Abstract
The single- and repeat-dose toxicity profile of IDX14184, a novel guanosine nucleotide prodrug with antiviral activity against hepatitis C viral infection, was characterized following once daily oral administration for durations up to 13, 26, and 32 weeks in mouse, rat, and cynomolgus monkey, respectively. The heart, liver, kidney, skeletal muscles, and lower gastrointestinal tract (cecum, colon, and/or rectum) were identified as the primary toxicity targets in these nonclinical species. The mouse was relatively insensitive to IDX14184-induced cardiac toxicity and hepatotoxicity. The rat was very sensitive to IDX14184-induced skeletal muscle, liver, heart, and lower gastrointestinal tract toxicity but relatively insensitive to kidney toxicity. The monkey is a good animal species to detect IDX14184-induced toxicity in the cardiac and skeletal muscles, and in the liver and kidney, but not lower gastrointestinal tract toxicity. The toxicity profile of IDX14184 was most appropriately characterized in rats and monkeys. The conduct of a series of cardiac size and function assessments during a non-rodent toxicology study using echocardiography proved great utility in this work. IDX14184 clinical development was eventually terminated due to suboptimal efficacy and regulatory concerns on potential heart and kidney injury in patients, as seen with a different guanosine nucleotide prodrug, BMS-986094.
Keywords
Introduction
Hepatitis C virus (HCV) chronically infects 130–150 million people in the world: 15–30% of the chronically infected develop cirrhosis and 350,000–500,000 people die each year from HCV-related liver diseases. 1 There are at least six different genotypes of HCV. Approximately 75% of Americans with HCV have genotype 1 virus (subtypes 1a or 1b), and 20–25% have genotype 2 or 3, with small numbers of patients infected with genotype 4, 5, or 6. 2 Genotype 4 is much more common in Africa than in many other parts of the world, and genotype 6 is common in Southeast Asia, and each area of the world has its own distribution of genotypes. 3
The mainstay of HCV treatment until recently was the combination of pegylated α-interferon (PEG-IFN) and ribavirin (RBV), which resulted in an overall sustained virologic response rate of approximately 50% for genotype 1 HCV 4 –6 and rather higher for genotypes 2 and 3. 7 However, this therapy typically requires 24–48 weeks of therapy, is not very convenient (IFN is an injectable), and is poorly tolerated by many patients due to significant side effects.
Recent research in HCV drug development have focused on identifying direct-acting antiviral agents (DAAs) that, unlike IFN and RBV, are designed to inhibit HCV propagation by binding to specific HCV nonstructural (NS) protein targets, in particular the NS3-4A serine protease, the NS protein 5A (NS5A), and the NS5B RNA-dependent RNA polymerase. Drugs targeting the NS5B RNA-dependent RNA polymerase fall into two classes: nucleoside or nucleotide polymerase inhibitors (NIs) and non-nucleoside polymerase inhibitors (NNIs). NIs are analogs of natural substrates that bind to the active site of NS5B and terminate viral RNA chain elongation, 8 while NNIs bind at allosteric sites on the polymerase and induce conformational changes.
Several DAAs have been approved since 2011, including the NS3-4 protease inhibitors telaprevir, boceprevir, and simeprevir and the nucleotide prodrug sofosbuvir that inhibits the NS5B polymerase. The DAA classes differ somewhat with respect to their genotypic coverage and genetic barrier to resistance. Protease inhibitors show high efficacy in limited genotypes and provide a low to intermediate barrier to viral resistance. 9,10 NS5A inhibitors can be highly active against multiple genotypes but provide a low barrier to resistance. NNIs show HCV genotype-specific activity and have a low barrier to resistance. 10 Only nucleotides couple good antiviral efficacy in all genotypes with a high genetic barrier to resistance. 9 –11 Thus, nucleoside/nucleotide analogs such as sofosbuvir (GS-7977) and MK-3682, formerly IDX21437, 12 provide optimal backbone candidates for all-oral therapies that utilize a combination of different DAAs to hit HCV at different stages in the replication cycle.
IDX14184 (3-hydroxy-2,2-dimethyl-thiopropionic acid S-(2-{[(2 R,3 R,4 R,5 R)-5-(2-amino-6-oxo-1,6-dihydro-purin-9-yl)-3,4-dihydroxy-4-methyl-tetrahydro-furan-2-ylmethoxy]-benzylamino-phosphoryloxy}-ethyl) ester (Figure 1) is an early nucleotide prodrug developed by Idenix Pharmaceuticals. IDX14184 was designed as a liver-targeted prodrug that delivers 2′-C-methylguanosine monophosphate (2′-MeGMP), the monophosphate form of the corresponding nucleoside, 2′-C-methylguanosine (2′-MeG) to the liver after oral administration. 11,13 –17 Liver targeting results from the fact that IDX14184 is (a) subject to very efficient hepatic extraction (>95% in monkeys) and (b) is then cleaved, releasing 2′-MeGMP in a reaction that occurs preferentially in hepatocytes but appears to be considerably less efficient in cells of non-hepatocyte origin. The liberated 2′-MeGMP is readily converted to high levels of the active 2′-MeG triphosphate (2′-MeGTP) by intracellular kinases.
