Abstract
Bisphenol-A (BPA) is a chemical substance that is widely used in industry for manufacturing of plastic bottles and resins. Recent reports found that BPA may mimic the effects of estrogen to a great manner that might disrupt the normal hormonal balance in the human body. Fluvastatin is an agent used for treatment of hypercholesterolemia that was proven to possess promising antioxidant ant anti-inflammatory properties. Taxifolin is a polyphenolic compound with potential antioxidant and antiestrogenic effects. The present study investigated the prospect of fluvastatin with or without taxifolin to mitigate testicular dysfunction elicited by BPA in rats. In a model of BPA-induced testicular toxicity, the hormonal profile was assessed and the testicular tissues were examined by biochemical analysis, histopathology, and immunohistochemistry. Fluvastatin with or without taxifolin improved the body weight gain, hormonal profile, testicular weight and functions, sperm characteristics, the antioxidant status, and the anti-inflammatory mechanisms together with enhancement of autophagy and suppression of the proapoptotic events induced by BPA in the testicular tissues. In addition, fluvastatin with or without taxifolin significantly mitigated the histopathological and the immunohistochemical changes induced by BPA in the testicular tissues. These desirable effects were more pronounced with fluvastatin/taxifolin combination relative to the use of each of these agents alone. In tandem, fluvastatin/taxifolin combination might counteract the pathogenic events induced by BPA in the testicular tissues which may be considered as a novel strategy for amelioration of these disorders.
Introduction
Bisphenol-A (BPA) is a synthetic diphenylmethane derivative that is used for the manufacturing of certain types of plastics and resins that represent the containers that store a wide range of foods and beverages. 1 BPA is considered as one of the highest volume of chemicals produced and used all over the world, particularly in the water bottles. 2 Recent reports paid the attention towards the findings that chronic exposure to BPA may cause endocrinal dysfunction.3,4 These findings might be attributed to the close similarity between the phenol groups in both BPA and estradiol, which may confer an effect to BPA on the estrogenic pathways in the body. 5 In addition, high concentrations of BPA were proven to block the androgen receptors (AR), modulate Leydig cell steroidogenesis, and hinder LH receptor-ligand binding. 4 Also, massive generation of reactive oxygen species (ROS) induced by BPA with subsequent apoptotic changes associated with suppression of autophagy were proposed as possible underlying mechanisms. 6 Together, these effects attracted the attention of the authorities to the possible reproductive toxicity induced by BPA. 7
Fluvastatin is one of the agents that restore the normal serum cholesterol levels in cases of hypercholesterolemia. 8 Recent trials postulated that fluvastatin has the ability to clean hydroxyl and superoxide radicals from the body and augment the antioxidant defense mechanisms. 9 In addition, fluvastatin was proven to modulate the mammalian target of rapamycin (mTOR) signaling pathway in a manner that may significantly ameliorate toxin-induced testicular damage. 10 Moreover, fluvastatin was proposed as a potentially vital agent that restores the balance between the pro-apoptotic and the anti-apoptotic proteins which is usually lost in toxin-induced gonadal dysfunction. 11
Taxifolin is a flavonoid that was proven to have potent radical scavenging activities in various tissues of the body. 12 Taxifolin was reported to have endogenous antioxidant properties which may enhance the activity of the antioxidant enzymes and ameliorate the deleterious effects of the different types of ROS on body tissues. 13 In addition, the proven effects of taxifolin on the inflammatory and the apoptotic pathways together with its anti-estrogenic effects may make it a potential candidate for amelioration of BPA-induced testicular dysfunction. 14 The main target of the present trial was to investigate the prospect of fluvastatin with or without taxifolin to mitigate the testicular dysfunction elicited by BPA in rats.
Materials and methods
Chemicals and reagents
BPA was supplied by Sigma Aldrich Co., USA (CAS # 80-05-7). Fluvastatin was provided by a commercial vendor (Cayman Chemical, Michigan, USA; CAS # 93957-54-1). Taxifolin and methyl cellulose were obtained from Santa Cruz Biotechnology, Heidelberg, Germany (CAS # 480-18-2 and 9004-65-3, respectively). The other reagents were supplied as a kind gift from RayBiotech, GA, USA and were of analytical grade. BPA was dissolved in olive oil. Fluvastatin and taxifolin were administered as a suspension in 1% methyl cellulose solution.
