Abstract
The present study was premeditated to examine the radioprotective effects of aqueous Aloe vera gel extract against whole-body X-ray irradiation–induced hematological alterations and splenic tissue injury in mice. Healthy male balb/c mice were divided into four groups: group 1, control; group 2, A. vera (50 mg/kg body weight) administered per oral on alternate days for 30 days (15 times); group 3, X-ray exposure of 2 Gy (0.25 Gy twice a day for four consecutive days in the last week of the experimental protocol); and group 4, A. vera + X-ray. X-ray exposure caused alterations in histoarchitecture of spleen along with enhanced clastogenic damage as assessed by micronucleus formation and apoptotic index. Irradiation caused an elevation in proinflammatory cytokines like tumor necrosis factor and interleukin-6, total leucocyte counts, neutrophil counts and decreased platelet counts along with unaltered red blood cell counts and hemoglobin. Irradiation also caused an elevation in reactive oxygen species (ROS), lipid peroxidation (LPO) levels, lactate dehydrogenase activity and alterations in enzymatic and nonenzymatic antioxidant defense mechanism in plasma and spleen. However, administration of A. vera gel extract ameliorated X-ray irradiation–induced elevation in ROS/LPO levels, histopathological and clastogenic damage. It also modulated biochemical indices, inflammatory markers, and hematological parameters. These results collectively indicated that the A. vera gel extract offers protection against whole-body X-ray exposure by virtue of its antioxidant, anti-inflammatory and anti-apoptotic potential.
Introduction
Although ionizing radiation has tremendous therapeutic benefits for humans, it is also associated with the risk of serious adverse effects. 1 Ionizing radiation inflicts damage to biological systems through direct deposition of energy into macromolecules or by radiolysis of water, resulting in excessive generation of reactive oxygen species (ROS) like superoxide radicals (O2·−), hydroperoxyl radicals (HO2·), hydroxyl radicals (OH·), and so on. 2 These ROS in turn react with biomolecules and cause oxidative damage to them. There is a wide range of response to ionizing radiation exposure, depending on absorbed dose, duration of exposure, interval after exposure, and susceptibility of tissues. 3
After whole-body radiation exposure, manifestation of injury is clearly reflected in peripheral blood. 4 Numerous studies on radiation exposure have yielded information on its effects on the hematological system. 5,6 The damaging effects of radiation on stem cells of the hematopoietic system can cause bone marrow suppression, leukemia, anemia, and so on, as observed in the victims of Hiroshima and Nagasaki, Japan. 7,8 Ionizing radiation can cause physiological responses such as nausea, vomiting, loss of appetite, decreased leucocyte count, and weakened immune function. 9 Alterations in blood cell counts are considered as a diagnostic marker for exposure to internal and external irradiation. 10 The hematopoietic system is among the most radiosensitive tissue in the body as it has the highest cell turnover. 11,12 Ionizing radiation has sufficient amount of energy to induce acute radiation syndrome, which includes anorexia, nausea, vomiting, and so on, within minutes of exposure at the dose of >0.7 Gy. 13
The spleen is the largest peripheral immune organ and it plays an important role in regulating immune responses. 14 The macrophages in the spleen respond to a variety of stress stimuli with the excessive production of ROS. 15 Due to leukocyte activity, the spleen has a high metabolic rate and thus is vulnerable to oxidative stress and damage. 16,17 It has been demonstrated that the interaction of ionizing radiation with the biological system leads to increased intracellular ROS resulting in mutations and chromosomal aberrations in bone marrow and spleen of mice. 18 Histomorphological alterations have also been reported in the spleen of rats exposed to gamma radiation. 19 Some reports reveal a decline in splenic lymphocytes after X-ray exposure in mice. 20 Epidemiological studies have demonstrated that ionizing radiation could induce a dose-dependent impairment of the immune response as well as a persistent inflammatory status along with deregulation of cytokine production. 21,22 Dainiak et al. 23 reported that ionizing radiation could result in a massive destruction of blood cells, leading to suppression of immune function and increasing the risk of infection while impairing wound healing. Besides immunosuppression, ionizing radiation can also induce the release of proinflammatory cytokines like tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) leading to inflammatory disorders. 24
The search for safe and effective radioprotectors has been ongoing for several decades. 25,26 Recently, much attention has been focused on plant extracts as radioprotectors, which are capable of modifying immune and radiation responses with minimum or no side effects. 24 Aloe vera (syn. Aloe barbadensis miller, family Liliaceae) is a cactus-like perennial plant, which has been used medicinally for centuries. 27 It is rich in various constituents including vitamins, minerals, amino acids, enzymes, polysaccharides, fatty acids, lignin, saponins, salicylic acid, and so on, used for a wide range of remedies. 28 Various studies have revealed that A. vera possesses antioxidant, immunomodulatory, and radioprotective properties. 29 The hematoprotective role of A. vera was observed after gamma-ray irradiation and cadmium-treated animals. 25 The modulatory effect of A. vera has been documented against radiation-induced changes in differential leucocyte counts in mice. 8 Previous studies, including ours, have demonstrated the radioprotective potential of A. vera against radiation-induced damage in hepatic, renal, and testicular tissues of mice. 29 –31 The hematopoietic system is extremely sensitive to radiation and it readily manifests changes upon radiation exposure. Thus, investigating the modulatory effects of A. vera on radiation-induced changes in the hematopoietic system could yield more information about its effectiveness as a radioprotector. Therefore, the present study was carried out to explore the potential of aqueous A. vera gel extract in mitigating damage induced to the hematological system upon radiation exposure.
