Abstract
Doxorubicin (DOX) is an anthracycline derivative antibiotic that still frequently used in the treatment of solid tumors and hematological malignancies. The clinical use of DOX is largely restricted due to acute and chronic renal, cardiac, hematological, and testicular toxicities. Previous studies have indicated that oxidative stress, lipid peroxidation, and apoptosis in germ cells are the main factors in DOX-induced testicular toxicity, but the entire molecular mechanisms that responsible for DOX-induced testicular damage are not yet fully understood. Fluvastatin is a cholesterol-lowering agent that acts by inhibiting hydroxylmethyl glutaryl coenzyme A, the key enzyme for cholesterol biosynthesis. In addition to its cholesterol-lowering effect, fluvastatin showed an antioxidant effect by cleaning hydroxyl and superoxide radicals and this drug could have a protective effect by acting on the mammalian target of rapamycin (mTOR) signal pathway in testicular damage caused by obesity. This study aimed to investigate the possible protective and therapeutic effects of fluvastatin on the DOX-induced testicular toxicity model by histochemical, immunohistochemical, biochemical, and real-time polymerase chain reaction analyses. The present study indicates that fluvastatin may have a protective and therapeutic effect by removing reactive oxygen species and by regulating the mTOR, connexin 43, and matrix metalloproteinase 9 protein and messenger ribonucleic acid expressions, which play an important role in regulating the blood–testis barrier. On the other hand, the use of fluvastatin as a protective/prophylactic agent was found to be more effective than the use of this drug for treatment. In light of this information, fluvastatin may be a candidate agent that can be used to prevent testicular toxicity observed in men receiving DOX treatment.
Introduction
Cancer is a disease characterized by abnormal and uncontrolled cell proliferation and suppressed apoptosis and metastasis. 1 Surgery, radiotherapy, and chemotherapy are classically used methods in the treatment of cancer, which is one of the leading death causes of humanity in the last century. 2,3 Advances in these methods have significantly increased the success rate in cancer treatment, whereas especially chemotherapy protocols using potent cytotoxic drugs cause serious side effects and tissue damage in nontarget organs. 4,5 For example, testicular toxicity is one of the most common side effects of chemotherapeutic drugs. 6 –8 In fact, many studies in the past presented that chemotherapeutics could significantly affect spermatogenesis and thus male fertility 9 –11 For example, doxorubicin (DOX), an anthracycline-derived antibiotic that used successfully in the treatment of lung, prostate, and esophageal cancers as well as malignancies, such as lymphoma, leukemia, and sarcoma, is known to be able to trigger testicular toxicity and thus short- or long-term male infertility. 12 –15
Oxidative stress and increased production of reactive oxygen species (ROS) are regarded as the main phenomena responsible for DOX-induced testicular toxicity. Increased production of ROS in relation to DOX cytotoxicity leads to lipid peroxidation, deoxyribonucleic acid (DNA) fragmentation, and consequently necrosis and apoptosis in spermatogonial cells. 16 –18 The seminiferous epithelial thinning due to necrosis and apoptosis observed in spermatogonia and spermatocytes related to DOX-induced oxidative stress, the decrease in epididymal sperm count, and the increase in abnormal sperm morphology are important histopathological changes that after shown DOX cytotoxicity. 19 –21 Also, DOX-induced oxidative stress can cause serious side effects, such as cardiotoxicity and nephrotoxicity as well as testicular toxicity, thus the clinical use of this drug is restricted. 22,23 For this reason, research on natural or synthetic antioxidant substances that can be available to prevent or mitigate the oxidative damage caused by DOX is important in reducing the complications associated with this drug. 24,25
The mammalian target of rapamycin (mTOR) is a conserved and ubiquitous serine/threonine kinase that regulates numerous cellular function including cell growth, cell survival, proliferation, and autophagy. 26 In the studies on mTOR after discovery, this kinase has been shown to form two different complexes in mTORC1 and mTORC2 in mammalian cells. 27 From these complexes, mTORC1 plays a role in transcription, translation, cell growth, and cellular differentiation by interacting with nutrients, growth factors, hormones, and other proteins. On the other hand, the mTORC2 complex is activated by growth factors and regulates the organization of the cell framework and cellular survival mechanisms via Akt phosphorylation, another member of the phosphoinositol-3-phosphate signaling pathway. 28 In addition to their role in cell physiology, mTOR complexes play a role in “reshaping” the blood–testis barrier (BTB) during spermatogenesis. 29 From these complexes, mTORC1 facilitates the relaxation of the BTB by inducing the production of matrix metalloproteinase 9 (MMP-9) in the Sertoli cells, thus allowing the spermatocytes to migrate toward the adluminal compartment and preparation for meiosis divisions. 30 In contrast, mTORC2 induces the production of proteins, such as connexin 33 (Cx33) and Cx43, which are found in the structure of gap junction complexes in the BTB, thereby retightening the BTB during spermatogenesis. 31
