Abstract
Objective:
The present study was designed to highlight the toxic impact of baclofen on both biochemical and histopathological aspects in rats’ liver, gastric, lung, kidney, and brain tissues.
Methods:
The study was performed on 30 healthy adult male albino rats divided into four groups with 5 rats in each control group, and 10 rats in either experimental groups (two experimental and two control groups). Five rats (negative control) were kept in a quite non-stressful environment, provided with food ad libitum and free access to water. Normal saline (1 ml) was given orally as placebo in the positive control group (n = 5). Experimental group III, baclofen acute toxicity group (10 rats): Each animal received a single dose of lethal dose (LD50) of baclofen orally by gavage. It equals 145 mg/kg body weight. The rats were observed for acute toxicity manifestations as well as for LD50 deaths. Group IV, (baclofen-dependent group, 10 rats): Each animal received baclofen (1/10th LD50) in gradually increasing doses for 1 month.
Results:
The levels of blood urea nitrogen, creatinine kinase, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, γ-glutamyl transpeptidase, cardiac troponin I, and prothrombin time in both baclofen-treated groups showed significant elevation when compared to controls. There were brain, lung, gastric, hepatic, and renal histopathological changes in baclofen-treated rats whose severity varied between the two experimental groups.
Conclusion and recommendation:
Baclofen toxicity is an under diagnosed emergency. Physicians should consider baclofen toxicity in users having hepatorenal dysfunction, presenting with altered mental status, bradycardia, and hypotension.
Introduction
Baclofen (parachlorophenyl γ-aminobutyric acid) is a potent agonist of the neurotransmitter γ-aminobutyric acid (GABA). 1 The centrally acting effect of the drug has been used mainly to treat spasticity of spinal origin, in addition to the treatment of alcohol dependence. 2 More recently, off-label use of high-dose baclofen markedly increased. It has been lately used to treat chronic hiccups of different etiologies as well as anxiety disorders. 3,4 Baclofen is well absorbed in the gastrointestinal tract, producing the determined therapeutic levels of 80–400 ng/ml. 5 The main method of drug excretion is through the kidney (70–80%), whereas the rest is metabolized in the liver or processed through the gastrointestinal tract. 4 Minute fraction of the drug only is capable of crossing the blood–brain barrier, leading to the desired effects. The drug’s half-life is nearly 6.8 h. 4 Patients with known impairment of renal functions have longer half-life with more crossing of the blood–brain barrier. The generalized central nervous system (CNS) depression effects will lead to fatigue, syncope, hypotension, ataxia, psychological disturbances, and cardiovascular and respiratory depression. 3,5
In patients with inadequate renal function, reports of toxic side effects with the initial dosing of 5 mg three times a day within only a few days have been reported. 6 Baclofen toxicity usually improves with hemodialysis with excretion rates similar to normal renal function. 7 Every drug has been associated with hepatotoxicity most likely due to the fundamental role of the liver in drug metabolism. Drug metabolites excreted from kidneys may also cause cellular damage ending in renal dysfunction. 8
Aim of the work
The aim of this work was to investigate the toxicity of baclofen on both biochemical and different histopathological changes in rats.
Materials and methods
Animals
Thirty healthy adult male albino rats weighing about 150–170 g were obtained from the animal house in faculty of Agriculture, Alexandria University. All animals were allowed free access to distilled water and laboratory chow ad libitum. To avoid stress of isolation or overcrowdings, five rats were housed per cage. They were left freely moving in their cages for 2 weeks with 12-h dark to 12-h light cycle for acclimatization before starting the experiment. Experimental procedures were performed in accordance with the guide of the care and use of laboratory animals approved by the committee of Alexandria University, the fewest number of animals estimated to obtain valid results were used and painful procedures were conducted with appropriate sedation to avoid pain and stress.
Drug
Baclofen was purchased from Sigma Chemical Company. All doses of baclofen were delivered to rats by using the gavage technique. At the end of each experimental time and using ether anesthesia, all animals were sacrificed after 24 h of the last dose.
Experimental design
Group I (negative control): Five rats were kept in a quite group non-stressful environment, provided with food ad libitum and free access to water for 1 month.
Group II (positive control): Each animal (five rats) received 1 ml/day normal saline 0.9% orally. They were kept throughout the experiment under the same conditions for 1 month.