Materials and methods
Chemicals
Glutathione oxidized (GSSG), glutathione reduced (GSH), nicotinamide adenine dinucleotide phosphate reduced (NADPH), thiobarbituric acid (TBA), 5′-5′-dithiobis-2-nitrobenzoic acid (DTNB or Ellman’s reagent), 1-chloro-2,4-dinitrobenzene, bovine serum albumin (BSA), and nitroblue tetrazolium (NBT) were purchased from reputed Indian manufacturers (Sisco Research Laboratory Pvt. Ltd., Central Drug House Pvt. Ltd., Himedia Pvt. Ltd., India). Dichloro-dihydrofluorescein diacetate (DCFH-DA) and ethidium bromide were obtained from Sigma Chemicals Co. (St. Louis, Missouri, USA). TNF-α and IL-6 kits were obtained from Ray Biotech, USA. Other chemicals used were procured from Indian firms and were of the highest purity and analytical grade.
A. vera gel extract preparation
Aqueous A. vera gel extract was prepared in our laboratory according to the method described previously. Carbohydrates, tannins, and anthraquinones were found to be present in the extract as revealed by the phytochemical analysis in our previous study. 32
Whole-body X-ray irradiation
X-ray whole-body irradiation was carried out using an X-ray machine (Philips, Model no. 9890 000 86 101) at the Department of Radiodiagnosis and Imaging, PGIMER, Chandigarh (India). For irradiation, animals were restrained in a well-ventilated perspex box with dose area product of 776.57 dGycm2 (deci Gray centimeter square) at 90 kV, 500 mAs and source to surface distance was 49 cm, and these parameters were kept constant throughout the experiment. These animals were exposed to a cumulative dose of 2 Gy (0.258 Gy twice a day for 4 days in the last week) and the dose of X-ray was standardized in the Department of Radiodiagnosis and Imaging, PGIMER, Chandigarh. 30,31
Animal treatment and study design
Male balb/c mice (25–30 g) were selected from an inbred colony of central animal house, Panjab University (Chandigarh) maintained under the controlled conditions of temperature (23 ± 2°C), humidity (50 ± 5%), and 12 h of light and dark cycle, respectively. The animals were provided with a standard pellet diet as recommended by the Ashirwad Industries Ltd., Ropar, Punjab (India) and water ad libitum. All the experiments were conducted strictly adhering to the Indian National Science Academy guidelines. The experimental protocols were approved by the Institutional Ethics Committee of Panjab University, Chandigarh (PU/IAEC/S/14/23). The animals were kept in polypropylene cages containing clean rice husk. All the treated animals were continuously observed for any sign of radiation sickness, behavioral toxicity, body weight change, mortality, hair ruffling, and so on. Male balb/c mice were randomly assorted into four groups (n = 8–10 mice each). Group 1 served as control animals and received normal pellet diet and water ad libitium. Group 2 mice were orally administered with A. vera gel extract (AV) (50 mg/kg body weight) 0.2 ml on alternate days for 30 days. Group 3 mice were irradiated with whole-body X-ray exposure (0.25 Gy twice a day) for four consecutive days in the last week of the experimental protocol (X-ray). Group 4 mice were orally administered with A. vera gel extract (as in group 2) followed by whole-body X-ray exposure (as in group 3) (AV + X-ray). After scrutiny of various documented studies, dosage of the A. vera gel extract was standardized in our laboratory. 32
Preparation of serum and tissue samples
A drop of 0.5% proparacaine hydrochloride ophthalmic solution was used to anesthetize the retro-orbital plexus of mice and blood samples were collected after 24 h on the last day of study. Lactate dehydrogenase (LDH) activity, hematological parameters, inflammatory markers like TNF-α and IL-6, and antioxidant defense system were analyzed in serum and plasma. Spleen tissue was excised after cervical dislocation and examined for histoarchitectural alterations and antioxidant defense system. Micronucleus assay was done for the assessment of genotoxic damage and terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine, 5′-triphosphate (dUTP) nick end labeling (TUNEL) assay was employed in splenic tissue for detection of apoptosis.
Histopathological studies
After killing the animals, spleen tissue from different treatment groups was excised and immediately fixed in 10% formaldehyde solution. The tissue was washed in water to remove excessive fixative, gradually dehydrated in ascending series of alcohol, cleared in xylene, and embedded in paraffin wax. Sections were cut at 5–7 µm and stained with hematoxylin and eosin to examine any histopathological alteration. 33
Micronucleus assay
Micronucleus assay was performed according to the method described previously. 34 Briefly, tissue was homogenized in chilled homogenizing buffer (24 mM disodium ethylenediaminetetraacetate dihydrate Na2-EDTA buffer pH 7.5, containing 75 mM NaCl) and centrifuged at 5000 g for 10 min. The supernatant was discarded and the pellet was resuspended in fresh homogenizing buffer. A drop of suspension was spread into a smooth layer on grease-free microscopic slides using coverslip. The slides were then air dried in a dust-free environment for at least 12 h before staining. The splenocytes were then stained with May and Grunwald for 1–2 min followed by staining with Giemsa for 10 min. Maximum of 300 cells were counted per mice for the presence of micronuclei using a light microscope.
