Abstract
The present study was aimed to study protective effect of butylated hydroxyanisole (BHA), a phenolic antioxidant used in foods on ferric nitrilotriacetate (Fe-NTA)–induced nephrotoxicity. Male albino rats of Wistar strain (4–6 weeks old) weighing 125–150 g were used in this study. Animals were given a single dose of Fe-NTA (9 mg kg−1 body weight) after treatment with BHA (1 and 2 mg animal−1 day−1). Fe-NTA treatment enhanced ornithine decarboxylase (ODC) activity to 5.3-fold, and [3H]-thymidine incorporation in DNA to 2.5-fold in kidney compared with the corresponding saline-treated control, whereas glutathione (GSH) levels and the activities of antioxidant enzymes decreased to a range of 2- to 2.5-fold in kidney. These changes were reversed significantly in animals receiving a pretreatment of BHA. The enhanced ODC activity and DNA synthesis showed a reduction to 2.12-fold and 1.15-fold, respectively, at a higher dose of 2 mg BHA day−1 animal−1, compared with the Fe-NTA-treated groups. Pretreatment with BHA prior to Fe-NTA treatment increased GSH and the activities of antioxidant enzymes to a range of 1.5- to 2-fold in kidney. The results indicate that BHA suppresses Fe-NTA-induced nephrotoxicity in male Wistar rats.
Introduction
Many studies have been carried out linking oxidative stress and carcinogenesis in animal experiments. Butylated hydroxyanisole (BHA) as an antioxidant can mediate its antitumor-promoting effects by scavenging free radicals. 1 –7 The inhibitory effects of antioxidants on carcinogenesis may be due to their ability to prevent the in vivo activation of carcinogens and may be related to their ability to prevent the in vivo activation of carcinogens to proximate or ultimate forms or to an action in increasing detoxification of the reactive intermediate.
Ferric nitrilotriacetate (Fe-NTA) is a potent nephrotoxic agent. 8 The renal toxicity is assumed to be caused by an elevation in serum free iron concentration following its reduction at the luminal side of the proximal tubule, which generates reactive oxygen species (ROS), leading to enhanced lipid peroxidation with a concomitant decrease in tissue glutathione (GSH) level. 9,10 Repeated intraperitoneal administration of Fe-NTA produces acute and subacute proximal tubular necrosis associated with a high incidence of renal adenocarcinoma in male mice and rats. 11,12
Antioxidants when added to human and animal food lower the incidence of cancer caused by chemical compounds. They prevent cancer through interception of harmful free radicals or activating the detoxifying enzymes of the body, inhibiting the formation of ultimately carcinogenic metabolites and their binding to DNA. BHA, butylated hydroxytoluene, and propyl gallate are also known to inhibit the development of glutathione-S-transferase placental form positive foci, that is, heterocyclic amine-induced carcinogenesis. 2
The widely used antioxidant food additive BHA also has a number of interesting and potentially important pharmacological properties. 13,14 In previous studies, BHA has also been shown to protect against acetylaiton of 2-aminoflourine and DNA-2-aminoflourene adducts in the rats. 14 In this study, we show that BHA may attenuate Fe-NTA–induced renal injury by reducing induction of early tumor markers in kidney.
Materials and methods
Preparation of Fe-NTA
The Fe-NTA solution was prepared by the method followed by Awai et al. 8
Animals and treatments
Male albino rats of Wistar strain (4–6 weeks old) weighing 125–150 g were used in this study and were housed in an air-conditioned room and had free access to pellet diet and water ad libitum. Approval was obtained from the ethics committee of the University.
For various sets of biological studies, a total of 30 rats were used in the study. For studying the effect of BHA on Fe-NTA-mediated generation of renal oxidative stress, rats were divided into five groups with six rats in each group. Group I received saline and served as negative control. Group II received an oral (gavage) treatment of BHA (only higher dose) and also served as control. Group III received only corn oil (vehicle of BHA) for a period of 1 week through the gavage. Groups IV and V received 1 and 2 mg animal−1 day−1 in 0.2 ml of corn oil of BHA respectively daily for 1 week orally. Twenty-four hours after the last treatment of BHA or corn oil, the animals of groups III, IV, and V received an intraperitoneal (i.p.) injection of Fe-NTA (9 mg Fe kg−1 body weight). Kidneys were collected and processed for preparation of post-mitochondrial supernatant (PMS).