Experimental animals
The present study was conducted using 48 male 8-weeks old Wistar rats, weighing about 170–230 g. Rats had unrestricted access to the lab food and drinking water, under experimental conditions of 12 L/12D lighting system, with humidity of 55–68%, and temperature of 22–25°C throughout the experiment. Rats were allowed to acclimatize for 10 days before starting the experimental procedures. This study was approved by the Research Ethics Committee of Faculty of Medicine, Tanta University, Egypt (Approval code 33516/8). Animal handling was carried out by strictly following Helsinki declaration of animal ethics.
Experimental protocol
Animals were randomly divided into six equal groups of eight rats per group by a technician who was unaware of the identity of the experimental groups as follows: I) Control group, received 0.5 mL olive oil daily by oral gavage; II) BPA group, received BPA in a dose of 20 μg/kg by oral gavage 15 ; III) BPA + methyl cellulose group, received 1% methyl cellulose solution concomitantly with BPA by oral gavage; IV) BPA + Fluvastatin group, received fluvastatin (10 mg/kg/day) by oral gavage 16 concomitantly with BPA; V) BPA + Taxifolin group, received taxifolin (50 mg/kg/day) by oral gavage 17 concomitantly with BPA, and VI) BPA + Fluvastatin + Taxifolin group, received BPA, fluvastatin, and taxifolin in the fore-mentioned doses by oral gavage. Olive oil, BPA, methyl cellulose, fluvastatin, and taxifolin were given daily for 8 weeks. Body weight was measured at the start of the study and then every 7 days throughout the whole study.
Harvesting the blood and tissue samples
At the end of the experimental period, blood samples were obtained from the retro-orbital plexus, centrifuged and the sera were used for measurement of the hormonal profile. After animal euthanization, both testes from each rat were dissected and washed with saline. The right testes were weighed and kept at –80°C for further homogenization and assay of the biochemical measurements. The left testes were put in 10% formol-buffered saline (pH 7.4) for further histopathological and immunohistochemical assay.
Measurement of the hormonal profile
Serum follicle-stimulating hormone (FSH) was assayed using kits supplied by Elabscience, Texas, USA (CAT # E-EL-R0391). Also, ELISA kits produced by CUSABIO, Houston, TX, USA (CAT # CSB-E12654r) were utilized for measurement of serum luteinizing hormone (LH) levels according to the vendor’s protocol. In addition, kits purchased from Boster Biological Technology, Pleasanton, USA (CAT # EK7014) were used for assessment of serum levels of testosterone according to the vendor’s instructions. Following the manufacturer’s guide, assay of tissue 3-beta hydroxysteroid dehydrogenase type 1 (HSD3B1) and 17-beta hydroxysteroid dehydrogenase 1 (HSD17B1) was executed using kits purchased from MyBioSource, San Diego, USA (CAT # MBS456446 and HSD17B1, respectively).
Assay of the markers of the testicular functions
Tissue lactate dehydrogenase (LDH) was assayed using ELISA kits (CAT # MBS2018912, MyBioSource, San Diego, USA). The levels of alkaline phosphatase (ALP) and glucose-6-phosphate dehydrogenase (G6PD) activity were determined using kits purchased from BioVision, Milpitas, USA (CAT # E4575-100 and K757-100, respectively). ELISA kits supplied by AssayGenie, Dublin, Ireland (CAT # RTFI01238) were utilized for detection of tissue acid phosphatase (ACP). Assay of the markers of the testicular functions was performed as instructed by the vendor.
Assay of the antioxidant enzymes, nuclear factor erythroid 2-related factor 2 (Nrf2), and heme oxygenase-1 (HO-1) content in the testicular tissues
For assessment of the antioxidant status of the testicular tissues, kits supplied by MyBioSource, San Diego, USA (CAT # MBS726781 and MBS9308239) were utilized for measurement of tissue catalase (CAT) and glutathione reductase (GR), respectively, according to the vendor’s instructions. Nrf2 content of the testicular tissues was quantified using kits purchased from Novus Biologicals, USA (CAT # NBP3-08161) following the manufacturer’s guide. The vendor’s instructions of the kits supplied by CUSABIO, USA (CAT # CSB-E08267r) were followed to determine HO-1 content in the testicular tissues.