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling assay
Apoptosis was detected using a TUNEL assay kit (TACS-XL DAB) according to the manufacturer’s recommended protocol. The splenic section was deparaffinized in xylene and rehydrated through a graded series of alcohol. This assay relies on the ability of the enzyme terminal deoxynucleotidyl transferase to incorporate brominated nucleotide (BrdU) into free 3′-hydroxyl termini generated by the fragmentation of genomic DNA during apoptosis. Anti-BrdU antibody was used to detect Br-dUTP complex and was incubated with streptavidin–horseradish peroxidase. The apoptotic cells were visualized under light microscope using DAB-based detection system and these sections were counterstained with methyl green.
Lactate dehydrogenase
LDH activity was measured in serum and the splenic tissue according to the method described previously. 35 It relies on the principle that LDH catalyzes the oxidation of lactate to pyruvate accompanied by the simultaneous reduction of NAD+ to NADH. LDH activity is proportional to the decrease in absorbance due to the reduction of NAD+ and was measured at 340 nm using an extinction coefficient of 6.22 mM−1 cm−1. LDH activity was expressed as nanomoles of NADH consumed/min/mg protein.
Reactive oxygen species
The total ROS content in blood plasma and splenic tissue homogenate was estimated according to the method described previously. 36 The level of intracellular ROS was measured using DCFH-DA, which is an oxidation-sensitive fluorescence probe. DCFH-DA upon enzymatic hydrolysis by intracellular esterases forms nonfluorescent DCFH, which is subsequently oxidized to form highly fluorescent DCF in the presence of ROS and measured at excitation and emission wavelengths of 488 and 525 nm. The degree of DCF fluorescence intensity demonstrates the amount of ROS formed in the cells.
Lipid peroxidation
Lipid peroxidation (LPO) assay was detected in blood and splenic tissue by measuring the formation of TBA-reactive substances (TBA-RS), according to the previously described method. 37 Malondialdehyde (MDA), a secondary end product of the oxidation of polyunsaturated fatty acids, reacts with TBA yielding a pinkish red chromogen with an absorbance maximum at 532 nm. The extinction coefficient of 1.56 × 105 M−1 cm−1 was used to calculate MDA-TBA chromophore production and expressed as nanomoles of MDA-TBA chromophore formed per milligram of protein.
Reduced glutathione
GSH was estimated in blood plasma and splenic tissue by the method described previously. 38 The total nonprotein sulphydryl group in GSH reacts with DTNB to form deep yellow colored 2-nitro-5-mercaptobenzoic acid, which has absorption maxima at 412 nm. Reduced GSH was used as a standard to calculate nanomoles of GSH content per milligram of protein.
Glutathione reductase
Glutathione reductase (GR) activity was estimated in blood plasma and splenic tissue according to the method described previously. 39 The method is based on the measurement of GSH produced when the enzyme is incubated with GSSG in the presence of NADPH. Enzyme activity was quantitated by measuring the utilization of NADPH at 340 nm and was calculated as nanomoles of NADPH oxidized/min/mg protein using a molar extinction coefficient of 6.22 mM−1 cm−1.
Glutathione peroxidase
Glutathione peroxidase (GSH-Px) activity was measured in blood plasma and splenic tissue by the method as described previously. 40 GSH-Px reduces hydrogen peroxide (H2O2) to water and leads to the formation of GSSG from GSH. The decrease in absorbance due to the oxidation of NADPH to NADP+ was read at 340 nm and the enzyme activity was expressed as nanomoles of NADPH consumed per minute per milligram of protein using an extinction coefficient of 6.22 mM−1 cm−1.
Catalase
Catalase (CAT) activity was carried out in blood plasma and spleen tissue by the method described previously. 41 The rate of disintegration of H2O2 into water and oxygen is proportional to the concentration of CAT. One enzyme unit was calculated as the amount of enzyme required to decrease the absorbance at 240 nm by 0.05 units. The activity of the enzyme was expressed as IU of H2O2 reduced per minute per milligram of protein.
Superoxide dismutase
Superoxide dismutase (SOD) activity was estimated in blood plasma and spleen tissue according to the method described previously. 42 This method is based on the principle of the inhibitory effect of SOD on the reduction of NBT dye by superoxide anions, which are generated by photooxidation of hydroxylamine hydrochloride (NH2OH.HCl). The percentage inhibition in the rate of NBT reduction was noted at 560 nm and was expressed as IU per milligram of protein.
Assessment of hematological parameters
Total leucocyte counts or white blood cells
White blood cell (WBC) counting in blood was carried out by the previously described method 43 in which blood samples were diluted with Turk’s fluid (1% gentian violet and 2% acetic acid) present in the diluent to lyse the red blood cells (RBCs). All the nucleated cells were stained by crystal violet WBCs and were counted in a Neubauer’s chamber. WBC count in undiluted blood was calculated and was expressed as total leucocyte count (TLC) per cubic millimeter of blood.
Differential leukocyte counts
Differential leukocyte count (DLC) in the blood was also done by the previously described method. 43 A thin blood smear was stained with Giemsa’s or Leishman’s stain and different subtypes of the leukocytes were then identified and counted under light microscope. Percentage of each type of cell was calculated and the DLC was expressed as percentage (%).