Experimental design
Preparation of PMS
Kidneys were quickly removed, perfused immediately with ice-cold saline (0.85% w/v sodium chloride), and homogenized in chilled phosphate buffer (0.1 M, pH 7.4) that contained potassium chloride (KCl; 1.17% w/v), using a Potter Elvehjem homogenizer. The homogenate was filtered through a muslin cloth and was centrifuged at 800g for 5 min at 4°C to separate the nuclear debris. The aliquot obtained was centrifuged at 10,500g for 20 min at 4°C to obtain PMS, which was used as a source of enzymes. A portion of the PMS was centrifuged in an ultracentrifuge (Beckman, L7-55) at 105,000g for 60 min at 4°C. This pellet was considered to be the microsomal fraction and was suspended in phosphate buffer (0.1 M, pH 7.4) containing KCl (1.17% w/v).
Determination of ODC activity
Ornithine decarboxylase (ODC) activity was determined by utilizing 0.4 ml of renal 105,000 g supernatant fractions per assay tube and measuring the release of 14CO2 from
[3H]-Thymidine incorporation in renal DNA
For studying [3H]-thymidine incorporation in DNA, another 30 rats were used, and the animal treatment protocol and dose regimen were the same as described earlier. Additionally, the animals were given an i.p. injection of [3H]-thymidine (20 µCi/animal). After 2 h of administration of [3H]-thymidine, the animals were killed by cervical dislocation. Livers were quickly removed, cleaned free of extraneous material, and homogenized in distilled water for their further processing and the separation of DNA. The homogenate (10% w/v) was prepared in ice-cold water. The precipitate obtained was washed with cold trichloroacetic acid (5% w/v) and incubated with cold perchloric acid (PCA; 10%) at 4°C overnight. After incubation it was centrifuged, and the precipitate was washed with cold PCA (5%). The precipitate was dissolved in warm PCA (10% w/v), followed by incubation in a boiling water bath for 30 min and then filtered through Whattman 50 paper. The filtrate was counted for [3H] in a liquid scintillation counter (LKB-Wallace-1410, Pharmacia Biotech, Finland). The incorporation of [3H]-thymidine in DNA was done by the method followed by Smart et al. 16
Assay of antioxidant enzymes
Glutathione peroxidase activity
Glutathione peroxidase activity was measured according to the procedure described by Mohandas et al. 17 The reaction mixture consisted of 1.44 ml 0.05 M phosphate buffer, pH 7.0, 0.1 ml 1 mM EDTA, 0.10 ml 1 mM sodium azide, 0.05 ml 1 U ml−1 GSH reductase, 0.10 ml 1 mM GSH, 0.10 ml 2 mM nicotinamide adenine dinucleotide phosphate (NADPH), 0.01 ml 0.25 mM hydrogen peroxide (H2O2), and 0.10 ml 10% PMS in a total volume of 2.0 ml. Disappearance of NADPH at 340 nm was recorded at 25°C. Enzyme activity was calculated as nanomoles of NADPH oxidized per minute per milligram protein using a molar extinction coefficient of 6.223103 M−1 cm−1.
Glucose 6-phosphate dehydrogenase activity
The activity of glucose-6-phosphate dehydrogenase was assayed by the method described by Zaheer et al. 18 The reaction mixture in a total volume of 3.0 ml consisted of 0.3 ml Tris-HCl buffer (0.05 M, pH 7.6), 0.1 ml NADP (0.1 mM), 0.1 ml glucose-6-phosphate (0.8 mM), 0.1 ml magnesium chloride (8 mM), 0.3 ml of renal PMS (10% w/v), and 2.1 ml distilled water. The changes in absorbance were recorded at 340 nm, and the enzyme activity was calculated as nanomole NADP reduced per minute per milligram protein using a molar extinction coefficient of 6.223103 M−1 cm−1.
Catalase activity
Catalase activity was assayed by the method followed by Claiborne. 19 Briefly, the assay mixture consisted of 1.95 ml phosphate buffer (0.05 M, pH 7.0), 1.0 ml H2O2 (0.019 M), 0.05 ml, and 0.025 ml of renal PMS (10% w/v), respectively, in a final volume of 3.0 ml. Changes in absorbance were recorded at 240 nm. Catalase activity was calculated in terms of nanomole H2O2 consumed per minute per milligram protein.
Statistical analysis
Values were expressed as means ± SEM. The level of significance between different groups is based on Dunnett’s t-test, followed by analysis of variance (ANOVA). One-way ANOVA was used to calculate the statistical significance between various groups. The value of p < 0.05 was considered to be statistically significant.
Results
Fe-NTA increased renal ODC activity by 4.45-fold of their corresponding saline-treated controls. However, pretreatment with BHA prior to the administration of Fe-NTA depletes ODC activity in a dose-dependent manner. BHA (1 mg animal−1 day−1) showed a drop in ODC activity to 376% in kidney compared with the Fe-NTA–treated group. At a higher dose level of 2 mg animal−1 day−1 of BHA, it was reduced to approximately twofold in ODC activity as shown in Figure 1.