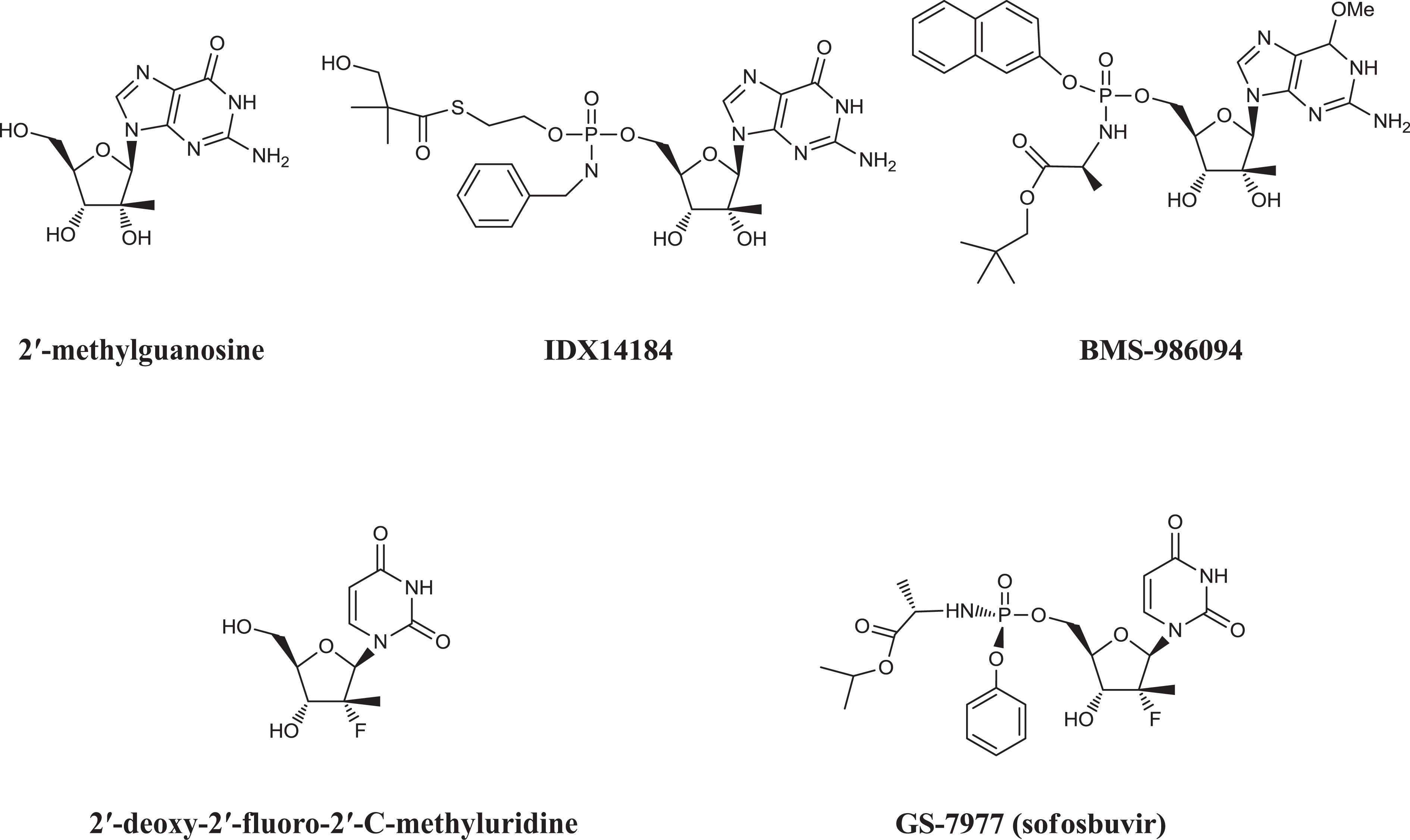
Structures of 2′-methylguanosine, IDX14184, BMS-986094 (INX-189), 2′-deoxy-2′-fluoro-2′-C-methyluridine, and GS-7977 (sofosbuvir).
BMS-986094 (formerly, INX-189) is another nucleotide prodrug developed by Bristol-Myers Squibb (BMS). Unlike GS-7977, which is a uridine analog, IDX14184 and BMS-986094 (Figure 1) are guanosine analogs. Although they carry different prodrug moieties, the two guanosine analogs generate two identical metabolites, 2′-MeGTP and the nucleoside 2′-MeG, which may arise by catabolism of the phosphorylated species. BMS voluntarily suspended phase II studies of BMS-986094 in 2012 and later terminated its development after the compound caused the death of one patient who had received a 200-mg dose of the drug 18 and severe heart and kidney damage to other patients at 100 and 200 mg doses. 18 –20 Although no such issues had emerged during phase II studies of IDX14184, the Idenix nucleotide was put on “partial clinical hold” by the US Food and Drug Administration in 2012, as a consequence of its close structural similarity between the two agents and their nucleotide and nucleoside metabolites. The clinical development of IDX14184 was eventually terminated due to suboptimal efficacy and regulatory concerns on potential heart and kidney injury in patients. In that context, this article reports the relevant target organ toxicity of IDX14184 following single- and repeat-dose oral administration to mice, rats, and cynomolgus monkeys for up to 32 weeks.
Materials and methods
IDX14184
IDX14184 was supplied by Idenix with a chemical purity ≥97%. IDX14184 was formulated in poly(ethylene glycol) 200 and administered at a dose volume up to 5 mL/kg in mice and rats or 3 mL/kg in monkeys.
Animals
CD-1 mice were obtained from Charles River Laboratories (Raleigh, North Carolina, USA and L’Arbresle, France). Sprague Dawley rats (Crl: CD®(SD)IGS BR) were obtained from Charles River Laboratories (Raleigh, North Carolina, USA; Portage, Michigan, USA; Saint-Constant, Quebec, Canada; and L’Arbresle, France). Cynomolgus monkeys (
Toxicology studies
Toxicology studies designed to characterize the single- and repeat-dose toxicity profile of IDX14184 were performed in mice, rats, and cynomolgus monkeys with a range of daily administration durations from single dose in rats up to 13, 26, or 32 weeks in mice, rats, and monkeys, respectively, in order to support the intended 12-week use of IDX14184 in humans (Table 1). Animals were assigned to dose groups by stratification based on body weights using a computerized procedure. Males and females were randomized separately. The route of administration was oral or nasogastric gavage for all studies since the oral route was the intended route of administration in humans. Dose levels for each study were selected based on results obtained from prior studies in the same species. The high dose for this study was selected as the highest dose level, which would not be expected to produce overt toxicological effects. The low-dose level selected for this study is expected to have no observable effects, and the mid-dose level was chosen as an intermediate level to describe any potential dose–response relationships. At dose initiations, animals were approximately 6–8 weeks old for mice (weighing approximately 18–35 g), 5–8 weeks for rats (weighing approximately 100–385 g), and 2–5 years old for monkeys (weighing approximately 2–3.6 kg). Assessment of toxicity was based on mortality/moribundity, clinical observations, body weights, food consumption, ophthalmic examinations, hematology, coagulation, serum chemistry, urinalysis, organ weight, and/or histopathology.