Total platelet counts
Platelet count was estimated according to the previously described method 44 using ammonium solution as the diluting fluid. Platelet count was calculated and was expressed as platelet counts per cubic millimeter of blood.
Hemoglobin
Hemoglobin content was determined by the cyanmethemoglobin method according to the previously described method 43 using Drabkin’s solution as the diluting fluid. Hemoglobin is converted to cyanmethemoglobin in the presence of potassium ferricyanide and potassium cyanide. The absorbance of cyanmethemoglobin is proportional to the hemoglobin concentration and was estimated at 540 nm on a spectrophotometer. Hb levels in unknown samples were calculated and were expressed as grams per deciliter.
Red blood cells
The total number of RBCs was estimated according to the previously described method 43 using Hayem’s solution as the diluting fluid. Their number in undiluted blood was calculated using Neubaur’s chamber and was expressed as the total number of RBCs per cubic millimeter of blood.
Inflammatory markers
TNF-α and IL-6 levels were estimated in blood serum using in vitro enzyme-linked immunosorbent assay kit (ELISA) (Ray Biotech) as the detection system.
Protein estimation
Total protein content was determined according to the method described previously 45 using BSA as the standard.
Statistical analysis
Data were expressed as mean ± standard deviation (SD) and analyzed by one-way analysis of variance followed by the Least Significant Difference (LSD) post hoc test using Statistical Package for the Social Sciences.
Results
Effect of X-ray irradiation and/or aqueous A. vera gel extract on body weight, diet intake, and water consumption
X-ray irradiated animals exhibited some signs of radiation sickness during exposure period. The symptoms included decreased diet intake, irritability, hair ruffling, and behavioral alterations were also observed after irradiation. Body weight of control and A. vera-treated animals remained unaltered throughout the experiment period and showed a normal trend in maintaining body weight. However, a continuous decline in the body weight was observed in X-ray on the last week (i.e. fourth week) of the exposure in mice when compared to their initial body weight on the first week of the study as well as when compared to control and A. vera-treated groups. Also, the body weight of mice in A. vera pretreated and X-ray irradiated group increased significantly when compared to the X-ray irradiated group on the last week of the exposure (Figure 1). Water consumption remained unaltered in all the treatment groups.

Effect of X-ray irradiation and/or aqueous Aloe vera gel extract on body weight.
Histopathological analysis of spleen