Group III (baclofen acute toxicity group, 20 rats): Each animal received a single dose of lethal dose (LD50) of baclofen orally by gavage. It equals 145 mg/kg body weight. The rats were observed for acute toxicity manifestations as well as for LD50 deaths. 9
Group IV (baclofen dependence group, 20 rats): Each animal received baclofen (1/10th LD50) in gradually increasing doses for 1 month. Dependence was induced by giving the calculated dose of baclofen. The dose was then gradually increased by adding the initial calculated dose every 3 days till the end of the study month. 10
Biochemical assay
Three millilitres of blood were collected from all animals. The collected samples were centrifuged for 10 min at 5000 r/min. Separated clear serum was collected to assay liver function tests including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and γ-glutamyl transferase (GGT) as well as renal function tests, namely, serum creatinine (SC) and blood urea nitrogen (BUN), creatinine kinase (CK). This assay used enzyme linked immunosorbent assay (ELISA) manufactured by Ranbaxy Diagnostics Ltd (UK). Prothrombin time (PT) and partial thromboplastin time (PTT) were measured in seconds. Total protein (TP), cardiac troponin I (CTnl), and glucose level were measured in all studied groups. All studied biochemical laboratory findings following the study were given in Table 1.
Effect of baclofen administration on biochemical laboratory findings in adult male rats.
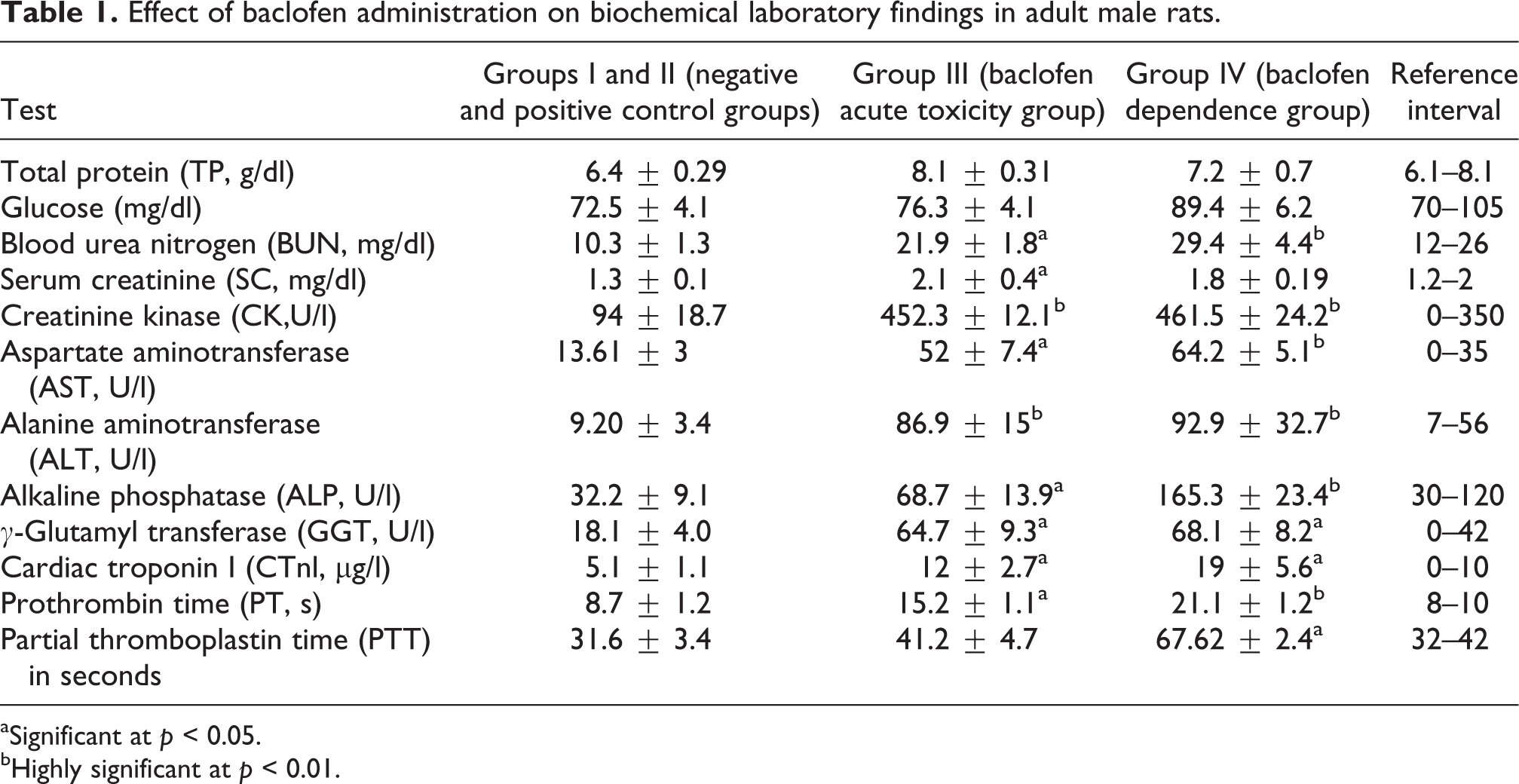
aSignificant at p < 0.05.
