Abstract
The purpose of the present study was to investigate the cardioprotective effects of Glycyrrhiza uralensis extract (GUE) against doxorubicin (DOX)-induced cardiotoxicity. Imprinting control region (ICR) mice were treated with saline, DOX (20 mg/kg intraperitoneal [ip] for once), GUE (100 mg/kg intragastric [ig] for 8 days), co-treatments with DOX and GUE (100 mg/kg ig for 8 days), and amifostine (100 mg/kg intravenous [iv] for once), respectively. Serum levels of lactate dehydrogenase (LDH) and creatine kinase isoenzyme (CK-MB), glutathione peroxidase (GSH-PX) activity, and glutathione (GSH) level in heart tissue were measured. Histopathologic analysis of heart tissue was also performed. Treatment with GUE significantly protected the mice from DOX-induced cardiotoxicity, indicated by decreased levels of serum LDH and CK-MB, improved heart morphology and increased GSH-PX activity and GSH level. Additionally, GUE did not compromise the tumor-inhibitory effect of DOX. In conclusion, our studies imply the potentially clinical application of GUE to overcome the cardiotoxicity of doxorubicin.
Keywords
Doxorubicin (DOX), an antitumor anthracycline antibiotic, is extremely effective in treating severe Hodgkin and non-Hodgkin lymphomas, breast cancer, and leukemia. 1 However, numerous studies have shown DOX-treatment-related free radical generation, membrane lipid peroxidation, and mitochondrial damage, 2,3 and its therapeutic usage is rigorously limited by late-onset, acute, or chronic cardiotoxicity.
Radix Glycyrrhizae is used in foods, beverages, candies, tobacco, and dietary supplements in the United States, Europe, the Middle East, and Russia. 4 It is considered to be the “King of Herbs” 5 and possesses various pharmaceutical anticarcinogenic, anti-inflammatory and antiulcer properties. 6 –8 Glycyrrhizin, the main water-soluble constituent of licorice, was used as antidote for saponin, alkaloid urethan, benzene, carbon tetrachloride-induced toxicity, 9 and exerted antigenotoxic and hepatoprotective effects. 10 Isoliquiritin apioside potentially combated oxidative stress-induced genotoxicity. 11 Glycyrrhizic acid was shown to protect against aflatoxin-induced oxidative stress with its capacity to inhibit the metabolic activation of hepatotoxin. 12 Lee et al showed that Glycyrrhizae inflata attenuates the toxicity of cisplatin without compromising its antitumor effects. 13 There are 3 species of plant material recorded as Radix Glycyrrhizae in Chinese Pharmacopeia. Among them, Glycyrrhiza uralensis is most commonly used in China. Very recently, Choi reported that hexane/ethanol extract of Glycyrrhiza uralensis suppresses DOX-induced apoptosis in H9c2 rat cardiac myoblasts. 14 However, few studies have been undertaken to evaluate the protective effect of Glycyrrhiza uralensis extracts (GUE) against DOX-induced cardiotoxicity as yet. The present study was designed to estimate the cardioprotection of GUE against DOX-induced toxicity. Meanwhile, antioxidant activity and the potential impact on antitumor activity of DOX were also studied. Amifostine, a cytoprotective agent, was also used as a positive control in our study.
Methods
Chemicals and Cell Line
Doxorubicin was provided by Hisun Pharmaceutical Co (Taizhou, China). Amifostine (AMF) was purchased from Dalian Merro Pharmaceutical Factory (Dalian, China). 2, 2-dipheny-1-picryl-hydrazylhydrate (DPPH) was purchased from Sigma–Aldrich Co (St. Louis, Missouri). Luminol was obtained from the Institute of Analytical Science of Xi’an Jiaotong University (Xi’an, China). The diagnostic kits for LDH, CK-MB were obtained from Asia-Pacific (Ningbo, China). The diagnostic kit for superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-PX) were obtained from Jiancheng (Nanjing, China). Murine hepatoma 22 cells (H22 cells) were purchased from Zhejiang Academy of Medical Sciences (Hangzhou, China). Analytical reagent-grade chemicals and double-distilled water were used to prepare all solutions.
Plant Material and Preparation of GUE
The root of Glycyrrhiza uralensis was purchased from Hangzhou Traditional Chinese Medicine Electuary Factory (080906, Hangzhou, China) and authorized by associate Prof Liurong Chen (College of Pharmaceutical Sciences, Zhejiang University, Hangzhou, China). A voucher specimen was deposited at the herbarium of Pharmaceutical Information Institute, College of Pharmaceutical Sciences, Zhejiang University, Hangzhou, China. Dried roots of Glycyrrhiza uralensis was extracted with 70% ethanol (10 mL × 2) by refluxing for 1.5 hours. The solution was filtered and then evaporated to dryness under vacuum. The residue was subjected to D101 porous resin column (Tianjin Resin Factory) and eluted with water and 70% ethanol. The eluant by 70% ethanol was evaporated to dry, and the residue was obtained as GUE. The extraction efficiency was 10.8%. Using licorice glycoside as the reference compound, the content of total flavonoids in GUE was 25.8% determined by Ultra Violet (UV) spectrophotometric method. Using glycyrrhizic acid as standard compound, the content of glycyrrhizic acid was 15.1% by High-Performance Liquid Chromatography (HPLC) analysis.
Liquid Chromatography/UltraViolet (LC/UV) chromatograms of GUE at 230 nm and 254 nm are shown in Figures 1 and 2 , respectively. Analysis was carried out using a Agilent 1100 series HPLC system (Waldbronn, Germany) equipped with a quaternary pump with on-line degasser, autosampler, column oven, and diode array detector (DAD). The chromatographic conditions were as follows: Zorbax StableBond C18 column (4.6 mm × 250 mm, 5 μm, Agilent); sample injection volume: 10 μL; the temperature of column oven: 35°C; flow rate: 1 mL/min; mobile phases: water with 0.01% acetic acid (A) and methanol (B). A gradient programmer was used according to the following profile: 0-15 minutes, hold on 20% B; 15-25 minutes linear increase to 25% B; 25-35 minutes hold on 25% B; 35-45 minutes linear increase to 40% B; 45-55 minutes hold on 40% B; 55-60 minutes increase to 90% B. UV spectra wavelengths were set at 230 nm and 254 nm. The identification of major compounds in GUE are listed in Table 1 . Chromatogram of GUE and structures of major compounds are shown in Figures 1 and 2.