Histopathological analysis of spleen from control and A. vera extract–treated animals revealed normal architecture consisting of areas of white pulp with germinal centers surrounded by lighter marginal zones and the red pulp. White pulp contained lymphoid aggregations, mostly lymphocytes and macrophages, which were arranged around the arteries. Red pulp had numerous vascular sinuses along with connective tissues termed as trabeculae (Figure 2(a) and (b)). X-ray irradiation caused a decrease in the amount of white pulp and an increase in the red pulp, a large number of macrophages, and reduction in a number of lymphocytes in the white pulp region, infiltration of red pulp in the white pulp area, and thickened trabeculae (Figure 2(c)). The histoarchitecture of spleen in the AV + X-ray group appeared to be normal like control animals (Figure 2(d)).
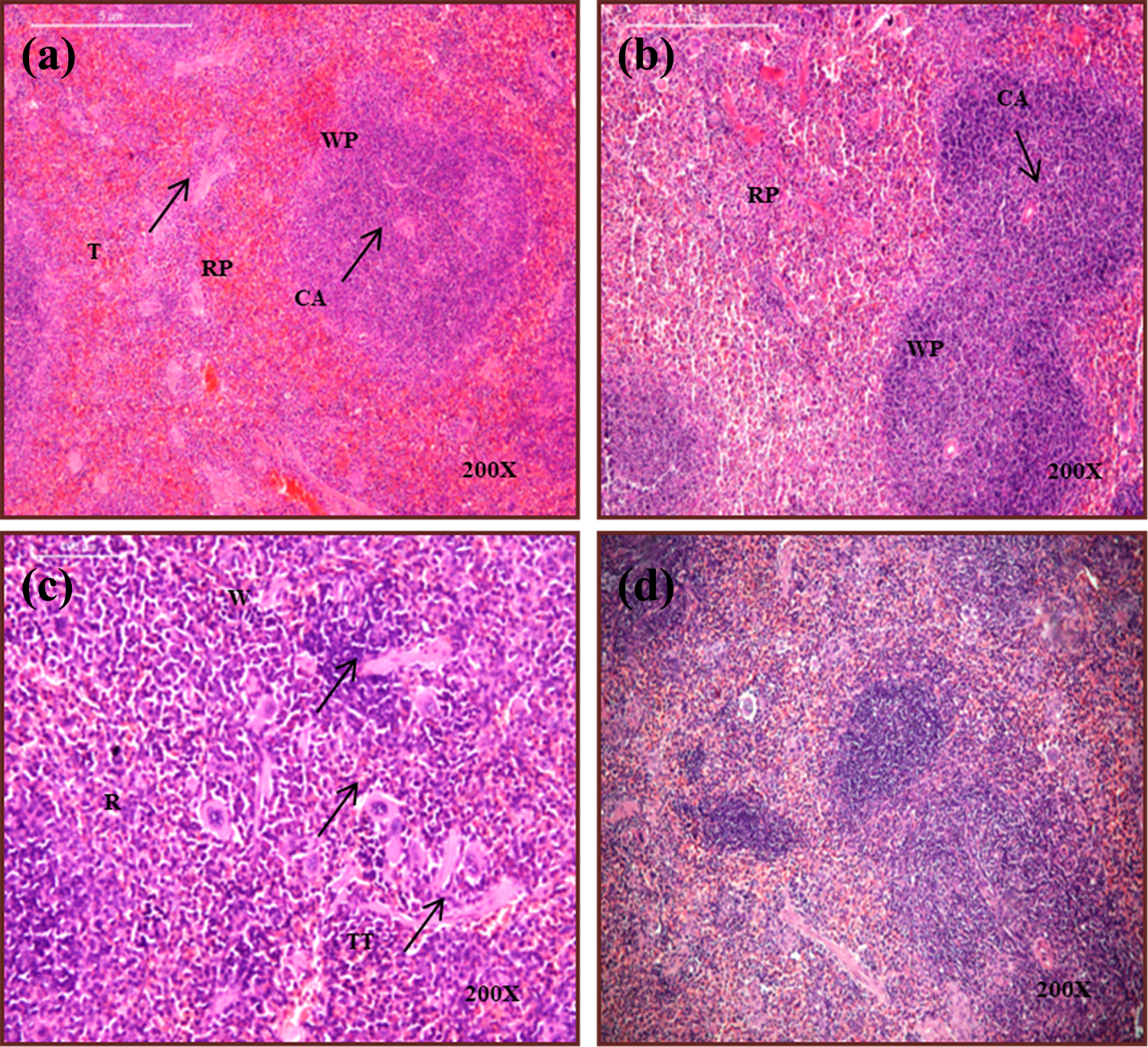
Photomicrograph of spleen section from (a) control animals, (b) aqueous A. vera gel extract, (c) X- ray irradiated animals, and (d) X-ray irradiated animals pretreated with aqueous A. vera gel extract. WP: white pulp; RP: red pulp; CA: central arteriole; T: trabeculae; GC: germinal center; W: decreased white pulp; R: increased red pulp; M: increased macrophages; I: infiltration in white pulp; TT: thickened trabeculae.
Evaluation of clastogenic damage by micronucleus assay
Some clastogenic damage was observed in control and A. vera-administered groups (Figure 3(a) and (b)]. However, a significant elevation in micronucleus formation was observed in splenic tissue of the X-ray irradiated group (Figure 3(c)) when compared with control animals (p ≤ 0.001) (Figure 3(a)). The AV + X-ray group showed a lesser amount of micronucleus formation as compared to the only X-ray irradiated group (p ≤ 0.01) (Figure 3(d) and (e)).
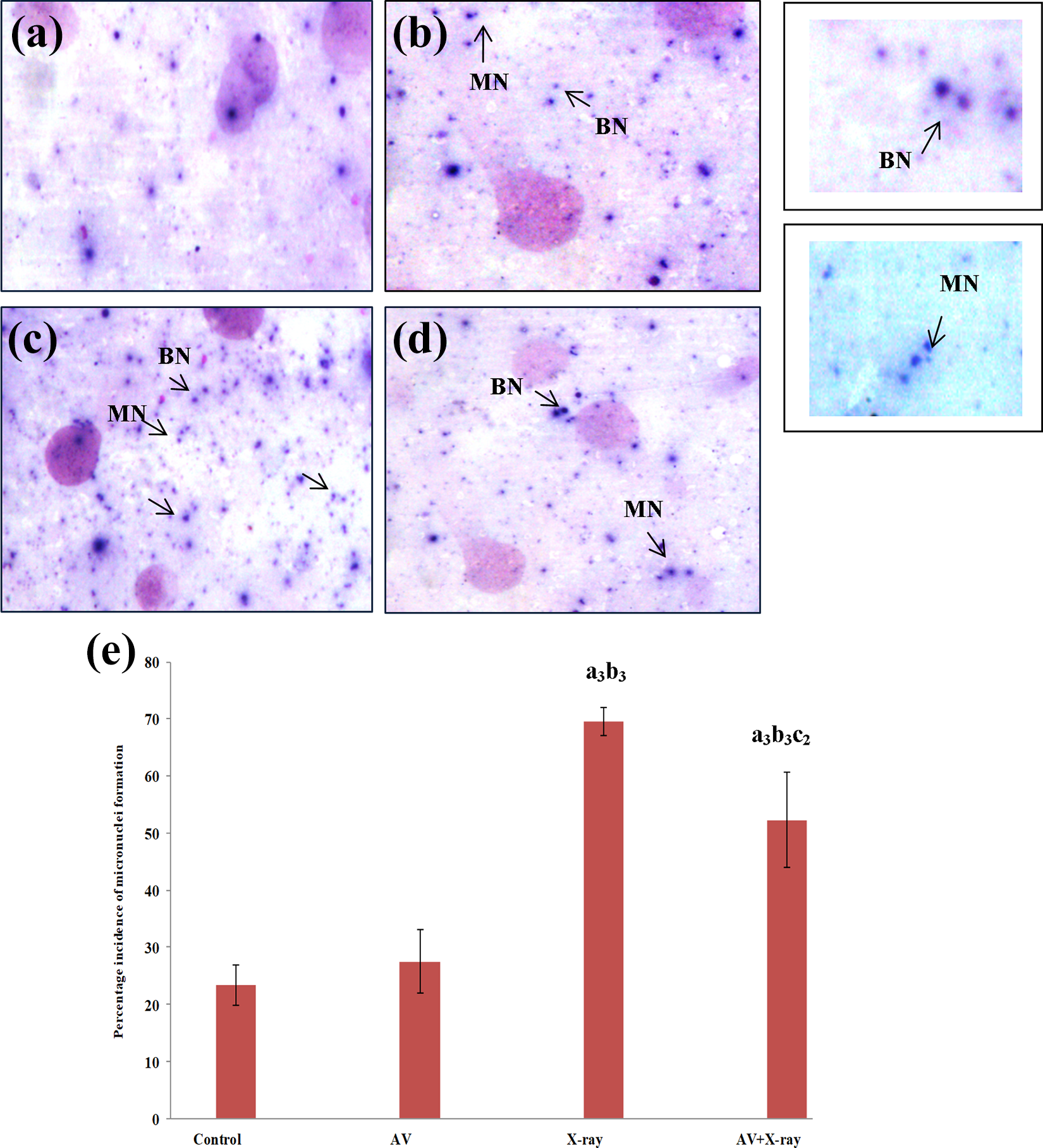
Effect of X-ray/or aqueous A. vera gel extract in the spleen on the basis of micronucleus assay. Photomicrograph of dividing cells of spleen (a) control, (b) A. vera, (c) X-ray irradiated group, (d) A. vera pretreated X-ray irradiated group (binucleated (BN) cells, micronucleus (MN) formation), (e) data are represented as mean ± SD (n = 5) and are analyzed by one-way ANOVA followed by post hoc test. a 3p ≤ 0.001 significant with respect to control group, b 3p ≤ 0.001 significant with respect to A. vera group, c 2p ≤ 0.01 significant with respect to X-ray irradiated group. ANOVA: analysis of variance; SD: standard deviation.
Evaluation of apoptosis by TUNEL assay
The X-ray irradiated group showed apoptotic cells as revealed by brown stained nucleus (Figure 4(c)). Control and aqueous A. vera gel extract–treated groups showed normal methyl green stained nucleus with some amount of brown stained nucleus (Figure 4(a) and (b)). Administration of A. vera gel extract did not cause any change in the apoptotic index when compared to the control group (Figure 4(e)). However, X-ray irradiation induced a significant increase in the percentage of apoptotic cell count when compared with its control counterparts (p ≤ 0.001). The AV + X-ray group showed a lesser amount of apoptotic cells percentage as compared to the only X-ray irradiated group (p ≤ 0.001) (Figure 4(d)).