Assay of transforming growth factor beta 1 (TGF-β1), interleukin 8 (IL-8), interleukin 18 (IL-18), and nucleotide-binding domain-like receptor family, pyrin domain-containing 3 (NLRP3) inflammasome in the testicular tissues
TGF-β1 levels were assayed using kits obtained from ThermoFisher Scientific, USA (CAT # BMS623-3). ELISA kits purchased from ABclonal, Woburn, MA, USA (CAT # ERI0063 and RK03757) were used to conduct assay of IL-8 and IL-18, respectively, following the vendor’s instructions. The levels of NLRP3 inflammasome were measured using kits provided by Abcam, USA (CAT # ab277086) according to the guidelines of the manufacturer.
Determination of testicular tissue phospho-mTOR, phosphoprotein kinase B (p-AKT), p38 mitogen-activated protein (p-MAP) kinase and beclin-1 levels
Phospho-mTOR levels were quantified using kits supplied by Cell Signaling, USA (CAT # 7976) with strict adherence to the provider’s guide. P-AKT was measured in the testicular tissues with the aid of ELISA kits (RayBiotech, USA, CAT # PEL-AKT-S473-T-1) according to the vendor’s instructions. ELISA kits purchased from MyBioSource, San Diego, USA (CAT # MBS720509 and MBS733192) was used to detect the levels of tissue p38 MAP kinase and beclin-1, respectively, following the provider’s instructions.
Assay of the apoptotic markers in the testicular tissues
Caspase 3, Bax, and Bcl-2 in the testicular tissues were quantified using kits supplied by MyBioSource, USA (CAT # MBS018987, MBS935667 and MBS2881713, respectively) according to the protocol provided by the vendor.
Determination of the characteristics of the spermatozoa
The epididymis was sliced in 5 mL of Ham’s F10 and incubated for 5 min at 37°C in 5% CO2. This allows the sperms to swim out of the epididymal tubules. Immediately after that, one drop of the suspension was placed on a glass slide with a cover slip and the percentage of the motile sperms was counted microscopically within 2–4 min of their isolation using 10 microscopic fields at ×400 magnification. 18
The number of the epididymal sperms was counted by following the method of Wang. 19 Also, smears of the epididymal suspension were prepared on glass slides and left to dry overnight, after which they were stained with 1% eosin-Y/5% nigrosine. Then, these slides were examined at 400× for detection of the morphological abnormalities of the spermatozoa. 20
Histopathological evaluation of the testicular tissues
The left testes were immersed in 10% formalin solution for 24 h, dehydrated in ethanol then embedded in paraffin. After that, tissue sections were cut, put on glass slides, stained with hematoxylin and eosin and inspected using the light microscope. Spermatogenesis was assessed using Johnsen’s 21 tubular biopsy score (JTBS). Also, seminiferous tubules diameter (STD) and the thickness of the germinal epithelium were evaluated according to Erfani Majd et al. 22 under a light microscope (Olympus Optical Co., Japan) using Image J Software (1.49v), NIH, USA.
Evaluation of the expression of Ki-67 in the testicular tissues
Five μm sections of the left testes were dewaxed, then rehydrated in xylene and distilled water. Then, retrieval of the antigen was executed in a microwave oven. Endogenous peroxidase activity was quenched by 3% hydrogen peroxide solution at 25°C for 30 min. After that, these sections were blocked using 5% bovine serum for 15 min. Then, sections were incubated with specific primary antibodies against Ki-67 (GB11010; Wuhan Saiweier Biological Technology, China, dilution 1:100) overnight at 4°C in a humidified chamber. Then, sections were washed and incubated with secondary antibody, and stained by 3, 3′-diaminobenzidine. After that, sections were counterstained with hematoxylin and rinsed in tap water. The labeled sections were examined by a light microscope at a magnification of ×10 to detect areas with obvious Ki-67 staining. In every section, the most intense three areas stained with Ki-67 were detected and the ratio of the number of the nuclei positively stained with Ki-67 antigen was counted in each area using Image-J software (1.49v), NIH, USA. The average of these ratios indicated the degree of positive Ki-67 immunostaining. 23
Statistical analysis of the results
Analysis of the obtained results was carried out using GraphPad Software version 6.04 for Science Inc., CA, USA. Shapiro-Wilk test was utilized for examination of the parametric data. The statistical significance among the different groups was assessed by one-way analysis of variance (ANOVA) and Tukey’s test (Significant at p-value less than 0.05). The obtained data were presented as mean ± standard deviation (SD).