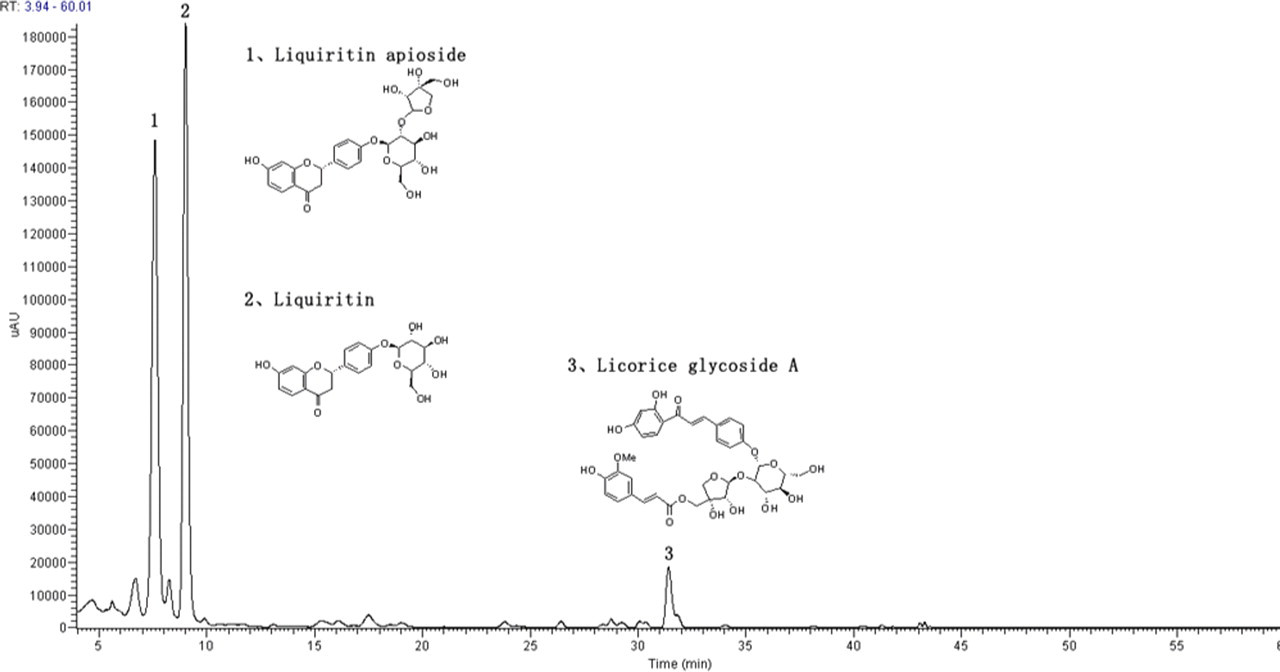
Chromatogram of Glycyrrhiza uralensis extract (GUE) by HPLC/DAD/MS analysis at 254 nm. The structures of 3 representative compounds were shown. Peaks 1, Liquiritin apioside; 2, Liquiritin; 3, Licorice glycoside A.
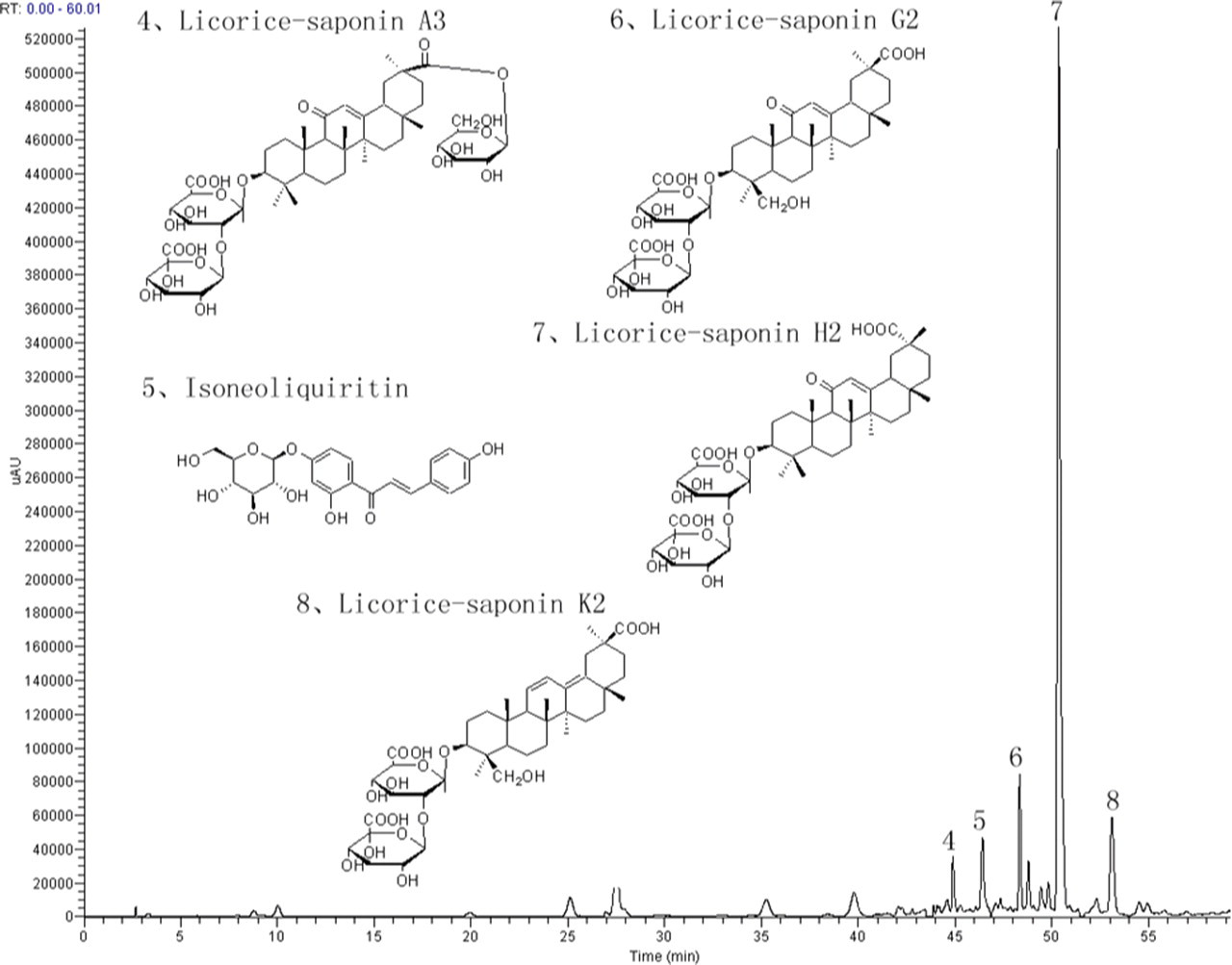
Chromatogram of Glycyrrhiza uralensis extract (GUE) by HPLC/DAD/MS analysis at 230 nm. The structures of 3 representative compounds were shown. Peaks 4, Licorice-saponin A3; 5, Isoneoliquiritin; 6, Licorice-saponin G2; 7, Licorice-saponin H2; 8, Licorice-saponin K2.