Statins (atorvastatin, simvastatin, lovastatin, pravastatin, rosuvastatin, and fluvastatin) are drugs that inhibit the hydroxylmethyl glutaryl coenzyme A reductase, an essential enzyme for cholesterol biosynthesis, and these drugs widely used to regulate serum cholesterol levels in hyperlipidemic patients. 32,33
Many clinical and experimental studies have shown that these drugs have anti-inflammatory, neuroprotective, immunomodulatory, and antioxidant effects, in addition effect of lowering blood cholesterol levels. 34 For example, fluvastatin, a fully synthetic statin, has been found to exert an antioxidant effect by cleaning the hydroxyl and superoxide radicals in the body and reducing cellular damage caused by ROS. 33,35 Also, fluvastatin has been shown to prevent apoptosis due to lipid peroxidation induced by hydrogen peroxide (H2O2) in endothelial cells. 36 In addition, this drug has been shown to have protective effects on diabetic nephropathy 37 and cardiomyopathy 38 and obesity-induced testicular damage in rats. 39
The aim of this study is to evaluate the possible protective and therapeutic effects of fluvastatin in DOX-induced testicular toxicity and to clarify the molecular mechanisms underlying these effects. In this context, immunohistochemistry and real-time polymerase chain reaction (PCR) analyses were carried out to investigate the effects of fluvastatin on mTOR, Cx43, and MMP9 genes that were responsible for the regulation of BTB in the DOX-induced testicular toxicity. In addition, biochemical, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL), and histochemical analyses were performed to investigate the change in oxidative stress and apoptosis.
Materials and methods
Animals
Thirty-five male Sprague Dawley rats aged 4 weeks, weighing between 200 g and 250 g, were used for this study. Rats caged in controlled rooms with 22 ± 3°C temperature and 12-h light/12-h dark cycle were fed by standard rat feed and water ad libitum. Experimental procedures used in this study were approved by Ege University, Local Ethics Committee for Animal Experiments (approval no: 2017-098). All procedures were carried out in strict compliance with the animal experiment guidelines prepared for the care and use of laboratory animals.
Experimental design
The rats used in this study were divided into five groups, each contained seven rats. In determining this value, the principle of 3 R (Replacement, Reduction, and Refinement) proposed by Russell and Burch was taken into consideration. 40 Groups and experimental procedures applied to each group are listed in the following.
Control group
On the first, fourth, and seventh days of the study, rats were administered 7.5-mg/kg intraperitoneal saline treatment three times. Rats in this group have not undergone any treatment or intervention other than this procedure.
Fluvastatin group
The rats in this group were administered 6 mg/kg of fluvastatin (Lescol XL 80 mg; Novartis AG, Basel, Switzerland) by gavage for 7 days. 39 Other than fluvastatin treatment, no additional treatment or intervention was administered to rats in this group.
DOX group
On the first, fourth, and seventh days of the study, rats were administered 7.5-mg/kg intraperitoneal DOX (Koçak Farma, Istanbul Turkey) treatment three times. 41,42 Rats in this group have not undergone any treatment or intervention other than DOX treatment.
Prevention/prophylaxis group
The rats in this group were administered 6 mg/kg of fluvastatin by gavage for 7 days. In addition to the treatment of fluvastatin in this group rats, on the first, fourth, and seventh days of the study, 7.5-mg/kg intraperitoneal DOX treatment was applied. DOX treatments were performed 1°h after fluvastatin administration. 42
Treatment group
On the first, fourth, and seventh days of the study, rats were administered 7.5-mg/kg intraperitoneal DOX treatment three times. The rats in this group were administered 6 mg/kg of fluvastatin by gavage for 7 days 24 h after the last DOX administration. 42
At the end of the experiment, the rats were killed under combined ketamine (60 mg/kg, Ege Vet, Alfamine®; Alfasan International B.V., Holland, the Netherlands) and xylazine (10 mg/kg, Ege Vet, Alfazyne®; Alfasan International B.V.) anesthesia. Before killing, 1 ml of blood samples was taken into the heparinized tubes by cardiac puncture from the rats, which were sedated under anesthesia. These samples were used for biochemical analysis. After bloodletting, testicular tissues of rats were rapidly dissected and rats were euthanized by cervical dislocation. In order to achieve standardization in analysis, the left testes were preserved at −80°C without fixation for real-time PCR analyses, while the right testes were fixed by modified Davidson’s fixative (mDF) for histopathological studies.