Results
Fluvastatin and/or taxifolin improved the body weight gain in BPA-treated animals
Significant decrease in the final body weight (0.35-fold change) was noted in rats treated with BPA relative to the control group. This decrease was not improved with administration of methyl cellulose but treatment with either fluvastatin or taxifolin improved the final body weight (1.17- and 1.21-fold change, respectively) versus animals treated with BPA alone. This improvement was significant with fluvastatin/taxifolin combination relative to treatment with either fluvastatin or taxifolin alone (1.16- and 1.13-fold change, respectively) (Figure 1). Fluvastatin with or without taxifolin affects the body weight gain in bisphenol-A treated animals (Mean ± SD). (a) Significant versus the control group (p-value less than 0.05), (b) Significant versus bisphenol-A group (p-value less than 0.05), (c) Significant versus bisphenol-A + Fluvastatin group (p-value less than 0.05), and (d) Significant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin combatted the hormonal changes induced by BPA
Effect of fluvastatin with or without taxifolin on the hormonal profile in bisphenol-A treated animals (mean ± SD).
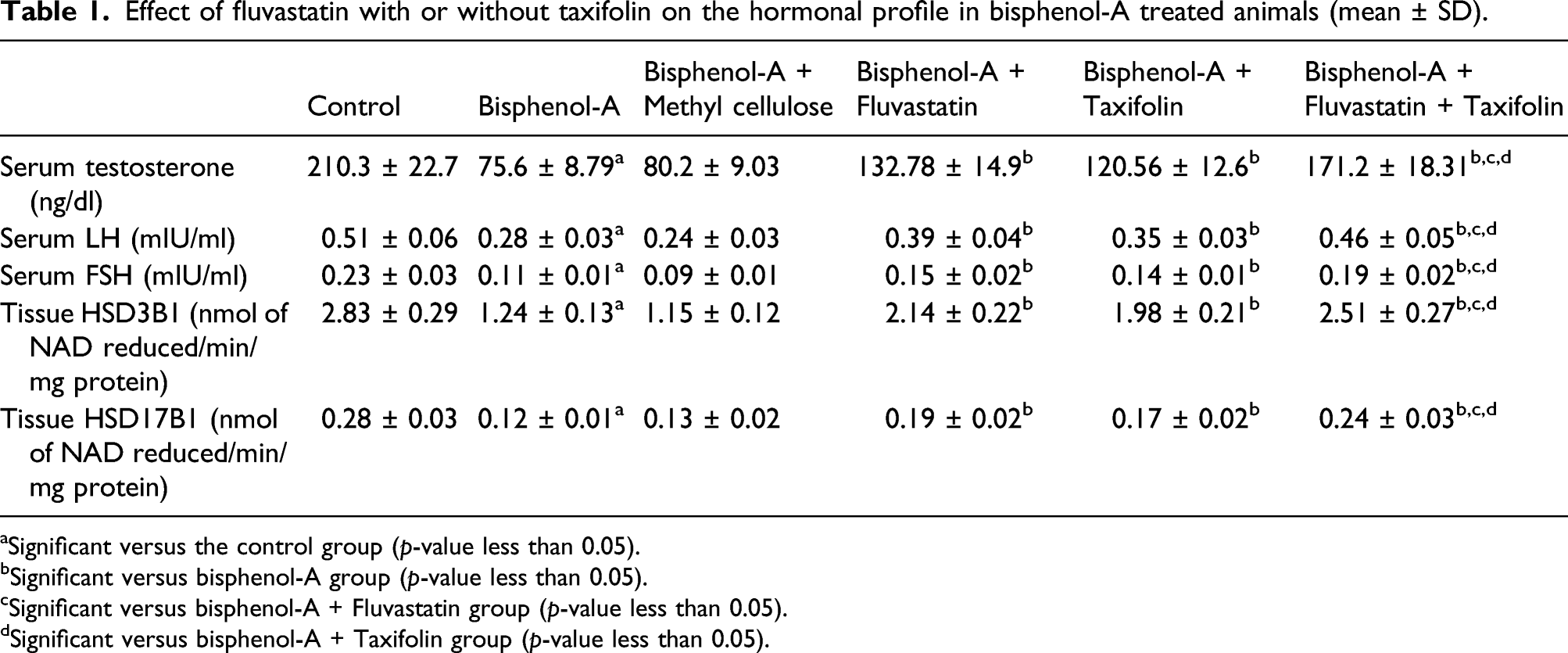
aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin restored the markers of testicular functions to the normal values compared to PBA-treated animals
Effect of fluvastatin with or without taxifolin on the testicular function tests in bisphenol-A treated animals (mean ± SD).

aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin improved the antioxidant status and mitigated the changes in Nrf2/HO-1 content in BPA-treated animals
Administration of BPA elicited significant decrease in tissue CAT, GR, Nrf2, and HO-1 (0.45-, 0.47-, 0.51- and 0.56-fold change, respectively) in the testicular tissues versus the control group. Methyl cellulose didn’t significantly affect these parameters compared to animals that received BPA alone. These changes were reversed in the groups treated with fluvastatin and/or taxifolin but rats treated with fluvastatin/taxifolin combination exhibited maximal improvement relative to groups treated with each of these agents alone (Figure 2). Effect of fluvastatin with or without taxifolin on tissue antioxidant enzymes and Nrf2/HO-1 content in bisphenol-A treated animals (Mean ± SD). (a) Significant versus the control group (p-value less than 0.05), (b) Significant versus bisphenol-A group (p-value less than 0.05), (c) Significant versus bisphenol-A + Fluvastatin group (p-value less than 0.05), and (d) Significant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin abrogated the changes related to the inflammatory cascade induced by BPA in the testicular tissues
Effect of fluvastatin with or without taxifolin on the inflammatory markers in bisphenol-A treated animals (mean ± SD).

aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin enhanced the autophagy-associated pathways in the testicular tissues of the studied groups
Effect of fluvastatin with or without taxifolin on testicular tissue phosphorylated AKT, mTOR, p38 MAP kinase, and beclin-1 levels in bisphenol-A treated animals (mean ± SD).

aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin significantly combatted the apoptotic changes induced by BPA in the testicular tissues
Animals treated with fluvastatin or taxifolin exhibited significant suppression of apoptosis evidenced by significant decrease in caspase-3 (0.34- and 0.24-fold change, respectively) and Bax (0.27- and 0.21-fold change, respectively) with significant increase in Bcl-2 levels (1.38- and 1.3-fold change, respectively) in the testicular tissues when compared to animals treated with BPA alone. The maximal inhibition of apoptosis was evidenced in rats that received fluvastatin/taxifolin combination. However, methyl cellulose didn’t exhibit any antiapoptotic effects when administered to BPA-treated rats (Figure 3). Effect of fluvastatin with or without taxifolin on caspase-3, Bax, and Bcl-2 levels in the testicular tissues of bisphenol-A treated animals (Mean ± SD). (a) Significant versus the control group (p-value less than 0.05), (b) Significant versus bisphenol-A group (p-value less than 0.05), (c) Significant versus bisphenol-A + Fluvastatin group (p-value less than 0.05), and (d) Significant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin mitigated the changes in the characteristics of the spermatozoa induced by BPA
Effect of fluvastatin with or without taxifolin on the sperm characteristics in bisphenol-A treated animals (mean ± SD).

aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).
Fluvastatin and/or taxifolin combatted the changes in the testicular morphometric characteristics and JTBS induced by BPA
Effect of fluvastatin with or without taxifolin on the testicular morphometric characteristics and Johnsen’s tubular biopsy score (JTBS) in bisphenol-A treated animals (mean ± SD).

aSignificant versus the control group (p-value less than 0.05).
bSignificant versus bisphenol-A group (p-value less than 0.05).
cSignificant versus bisphenol-A + Fluvastatin group (p-value less than 0.05).
dSignificant versus bisphenol-A + Taxifolin group (p-value less than 0.05).