Contents of the Major Compounds in Glycyrrhiza uralensis Extraction
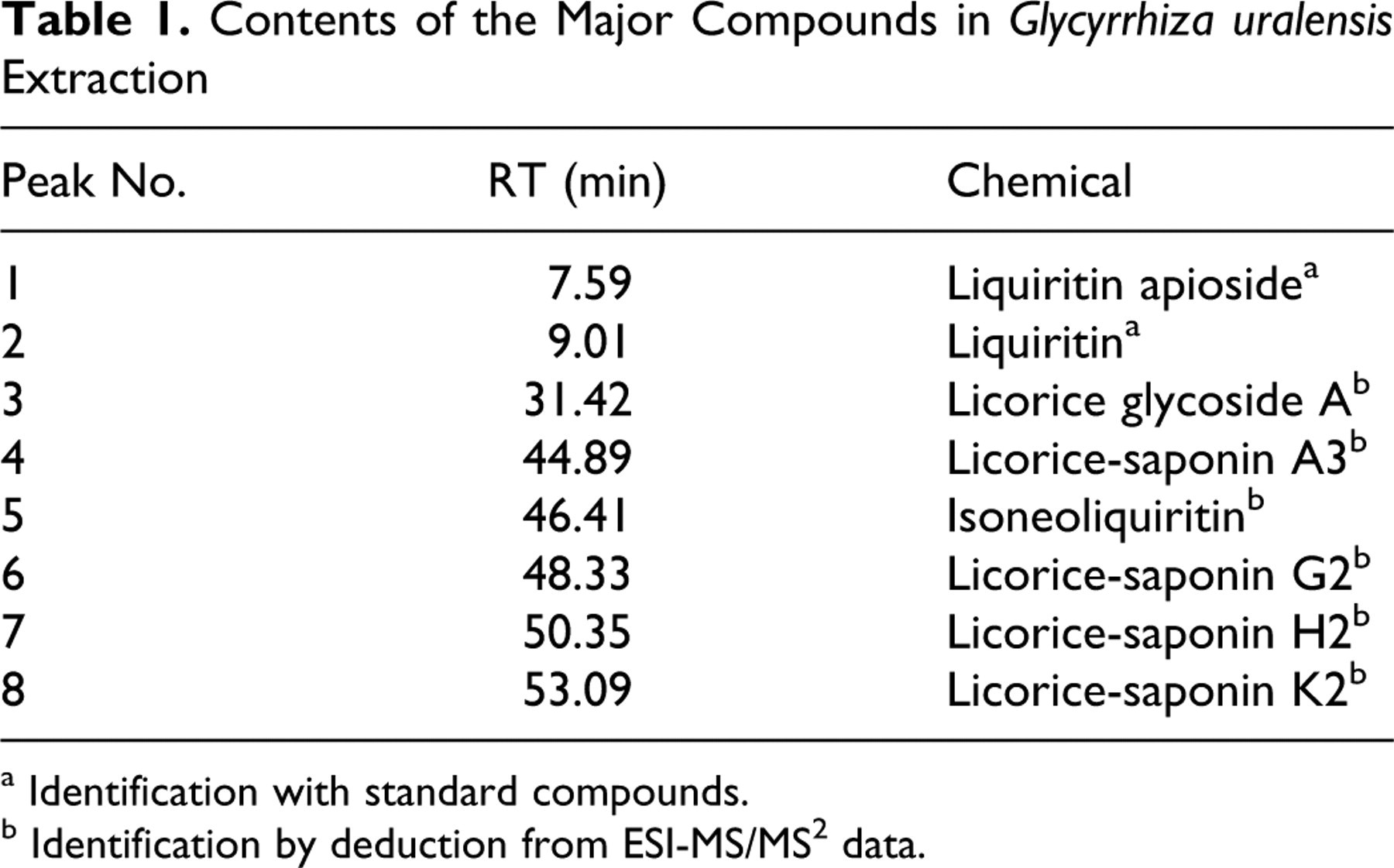
a Identification with standard compounds.
b Identification by deduction from ESI-MS/MS2 data.
Animal and Experimental Protocol
Male ICR mice (20-22 g) were purchased from Zhejiang Experimental Animal Center. The mice were fed with a standard laboratory diet and given free access to tap water, kept in a controlled room temperature (22 ± 1°C), humidity (65%-70%), and 12 hours dark/light cycles. The investigation conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). The Animal Ethic Review Committees of Zhejiang University approved all procedures.
Mice were divided into the following 5 groups, each group consisted of 10 mice: Control group (CON), DOX-treated group (DOX), DOX-treated group combined with amifostine (DOX + AMF), Glycyrrhiza uralensis extract-treated group (GUE), and DOX-treated group combined with GUE (DOX + GUE). The treatment protocol was as in Table 2 .
Treatment Protocol
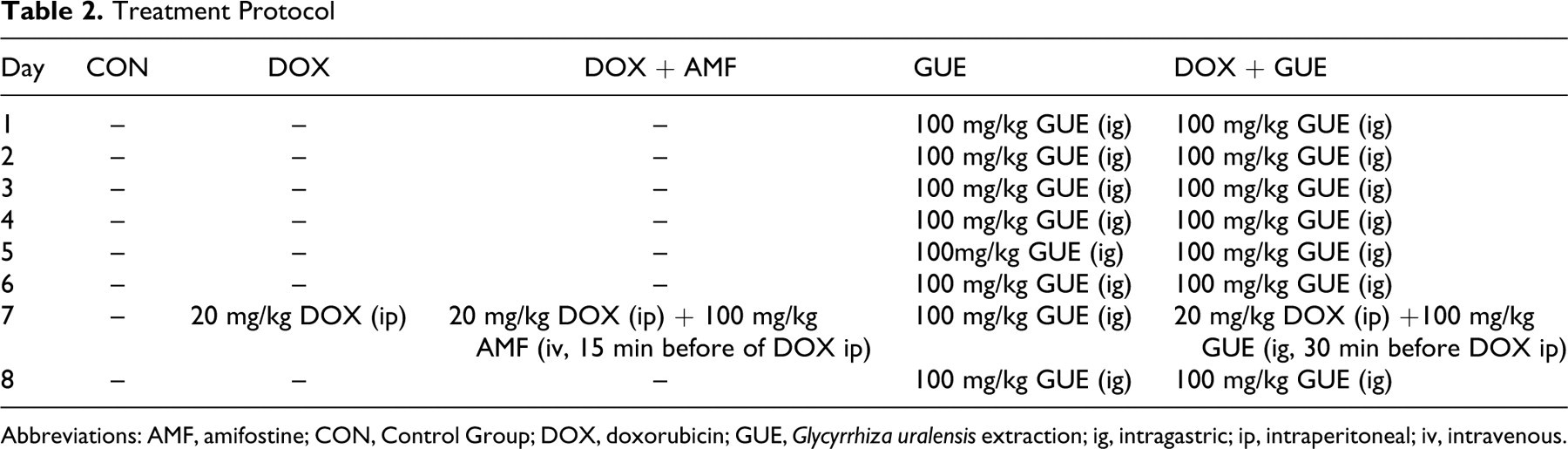
Abbreviations: AMF, amifostine; CON, Control Group; DOX, doxorubicin; GUE, Glycyrrhiza uralensis extraction; ig, intragastric; ip, intraperitoneal; iv, intravenous.