Summary of single- and repeat-dose toxicology studies with IDX14184 in mice, rats, and monkeys.

DRF: dose range finding; TK: toxicokinetics; GLP: Good Laboratory Practice.
aThe duration was shortened to 32 weeks since the development of IDX14184 was terminated.
In addition to these routine toxicology examinations, the following special examinations were performed in some studies to further characterize the toxicity profile or to investigate the mechanism of toxicity.
Biomarkers indicative of cardiac and/or skeletal muscle injury
Blood samples (0.5 mL) were collected from toxicity animals of the 26-week rat study via a jugular vein during week 20 of the dosing phase and on the days of scheduled terminal and recovery euthanasias. Plasma samples were analyzed for concentrations of cardiac troponin I (cTnI), cardiac troponin T (cTnT), fatty acid-binding protein 3 (Fabp3), myosin light chain 3 (Myl3), and skeletal troponin I (sTnI) using a Rat Muscle Injury Panel-1 Test® kits (Meso Scale Discovery, Gaithersburg, Maryland). Assays were performed according to the manufacturer’s instructions and validated for precision, accuracy, and recovery. The lower limit of quantitation (LLOQ) and upper limit of quantitation (ULOQ) of the assays were 0.136 and 33.2 ng/mL for cTnI, 2.38 and 192 ng/mL for cTnT, 1.57 and 127 ng/mL for Fabp3, 0.292 and 214 ng/mL for Myl3, and 1.09 and 264 ng/mL for sTnI, respectively.
Blood samples (1.0 mL) were collected from all animals of the chronic (32-week) monkey study via a femoral vein during weeks 9, 13, and 29 of the dosing phase for N-terminal prohormone of brain natriuretic peptide (NT-proBNP) analysis and during week 29 of the dosing phase for cTnI analysis. Serum cTnI levels were measured with a paramagnetic particle chemiluminescent method on the Beckman Access Analyzer using reagent Acc iTnI supplied by Beckman Coulter (Pasadena, California, USA). The LLOQ and ULOQ of the assays were 0.01–94.0 ng/mL. Serum NT-proBNP levels were measured with an enzyme-linked immunosorbent assay according to the manufacturer’s instructions. The LLOQ and ULOQ of the assays were 235 and 6400 pmol/L for NT-proBNP, respectively.
In addition, serum cTnI levels were determined in the 13-week study in monkeys on days 28, 56, and 92 and at unscheduled euthanasias on days 44, 72, and 84 using an immunologic method on the Siemens Immulite analyzer (Germany). The cTnI level was determined by a solid-phase chemiluminescent immunoassay (Siemens Healthcare, Germany). The LLOQ and ULOQ of the assays were 0.01 and 90.00 ng/mL for cTnI, respectively.
Echocardiogram
Echocardiogram evaluations were performed on animals of the 32-week monkey study. Animals were anesthetized with ketamine at weeks 9 and 29 of the dosing phase and week 13 of the recovery phase for the examinations. Principal measurements evaluated included left ventricular internal diameter during diastole and systole, left ventricular posterior wall thickness at end diastole and systole, aortic diameter (AO), diastolic left atrial diameter (LA), E-point septal separation, left ventricular cross-sectional chamber (short axis) area in diastole (LVAd) and systole (LVAs), cross-sectional area bounded by left ventricular epicardium (Epi), and left ventricular myocardial cross-sectional area (Myo). Subjective readings included paradoxical septal motion, varying degrees of right heart enlargement (mild, moderate, or severe), and pericardial effusion if identified. LVAd, LVAs, percent change in left ventricular chamber cross-sectional area, percent change in left ventricular internal diameter, left ventricular ejection fraction (EjFx), and LA to AO ratios were calculated.
EjFx was presented as the percent change in the Teicholz calculated left ventricular volume at the end of diastole and systole. Left ventricular volume was calculated as ([7 ×
Transmission electron microscopy examinations
Samples (5 to 6 per site) of liver, heart (from left and right ventricles and septum), and kidney (from cortical and medullary regions), approximately 1 mm3 each, were collected from all animals of the 13-week monkey study at terminal necropsy and fixed in Trump’s Fixative™ at 4°C. Selected samples from designated animals were examined for ultrastructural changes: 1 per sex from group 1 (control, heart, liver, and kidney) and 2 per sex from groups 2 (10 mg/kg; heart only), 3 (50 mg/kg; liver and kidney), 4 (100 mg/kg; heart, liver, and kidney), and 5 (300 mg/kg; heart, liver, and kidney). These animals were selected based on histomorphologic findings in order to maximize the possibility to detect any ultrastructural changes.
Samples were processed through an industry standard buffer rinse, osmium postfixation, graded alcohol–acetone dehydration series, and embedded in Spurr’s epoxy resin blocks. The blocks were thick sectioned on an ultramicrotome at approximately 1000 nm thickness, placed on glass slides, and stained with toluidine blue. After areas of interest were identified, thin sections at approximately 80–90 nm thickness were cut and placed on copper grids, stained with uranyl acetate and lead citrate, and examined on a Jeol JSM-100 S transmission electron microscope (Japan). Three photographs were taken from each sample to show both lower magnification (5600×) and higher magnification (22,400×) aspects of cells.
Statistical analyses
To determine an appropriate statistical test for continuous variables, each data set was subjected to a statistical decision tree using the SAS® System (version 8.0, SAS Institute, Cary, NC). A minimum of 3 animals/sex/group were required for statistical analysis. The data were initially tested for normality using the Shapiro–Wilk test followed by the Levene’s test for homogeneity of variance. If both assumptions were fulfilled with or without rank transformations, one-way analysis of variance (ANOVA) and pairwise comparisons (Dunnett’s test) were performed.
