. Values are expressed as mean (±standard deviation), n = 10 rats per group (one-way ANOVA Tukey’s post hoc test). (a) Hepatic MDA: ap < 0.001 as compared with the control group. bp < 0.01 as compared with the control group. cp < 0.01 as compared with the 2,4-D group. (b) Renal MDA: ap < 0.001 as compared with the control group. bp < 0.001 as compared with the 2,4-D group. 2,4-D: rats intoxicated with 2,4-D; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D; Mg: rats treated with Mg. 2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; MDA: malondialdehyde; ANOVA: analysis of variance.

(a) Hepatic and (b) renal SOD activity of rats of four treated groups. Values are expressed as mean (±standard deviation), n = 10 rats per group (one-way ANOVA Tukey’s post hoc test). (a) Hepatic SOD: ap < 0.001 as compared with the control group. bp < 0.001 as compared with the 2,4-D group. (b) Renal SOD: ap < 0.001 as compared with the control group. bp < 0.001 as compared with the 2,4-D group. cp < 0.01 as compared with the control group. 2,4-D: intoxicated with 2,4-D; Mg: rats treated with Mg; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D. 2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; SOD: superoxide dismutase; ANOVA: analysis of variance.

(a) Hepatic and (b) renal CAT activity of rats of four treated groups. Values are expressed as mean (±standard deviation), n = 10 rats per group (one-way ANOVA Tukey’s post hoc test). (a) Hepatic CAT: ap < 0.001 as compared with the control group. bp < 0.05 as compared with the 2,4-D group.
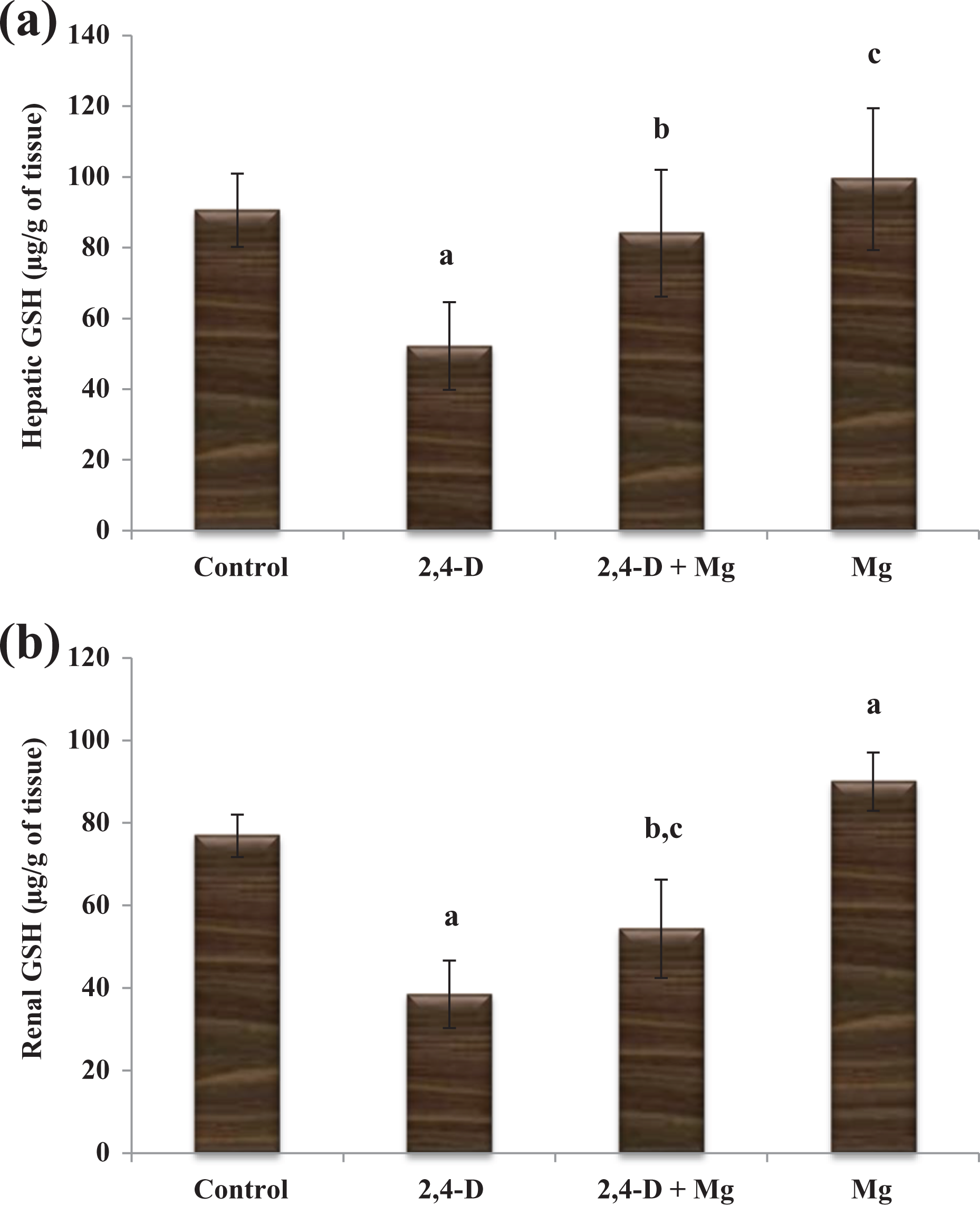
(a) Hepatic and (b) renal GSH activity of rats of four treated groups. Values are expressed as mean (±standard deviation), n = 10 rats per group (one-way ANOVA Tukey’s post hoc test). (a) HepaticGSH: ap < 0.001 as compared to the control group. bp < 0.001 as compared with the 2,4-D group. cp < 0.05 as compared to the control group. (b) Renal GSH: ap < 0.001 as compared to the control group. bp < 0.001 as compared with the 2,4-D group. cp < 0.01 as compared to the control group. 2,4-D: intoxicated with 2,4-D; Mg: rats treated with Mg; 2,4-D + Mg: rats treated with Mg and intoxicated with 2,4-D. 2,4-D: 2,4-dichlorophenoxyacetic acid; Mg: magnesium; GSH: glutathione reductase; ANOVA: analysis of variance.
Renal oxidative stress biomarkers of control and treated groups
Renal oxidative stress markers are shown in Figures 1(b) to 4(b). 2,4-D exposition caused a remarkable enhancement in renal MDA levels of the 2,4-D exposed group in comparison with the control group (p < 0.001). However, the 2,4-D + Mg group showed a remarkable decrease in MDA levels compared to the 2,4-D exposed group (p < 0.001; Figure 1(b)). Renal SOD activity was considerably reduced in the 2,4-D intoxicated group when compared with control one (p < 0.001). Likewise, SOD activity was also decreased in the 2,4-D + Mg group when compared with the control group (p < 0.01). However, treatment provided to the 2,4-D group showed a noticeable recovery in SOD activity as compared with 2,4-D (p < 0.001; Figure 2(b)). A decline activity of CAT in renal tissues was observed in the 2,4-D exposed group when compared to the control group (p < 0.01). On the other hand, 2,4-D + Mg showed a pronounced increase in CAT activity than in the 2,4-D group (p < 0.01) (Figure 3(b)). Decline in GSH activity was provoked by 2,4-D exposition. As a result, GSH activity was remarkably reduced in the 2,4-D and 2,4-D + Mg exposed groups when compared to the control group (p < 0.001, p < 0.01). However, a significant improvement in renal GSH activity was observed in 2,4-D + Mg when compared with the 2,4-D alone group (p < 0.001). An incredible increase in renal GSH activity was also observed in the alone Mg-treated group when compared to the control group (p < 0.001; Figure 4(b)).