Determination of Serum Enzymes
After 8 days of GUE treatment, blood was collected from the inferior vena cava. The serum was separated by centrifugation at 3000 rpm for 15 minutes and serum concentrations of LDH and CK-MB were determined by the use of diagnostic kit according to the manufacturer’s instructions.
Antioxidant Activity of GUE In Vivo
Ten hearts of each group were weighed and homogenized in 10 mmol/L Tris-HCl of ice-cold phosphate-buffered saline (PBS; 50 mg/mL; pH 7.4) and centrifuged for 5 minutes at 5000 rpm. The protein content of the supernatants was determined by the Bradford protein assay. The activities of CAT and SOD were assayed by ammonium molybdate and xanthine-xanthine oxidase spectrophotometric methods. Glutathione peroxidase was assayed by enzymatic reaction and GSH was estimated at 412 nm colorimetrically by (5, 5'-dithiobis [2-nitrobenzoic acid]) (DTNB) reduction method. The GSH level and antioxidant enzymes were measured by the use of diagnostic kits according to the manufacturer’s instructions. Data were normalized to the mean control value.
Histopathologic Evaluation
Another 4 hearts of each group were rapidly excised fixed in 10% formalin and embedded in paraffin. A slice containing the left and right ventricules was cut at a thickness of 5 μm and stained with hematoxylin and eosin for morphometric analysis. The stained sections were examined at ×400 magnification to analyze the severity of myocardial lesions.
DPPH• Scavenging Activity
DPPH• scavenging activity of GUE was measured by the luminol chemiluminescence (CL) system according to the method of Yao et al.
15
Briefly, solutions of GUE and vitamin C with a series of concentrations (1, 10, 50, and 100 μg mL–1) were measured by an Infinite F200 microplate reader instrument (Tecan Group Ltd, Switzerland). The water and DPPH• solution were used as control and water was used as background. The test of DPPH• scavenging activity for all sample solutions, the controls, and the background were repeated 3 times. The formula of inhibition percentage of DPPH•–luminol CL by the sample solution was
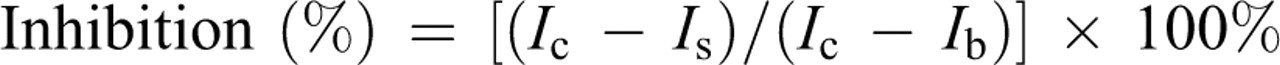
O2 − • Scavenging Activity
O2
−
• scavenging activity of GUE was assayed by SOD diagnostic kit. Briefly, solutions of GUE with a series of concentrations (20, 100, 200, 400, and 1000 μg mL–1) and vit C with a series of concentrations (4, 20, 100, 200, and 400 μg mL–1) were measured at 550 nm. The reaction system without xanthine oxidase and sample was used as 100% clearance (A100), the reaction system only without sample was used as 0% clearance (A0). The test of O2
−
• scavenging activity for all sample solutions was repeated 3 times. The formula of clearance rate of O2
−
• by the sample solution was
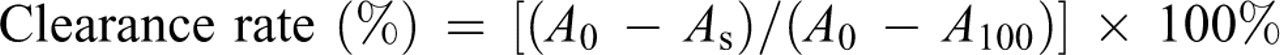
The Effect on Antitumor Activity of DOX In Vivo
An independent experiment was conducted to evaluate the potential effect of GUE on antitumor activity of DOX. H22 cells were propagated by serial intraperitoneal transplantation in an aseptic environment, then, 8 × 105 H22 ascites tumor cells were inoculated subcutaneously (sc) on the left side of the axilla of mice, according to the methods of Chen et al.
16
Glycyrrhiza uralensis extract treatment was started when the tumor size was approximately 100 mm3. The first treatment day was designated as day 1. Treatment and control groups consisted of 10 mice each. Treatments were as follows: control group, no treatment; DOX group, 7.5 mg/kg of DOX intraperitoneal (ip) on days 4 and 5; DOX + AMF group, 7.5 mg/kg of DOX ip on days 4 and 5, 100 mg/kg amifostine iv on days 4 and 5 (15 minutes before DOX injection); DOX + GUE group 7.5 mg/kg of DOX ip on days 4 and 5, 100 mg/kg GUE intragastric (ig) once daily consecutively for 8 days (30 minutes before DOX injection); GUE group, 100 mg/kg GUE ig once daily consecutively for 8 days. After 8 days of GUE treatment, the tumors were weighed. The antitumor activity was assessed on the basis of tumor weight and the tumor inhibition, the tumor inhibition was calculated as:
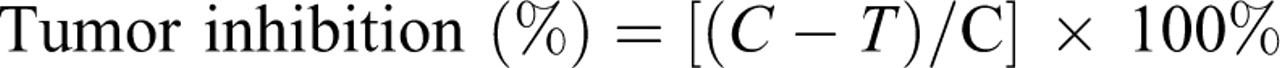
Data Analysis
All data were expressed as means ± SD and n, the number of individual experiments. One-way analysis of variance (ANOVA) was used for statistical analysis. A value of P < .05 was considered significant. All statistical analyses were performed using MINITAB 14 (Mintab Inc, State College, Pennsylvania).