If either of the parametric assumptions was not satisfied with rank transformations, then the Kruskal–Wallis nonparametric ANOVA procedure was used to evaluate intergroup differences (
Results
Mouse
In
In
In the animals that were sacrificed at the scheduled terminal necropsy, lower food consumption (decreased by 21%) was observed between days 11 and 14 in males dosed at 500 mg/kg/day. Histomorphologic findings at 500 mg/kg/day were observed in the heart (minimal to moderate cardiomyocyte degeneration/necrosis in females), kidney (diffuse moderate vacuolation of the cortical tubules in males), skeletal muscles (minimal to moderate myofiber degeneration/regeneration in the diaphragm in males and females), and cecum and colon (minimal increases in enterocyte apoptosis in females). Minimal to moderate myofiber degeneration/regeneration was also seen in animals dosed at 150 mg/kg/day.
Following a 28-day recovery, findings in the heart (minimal to mild vacuolation and myofibrosis in females), kidney (moderate vacuolation of inner cortical tubules in females), diaphragm (minimal to marked myofiber degeneration/regeneration), tongue (minimal to mild myofiber degeneration/regeneration), and cecum and colon (minimally increased enterocyte apoptosis in females) were limited to animals at 500 mg/kg/day. The no-observed-adverse-effect level (NOAEL) was 50 mg/kg/day.
In
In the animals that were sacrificed at the terminal euthanasia, test article-related histomorphologic findings were limited to the spleen and skeletal muscles (rhabdomyopathy) at 75 mg/kg/day. Lower spleen weights, correlating with splenic contraction histomorphologically, were noted in females at 75 mg/kg/day. Minimal to severe rhabdomyopathy was seen in the diaphragm, tongue, extraocular muscles, subcutaneous muscles, gastrocnemius, quadriceps, and biceps femoris with greater incidence and severity noted in females than in males. Following a 28-day recovery, minimal to mild rhabdomyopathy was observed in the diaphragm, tongue, and gastrocnemius of females at 75 mg/kg/day. There were no skeletal muscle findings in mice dosed at 2.5, 10, and 25 mg/kg/day or in males dosed at 75 mg/kg/day. The NOAEL was 25 mg/kg/day, corresponding to
Rat
In
In
For the remaining animals at 600 mg/kg/day, increases in plasma AST and ALT levels were noted (5 to 7 times the upper limit of historical control values). Other changes in clinical chemistry included mild increases in urea, alkaline phosphatase (males only), and amylase levels. Histomorphologic findings were reported in the liver (minimal to moderate bile duct degeneration/necrosis and hyperplasia surrounded by portal edema and mononuclear cell infiltrates, and minimal to mild single hepatocyte necrosis and localized coagulative necrosis), kidneys (moderately enlarged and lightly eosinophilic nuclei with marginated nucleoli in the proximal tubules, mild cytoplasmic vacuolation, minimal to mild cell necrosis, and minimally increased mitotic figures in the proximal tubules), heart (diffuse minimal to mild mononuclear cell infiltrates), skeletal muscles (minimal mononuclear cell infiltrates with isolated myofiber degeneration/necrosis in the biceps, gastrocnemius, and quadriceps), and cecum (moderate necrotizing inflammation).
At 300 mg/kg/day, the magnitudes of changes in plasma AST and ALT were smaller than those noted at 600 mg/kg/day. A mild increase in plasma UN was limited to one female rat, and a transient, mild increase in alkaline phosphatase was observed in male rats only. Less severe histomorphologic findings were seen in the liver, kidneys, heart, skeletal muscle, and cecum.
At 150 mg/kg/day, plasma AST and ALT levels remained elevated. Sporadic histomorphologic findings, generally less pervasive and less severe, were observed in the liver (both males and females) and kidneys (females only). Based on the aforementioned results, the NOAEL was below 150 mg/kg/day, and the maximum tolerated dose was 300 mg/kg/day.
In
In
Test article-related histomorphologic findings and potential cause of death for rats from the 13-week study.a

aSeverity of histomorphologic findings: G1 =
For animals that survived the terminal euthanasia, increased incidence of dehydration was observed at 10 mg/kg/day. Mean body weights in males and females at 10 mg/kg/day were lower than controls starting from day 42 to day 49, respectively. Consistent with the decreases in body weight, lower food consumption was observed. Following the 4-week recovery period, mean body weights were comparable to those of controls.
Test article-related findings in clinical chemistry at the terminal necropsy included dose-dependent increases in serum AST at 5 (by 0.93-fold in males and 1.68-fold in females) and 10 mg/kg/day (by 2.92-fold in males and 3.37-fold in females) and in serum ALT at 5 (by 1.22-fold in males and 2.07-fold in females) and 10 mg/kg/day (by 4.22-fold in males and 7.40-fold in females). Following the 4-week recovery period, serum AST and ALT levels were generally comparable to controls.
Test article-related histomorphologic findings were observed in the muscles of the biceps femoris, diaphragm, esophagus, gastrocnemius, quadriceps, and tongue (minimal to marked myofiber degeneration/regeneration) at ≥0.5 mg/kg/day; in the cecum (minimal crypt epithelial hyperplasia, degeneration, and/or inflammation), colon (minimal to mild crypt epithelial hyperplasia and minimal epithelial degeneration), and rectum (minimal crypt epithelial degeneration and erosion) at ≥1 mg/kg/day; in the liver (minimal to mild hepatocellular vacuolation and minimal to moderate hepatocellular necrosis) at ≥0.5 mg/kg/day; and in the heart (increased incidence of minimal myocardial degeneration/necrosis) at 10 mg/kg/day (Table 3). Myofiber degeneration/regeneration was characterized by hypereosinophilic/fragmented and/or basophilic muscle fibers. At the end of the 4-week recovery, no test article-related histomorphologic findings were observed in the cecum, colon, or rectum, indicating complete reversibility, but only partial reversibility was seen in the muscles, liver, and heart (Table 4). Hepatocellular necrosis was observed only in males. The study NOAEL was less than 0.5 mg/kg/day for males and 1 mg/kg/day for females.