Biochemical analyses
Analysis of lipid peroxidation
Blood samples collected by cardiac puncture under sterile conditions were centrifuged at +4°C temperature and at 1000 × g for 15 min, so that blood plasmas were obtained. Plasma samples frozen rapidly on dry ice were stored at −80°C until that were used. Lipid peroxidation was determined by measuring malondialdehyde (MDA) levels in plasma samples. 43 In this respect, MDA levels were determined in accordance with the instructions of the commercially available lipid peroxidation Colorimetric/Fluorometric Assay kit (BioVision®, Milpitas, California, USA). The absorbance of each sample was measured at 532 nm with the enzyme-linked immunosorbent assay (ELISA) plate reader (POLARstar Omega; BMG LABTECH, Ortenberg, Germany) and results were obtained.
Evaluation of serum SOD activity
Blood samples collected by cardiac puncture under sterile conditions were centrifuged at +4°C temperature and at 1000 × g for 15 min, so that blood plasmas were obtained. Plasma samples were frozen rapidly on dry ice and stored at −80°C until they were used. The superoxide dismutase (SOD) levels of groups were determined according to the commercially available SOD Activity Assay Kit (BioVision®). The absorbance of each sample was measured at 450 nm with ELISA plate reader (POLARstar Omega; BMG LABTECH) and results were obtained.
Analysis of blood plasma testosterone levels
Blood samples collected by cardiac puncture under sterile conditions were centrifuged at +4°C temperature and at 1000 × g for 15 min and blood plasmas were obtained. Plasma samples were frozen rapidly on dry ice. These samples were stored at −80°C until they were used. The testosterone levels of groups were determined according to the commercially available Rat Testosterone ELISA Kit (CUSABIO, Wuhan, People’s Republic of China). The absorbance of each sample was measured at 450 nm with ELISA plate reader (POLARstar Omega, BMG LABTECH) and results were obtained.
Histopathological evaluation of testicular tissues
Testicular tissues were fixed for 48 h in mDF. After fixation, the tissues were postfixed with neutral buffered formalin for 1 h and then removed from fixative by washing in phosphate buffer solution (PBS) for 24 h. 44 Afterwards, 5-μm sections were taken from the tissues embedded in paraffin blocks using routine protocols. Sections were stained with hematoxylin and eosin (H&E) after deparaffinization and dehydration. The tissues were photographed with a digital camera (C-5050; Olympus, Tokyo, Japan) mounted on the microscope (BX5; Olympus) after staining.
Immunoexpressions of the mTOR, Cx43, and MMP-9
Sections were incubated with 10% H2O2 (Sigma-Aldrich, Inc., St Louis, Missouri, USA) for 30 min for endogenous peroxidase blockade. To prevent nonspecific antibody-antigen binding, sections were incubated with Super Block (Scytec Consulting Inc., Greenwood Village, Colorado, USA) for 1 h at room temperature and washed with PBS. After this step, sections were incubated with 1:200 diluted primary antibodies (mTOR, MMP-9, and Cx43; Santa Cruz, California, USA) for 24 h at +4°C. At the end of this time, the sections were respectively incubated with biotinylated secondary antibody (Scytec Consulting Inc.) and horseradish peroxidase-conjugated streptavidin (Scytec Consulting Inc). Finally, the contrast staining of the sections incubated with diaminobenzidine was performed with Mayer Hematoxylin (Merck, Germany). Sections cleaned with xylol and then mounted with the Entellan (Merck). 22
Sperm parameters
Preparation of sperm samples
To ascertain sperm parameters, epididymal sperms were collected by slicing the cauda of left epididymes in 10 mL of saline and incubating for 15 min at 37°C to allow sperm to swim out of the epididymal tubules. The incubated samples were gently shaken several times to obtain a homogenous sperm suspension and mixed several times with pasteur pipette. Subsequently, 0.5 ml of the suspension was transferred into Falcon’s tubes which contain 2 ml of saline and it was centrifuged at 1000 × g for 5 min. After centrifugation, the supernatant was discarded and the pellet dissolved in 1 ml of saline. 45 These samples were used for sperm count and sperm morphology analyses.