bHighly significant at p < 0.01.
Histopathological studies
At the end of the designed study, scarification and decapitation of treated rats were done. The right lobe of the liver and right kidney, lungs, stomach, and brain tissues of all rats were excised, fixed in buffered 10% formalin solution for 24 h and embedded in paraffin wax, and then sectioned and stained with haematoxilin and eosin stain (H&E) for histological examination under light microscope.
For comparison between groups in histopathological findings, qualitative diagnosis was used based on the sum of observable changes in the morphology of the analyzed tissue. The cognition of these changes was based on the pattern recognition of the observer and the comparison of these patterns with the known physiologic variation in tissue morphology in the respective species. Different grades used for histopathological evaluation in different studied tissues were given in details in Table 2. Histopathological qualitative changes in all studied tissues were summarized in Table 3.
Different grades used for histopathological evaluation in different studied tissues.

Summary of histopathological qualitative changes in each figure.

H&E: haematoxilin and eosin stain.
Statistical analysis
Data were presented as the mean ± standard deviation. Statistical analysis was performed using SPSS V 22. The 0.05 level of probability was used as the criterion of significance.
Results and discussion
The key role of liver and kidney in drug metabolism makes them prone to toxic injuries. However, elevated hepatic enzymes have also been documented in non-liver injury conditions as muscle injury. 11 Eighty-five percent is excreted unchanged in the urine and the remaining 15% is changed into its inactive, deaminated metabolite in the liver and excreted in the stool. 12
Manifestations of baclofen toxicity include delirium, seizures, excessive salivation, dizziness, nausea, vomiting, somnolence, hypotonia, and coma. Tachycardia and hypertension are considerably more common, especially at high doses, but bradycardia and hypotension can also occur with either high or low doses. 11
Highly significant elevation (p < 0.0l) in the level of CK was observed in both groups treated with baclofen. Elevated level of BUN and SC were recorded in group III when compared to all other groups. A significant difference in BUN, SC, and biochemistry parameters was detected between the control group and all other groups as given in Table 1.
The renal tissue histopathological examination in the current study revealed that eosinophilic intranuclear inclusions in tubular cells found in the majority of proximal tubular cells that are not associated with degenerative or functional changes in group III, as shown in Figure 1(c). Group IV rats showed tubular degeneration with ongoing regenerative hyperplasia, showing epithelial multilayering (Figure 1(d)).

Renal tissue in different groups: (a) Grade 0.0: normal architecture of the renal tissue in group I (negative control; H&E, ×200). (b) Grade 0.0: normal architecture of the renal tissue in group II (positive control; H&E, ×250). (C) Grade 2A.3: eosinophilic intranuclear inclusions in tubular cells found in the majority of proximal tubular cells that are not associated with degenerative or functional changes in group III (baclofen acute toxicity; H&E, ×400 magnification to show eosinophilic inclusions). (d) Grade 5.1: tubular degeneration with ongoing regenerative hyperplasia in group IV (baclofen dependence) showing hyperplastic tubules with epithelial multilayering (H&E, ×150). H&E: haematoxilin and eosin stain.
In accordance with our results, Donato et al. 13 demonstrated that acute in vivo administration of the GABA-B agonist, baclofen, lead to renal tubular transport impairments, manifested as diuresis, kaliuresis, natriuresis, decreased urine concentrating capacity, and altered glucose transport.
The same study revealed absent pathological changes, indicating that chemical observed effects with baclofen treatment would not be the consequence of cytotoxicity on renal cells. An impaired renal glucose transport had been previously described by Monasterolo et al. 14 using an isolated rat kidney model signifying that the renal proximal tubule could be a target for the GABA-B agonist baclofen actions.