Test article-related histomorphologic findings at terminal necropsy in rats from the 13-week study (excluding the early deaths).a

aSeverity of histomorphologic findings: G1 =
bNo samples were examined histomorphologically as per protocol.
Test article-related histomorphologic findings at recovery necropsy in rats from the 13-week study (excluding the early deaths).a

aSeverity of histomorphologic findings: G1 =
bNo samples were examined histomorphologically as per protocol.
In
IDX14184-related clinical pathology changes were limited to increases in serum aldolase activity, plasma sTnI, Fabp3, and Myl3 levels without changes in cTnI or cTnT at 3 mg/kg/day (Table 5).
Changes in muscle-related biomarkers in rats from the 26-week study.a

Fabp3: fatty acid-binding protein 3; cTnT: cardiac troponin T; cTnI: cardiac troponin I; LLOQ: lower limit of quantitation; Myl3: myosin light chain 3; sTnI: skeletal troponin I.
aLLOQ: 0.136 ng/mL for cTnI, 2.38 ng/mL for cTnT, 1.57 ng/mL for Fabp3, 0.292 ng/mL for Myl3, 1.09 ng/mL for sTnI.
Data are presented as group mean for each sex.
b
cRecovery day 57.
IDX14184-related histomorphologic effects on skeletal muscles included minimal to moderate myofiber degeneration/regeneration in animals dosed at ≥0.3 mg/kg/day. Affected muscle myofibers were noted in the biceps femoris, diaphragm, esophagus, gastrocnemius, psoas, quadriceps, and tongue. Moderate myofiber degeneration/regeneration occurred in animals dosed at 3 mg/kg/day, which was considered adverse. A complete recovery was observed in animals dosed at ≤1 mg/kg/day but only partial recovery at 3 mg/kg/day.
In the liver, increased incidence of minimal hepatocyte vacuolation was noted in males dosed at 0.3 or 3 mg/kg/day. This finding was reversible and considered non-adverse. Based on the adverse effects in the muscles observed at 3 mg/kg/day, the NOAEL was 1 mg/kg/day, corresponding to
Monkey
In
In
IDX14184-related changes in plasma chemistry at terminal necropsy included increases in plasma ALT, AST, and creatinine in both males and females at 500 mg/kg/day and increases in γ-glutamyl transferase and total bilirubin in females at 500 mg/kg/day. Following the 28-day recovery period, plasma AST, total bilirubin, and creatinine levels recovered to control values in the two female monkeys. ALT and γ-glutamyl transferase activities declined over time but were still higher than respective control values at the end of recovery.
Macroscopic necropsy findings were noted in the found dead recovery male (red fluid in the abdominal cavity) and in the euthanized recovery male (red fluid in the thoracic and abdominal cavities, a pale liver with an accentuated lobular pattern, and pale kidneys).
IDX14184-related histomorphologic findings were noted in the liver, kidney, and heart of the 500 mg/kg/day group. Liver histomorphologic findings included minimal to moderate single hepatocellular necrosis, minimal or mild green pigment in hepatocytes and Kupffer cells, mild or moderate hepatocellular nuclear paleness, and moderate vacuolation of periportal hepatocytes. Marked, zonal hepatocellular necrosis occurred in the centrilobular to midzonal lobules of both early death animals.
Kidney histomorphologic findings included minimal to moderate tubular necrosis, dilatation, casts, and vacuolation. Tubular vacuolation in the cortex was characterized by the enlargement of the lining cells that contained a dark condensed nucleus and several large, clear cytoplasmic vacuoles. Additional cortical tubular findings included mild to moderate karyocytomegaly and minimal to moderate nuclear paleness.
Minimal to moderate myocardial necrosis comprising multifocal to coalescing foci of necrosis was observed in the left ventricle, papillary muscles, and septum, and to a lesser extent in the right ventricle. Moderate myocardial necrosis was also noted in the found dead male monkey.
Minimal tubular dilatation, casts, and mild to moderate tubular vacuolation were noted in the kidneys at 150 mg/kg/day. No test article-related findings were observed at 50 mg/kg/day. The no-observed-effect level (NOEL) was 50 mg/kg/day.
In
Test article-related histomorphologic findings and potential cause of death for monkeys from the 13-week study.a

aSeverity of histomorphologic findings: G1 =
Test article-related clinical pathology changes in monkeys euthanized early.

ALT: alanine transaminase; AST: aspartate transaminase; APTT: activated partial thromboplastin time; BUN: blood urea nitrogen; CRE: creatinine; FIB, fibrinogen; GGT: γ-glutamyltransferase; PHOS: phosphorus; PT: prothrombin time; TBIL: total bilirubin.
aData are expressed as fold of the upper limit of the baseline values (the lower limit of the baseline values was used for TP, ALB, GLOB, and FIB). Fold values in italics indicate test article-related changes; fold values in regular text were not considered test article-related as the fold values were too small or the values on the day of euthanasia remained within the historical range.
In all monkeys that were sacrificed at the terminal euthanasia on day 92, IDX14184-related clinical chemistry findings included elevated serum ALT (1.3- to 4.9-fold concurrent control value) and γ-glutamyl transferase (1.4- to 2.8-fold concurrent control value) in male and female monkeys dosed at 300 mg/kg/day on days 28, 49, 70, and 92. IDX14184-related histomorphologic findings included degeneration in the heart and skeletal muscles. A minimal cardiac degeneration was observed at ≥50 mg/kg/day. In addition, minimal degeneration was present in the quadriceps at 100 mg/kg/day and in the gastrocnemius at 300 mg/kg/day. There were no IDX14184-related histomorphologic findings in animals dosed at 10 mg/kg/day. Following a 28-day recovery, minimal to mild muscle degeneration was observed in the biceps femoris at ≥100 mg/kg/day and in the gastrocnemius at 300 mg/kg/day. This finding was not considered adverse since mild degeneration was also present in the biceps femoris of one control female on day 92.