TUNEL-stained section of spleen from (a) control group, (b) aqueous A. vera gel extract, (c) X-ray irradiated group, (d) A. vera pretreated X-ray irradiated group (methyl green stained cells are nonapoptotic, brown stained cells are apoptotic (arrow)), (e) data are represented as mean ± SD (n = 5) and are analyzed by one-way ANOVA followed by post hoc test. a3p ≤ 0.001, significant with respect to the control group, b3p ≤ 0.001 significant with respect to A. vera group, c3p ≤ 0.001 significant with respect to X-ray irradiated group. ANOVA: analysis of variance; SD: standard deviation; TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling.
Assessment of tissue injury by serum LDH analysis
No change in LDH activity was observed in control and A. vera gel extract–administered group. However, X-ray exposure caused a significant rise in serum LDH activity when compared with the control group (p ≤ 0.001). AV + X-ray group caused a significant improvement in LDH activity when compared to the X-ray irradiated group (p ≤ 0.001) (Figure 5).

Effect of X-ray and/or aqueous A. vera gel extract on lactate dehydrogenase activity in serum. Data are represented as mean ± SD (n = 8) and are analyzed by one-way ANOVA followed by post hoc test. a 3p ≤ 0.001 significant with respect to control group, b 3p ≤ 0.001 significant with respect to aqueous A. vera gel extract group, c 3p ≤ 0.001 significant with respect to X-ray irradiated group. Units: nmoles of pyruvate formed per milligram of protein per minute. ANOVA: analysis of variance; SD: standard deviation.
Evaluation of peroxidative damage by ROS and LPO levels
No change was observed in ROS and LPO levels in A. vera gel extract–administered group when compared to control groups. However, X-ray irradiation caused a significant elevation in ROS and LPO levels in blood plasma (p ≤ 0.001) and spleen (p ≤ 0.001) when compared to the control groups (Figure 6(a) to (d)). A significant reduction was observed in ROS and LPO levels in the AV + X-ray group when compared with X-ray irradiated groups (p ≤ 0.01).

(a) and (b) Effect of the X-ray and/ or A. vera on ROS formation in plasma and spleen. Data are represented as mean ± SD (n = 6) and are analyzed by one-way ANOVA followed by post hoc test. a 2p ≤ 0.01 significant with respect to control group, b 2p ≤ 0.01 significant with respect to A. vera group, c 2p ≤ 0.01 significant with respect to X-ray irradiated group. Units: Fluorescence intensity per milligram of protein. (c) and (d) Effect of X-ray and/or aqueous A. vera gel extract on lipid peroxidation in blood and spleen. Data are represented as mean ± SD (n = 8) and are analyzed by one-way ANOVA followed by post hoc test. a 3p ≤ 0.001 significant with respect to control group, b 3p ≤ 0.001 significant with respect to A. vera group, c 3p ≤ 0.001 significant with respect to X-ray irradiated group. Units: nanomoles of TBA-MDA chromophore formed per milligram of protein. ANOVA: analysis of variance; SD: standard deviation.