Inhibitory effects on BHA on Fe-NTA–induced renal (ODC activity. I: saline; II: BHA (D2); III: Fe-NTA; IV: BHA (D1) + Fe-NTA; V: BHA (D2) + Fe-NTA. Data represent mean ± SE of six animals. Saline-treated group served as control for group III. Fe-NTA–treated group served as control for groups IV and V. Dose regimen and treatment protocol are described in the text. *p < 0.05: statistically significant. BHA: butylated hydroxyanisole; Fe-NTA: ferric nitrilotriacetate; ODC: ornithine decarboxylase; D2: dose 2; D1: dose 1.
Fe-NTA enhances renal DNA synthesis to ∼2.5-fold of their corresponding saline-treated controls. The decrease in DNA synthesis was dependent on the dose of BHA used as has been observed with the ODC induction studies. In kidney, the decrease was significant (p < 0.001) at higher dose level as compared to the Fe-NTA–treated group as shown in Figure 2.
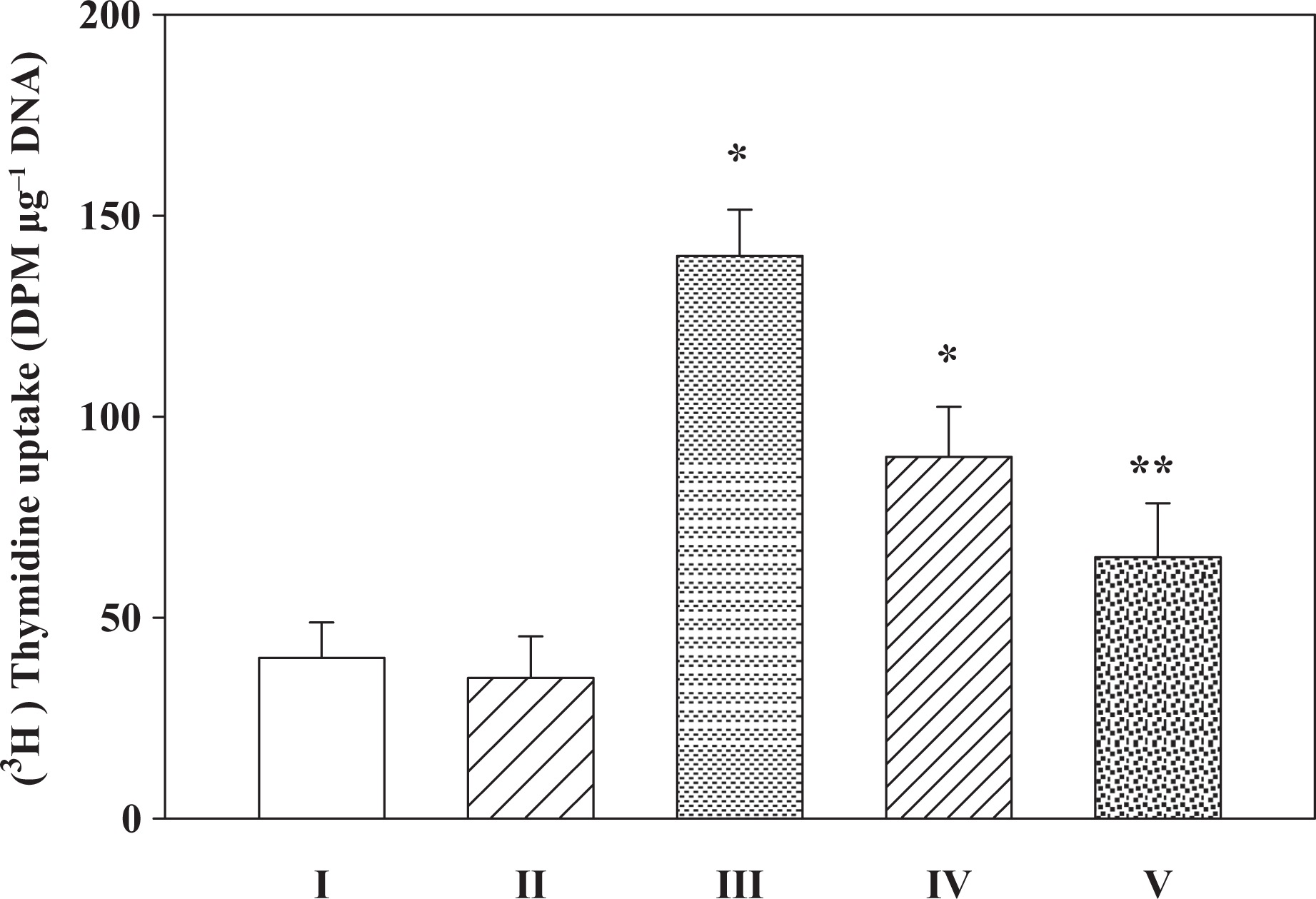
Inhibitory effects on BHA on Fe-NTA–induced renal enhanced DNA synthesis. I: saline; II: BHA (D2); III: Fe-NTA; IV: BHA (D1) + Fe-NTA; V: BHA (D2) + Fe-NTA. Data represent mean ± SE of six animals. Dose regimen and treatment protocol are described in the text. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V. * p < 0.05; **p < 0.001: statistically significant. BHA: butylated hydroxyanisole; Fe-NTA: ferric nitrilotriacetate; D2: dose 2; D1: dose 1.