Transmission electron microscopy (TEM) analysis was performed on the heart (at doses of 10, 100, and 300), kidney, and liver (at doses of 50, 100, and 300) to evaluate mitochondrial toxicity. The doses of 10 and 50 mg/kg/day were selected for analysis because they represented the NOEL for the heart (10 mg/kg/day) and kidney and liver (50 mg/kg/day). In the male dosed at 300 mg/kg/day and euthanized on day 50 (no 5003), mildly swollen mitochondria in the cytoplasm of cells in parts of two proximal convoluted tubule S3 (pars recta) segments were seen in the kidney cortex. The organelle area for a circular section through affected mitochondria was 10–20% greater compared to unaffected mitochondria in the same monkey or control monkeys. In contrast to unaffected mitochondria, swollen mitochondria showed clear spaces between the cristae. A relatively increased density of mitochondria was seen in the cytoplasm of hepatocytes compared with the control male. However, this increased density was considered a structural reflection of the absence of glycogen and very small amount of endoplasmic reticulum in the cytoplasm compared to that of hepatocytes assigned to the control group. At the same magnification, mitochondria in hepatocytes of this animal were larger than those in hepatocytes of the control male. The hepatocytes with swollen mitochondria also tended to show an increased density of lipid vacuoles as compared to hepatocytes in the control male.
In the female dosed at 300 mg/kg/day and euthanized on day 84 (no 5105), mild mitochondria swelling (characterized as increased clear space between cristae) was seen in a proximal convoluted tubule epithelial cell, in the basal processes of an adjacent epithelial cell of the kidney cortex and in epithelial cells of the ascending limb of the loop of Henle. Swollen mitochondria were intermixed with well-formed mitochondria. Some cells with condensed (electron-dense) cytosol that were adjacent to epithelial cells with normal mitochondria also showed swollen mitochondria. Mild mitochondrial swelling was also seen in hepatocytes. No toxicologically significant mitochondrial changes or other ultrastructural changes were observed in the heart. In animals dosed at 10 (heart only), 50 (kidney and liver only), or 100 mg/kg/day (heart, kidney, or liver) including the male euthanized on day 44, there were no IDX14184-induced ultrastructural changes in mitochondria or other organelles.
For animals dosed at 100 mg/kg/day, increased smooth endoplasmic reticulum was seen in the liver of one male (no 4002) and two females (Nos. 4102 and 4103). However, this finding was considered to be an adaptive response to IDX14184.
The NOAEL was 50 mg/kg/day since the minimal and sporadic cardiac finding at that dose was considered negligible toxicological significance. TEM analysis did not disclose any IDX14184-induced ultrastructural changes that would lower the NOAEL based on the routine in-life and postmortem histomorphologic analyses.
In
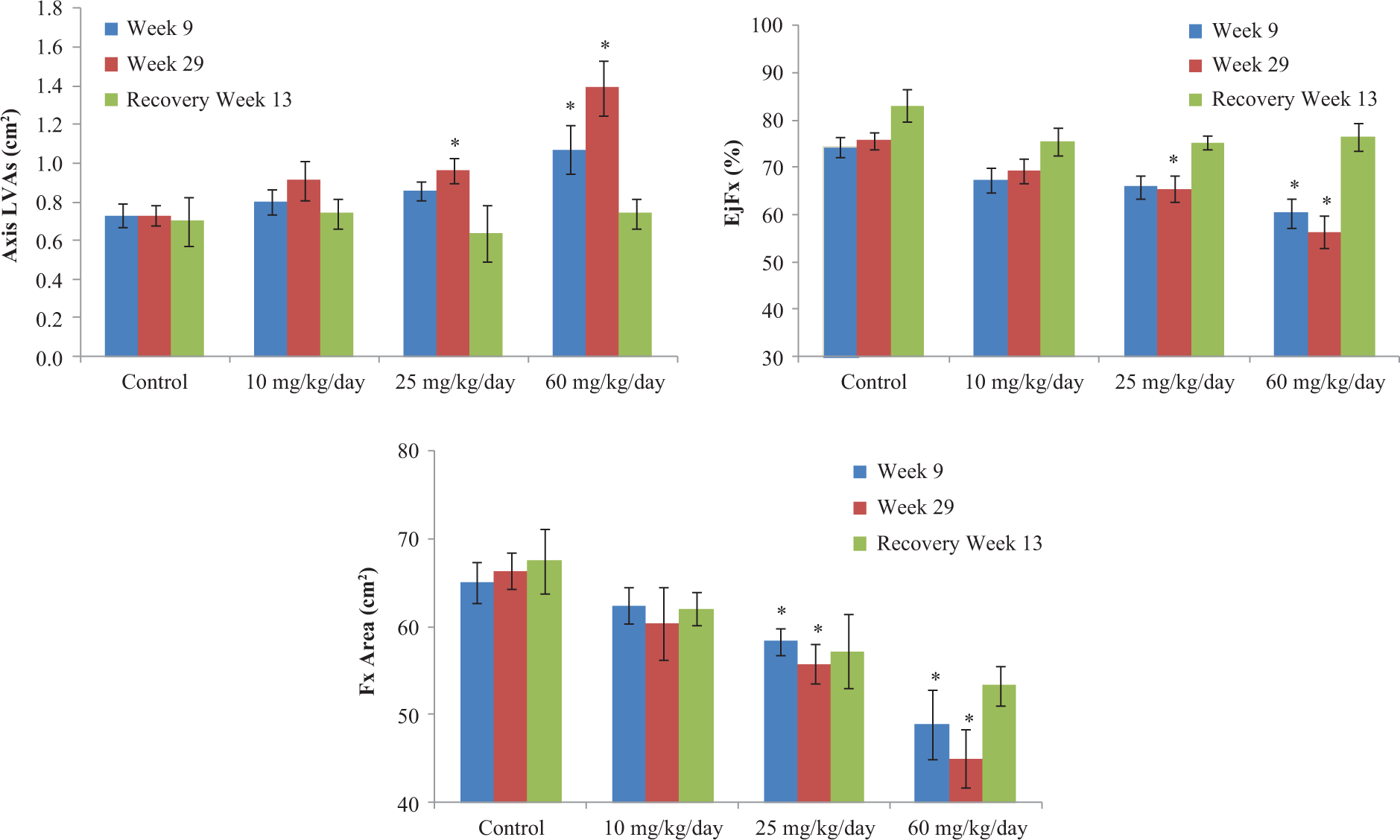
IDX14184-related effects on echocardiogram parameters in monkeys of the 32-week chronic toxicology study. *
IDX14184-related clinical pathology effects were limited to minimally increased aldolase for females dosed at 25 or 60 mg/kg/day, minimally increased ALT for males dosed at 25 or 60 mg/kg/day and females dosed at 25 mg/kg/day, and minimally decreased creatinine concentration for females dosed at 60 mg/kg/day. The effects on aldolase activity and possibly creatinine concentration were consistent with IDX14184-related effects on muscles. Of uncertain relationship to IDX14184 were minimally increased ALT activities through much of the dosing phase for the same two females dosed at 60 mg/kg/day. No correlative histomorphologic findings were observed in the liver. All IDX14184-related clinical pathology effects exhibited reversibility at the end of the recovery phase. IDX14184 administration had no effects on hematology, coagulation, or urinalysis test results. There were no IDX14184-related effects on serum cTnI and NT-proBNP concentrations (Figure 3).