Antioxidant defense mechanism
No change was observed in GSH levels in A. vera gel extract–administered group when compared to the control group. However, GSH levels in the X-ray irradiated group significantly declined when compared to the control group in blood plasma (p ≤ 0.001) and spleen (p ≤ 0.001). Significant elevation in GSH content was observed in the AV + X-ray group when compared to X-ray irradiation only (p ≤ 0.01) (Table 1).
Effect of X-ray and/or aqueous Aloe vera gel extract on antioxidant defense system in plasma.a

GSH: glutathione reduced; GR: glutathione reductase; GSH-Px: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase ; NADPH: nicotinamide adenine dinucleotide phosphate reduced; ANOVA: analysis of variance; SD: standard deviation.
aData are represented as mean ± SD (n = 8) and are analyzed by one-way ANOVA followed by post hoc test.
bp ≤ 0.01 significant with respect to control group.
cp ≤ 0.01 significant with respect to aqueous A. vera gel extract group.
dp ≤ 0.05 significant with respect to X-ray irradiated group
ep ≤ 0.001 significant with respect to aqueous A. vera gel extract group
fp ≤ 0.05 significant with respect to aqueous A. vera gel extract group
gp ≤ 0.01 significant with respect to X-ray irradiated group.
hp ≤ 0.001 significant with respect to X-ray irradiated group.
ip ≤ 0.001 significant with respect to control group.
No change was observed in GR, GSH-Px, and CAT activities in A. vera gel extract–administered group when compared to the control group. X-ray irradiation caused significant enhancement in GR, GSH-Px, and CAT activities when compared to control in blood plasma (p ≤ 0.001) and splenic tissue (p ≤ 0.001). AV + X-ray group showed significant improvement in these enzymatic activities when compared with the X-ray exposed group (p ≤ 0.01) (Table 1).
A. vera gel extract administration did not cause any change in SOD activity when compared to the control group. The X-ray exposed group showed a significant enhancement in SOD activity of blood plasma (p ≤ 0.001), while decline in the activity of splenic tissue (p ≤ 0.001) was observed when compared with the control group. However, the AV + X-ray group caused a significant improvement in SOD activity when compared with the X-ray exposed group in blood plasma (p ≤ 0.01) and splenic tissue of mice (p ≤ 0.01) (Table 1).
Effect of X-ray and/or aqueous A. vera gel extract on hematological parameters
No change was observed in neutrophil counts and TLC levels in A. vera gel extract administration when compared to the control group. X-ray exposure led to a significant enhancement in neutrophil counts and TLC when compared to the control group (p ≤ 0.001). In contrast, AV + X-ray group induced a marked reduction in neutrophil counts and TLC in comparison to X-ray irradiated group (p ≤ 0.01). Lymphocytes, RBCs, and Hb remain unaltered in X-ray irradiated, control, AV + X-ray groups (Table 2).
Effect of X-ray and/ or aqueous Aloe vera gel extract on hematological parameters.a

TLC: total leucocyte count; DLC: differential leukocyte counts; RBC: red blood cells; Hb: hemoglobin; ANOVA: analysis of variance; SD: standard deviation.
aData are represented as mean ± SD (n = 6) and are analyzed by one-way ANOVA followed by post hoc test.
bp ≤ 0.001 significant with respect to control group.
cp ≤ 0.001 significant with respect to A. vera group
dp ≤ 0.001 significant with respect to X-ray irradiated group.
ep ≤ 0.01 significant with respect to control group.
fp ≤ 0.01 significant with respect to A. vera group.
gp ≤ 0.01 significant with respect to X-ray irradiated group.
hp ≤ 0.05 significant with respect to A. vera group.
Administration of A. vera gel extract did not cause any significant difference in platelet counts when compared to the control group. However, platelet counts were significantly declined by X-ray irradiation when compared to the control group (p ≤ 0.001). AV + X-ray group showed a significant improvement in platelet counts when compared to X-ray irradiated group (p ≤ 0.01) (Table 2).
Effect of X-ray and/ or aqueous A. vera gel extract on inflammatory markers
Administration of A. vera gel extract did not cause any change in TNF-α and IL-6 levels when compared to the control group. However, inflammatory markers (TNF-α and IL-6) were significantly elevated by X-ray irradiation when compared to the control group (p ≤ 0.001, p ≤ 0.001). AV + X-ray group showed a significant improvement when compared to X-ray irradiated group (p ≤ 0.01, p ≤ 0.01) (Figure 7(a) and (b)).