Hematoxylin and eosin-stained specimens of the testicles (x200) of (a) Control group with normal structure of the testis with well-formed seminiferous tubules which contain layers of the spermatogenic cells and spermatozoa, (b) Bisphenol A (BPA) treated group showing loss of the spermatogenic cells from the seminiferous tubules with detachment of some of them in the lumen (Arrow head). The interstitial tissue shows coagulative necrosis (Thick arrow) with severe vascular congestion (Thin arrow), (c) BPA + methyl cellulose group showing dilated empty lumen of most seminiferous tubules with cellular detachment in the lumen (Arrow head), profuse vascular congestion (Thin arrow) and coagulative necrosis of the interstitium (Thick arrow), (d) BPA + Fluvastatin group exhibiting significant decrement of the coagulative necrosis (Thick arrow) and vascular congestion (Thin arrow) of the interstitium with most seminiferous tubules showing well organized spermatogenic cells (Arrow head), (e) BPA + taxifolin group with significant improvement in the architecture of the seminiferous tubules (Arrow head) with significant decrease in the vascular congestion (Thin arrow) and scanty areas of coagulative necrosis of the interstitial tissue (Thick arrow), and (f) BPA + fluvastatin + taxifolin group exhibiting apparently normal testicular architecture with well-formed seminiferous tubules (Arrow head) and interstitium (Thick arrow) with significant diminution of vascular congestion (Thin arrow).
Fluvastatin and/or taxifolin mitigated the increase in Ki-67 immunoexpression induced by BPA in the testicular tissues
Significant increase in Ki-67 immunoexpression was noticeable in BPA-treated group relative to the control group (Figures 5(a) and (b)). Addition of methyl cellulose didn’t significantly affect Ki-67 immunoexpression versus animals treated with BPA alone (Figure 5(c)). Administration of either fluvastatin or taxifolin to BPA-treated rats resulted in significant increment of Ki-67 immunostaining compared versus rats treated with BPA alone (Figures 5(d) and (e)) but fluvastatin/taxifolin combination group exhibited the maximal inhibition of Ki-67 immunoexpression (Figure 5(f)). Immunohistochemical staining for Ki-67 of testicular specimens from (a) The control group exhibiting absence of immunostaining of Ki-67, (b) BPA group showing markedly positive immunostaining of Ki-67 (+++), (c) BPA group treated with methyl cellulose with markedly positive immunostaining of Ki-67 (+++), (d) BPA group treated with fluvastatin exhibiting moderate positive immunostaining of Ki-67 (++), (e) BPA group treated with taxifolin revealing moderate positive immunoexpression of Ki-67 (++), and (f
Discussion
In the last decades, diphenylmethane derivatives attained an increasing importance for the industry of certain types of plastics and resins. 24 Among these derivatives, recent studies were directed towards BPA which is a synthetic organic substance which is the precursor of certain polycarbons and epoxy resins that are used for the manufacturing of the plastic containers of foods and drinks. 25 Interestingly, some recent trials aroused the attention towards the toxic effects of the long use of BPA on the testicular functions. 26 This hypothesis relied on the finding that BPA is a xenoestrogen that may exert estrogen-mimicking effects. 4 These effects together with the pro-oxidant, proinflammatory, and apoptosis promoting properties of BPA might induce deleterious effects on the testicular microenvironment which in turn may significantly affect the reproductive functions in males. 27 This was in accordance with the data obtained from the present study where BPA elicited significant decrease in the body weight gain, the testicular morphometric measurements and the markers of testicular functions. This was accompanied with significant decrease in serum testosterone, LH, FSH, HSD3B1, and HSD17B1 secretion and markedly affected the sperm characteristics and the histopathological picture when compared versus the control group.