Determination of epididymal sperm count
The hemocytometer was placed under a phase contrast microscope, was focused on the upper left square of hemocytometer, and then counting was started. The sperm extending along the left and upper corners of each secondary frame, or any part of them, were implicated in the counting process, while the sperm extending or coming into contact with the right and bottom corners of the secondary squares were removed from the count. In this way, the counting process was repeated by three blind researchers and the counting results and the average of these results were recorded. Then, the sperm count of each group was calculated using the calculation system proposed by Wang. 45
Sperm morphology analysis
The epididymis sperm prepared as in the section “Preparation of sperm samples” was drawn on clean glass slides. This slides air dried, fixed in methanol, and stained with Giemsa for 35 min. The slides were washed under running tap water to remove the excess of the stain. Air-dried slides were mounted with Entellan. 46 For each slide prepared in this way, 250 spermatozoa were randomly examined by three different researchers. The sperm counts in abnormal morphology were recorded and the percentage of abnormal sperm was calculated. 47
Real-time PCR analysis
During killing, 50 mg of testicular samples were taken into sterile isolation tubes. One milliliter of TriPure Isolation Reagent (Roche Applied Science, Penzberg, Germany) with guanidinium thiocyanate was added to the samples and stored at −20°C until used. The thawed tissues were homogenized with a glass-teflon homogenizer and the ribonucleic acid (RNA) isolation was carried out according to the directives of the TriPure Isolation Reagent Kit. The resulting RNA pellet was resuspended with diethylpyrocarbonate—treated 0.5% of SDS and incubated for 10–15 min at 55–60°C. In the next step, complementary DNA (cDNA) synthesis was carried out the following routine procedures. After cDNA synthesis, real-time PCR analysis was performed using SYBR® Green PCR Master Mix (ThermoFisher, Waltham, Massachusetts, USA) and Light Cycler 480 (Roche, Germany). Changes in gene expression were calculated by the 2−ΔΔCt method. 48 The primary sequences 49 –51 used in real-time PCR analysis are given in Table 1.
Primary sequences of genes used in real-time PCR analysis.

PCR: polymerase chain reaction; mTOR: mammalian target of rapamycin; Cx43: connexin 43; MMP-9: matrix metalloproteinase 9; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Analysis of TUNEL
TUNEL analysis was performed to determine apoptosis 52 in testicular tissues belonging to groups. After TUNEL staining performed according to the procedure of the ApopTag® Peroxidase In Situ Apoptosis Detection Kit (Merck), the section was photographed and the percentages of TUNEL positive cells between all experimental groups were compared.
Statistical analysis
Data analysis was performed with SPSS version 15.0 for Windows software (IBM Corp., Armonk, New York, USA). Comparisons were then made between the control and treatment groups using one-way analysis of variance followed by a Tukey’s post hoc test. Values were presented with standard error of the mean and p < 0.05 was considered statistically significant.
Results
Evaluation of biochemical oxidative stress markers in blood plasma
The results obtained from biochemical analyses show that the change of oxidative stress markers among the groups revealed that SOD levels were significantly lower in the DOX-treated groups (DOX, prophylaxis, and treatment) than the control group (p < 0.05). However, SOD activity was significantly increased in prophylaxis and treatment groups compared to the DOX group (p < 0.05). When prophylaxis and treatment groups were evaluated among themselves, SOD activity was found significantly higher in the prophylaxis group compared to the treatment group (p < 0.05). On the other hand, MDA levels were found higher in the DOX group compared to the control group. In addition, we found that MDA levels decreased significantly in the prophylaxis and treatment groups compared to the DOX group (p < 0.05). The evaluation between prophylaxis and treatment groups revealed that MDA levels were higher in the treatment group than in the prophylaxis group (p < 0.05). The changes in SOD activity and MDA levels between groups are shown in Figure 1.

SOD and MDA values of rat blood plasmas. Values are presented as mean ± SEM. Ι: statistically significant compared to the control group (p < 0.05). ΙΙ: statistically significant compared to the fluvastatin group (only fluvastatin-administered group; p < 0.05). ΙΙΙ: statistically significant compared to the DOX group (only DOX-administered group; p < 0.05). ΙV: statistically significant compared to treatment (the group-administered fluvastatin after doxorubicin exposure; p < 0.05). SOD: superoxide dismutase; MDA: malondialdehyde; SEM: standard error of the mean; DOX: doxorubicin.
Blood plasma testosterone level findings
Serum testosterone level analyses showed that there was a significant decrease in serum testosterone level in the DOX-treated groups (DOX, prophylaxis, and treatment) compared to the control group (p < 0.05). In addition, when the DOX-treated groups were evaluated among themselves, testosterone levels were significantly higher in the prophylaxis and treatment groups compared to the DOX group (p < 0.05). On the other hand, fluvastatin group was found an increase in testosterone level compared to the control group statistically (p < 0.05; Figure 2).

Blood plasma testosterone levels of all groups. Values are presented as mean ± SEM. Ι: statistically significant compared to the control group (p < 0.05). ΙΙ: statistically significant compared to fluvastatin group (only fluvastatin-administered group; p < 0.05). ΙΙΙ: statistically significant compared to the DOX group (only DOX-administered group; p < 0.05). ΙV: Statistically significant compared to the prophylaxis group (the group-administered fluvastatin before doxorubicin exposure; p < 0.05). SEM: standard error of the mean; DOX: doxorubicin.