Results
Effect of GUE on Serum LDH and CK-MB Level
Creatine kinase isoenzyme and LDH were measured in the serum as indicators of cardiovascular damage. Figure 3 shows the effect of GUE on DOX-induced biochemical changes in the serum of mice. Compared with control group, administration of DOX significantly elevated serum level of LDH and CK-MB (P < .01, respectively), which indicated DOX-induced cardiotoxicity. Injection of AMF reduced the level of LDH and CK-MB, but there were no statistically significant differences. Oral treatment with GUE for 8 days significantly prevented the increase of LDH and CK-MB caused by DOX (P < .05, respectively; Figure 3).
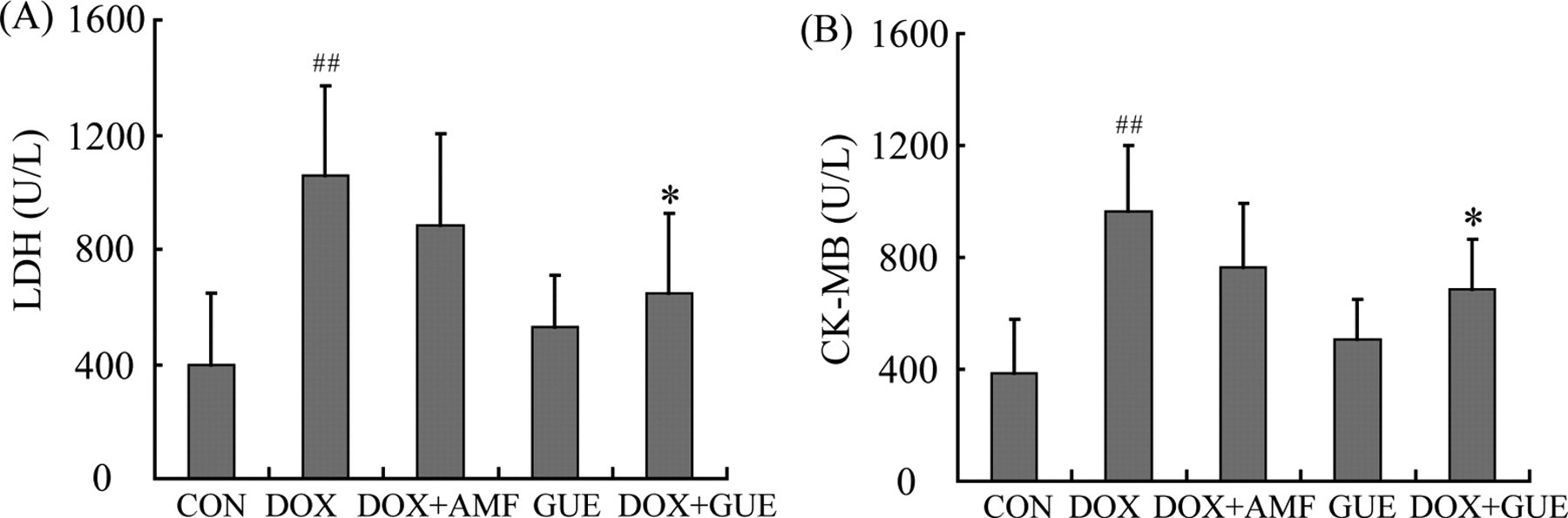
Serum enzyme level in experimental groups. (A) lactate dehydrogenase (LDH) level; (B) creatine kinase isoenzyme (CK-MB) level. Glycyrrhiza uralensis extract (GUE) alone group had no statistical difference in the serum level of LDH and CK-MB compared with control group. Compared with doxorubicin (DOX) group, DOX + GUE group significantly decreased the activity of LDH and CK-MB, while DOX + amifostine (AMF) group had no statistically significant differences. Values are means ± SD. n ≥ 7. ## P < .01 compared to control group; *P < .05 compared to DOX group.
Antioxidant Activity of GUE In Vivo
We also investigated the effect of GUE on the activity of antioxidant enzymes in heart tissue in vivo. With the exception of the increased level of SOD in GUE + DOX group, there was no significant change in the activities of CAT and SOD among these groups. In contrast to CAT and SOD, the level of GSH and the activity of GSH-PX were notably reduced in DOX group compared with control group (P < .05, respectively; Figure 4 ). Compared with DOX group, the activity of GSH-PX and the level of GSH were markedly restored in the AMF-treated group (P < .01 and P < .05; Figure 4). Glycyrrhiza uralensis extract significantly increased the activities of GSH-Px and the level of GSH in DOX-treated mice (P < .01 and P < .05; Figure 4).