Highly significant elevation (p < 0.0l) in the level of ALT was observed in both the groups treated with baclofen. A significant difference in AST, CK, GGT, and ALP biochemistry parameters was detected between the control group and all other groups, indicating baclofen-induced toxicity. Group IV rats showed a highly significant increase in ALP and a significant increase in PTT when compared to all other groups as given in Table 1. Liver histopathology in group III rats demonstrated coagulative necrosis of centrilobular hepatocytes with absence of any inflammatory response (Figure 2(c)), while group IV rats showed centrilobular (periacinar) liver necrosis, with perivenous area of coagulative necrosis and occasional cells with cytoplasmic vacuolation and ballooning. The affected areas in group IV revealed minimal inflammatory reaction (Figure 2(d)).

Liver tissue in different groups: (a) Grade 0.0: normal architecture in group I (negative control; H&E, ×200). (b) Grade 0.0: normal architecture in group II (positive control; H&E, ×200). (c) Grade 4.2: coagulative necrosis of centrilobular hepatocytes with absence of inflammatory response in group III (baclofen acute toxicity; H&E, ×100). (d) Grade 4.3: centrilobular (periacinar) necrosis. Marked perivenous area of coagulative necrosis and occasional cells with cytoplasmic vacuolation and ballooning, with minimal inflammatory reaction in group IV (baclofen dependence; H&E, ×200). H&E: haematoxilin and eosin stain.
Baclofen has low liver metabolism (about 15%) and is mainly eliminated unmodified by the kidney. 15 In contrast to our results, no hepatic side effects related to baclofen have been reported either in patients dependent of alcohol or those with neurological disorders by many researchers. 16 In accordance with our results, Macaigne et al. 17 reported a significant elevation of serum transaminases activity observed 4 days after initiation of treatment with baclofen. Chui and Pelot 18 reported that some baclofen-treated patients developed AST elevations, but no case of overt hepatitis was reported. Only one patient in their study with traumatic quadriplegia and spasticity developed ALT elevations after 6 weeks of escalating doses of baclofen without symptoms or jaundice, which fell to normal with decrease in dose. Caruso et al. 19 reported baclofen induced rash, eosinophilia, and jaundice 8 weeks after starting drug use that resolved on stopping it.
There was no detected significant difference between all studied groups with regard to TP and glucose level, as shown in Table 1, which is in contrast to Bonaventura et al. 20 who stated that baclofen did alter glucose homeostasis, alerting to the need to evaluate glucose metabolism during the clinical use of GABA-related drugs as baclofen. Renal glucose transport impairment was described by Monasterolo et al., 12 proving that the renal proximal tubules function might be targeted by the GABA-B agonist baclofen actions.
Interestingly, several authors 20 –23 demonstrated that acute or chronic administration of drugs acting through GABA-B receptors induced alterations in glucose homeostasis by affecting pancreatic function. In recent years, increased attention has been focused on the role of kidney in glucose homeostasis. 24
Also, there was a significant increase (p < 0.05) in CTnl in both baclofen-treated groups, as shown in Table 1. Baclofen toxicities had many serious reported cardiac adverse effects including prolonged QTc, first-degree heart block, premature atrial contractions, and supraventricular tachycardia. 25
Baclofen is considered as a proconvulsant drug, resulting from complex regulation by GABA-B of both the GABA-ergic and glutamatergic systems. 26 Experimental data suggested that activating the GABA-B receptors could accentuate neural excitation contrast in some parts of the brain. 27 In addition, akinetic mutism can occur through interruption of the thalamo-cortical limbic pathway, a consequence of toxicity that can mimic brain death. 28 Baclofen has anxiolytic effect reflecting GABA-B receptor stimulation within the amygdala. 29 Lanoux et al. 30 stated that EEG performed baclofen poisoning showed generalized slowdown and pseudo-periodic triphasic paroxystic pattern in favour of toxic encephalopathy. Histopathological results in the current research showed, some changes in brain tissues manifested by shrunken eosinophilic neurons with pyknotic nuclei in the substantia nigra in group IV treated rats only, while controls, and group III rats showed normal brain histopathology (Figure 3(d)), while all other groups showed normal histopathological brain appearance. Baclofen penetrates the blood–brain barrier leading to depression of neuronal activity in the CNS.