Histopathological findings
Testicular tissues belonging to all experimental groups were stained with H&E for general histopathological parameters. In the assessment, testicular tissues of the fluvastatin group showed that normal seminiferous tubule conformations and spermatogenic cells (spermatogonia, spermatocytes, spermatitis, and sperm) located in the seminiferous tubule. When the testicular tissues of the DOX group examined, we were observed that most of the seminiferous tubules were atrophied and there were losses in the spermatogenic cells. Seminiferous tubule vacuolization and multinuclear giant cells are the other degeneration observed in the stroma of testicular tissues belonging to this group. Intensive edema in interstitial areas, loss of Leydig cells, and repletion of capillary vessels are other histopathological changes detected in the DOX group. In the prophylaxis and treatment groups compared with the DOX group, we did not observe most of the histopathological changes. In the treatment group, the number of degenerated tubules was increased compared with control, fluvastatin, and prophylaxis groups. However, there was a significant decrease in the number of degenerative tubules compared with the DOX group (Figure 3). Johnsen testicular biopsy scores 53 are given in Table 2 with p-values.
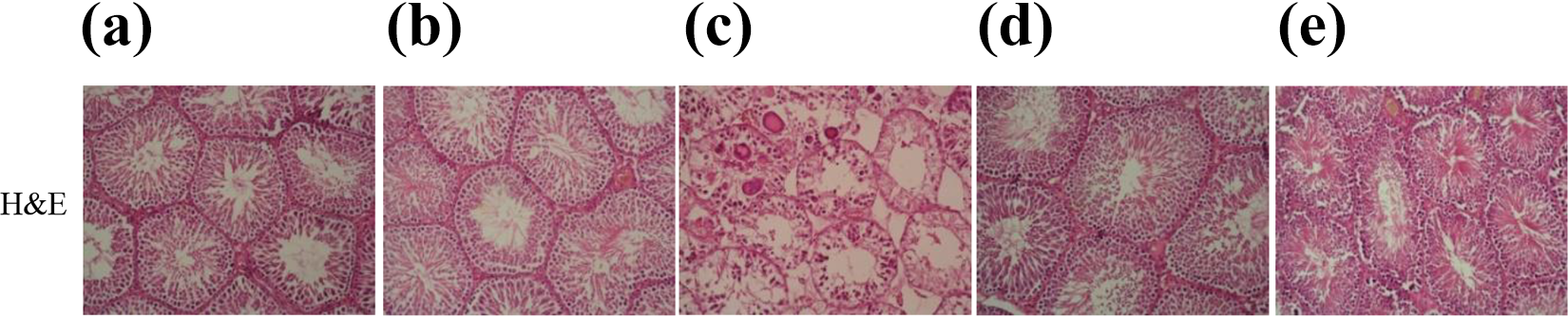
Hematoxylin and eosine (H&E) staining of sections from control and other experimental groups. Control (a) and only fluvastatin-administered groups (b) testes showed normal seminiferous tubules. Only DOX-administered group (c) testes showed a number of histopathological changes, such as spermatogenic and Leydig cells degeneration, seminiferous tubule vacuolization, and multinuclear giant cells. Prophylaxis group that administered fluvastatin before doxorubicin exposure (d) testes showed a decrease in spermatogenic and Leydig cells degeneration, seminiferous tubule vacuolization, and there were no multinuclear giant cells in these groups. In the treatment group that administered fluvastatin after doxorubicin exposure (e), the number of degenerated tubules was increased compared with the prophylaxis group. However, there was a significant decreased in the number of degenerative tubules compared with the DOX group (×20 magnification). DOX: doxorubicin.
Histological scores and immunoexpression levels of control and other experimental groups.a

mTOR: mammalian target of rapamycin; Cx43: connexin 43; MMP-9: matrix metalloproteinase 9; DOX: doxorubicin; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling; JTBS: Johnsen testicular biopsy scores; SEM: standard error of the mean.
aValues are presented as mean ± SEM. Ι: statistically significant compared to the control group (p < 0.05). ΙΙ: statistically significant compared to fluvastatin group (p < 0.05). ΙΙΙ: statistically significant compared to the DOX group (p < 0.05). ΙV: statistically significant compared to treatment (p < 0.05).