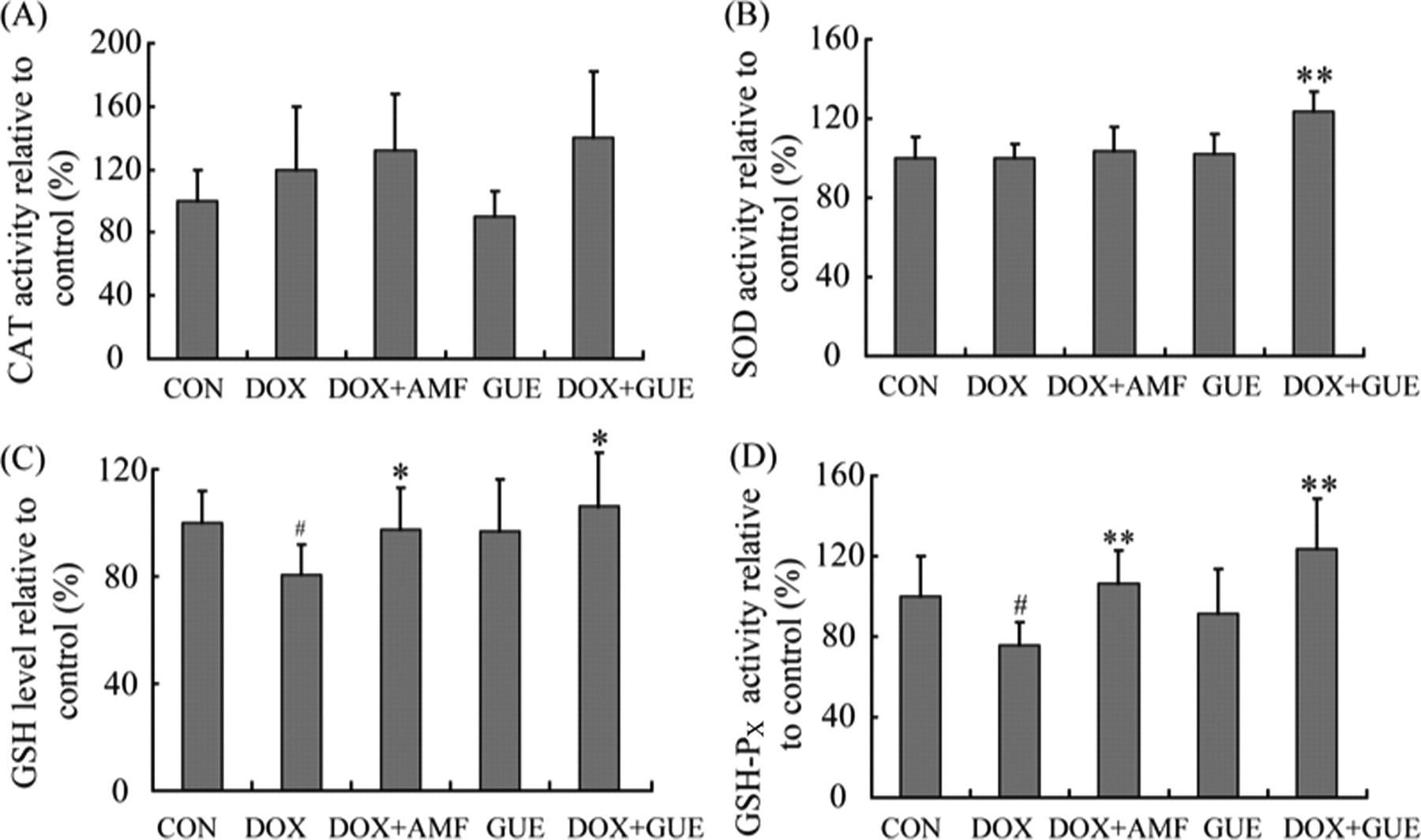
Antioxidant activity of Glycyrrhiza uralensis extract (GUE) in vivo. (A) catalase (CAT) activity; (B) superoxide dismutase (SOD) activity; (C) glutathione (GSH) level. Glycyrrhiza uralensis extract significantly increased the level of GSH in doxorubicin (DOX)-treated mice; (D) glutathione peroxidase (GSH-Px) activity. The activity of GSH-PX was markedly increased in GUE-treated group compared with DOX group. Data are normalized to the mean control value. Values are means ± SD. n ≥ 7. # P < .05 compared to control group; **P < .01 compared to DOX group; *P < .05 compared to DOX group.
Effect of GUE on Histopathological Change
Microscope analysis of the heart tissues was performed in Control, DOX, DOX + AMF, GUE, and DOX + GUE groups. Normal myocardium morphology was observed in control and GUE groups (Figures 5A and 5E ), whereas cytoplasmic vacuolation in DOX-treated animals was very prominent (Figure 5B). After GUE and AMF treatment in DOX-administrated mice, a significant reduction in cytoplasmic vacuolation was observed (Figure 5C and 5D), indicating a protective effect of GUE on DOX-induced cardiotoxicity.
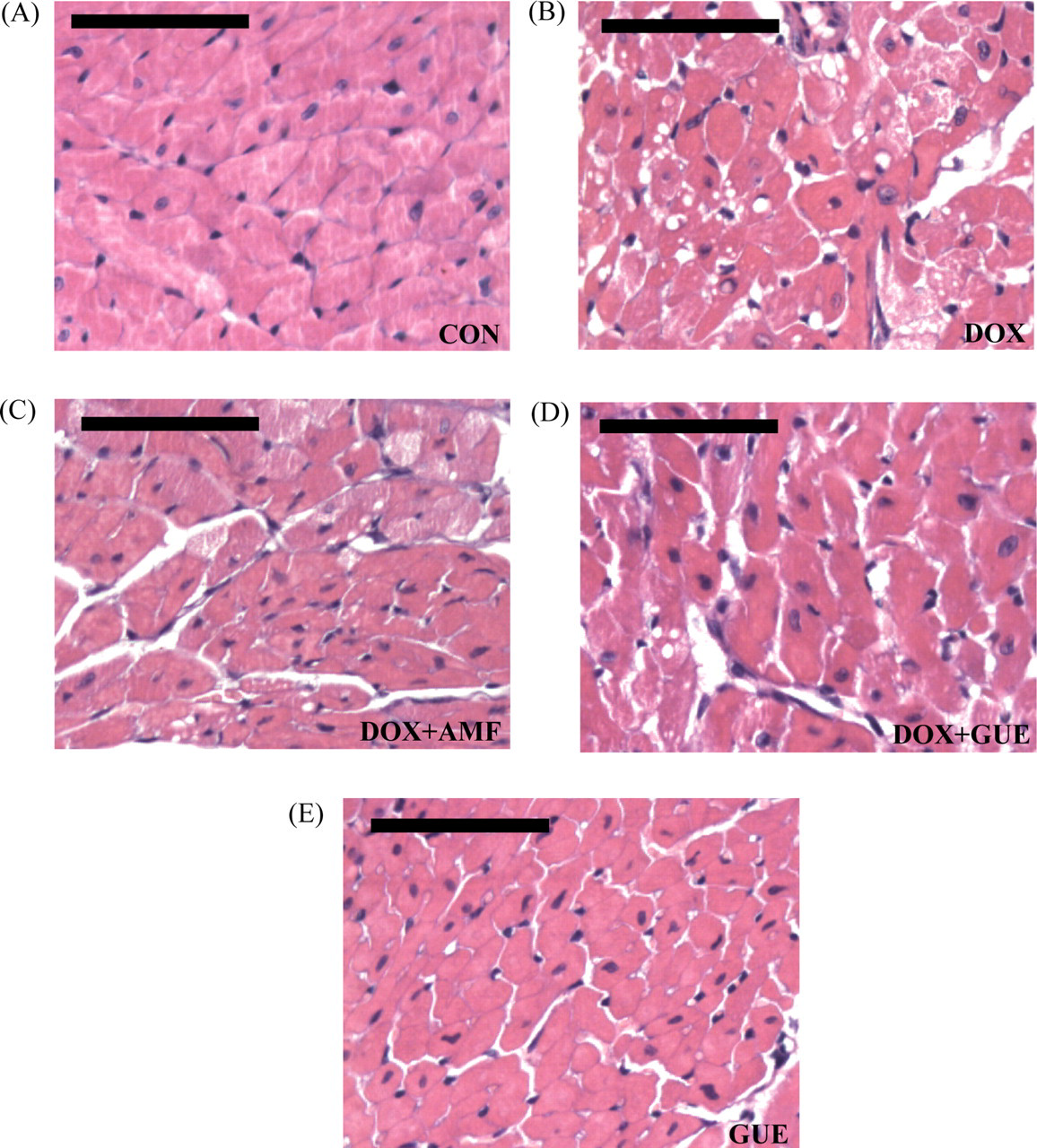
Histopathological examination. Hematoxylin and Eosin (H&E) staining for (A) Control, (B) doxorubicin (DOX), (C) DOX + amifostine (AMF), (D) DOX + GUE, and (E) Glycyrrhiza uralensis extract (GUE) group. Normal myocardium morphology was shown in control group, a significant reduction in cytoplasmic vacuolation formation was observed in GUE and AMF treatment group compared with DOX group. Original magnification ×400. Scale bar = 100 μm.