The balance between the pro-oxidant and the antioxidant factors was reported to play a fundamental role in the testicular toxicity induced by BPA. 28 Coinciding with the results of the present work, Rahman and Pang. 29 stated that ROS generated by BPA in the testicular tissues have a detrimental effect on the spermatogenic functions of the testis, thus inducing reproductive dysfunction. BPA was found to repress the main enzymes responsible for the antioxidant defense mechanisms in the testicular tissues. 28 In addition, BPA was reported to enhance the generation of free radicals, which in turn will decrease Nrf2 content and attenuate the activity of HO-1 in the testicular tissues with the end result of deprivation of the testis from an important defense mechanism against oxidative stress. 30
In the present work, rats treated with fluvastatin/BPA combination exhibited significant improvement of the activity of the antioxidant enzymes with restoration of Nrf2/HO-1 content of the testicular tissues when compared versus rats treated with BPA alone. This was explained by Gurel et al. 10 who proved that fluvastatin has detrimental effects on the pro-oxidant/antioxidant pathways in the testicular tissues. Ramanjaneyulu et al. 31 reported a significant increase in the total antioxidant capacity together with significant decline in malondialdehyde levels in the testicular tissues with administration of statins. These effects may be explained by the well documented effects of fluvastatin on Nrf2/HO-1 pathway which subsequently affect the activity of the antioxidant enzymes at the gene expression levels. 32 Interestingly, taxifolin in the present study was able to restore the antioxidant enzyme levels and mitigate the decremental effect of BPA on Nrf2/HO-1 content of the testicular tissues. This was confirmed by the well-proven antioxidant effects of taxifolin which were attributed in a large proportion to its free radical scavenging and singlet oxygen quenching mechanisms. 33
Recent reports threw light on the role of NLRP3 inflammasome in the pathogenic events that take place in the testicular tissue as a consequence of BPA administration. 28 Wang et al. 34 stated that ROS generated by BPA enhance the binding of danger-associated proteins to toll-like receptor 4 (TLR4). It is evident that generation of the intracellular ROS activates nuclear factor kappa B (NF-κB), TGF-β1, and caspase three followed by alteration of the Bax/Bcl-2 ratio to cause systemic damage in rat model; such condition ultimately leads to inflammation and apoptosis.35,36 Enhancement of the activity of NF-κB/TGF-β1 signaling pathways consequently upregulates the transcription of NLRP3 inflammasome. 37 This in turn was suggested to promote the proteolytic cleavage of pro-IL-8 and pro-IL-18 resulting in propagation of the inflammatory process in the testicular tissues and induction of apoptosis. 38 This coincided with the data obtained from the present study where BPA induced significant increase in TLR4 and NLRP3 inflammasome expression with subsequent increase in the inflammatory mediators including TGF-β1, IL-8, and IL-18 relative to the corresponding levels in the control group. This was also evidenced by the changes of the histopathological picture of the testicular tissues which were subjected to massive inflammatory cellular infiltration.
An interesting finding in the present study was that fluvastatin administration elicited a mitigating effect on the expression of NLRP3 inflammasome which subsequently abrogated the inflammatory cascade and combatted the expression of the proapoptotic proteins. This was attributed by Wang et al. 39 to the inhibitory effects of statins on the production of ROS which consequently deprives the NF-κB/TGF-β1 signaling pathway from an important triggering factor. Also, fluvastatin was proven to activate peroxisome proliferator activated receptor gamma (PPAR-γ) which plays a fundamental role in regulation of NLRP3 expression and modulation of the inflammatory process. 40
The present data strongly established an important link between NLRP3 expression and the anti-inflammatory effects of taxifolin on the testicular tissues. Ponte et al. 41 stated that taxifolin may modulate the expression of NF-κB and decrease TLR4 levels, which in turn decrease the tissue levels of NLRP3 inflammasome. In addition, taxifolin was found to combat the binding of danger-associated proteins to TLR4 which subsequently deprives NLRP3 inflammasome from an important step in the pathway of its activation.42,43 Moreover, taxifolin was proven to suppress the activity of the genes responsible for TGF-β1 expression, thus decreasing production of the proinflammatory cytokines and abrogated the inflammatory changes in the testicular microenvironment. 44
The interplay between the different pathways related to autophagy and apoptosis might represent the cornerstone in the pathophysiologic events that were observed in the gonads in response to prolonged exposure to BPA. 45 In the present work, rats treated with BPA exhibited significant decline in tissue p-MAPK and beclin-l with significant increase in tissue p-mTOR and p-AKT levels relative to the control group. This was in agreement with Anand et al. 46 who stated that there is an inverse relationship between the levels of p-mTOR and p-AKT on one hand and the expression of p-MAPK and beclin-l on the other hand. BPA was proven to enhance the activity of mTOR/AKT signaling pathways which in turn suppress the activity of MAP kinase enzyme. 47 This together with the resultant decrease in beclin-1 levels leads to defective autophagy. 48 In addition, modulation of mTOR/AKT/MAPK signaling in response to BPA administration was reported to enhance the expression of the proapoptotic proteins and decrement the tissue levels of the antiapoptotic molecules, particularly Bcl2. 45 This balance between autophagy and apoptosis was restored in the current study with administration of fluvastatin and/or taxifolin with induction of autophagy and decreased expression of the proapoptotic molecules. Cui et al. 49 attributed these events to the inhibitory effects that were exerted by fluvastatin on mTOR/AKT signaling which subsequently initiate the events that enhance autophagy and protect the testicular tissues from the pathogenic consequences of apoptosis. Also, Ponte et al. 41 established a strong relation between taxifolin-mediated increase in beclin-1 and c-Jun N-terminal kinase and protection from the deleterious effects of apoptosis which coincided with the data obtained from the current work.