Immunohistochemical findings
In the immunohistochemical assessment, there were no significant differences between control and fluvastatin groups in terms of mTOR, Cx43, and MMP-9 protein expressions. However, mTOR and Cx43 expressions decreased in the other three groups compared with the control group, while the MMP-9 expression increased. When these three groups were evaluated, a significant increase in mTOR and Cx43 protein expressions and a significant decrease in MMP9 expression were observed in the prophylaxis and treatment groups compared to the DOX group (Figure 4). Immunoexpression levels (H-score) were calculated using the following equation
54
:


mTOR, Cx43, and MMP-9 immunostaining of all experimental groups testes. There was no significant difference between control (a) and only fluvastatin-administered groups (b) (p > 0.05) in terms of mTOR, Cx43, and MMP-9 protein expressions. On the other hand, mTOR and Cx43 expressions significantly decreased compared to the control group in the other three groups (p < 0.05); (c): only DOX-administered group; (d): prophylaxis group that administered fluvastatin before doxorubicin exposure; and (e): treatment group that administered fluvastatin after doxorubicin exposure, while the MMP-9 expression significantly increased (×40 magnification). mTOR: mammalian target of rapamycin; Cx43: connexin 43; MMP-9: matrix metalloproteinase 9; DOX: doxorubicin.
where I is the intensity of staining with a value of 1 (weak), 2 (moderate), or 3 (strong) and Pi is the percent of stained Sertoli or primary spermatocyte cells for each intensity varying from 0% to 100%.
The immunoexpression levels (H-score) and p-values of the groups are given in Table 2.
TUNEL staining findings
A small number of TUNEL positive cells were observed in both parenchymal and stromal areas of testicular tissues of the control and fluvastatin groups. When testicular tissue belonging to the DOX group was evaluated, it is noted that the number of TUNEL positive cell in primary spermatocytes and spermatids is quite high compared to the control group. In the assessment of prophylaxis and treatment groups, the number of TUNEL positive cells was higher when compared the control group, but the number of TUNEL positive cells in these groups decreased significantly compared to the DOX group. When prophylaxis and treatment groups were compared among themselves, TUNEL positive cell number in spermatogenic cells was found higher in the treatment group compared to the prophylaxis group (Figure 5). TUNEL scores and p-values of the groups are given in Table 2.

TUNEL staining of all experimental groups testes (×40 magnification). Control (a) and only fluvastatin-administered groups (b) testes showed a few TUNEL positive cells. Only DOX-administered group (c) testes showed a large number of TUNEL positive cells. In particular, TUNEL positive primary spermatocytes and spermatids are quite high compared to the control group (p < 0.05). TUNEL positive cells in the prophylaxis group that administered fluvastatin after doxorubicin exposure (d) were higher compared to the control group, but TUNEL positive cells in these groups decreased significantly compared to the only DOX-administered group (p < 0.05). TUNEL positive cell in spermatogenic cells was found higher in the treatment group that administered fluvastatin after doxorubicin exposure (e) compared to the prophylaxis group that administered fluvastatin before doxorubicin exposure (f) (p < 0.05). TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling; DOX: doxorubicin.
Findings of epididymal sperm parameters
In the epididymal sperm count, there was a significant decrease in the DOX group compared to the other groups (p < 0.05). In the treatment and prophylaxis groups, there was a significant increase in sperm count compared to the DOX group (p < 0.05). Also, the epididymal sperm count in control and fluvastatin group was similar (Table 3).
Findings of epididymal sperm count and sperm morphology analysis of all groups.

DOX: doxorubicin; SEM: standard error of the mean. Values are presented as mean ± SEM. Ι: statistically significant decrease compared to the control group (p < 0.05). ΙΙ: statistically significant decrease compared to the fluvastatin group (p < 0.05). ΙΙΙ: statistically significant increase compared to the DOX group (p < 0.05). ΙV: statistically significant increase in treatment compared to group (p < 0.05).
Findings from sperm morphology analysis showed that the percentage of abnormal sperm morphology in DOX-treated groups showed a significant increase when compared to the control group (p < 0.05). The percentage of sperm with abnormal morphology in the DOX group was 23.6%, while this percentage was 10.8% in the treatment group and 9.2% in the prophylaxis group. In addition, the percentage of abnormal sperm in the control and fluvastatin group was similar (Table 3).
Real-time PCR findings
Real-time PCR analysis revealed that mTOR and Cx43 messenger RNA (mRNA) expressions decreased while MMP-9 mRNA expression increased in the DOX group compared to other groups. In fact, mTOR and Cx43 mRNA expressions were downregulated 5.26-fold and 14.9-fold, respectively, in the DOX group compared to the control group. However, in other DOX-treated groups (prophylaxis and treatment), mTOR and Cx43 mRNA expressions were decreased, whereas MMP-9 mRNA expression was increased. On the other hand, compared to the DOX group, mTOR and Cx43 mRNA expressions were increased and MMP-9 mRNA expression decreased in prophylaxis and treatment groups. In addition, mRNA expression patterns were found closer to the control group in the prophylaxis group than the treatment group. The results of real-time PCR analysis are given in Table 4.