Serum NT-proBNP levels in monkeys of the chronic toxicology study. NT-proBNP: N-terminal prohormone of brain natriuretic peptide.
IDX14184-related histomorphologic changes occurred in the skeletal and cardiac muscles at the terminal necropsy and consisted of minimal myocardial hypertrophy in the heart of animals dosed at 60 mg/kg/day and degeneration in examined skeletal muscles (biceps femoris, gastrocnemius, psoas, quadriceps, diaphragm, and/or skeletal muscle around the larynx) in animals dosed at
IDX14184-related histomorphologic findings at terminal necropsy in monkeys from the 32-week study.

aSeverity of histomorphologic findings: G1 =
Discussion
The single- and repeat-dose toxicity profile of IDX14184 was characterized following single- and repeat-dose administration via oral gavage for durations up to 13 weeks in mice, 26 weeks in rats, and 32 weeks in monkeys. The heart, liver, kidney, skeletal muscles, and lower gastrointestinal tract (cecum, colon, and/or rectum) were identified as the primary toxicity targets in these nonclinical species. The mouse was relatively insensitive to IDX14184-induced cardiac toxicity and hepatotoxicity. The rat was very sensitive to IDX14184-induced skeletal muscle, liver, heart, and lower gastrointestinal tract toxicity but relatively insensitive to kidney toxicity. The monkey is a good animal species to detect IDX14184-induced toxicity in the cardiac and skeletal muscles, and in the liver and kidney, but not lower gastrointestinal tract toxicity. The toxicity profile of IDX14184 was most appropriately characterized in rats and monkeys.
The development of BMS-986094, a structurally related guanosine nucleotide prodrug (Figure 1), was terminated in 2012 due to severe cardiac and kidney toxicity in patients. 20,21 In a phase II clinical trial, BMS-986094 led to the death of one patient due to congestive heart failure and severe heart and kidney injury to other patients who received 100 and 200 mg doses. 19,20 Of 34 patients who received BMS-986094 for 1–6 weeks, 11 were hospitalized for suspected cardiotoxicity. 20 Although no severe cardiac or renal toxicity had been observed in human subjects receiving IDX14184, IDX14184 was put on partial clinical hold in 2012 due to concerns arising from the fact that the two drugs shared common metabolites that could be responsible for the clinical toxicities (see subsequently).
In both patients and monkeys, echocardiography proved to be a valuable tool for evaluating HCV nucleotide-related cardiac changes via a series of cardiac size and function assessments. For BMS-986094, 14 of the 34 treated patients showed evidence of severe or moderate left ventricular dysfunction. 20 Follow-up assessments of the cardiac function of patients participating in a 12-week phase IIb trial of IDX14184 are still ongoing as a result of the BMS-986094 experience in patients. However, the review of interim data did not identify any subjects with echocardiographic or biomarker changes outside of those expected in an aging population with underlying disease and multiple health issues (unpublished data).
In monkeys of the 32-week chronic toxicology study, IDX14184-induced echocardiographic changes observed at Weeks 9 and 29 were decreases in Fx Area, EjFx, and increases in LVAs, indicative of depression of left ventricle systolic function and left ventricle muscle compensatory enlargement. These changes were small in magnitude and asymptomatic clinically but were statistically significant at 25 and 60 mg/kg/day at Week 29. Echocardiographic changes at 60 mg/kg/day were consistent with minimal myocardial hypertrophy of the left ventricle histomorphologically and were generally reversible following a 13-week recovery period. In BMS-986094 monkey studies, a variety of dose- and time-dependent echocardiogram changes were seen starting at Day 32 at doses of 15 and 30 mg/kg/day: Changes at 30 mg/kg/day were profound and two animals in this group had to be euthanized at Day 32 with hemodynamic and anatomic pathology changes suggesting heart involvement. 22,23 Collectively, the long-term data collected from both patients and monkeys would suggest that IDX14184 possesses much better tolerability for patient treatment.
Although IDX14184 did not cause the severe injuries seen in human subjects with BMS-986094, cardiac and kidney toxicities were seen in the mouse, rat, and/or monkeys with both BMS-986094 and IDX14184. IDX14184-induced cardiac toxicity was manifested as acute cardiomyopathy characterized by hemorrhage, degeneration/necrosis and myocardial inflammatory infiltrates, and edema in female mice following oral administration of 500 mg/kg/day for 14 days; as cardiomyocyte vacuolation, hypertrophy, degeneration, and necrosis in the rat at doses ≥10 mg/kg/day in the 14-day study and ≥5 mg/kg/day in the 13-week study; and as cardiomyocyte degeneration, necrosis, and hypertrophy in cynomolgus monkeys at doses ≥60 mg/kg/day in the 2- to 32-week studies.