Ki-67 is one of the proliferation factors that are widely distributed in the body tissues, including the testis. 50 In the same line with the data obtained from the current study, it was reported that positive Ki-67 expression in the testis has a positive correlation with defective spermatogenesis in BPA-treated rats. 23 Also, positive Ki-67 expression was associated with extensive damage to the testicular architecture and induction of spermatogenic cell apoptotic pathways in rats. 51 Moreover, increased Ki-67 expression induced by BPA administration was incriminated to be a major contributing factor to NF-κB mediated inflammation and apoptosis. 52 These events were mitigated in the present study with administration of either fluvastatin or taxifolin, raising a high possibility that their protective effects against BPA-induced testicular dysfunction may be mediated, at least in part, via inhibition of Ki-67 expression with subsequent decrement of inflammation and apoptosis.53,54
Fluvastatin/taxifolin combination given to rats treated with BPA in the current work elicited significant improvement of the body weight gain, hormonal profile, testicular morphometric measurements, sperm characteristics, the antioxidant status, and the anti-inflammatory mechanisms together with enhancement of autophagy and suppression of apoptosis in the testicular tissues relative to BPA-groups treated with either Fluvastatin or taxifolin alone. This may be explained, as illustrated in Figure 6, by the complementary effects of both agents on the ROS production, mTOR/AKT/MAPK signaling pathways together with their ability to combat BPA-induced activation of NLRP3 inflammasome which has an outstanding role in suppression of the proapoptotic pathways and induction of autophagy. Summary of the mechanisms by which fluvastatin and/or taxifolin might mitigate bisphenol A-induced testicular dysfunction.
P-glycoprotein is an efflux protein located in different tissues of the body including the gastrointestinal tract, brain, kidney and the gonads. 55 P-glycoprotein was reported to mediate resistance of the cells to certain members of statins, namely pravastatin and fluvastatin. 56 The synergistic effect encountered in the current work between taxifolin and fluvastatin may be explained by the ability of taxifolin to inhibit P-glycoprotein, thus facilitating entry and promoting the binding of fluvastatin to its target sites. 57 This together with the recent findings that taxifolin has a regulatory function on PPAR-γ pathway may add efficient anti-inflammatory and anti-apoptotic properties to fluvastatin.58,59
Conclusion
Fluvastatin/taxifolin combination may provide a new remedy for combating the harmful effects of BPA on the gonadal functions in males. Attenuation of ROS production, modulation of autophagy, and apoptosis in addition to suppression of NLRP3 inflammasome are possible contributing mechanisms. Application of these findings in the clinical settings may significantly mitigate BPA-induced testicular toxicity in a large number of patients. Further studies are essentially needed to fully explore the molecular pathways that may contribute to these admirable effects and to evaluate the consequences of long-term administration of fluvastatin and/or taxifolin.
Footnotes
Acknowledgments
We greatly thank Taif University for providing fund for this work through Taif University Researcher Supporting Project number (TURSP-2020/52), Taif University, Taif, Saudi Arabia.
Authors contributions
Conceptualization: Ahmed M. Kabel; Methodology, Investigation, Formal analysis, Validation, Visualization: Ahmed M. Kabel, Samir A. Salama, Hany M. Borg, Dina A. Ali, Maaly A. Abd Elmaaboud; Funding Acquisition: Samir A. Salama; Writing- Original draft preparation: Ahmed M. Kabel; Writing- Reviewing and Editing: Ahmed M. Kabel.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taif University (TURSP-2020/52).
Data availability
Data used and/or analyzed during this study are available from the corresponding author upon reasonable request.