Discussion
The current study noticeably demonstrated the protective role of Mg upon 2,4-D-induced oxidative damage in the liver and kidneys of experimental rats. Present results exhibited that rats under 2,4-D exposition displayed the severe symptoms of toxicity in the form of mark down activity, weakness, abdominal molting, hair loss, and nasal bleeding during 28 consecutive days. These observations corroborate earlier reports on excess dosages of 2,4-D produced overt signs of toxicity. 48 It has been suggested that there is a positive association between a decline in body weight gain and the administered pesticide doses. In the current study, 2,4-D treatment significantly induced a marked decline in % body weight change as compared with control one (Table 1). This reduction in % body weight change was due to less food and water consumption. 49,50 Organ and relative organ weights are the main criteria for assessment of organ toxicity in toxicological studies. 51 In our study, an incredible increase in both liver and kidney relative weights of 2,4-D-treated rats was observed (Table 1) which could be due to the cell swelling (edema) and inflammation in both tissues. 52,53 2,4-D-induced necrobiotic changes are responsible for cytoplasmic swelling in hepatic cells, 22 while mesangial and epithelial cells proliferation is responsible for swelling in renal cells. 49
Coadministration of Mg and 2,4-D in rats showed improvement in body weight and decrease in liver and kidney relative weights as compared with the 2,4-D exposed group. These findings seem to suggest that the probable protective role of Mg may be associated with its antioxidant property via Mg stimulatory effect in denovo GSH synthesis 54 and reducing accumulation of toxic substance via increased Mg in the lumen of the distal nephron and disable the uptake of toxic substance by intercellular transport. 55,56 However, Mg treatment slightly reduced the relative kidney weights when compared with the control group. As mentioned earlier, Mg therapy itself influences the gastrointestinal (GI) system, so this reduction in our model may be associated with GI disturbance. 57 –59
Kidney is a potential target organ for chronic exposure of phenoxy acids, as they are regarded as inhibitors of chloride ion channels in tubular cells of the kidney which leads to develop certain changes in the output of urine. 60 Plasma urea, creatinine, and BUN levels are used as biomarkers of kidney function. Under our experimental conditions, renal dysfunction of 2,4-D exposed was illustrated by elevated levels of plasma urea, creatinine, and BUN (Table 2) which could be due to a reduction in glomerular filtration rate. 22 These observations speculate the diagnosis of impaired renal functions. 61 Mg treatment tended to prevent the 2,4-D causing renal impairments as illustrated by decreased levels of urea and creatinine. This may be due to the fact that Mg sustains the normal activity of nitric oxide (NO) synthase, a potent dilator NO by diminishing the impairments induced in the response of highly reactive radicals and superoxide as an oxygen in free-radical scavenging. 58
In the present study, potentiality of 2,4-D toxicity on liver was clearly observed by ALP, ALT, and AST biomarkers as they are good indicators of liver functional disorders. 62 2,4-D treatment markedly increased in marker enzymes such as ALP, ALT, and AST in comparison with the control group (Table 3). These incredible alterations in biomarkers indicated the hepatic cell damage which can be due to hepatic bile duct proliferation. 63