The fold change of mTOR, Cx43, and MMP-9 genes expressions in rat testes.

mTOR: mammalian target of rapamycin; Cx43: connexin 43; MMP-9: matrix metalloproteinase 9; DOX: doxorubicin. Values greater than 2 and less than −2 were considered significantly.
Discussion
DOX is an anthracycline-derived antibiotic that is currently preferred in the treatment of solid tumors and hematological malignancies. 55,56 This drug, which is restricted clinical use due to its destructive effect, especially in the heart and kidneys, causes severe cytotoxicity in the testis. 57
Experimental studies on DOX-induced testicular toxicity suggest that this drug causes significant histopathological changes in the stromal and parenchymal areas of the testis. 9 For example, Divya et al. shown that pathological changes, such as loss of spermatogenic cells, seminiferous tubules vacuolization, necrosis, atrophy, and the multinucleated giant cells in the stromal areas of testicular tissue in DOX-treated rats. In the same study, edema, vacuolization, and atrophy of Leydig cells were noted in the parenchymal areas. 58 In the histochemical evaluation, we found that in the DOX group, histopathological changes, such as loss in spermatogenic cells, the dissociation between cells, atrophy in some tubules, atypical cells in lumen, and multinuclear giant cells, were observed parallel to the data in the literature. The multinucleated giant cells detected in the DOX group arising from degeneration of spermatids or spermatocytes and the enlargement of intercellular bridges between these cells. 59 Many studies have demonstrated that these cells are associated with seminiferous tubular degeneration that results from the use of chemotherapeutic drugs, such as 5-fluorouracil and tamoxifen, as well as DOX. 60,61 Given our literature and findings, the fact that most pathologies, such as multinucleated giant cells observed in the DOX group, are not observed in prophylaxis and treatment groups suggest that fluvastatin protects testicular tissues of these groups from the side effects of DOX. However, the fact that the number of degenerate tubules in the prophylaxis group is less than in the treatment group suggests that prophylactic use of fluvastatin in the prevention of DOX-induced testicular toxicity may be more effective than in the therapeutic use.
Other important fact that threaten male fertility in DOX-induced testicular toxicity is the decrease in testosterone level and total epididymal sperm count. 47,62 It is known that the decrease in testosterone level following DOX application is due to the decline in mRNA expression and activation of the enzymes responsible for androgenesis in the testes. 63,64 Considering the importance of testosterone for the continuity of spermatogenesis, sperm production, and sperm maturation, 65 –67 the decrease in the number of epididymal sperm after DOX treatment becomes more meaningful. In fact, many studies shown that there was a decrease in epididymal sperm count in parallel with the decrease in testosterone level after DOX exposure. 47,62,65,68 In our study, we found a significant decrease in the levels of testosterone and epididymal sperm counts in DOX-treated groups in parallel with the literature. On the other hand, the fact that testosterone levels and epididymal sperm counts were significantly higher in the prophylaxis and treatment groups compared to the DOX group indicates that fluvastatin applied to these groups may have a protective effect on spermatogenesis in testicular toxicity after DOX exposure. Cui et al. showed that fluvastatin application in testicular dysfunction due to obesity restored hormone concentrations, such as follicle-stimulating hormone, luteinizing hormone, and testosterone and brought its closer to normal. 39 When these findings are evaluated together, it reinforces the idea that fluvastatin may have protective effects on spermatogenesis in testicular injury.
Although entire mechanisms responsible for DOX-induced testicular toxicity have still not been absolutely resolved, studies have shown that lipid peroxidation and apoptosis caused by oxidative stress in germ cells are the predominant mechanisms for testicular toxicity arise out of the use of this drug. 69 –71 In addition, many studies have shown that DOX application leads to a decrease in the activity of antioxidant enzymes, such as SOD, as well as an increase in lipid peroxidation products, such as MDA in testis tissue. These findings indeed support the argument that oxidative stress and lipid peroxidation are the main mechanisms involved in testicular toxicity. 72,73 Biochemical analysis showed a significant increase in MDA level and a significant decrease in SOD activity in the DOX group. Also, a significant decrease in MDA level and a significant increase in SOD activity were found at prophylaxis and treatment groups in contrast with the DOX group. In the literature, it has been shown that fluvastatin may have an antioxidant effect by increasing the activity of enzymes, such as SOD, as well as cleaning the hydroxyl and superoxide radicals. 35,73,74 These results considering with the literature, fluvastatin may have an antioxidant effect against DOX toxicity in testis.