Brain tissue in different groups: (a) Grade 0.0: normal architecture of the brain tissue in the substantia nigra in group I (negative control; H&E, ×200). (b) Grade 0.0: normal architecture of the brain tissue in the substantia nigra in group II (positive control; H&E, ×200). (c) Grade 1.2: normal architecture of the brain tissue in the substantia nigra in group III with mild congestion (baclofen acute toxicity; H&E, ×200). (d) Grade 5.3: Shrunken eosinophilic neurones with pyknotic nuclei in the substantia nigra in group IV (baclofen dependence; H&E, ×100). H&E: haematoxilin and eosin stain.
Although the brain concentration of baclofen is lower than that in serum, it is eliminated more slowly from the CNS than from serum. CNS depression therefore persists for several days, even after plasma baclofen levels return to normal indicating pronounced neurotoxicity. 31 Baclofen high lipophilicity explains prolonged coma after baclofen overdose. 28
Drug-induced pulmonary injury may involve the lung parenchyma, airways, mediastinum, pleura, pulmonary vasculature, and/or the neuromuscular system. 32 Drug administration through both oral and parenteral routes is recurrently cited as cause of drug-induced interstitial lung disease; however, nebulized and intrathecal administration have been also concerned. 33 Toxic pulmonary changes may result from a direct or indirect drug effect. Direct effects may be either idiosyncratic or as a result of a toxic reaction of the parent drug or one of its metabolites. 34
Diagnosis of drug-induced lung disease, however, is easier said than done because the clinical, radiological, and histological findings are nonspecific. The link to drug use and the development of drug-related inflammatory damage or idiosyncratic toxicities are hard to objectify and recognize especially in cases using multiple drugs. 35 Normal H&E sections for all studied tissues in both control groups were seen in lung tissues. Pulmonary tissues in group III showed pulmonary oedema. The alveoli are filled with weakly eosinophilic material. The alveolar wall capillaries appeared dilated, with slight inflammatory reaction but little evidence of degenerative change, as shown in Figure 4(c)). Group IV showed congestion, inflammation, alveolar wall degeneration, and focal fibroblast proliferation (Figure 4(d)). Baclofen induced hypotension, pulmonary hypertension, and oxygen desaturation, which has been reported by some researchers. 36

Lung tissue in different groups: (a) Grade 0.0: normal architecture in group I (negative control; H&E, ×400). (b) Grade 0.0: normal architecture of the pulmonary tissue in group II (positive control; H&E, ×400). (c) Grade 4.3: Pulmonary oedema, alveoli are filled with weakly eosinophilic material and alveolar wall capillaries appear dilated with slight inflammatory reaction and little evidence of degenerative destructive changes in group III (baclofen acute toxicity; H&E, ×200 magnification). (d) Grade 5.2: congestion, inflammation, alveolar wall degeneration, and focal fibroblast proliferation in group IV (baclofen dependence; H&E, ×200). H&E: haematoxilin and eosin stain.
Animal studies have shown that baclofen accentuates gastric tone and increases the frequency and amplitude of gastric and duodenal contractions. 37 –41 Human studies have established that baclofen therapy reduces reflux symptoms in patients with reflux disease. 42–45, 3 The histopathology of gastric tissues in group IV only showed superficial mucosal necrosis in the glandular stomach, with edematous sub mucosa as demonstrated in Figure 5(d), while controls and group III rats showed normal gastric histopathology. Intrathecal baclofen effect on gastrointestinal motility has been proposed as demonstrated by gastrointestinal function deterioration in some reported cases. 46,47

Gastric tissue in different groups: (a) Grade 0.0: normal architecture of the gastric tissue in group I (negative control; H&E, ×200). (b) Grade 0.0: normal architecture of the gastric tissue in group II (positive control; H&E, ×250). (c) Grade 0.0: normal architecture of the gastric tissue in group III (baclofen acute toxicity; H&E, ×250). (d) Grade 5.2: superficial mucosal necrosis in the glandular stomach of group IV rats (baclofen dependence; H&E, ×200). The sub mucosa is edematous. H&E: haematoxilin and eosin stain.
Conclusions
Baclofen toxicity is an under diagnosed emergency. Physicians should consider baclofen toxicity in users having hepatorenal dysfunction, presenting with altered mental status, bradycardia, and hypotension. To our knowledge, this is the first research studying histopathological toxicity findings in experimental rats. Further investigations are recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