Measurements of DPPH• and O2−• scavenging activity
We investigated the radical (DPPH•) scavenging activities of GUE (a series of concentrations) by DPPH• –luminol CL inhibition rate (%), and the result was compared with a standard antioxidant, vitamin C. Vitamin C had stronger radical (DPPH•) scavenging activities than GUE (Figure 6A ). The IC50 of GUE and vitamin C were 3.71 and 0.56 μg/mL respectively.
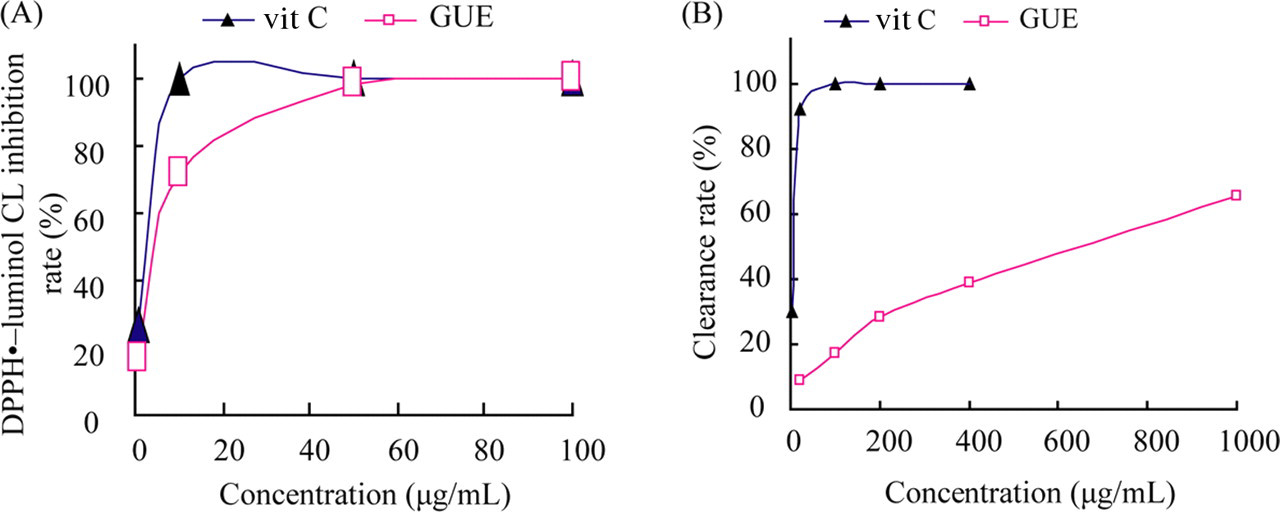
Effect of Glycyrrhiza uralensis extract (GUE) on DPPH• and O2 − • scavenging activity. (A) DPPH• scavenging activity of GUE and vit C with a series concentrations; (B) O2 − • scavenging activity of GUE and vit C with a series concentrations. The radical (DPPH•) scavenging activities and clearance rate of O2 − • induced by GUE was less than vit C.
The results for O2 − • scavenging activity showed that vitamin C as a standard antioxidant had much higher clearance rate of O2 − • than GUE (Figure 6B). The IC50 of vitamin C is 5.07 μg/mL, and IC50 of GUE is 599.80 μg/mL.
Effect of GUE on antitumor activity of DOX
No spontaneous regression was observed in the mice injected with H22 cells. Compared with the control, treatment of DOX significantly inhibited the tumor weight (P < .01), indicating an arrest of tumor growth (Figure 7 ). The tumor weight in DOX + GUE and DOX + AMF groups also decreased compared with control group (P < .01 respectively) (Figure 7). The tumor inhibitions of DOX, DOX + AMF and DOX + GUE group were 47.22%, 38.50%, and 48.39% respectively. The result showed that GUE did not compromise the antineoplastic activity of DOX.
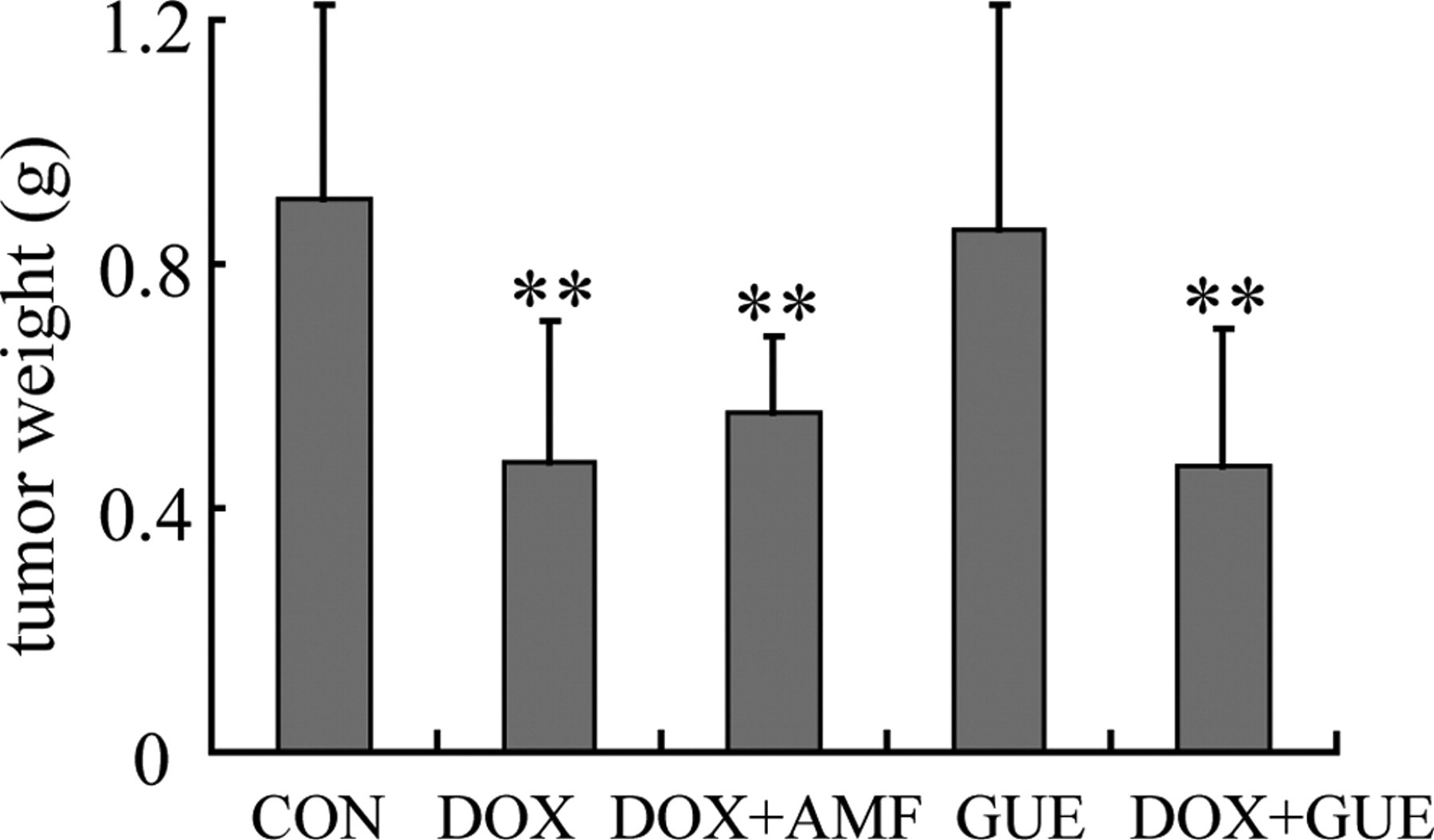
Effect of Glycyrrhiza uralensis extract (GUE) on tumor weight. GUE did not compromise the antineoplastic activity of doxorubicin (DOX). Values are means ± SD. n ≥ 8. **P < .01 compared to control group.