Thus, cardiotoxicity is apparent in the preclinical toxicology profiles of both compounds leading to the question of whether a common molecular feature(s) might be responsible for this phenomenon. IDX14184, like BMS-986094, is a 2′hydroxy, 2′methylguanosine analog, but the two drugs have very different prodrug structures (Figure 1) with different prodrug components and different chemistries: BMS-986094 is a phosphoramidate prodrug while IDX14184 is an S-acyl-2-thioethyl prodrug. In principle, the whole molecule or any of the component parts could be responsible for the observed toxicities, and the tissue site of processing of the prodrug may also be an important factor, while the IDX14184 prodrug is cleaved preferentially in the liver by carboxylesterase-2 and the most abundant cytochrome P450, CYP3A4, whereas BMS-986094 is processed by multiple enzymes
24
including cathepsin A which occurs in most tissues including the heart.
25
The marketed nucleoside prodrug GS-7977 (sofosbuvir or Solvaldi™; Figure 1), which has proven to be safe and effective in patients,
26
is a phosphoramidate prodrug like BMS-986094 (albeit with different prodrug moieties), suggesting that there is not a problem with phosphoramidate prodrugs per se. A more important difference between sofosbuvir and the other two drugs is that it possesses a 2′fluoro, 2′methyluridine rather than a guanosine base. To date, all HCV drugs containing a guanosine base have been withdrawn from further development including BMS-986094 and IDX14184 as well as IDX19368 (a 2′MeGMP analog with a different type of prodrug), GS-352938 (a cyclic phosphate prodrug of a 2′F, 2′MeGMP), and PSI-353661 (a phosphoramidate nucleotide of 2′F, 2′MeGMP)
27,28
This common thread seems to implicate 2′MeG and 2′MeGTP and the corresponding fluoro analogs as likely sources of toxicity, presumably due to their close structural relationship to GTP which is a key molecule in many GTPase-regulated cellular processes. One differentiator between BMS-986094 and IDX14184 may be 2′MeG exposure: The AUC0–24h and
The case for advancing a candidate such as IDX14184 in the clinic would be made easier by the identification of biomarkers that can reliably predict the emergence of cardiotoxicity in patients. Unfortunately, biomarkers of cardiac injury such as serum cTnI and cTnT levels or serum NT-proBNP proved to be uninformative for BMS-986094 in the preclinical species or in patients. Similarly, there were no IDX14184-induced changes in serum cTnI levels in the monkeys of the 13-week study, despite the observed cardiomyocyte degeneration and plasma NT-proBNP data were not informative in monkeys of the 32-week chronic toxicology study although myocardial hypertrophy was observed. Whether these biomarkers lacked predictivity for cardiac injury in monkeys, or their period of change was missed, or the cardiac pathology was subthreshold is not known.
Nucleoside/nucleotide analogs have been proven to be effective for the treatment of viral infections including HIV, hepatitis B virus, and HCV, but only one uridine nucleotide (sofosbuvir) has been approved to date for the treatment of HCV by the US Food and Drug Administration. Many other nucleoside/nucleotide analogs or prodrugs have failed in clinical or preclinical development, presumably due to toxicity. These include valopicitabine (NM283, 2′-C-methylcytidine analog) due to gastrointestinal toxicity, balapiravir (R1626, cytidine analog) due to lymphocytopenia, BMS-986094, IDX14184, and IDX19368 (2′-MeGMP analogs) due to cardiotoxicity and kidney toxicity, and GS-352938 (2′-MeGMP analog) and VX-135 (UMP analog) due to hepatotoxicity. 27,28
A major safety concern for nucleoside drugs is mitochondrial toxicity, which is manifested as hepatic failure, nephrotoxicity, pancreatitis, neuropathy, myopathy, and lactic acidosis. 31 –36 This issue was investigated for IDX14184 in a careful ultrastructural study in conjunction with the 13-week study in monkeys. No ultrastructural changes were observed in the heart of any monkey given IDX14184. Mildly swollen mitochondria were limited to the kidney and liver of one male and one female at 300 mg/kg/day that were euthanized on days 50 and 84, respectively. Overall, the ultrastructural results of the heart, liver, and kidney from the 13-week monkey study did not suggest that IDX14184 causes mitochondrial toxicity. This conclusion is further supported by the negative results of a series of biochemical assays designed to evaluate IDX14184 mitochondrial toxicity (unpublished results) and by studies showing that BMS-986094 does not appear to be a direct mitochondrial toxicant. 37
In conclusion, the toxicity profile of IDX14184 was characterized following single- and repeat-dose administration via oral gavage for durations up to 13, 26, and 32 weeks in mice, rats, and monkeys, respectively. The primary toxicity targets of IDX14184 were identified as the heart (rats, monkeys, and female mice), liver (rats and monkeys), kidney (mice, rats, and monkeys), skeletal muscles (mice, rats, and monkeys), and lower gastrointestinal tract including cecum, colon, and/or rectum (mice and rats). In relation to BMS-986094, the cardiac effects were minimal and asymptomatic. Nevertheless, the clinical development of IDX14184 was eventually terminated due to suboptimal efficacy in patients and lingering regulatory concerns on potential heart and kidney injury fueled by the problems encountered with BMS-986094 and the lack of a suitable clinical biomarker to monitor potential injury.
Footnotes
Acknowledgments
We thank the staff of Covance Laboratories Inc., Charles River Laboratories, WIL Research, CiToxLAB, and Experimental Pathology Laboratories, Inc., for the excellent work they have accomplished. We also thank Dr Kusum Gupta for discussion and Mr David Everett for assistance in the preparation of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