Fe-NTA depletes the renal levels of antioxidant enzymes, namely, glutathione-6-phosphate dehydrogenase, catalase and glutathione peroxidase to 45%, 38%, and 44%, respectively, of their corresponding saline-treated control as shown in Table 1. However, both the doses of BHA used showed a significant recovery in the levels of these enzymatic and nonenzymatic molecules in the animals pretreated with BHA. BHA at a dose level of 2 mg animal−1 day−1 recovered 87–96% of the enzymatic activities of corresponding Fe-NTA–treated group.
Inhibitory effects of BHA on Fe-NTA–mediated renal levels of antioxidant enzymes.a

BHA: butylated hydroxyanisole; Fe-NTA: ferric nitrilotriacetate; D2: dose 2; D1: dose 1; NADPH: nicotinamide adenine dinucleotide phosphate; H2O2: hydrogen peroxide.
aEach value represents mean ± SE of six animals. D1 and D2 represent administration of 1 mg and 2 mg BHA animal−1 day−1 respectively given orally 1 week before the treatment of Fe-NTA. Saline-treated group served as control for group III. Fe-NTA–treated group served as control for groups IV and V.
bp < 0.05.
cp < 0.001.
Discussion
The results in the present study show that Fe-NTA has a comparable potent toxic effect in kidney assessed by the enhanced activity of ODC and DNA synthesis. In this study, the protective action of BHA on renal caused by a known renal carcinogen Fe-NTA has been shown. BHA, an antioxidant, is known to be an effective inhibitor of chemical carcinogenesis in many experimental cancer models. 20 , 21 The mechanism of protection appears to include increased activities of numerous enzymes concerned with non-oxidative metabolism of ROS.
ODC activity and [3H]-thymidine incorporation are widely used as biochemical markers to evaluate tumor-promoting potential of an agent. It has also been reported that induction of ODC activity and DNA synthesis plays an important role in carcinogenesis.15,22 It is known that ODC and polyamines are necessary for cellular proliferation, induction of ODC is also involved in tumor promotion and cell transformation, and cultured tumor cells often contain high levels of ODC. Therefore, ODC is considered as an attractive target in both chemotherapy and chemoprevention. Renal ODC activity and [3H]-thymidine incorporation is decreased by the pretreatment of BHA in Fe-NTA–mediated enhancement suggests that BHA protects against Fe-NTA–induced renal toxicity in rats.
Free radicals are known to be involved in various pathogenic processes including carcinogenesis, and it has shown that most chemopreventive agents afford protection against tumor promotion by inhibiting oxidative stress induced by tumor promoters. In present study, results show that Fe-NTA induces oxidative stress in the kidney and decreases antioxidant defenses as indicated by decrease in the activities of antioxidant enzymes. Earlier studies also indicated that Fe-NTA treatment to rats not only depletes GSH levels but also leads to depletion of key enzymes associated with GSH regulation. 23 Also it has also been shown in earlier studies that reduction of lipid peroxidation and H2O2 generation coupled with modulation of enzymatic and nonenzymatic antioxidant system by BHA indicates the involvement of ROS leading to tissue injury in the process. 24
Antioxidants have therefore been shown to be successful preventive agents against oxidative responses. A number of agents particularly those which are part of our diet afford protection against onset of various disease including cancer. The present study clearly shows that BHA is a promising antioxidant and can inhibit the enhanced tissue injury caused by Fe-NTA.
Conclusion
In summary, the present data suggest that BHA may be an effective chemopreventive agent and may offer protection against Fe-NTA–mediated renal damage in rats. But dosage will have to be taken into consideration in the design of clinical studies and in evaluating the role of BHA and its related compounds as chemopreventives.
Footnotes
Acknowledgment
The authors are thankful to be supported by the Research Center, Center for Female Scientific and Medical Colleges, Deanship of Scientific Research, King Saud University, Riyadh, Kingdom of Saudi Arabia.
Conflict of interest
The authors declare no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