Discussion
In this study, GUE alleviated DOX-induced cardiotoxicity in vivo by deceasing of serum LDH and CK-MB levels, both of which were indicators of cardiac myocyte injury, and preventing morphological changes in heart structure. Though GUE did not exert the stronger DPPH• and O2 − • scavenging activities compared to vitamin C, it recovered the activity of GSH-PX and the level of GSH reduced by DOX. The result suggested that the protective effect of GUE on DOX-induce cardiotoxicity might be caused by increasing the activity of GSH-PX and the level of GSH in vivo, which are more likely effects of less oxidative stress. Furthermore, our study indicated that GUE did not interfere with the antineoplastic effect of DOX on H22 tumors in mice.
The mechanism of cardiotoxicity induced by anthracyclines is not clearly known. Some investigations suggest that apoptosis plays a crucial role in the pathogenesis of DOX-induced cardiac damage. 17,18 The sensitization of Ca2+ release from sarcoplasmic reticulum channels 19 and alterations in the myocardial energy metabolism 20 are also involved in DOX-induced cardiomyopathy. Among these hypotheses, increased oxidative stress associated with an impaired antioxidant defense status is most widely accepted. 21,22 In cells or tissue, oxidative stress results in the enhanced generation of reactive oxygen species (ROS) and a depletion of the antioxidants in the defense system. Reactive oxygen species generated in the tissue can be efficiently scavenged by the enzymatic antioxidant system and nonenzymatic antioxidants. 23 Heart is particularly susceptible to free radical injury, due to lower levels of free radical detoxifying substances than are found in organs such as liver or kidney. 24
The glutathione redox cycle is one of the most important intracellular antioxidant systems. GSH-Px and GSH are the essential compounds for maintaining cell integrity of structure and function, and the elevation of GSH-Px activity and GSH synthesis has been shown to be associated with oxidative stress. 25 Selenium (Se) possesses important biochemical functions in GSH-Px. 26 GSH is the most abundant cellular thiol and an important protective antioxidant against oxidative stress that plays a role in numerous detoxifications, bioreduction, and conjugation reactions. 27,28 It is a substrate and cofactor of GSH-Px in degrading hydrogen peroxide (H2O2). Low level of GSH may cause increased oxidative stress. 29 H2O2 is known to be cytotoxic and causes damage to lipids. GSH-Px catalyzes the H2O2 to water, thus GSH-Px can inhibit tissue injury by removing H2O2. 30 In addition, GSH-Px can act on hydroperoxides of unsaturated fatty acids, thus it plays an important role in protecting cell membranes from oxidative disintegration. 26 Our study also showed that GUE protected the heart from DOX-induced injury by increasing the activity of GSH-PX and the level of GSH.
Some free radical scavengers listed by Wouters et al 31 have been shown to protect the heart against DOX-induced cardiotoxicity. In the heart, iron ions can react with H2O2 to form reactive hydroxyl radicals and play a crucial role in the generation of oxygen free radicals. In the clinic, iron chelators such as dexrazoxane, also known as ICRF-187, which is the only cardioprotective drug currently available, 32 is limited in clinical use because of hematological toxicity and renal toxicity. 33 Amifostine, a broad-spectrum cytoprotective agent, has been extensively used clinically against chemotherapy-related toxicity. The experiments showed that amifostine can reduce DOX-associated cardiotoxicity by scavenging oxygen radicals in cultured neonatal myocytes and in mice. 34,35
Radix Glycyrrhizae has been widely used in food and medicinal remedies for thousands of years. Recently, glycyrrhetinic acid was found to induce actin disruption and had tumor cell-selective toxic properties, thus, it could be utilized as a promising chemopreventive and therapeutic antitumor agent. 36,37 Glycyrrhizin prevented human endometrial cancer by significant decrease in the COX-2, IL-1α, and TNF-α mRNA expression. 38 The antiangiogenic activity of licochalcone A, a component of Radix Glycyrrhizae, supports it as a potential supplemental source for cancer therapy. 39 These advantages suggest that the addition of Radix Glycyrrhizae and its active compounds may have favorable protective effects during DOX chemotherapy.
The present findings demonstrate that GUE protects against DOX-induced cardiotoxicity without compromising the antitumor activity of DOX. Therefore, GUE may be considered as a potentially useful candidate in the combination with DOX chemotherapy.
Footnotes
Acknowledgments
We thank Associate Prof. Youfa Zhu (Department of Pathology, Zhejiang University School of Medicine) for technical helps.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by National S&T Major Project (No. 2009ZX09502-012 & 2009ZX09311-002) and the Zhejiang Provincial Natural Science Foundation of China (No. Y2080741).