It is assumed that the main reason for the sperm morphology anomalies that occur after DOX exposure is the permanent chromosomal anomalies in spermatogonial cells and the division defects observed in these cells 75 . Also, one of the reasons for the increase in the number of sperm with abnormal morphology in DOX-induced testicular toxicity is increased oxidative stress. 65,76 In our study, we found that the number of sperm with abnormal morphology increased parallel with the increased oxidative stress in the DOX-treated groups. In addition to this finding, the decrease in sperm count with abnormal morphology in prophylaxis and treatment groups may be the result of the antioxidant effect of fluvastatin as mentioned above.
As noted above, the other important mechanism involved in the DOX-induced testicular toxicity mechanism is apoptosis. Previous studies have shown that DOX-induced oxidative stress can trigger apoptosis in the testis via nuclear factor kappa B and mitochondrial p53 signaling pathways. 72,77 In our study, the apoptotic index increased dramatically in the DOX group compared to the other groups, but this index was found closer to the control group in the prophylaxis and treatment groups. Xu et al. showed that low-dose fluvastatin administered to human endothelial cells reduced the H2O2-induced apoptosis by increasing Bcl-2 expression in these cells. 36 Our findings support previous studies and suggest that fluvastatin can attenuate oxidative stress-induced apoptosis.
mTOR is a protein with serine/threonine kinase activity originally defined as the cellular target of rapamycin in yeasts. 78 mTOR complexes involved in biological processes, such as autophagy, apoptosis, and spermatogenesis in mammalians. 79,80 For example, Xu et al. emphasized that mTORC1 is a vital importance in the regulation of mitotic proliferation in spermatogonia and the maintenance of germ cell pool. 81 In addition, mTORC1 and mTORC2 are also known to be involved in the regulation of the BTB. 31 mTORC1, one of these complexes that are acting as opposed to each other, induces MMP-9 production in Sertoli cells, and MMP-9 lead to degradation of the proteins in the adhesion complex in the BTB, thus causing the BTB to relax. 30 In contrast, mTORC2 induces the expression of Cx43, which has a vital role in maintaining the communication between the gap and tight junction complexes and thus maintaining the compact structure of BTB. Therefore, activation of mTORC2 in the testicular tissue results in the “tightening” of BTB. 31 This arrangement of BTB by mTOR complexes facilitates the migration of spermatocytes to the adluminal compartment during preleptoten phase and the preparation for meiosis division in this compartment, thus plays an important role in the continuity of spermatogenesis. 82 The data obtained from immunohistochemical staining revealed that mTOR positive cell density decreased in the DOX-treated groups (DOX, prophylaxis, and treatment) compared to the control group. However, mTOR positive cell density was higher in prophylaxis and treatment groups than in the DOX group. In addition to these data, we obtained from real-time PCR analysis revealed that mTOR mRNA expression was downregulated in the DOX-treated group compared to the control group. However, mTOR mRNA expression in prophylaxis and treatment groups was found 4.26 and 2.81 times higher than the DOX group, respectively. Also, Cx43 mRNA and protein expression in DOX-treated groups were downregulated compared to the control group. In addition, Cx43 mRNA expression was found 10.6 times higher in the prophylaxis group and 6.7 times higher in the treatment group compared to the DOX group. Immunohistochemical and real-time PCR analyses for MMP-9 showed that MMP-9 mRNA and protein expressions were upregulated in DOX-treated groups compared to the control group. In the context of this data, it can be argued that DOX has an effect on the mTOR signaling pathway, causing disruption in the regulation of BTB. In histochemical analyses, the presence of seminiferous tubular conformational defects in the DOX group and the presence of spermatogonial cells that spilled into the lumen without completing their maturation support this idea. On the other hand, the mTOR, Cx43, and MMP-9 expression levels in the fluvastatin-treated groups were found closer to the control group and the lack of most of the pathologies observed in the DOX group. In view of all the data we have obtained, fluvastatin may lead or contribute to protective and therapeutic effects with the regulation of BTB in DOX-induced testicular toxicity.
In conclusion, the results obtained from our biochemical and histopathological analyses suggest that fluvastatin in DOX-induced testicular toxicity may prevent the pathologies caused by the use of this chemotherapeutic agent by reducing the lipid peroxidation and activating the antioxidant system. Real-time PCR and immunohistochemical analyses suggest that fluvastatin in DOX-induced testicular toxicity may have both prophylactic and therapeutic effects in testicular toxicity by regulating mTOR, Cx43, and MMP-9 proteins and mRNA expressions that play important roles in the regulation of BTB. In addition, the prophylactic use of this drug was observed to be more favorable than therapeutical.
Footnotes
Acknowledgement
The experimental procedures on the animals in this study were performed in Ege University, Drug Research and Development and Pharmacokinetic Applications (ARGEFAR).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