The exact mechanism of toxicity development by chlorophenoxy herbicide is not clarified, but may include intracellular membranes interference or oxidative phosphorylation decoupling. 8 Chlorophenoxy acids and its metabolites accumulation in kidney reduces oxygen consumption by the kidney cortex slices and results in mitochondrial uncoupling thus increasing the chances of renal toxicity development. 64 Furthermore, it is believed that lipid peroxidation which could be accredited to the extreme production of ROS is one of the mechanisms involved in hepatic and renal toxicity development. 17,65 2,4-D-induced ROS generation 66 and lipid peroxidation 67 have been well documented. In the present study, 2,4-D treatment noticeably increased the hepatic and renal MDA level (Figure 1(a) and (b)). These alterations implicated in liver and kidney dysfunction through oxidative stress and resulting lipid peroxidation. 21,68
Antioxidant enzymes that limit free-radical formation takes part in a crucial role of cell membrane protection in opposed to oxidative injury in all tissues, including liver and kidney. 69,70 In the current study, antioxidant enzyme including SOD, CAT, and GSH activities were considerably influenced in both tissues by 2,4-D exposition (Figures 2(a) to 4(a) and 2(b) to 4(b)). These unfavorable effects due to the production of ROS and the consequent peroxidation of membrane lipids could lead to increased vulnerability of tissue damage. 71 These findings are in consistent with the earlier findings that explained the changes in antioxidant levels are associated with the herbicide intoxication. 72,73
Mg is considered as an important cofactor in numerous enzyme systems activation and participation in the metabolism of biomolecules by directly collaborating with the enzyme or its substrate as well. 54 In fact, Mg acts as a cofactor of GSH enzyme 26 which serves several vital functions including antioxidant defense, xenobiotics detoxification and its related metabolites, cell cycle progression and programmed cell death regulation, and maintenance of redox potential. 74 There are various experimental evidences that explained the Mg antioxidant defensive role against various toxic substances. 54,75,76 Therefore, the current research was carried out to explore the cellular antioxidant potency of Mg supplementation on animal models of hepatic and renal toxicity provoked by 2,4-D. It is incredible to observe the beneficial effects of Mg in the present findings against toxicity development. The most pronounced changes in the form of decreased hepatic biomarkers and MDA level and increased antioxidant enzymes (SOD, CAT, and GSH) activities were noticed in the 2,4-D + Mg group. These remarkable alterations in hepatic enzymes level, MDA level, and antioxidant enzymes may be due to Mg role in modulating selenium-dependent GPX activity which decreases H2O2 and oxidative degradation product lipid peroxidation. 29,77 These evidences could account for the Mg antioxidant potency in restoring the oxidative stress-induced hepatic and renal damage.
Conclusion
Mg supplementation was found to be a potent cellular antioxidant agent to restore the hepatic and renal toxicity induced by 2,4-D thus, mediated the body and relative weights, hepatic and renal biomarkers, lipid peroxidation, and antioxidant enzymes activities. On the view of current outcomes, it is concluded that Mg supplementation can be used effectively to ameliorate the toxicity due to its antioxidant property. Further mechanistic approach in this regard is required to clarify the exact mechanism of Mg supplementation against 2,4-D toxicity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.