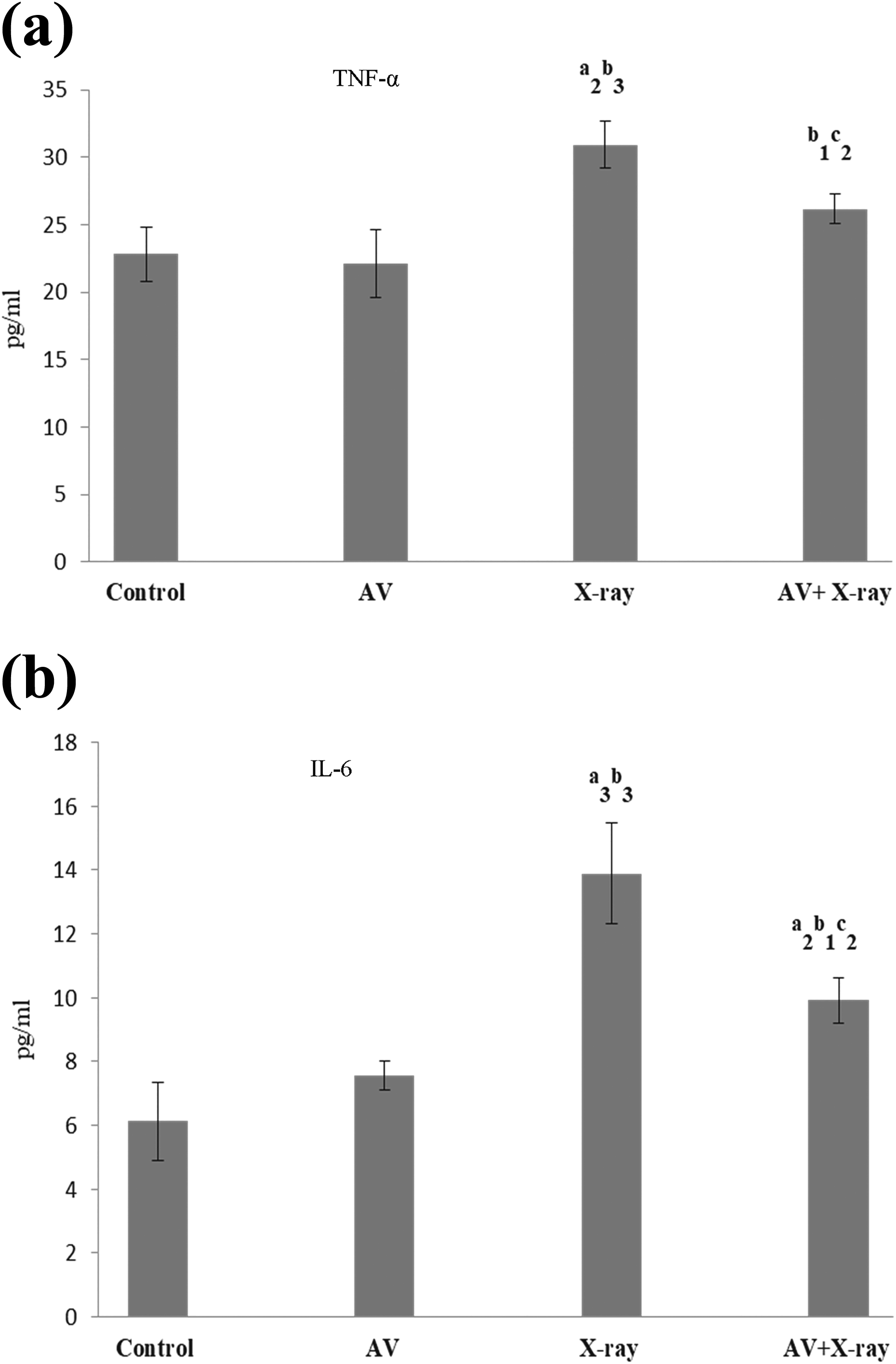
(a) and (b) Effect of X-ray and/ or aqueous A. vera gel extract on TNF-α and IL-6 in serum. Data are represented as mean ± SD (n = 5) and are analyzed by one-way ANOVA followed by post hoc test. a 3p ≤ 0.001, a 2p ≤ 0.01, significant with respect to control group, b 3p ≤ 0.001, b 1p ≤ 0.05 significant with respect to A. vera group, c 2p ≤ 0.01 significant with respect to X-ray irradiated group. ANOVA: analysis of variance; SD: standard deviation.
Discussion
The present study describes the potential of A. vera gel extract in protecting the hematopoietic system against injuries caused by X-ray irradiation. Ionizing radiation inflicts genotoxic damage causing chromosomal rearrangements and mutations. 13,46 Studies on accidental or intentional radiation exposure have revealed that hematopoietic tissues commonly suffer damage due to their radiosensitive nature. 47
Histopathological analysis of splenic tissue of mice exposed to X-ray irradiation exhibited a reduction in the amount of white pulp, increased red pulp area, increase in the number of macrophages, poor germinal centers associated with lighter marginal zones, and infiltration in white pulp along with thickened trabeculae. 48 Infiltration of red pulp into the white pulp region was associated with the accumulation of neutrophils in the red pulp of spleen of animals exposed to radiation. 21 Various histopathological alterations in splenic tissue have been reported after gamma-ray irradiation in rats. 49 In the present study, AV + X-ray-treated animals revealed a considerably reduced population of macrophages in splenic tissue when compared to X-ray irradiated animals. Previously, studies have demonstrated that radiation exposure-induced histopathological damage in the spleen was ameliorated by the administration of Adhatoda vasica and Pinus koraiensis extract. 50
The target for the direct or indirect effects of ionizing radiation is the genetic material of cells, which leads to the manifestation of their acute and chronic biological effects. 51 It has been demonstrated that X-ray irradiation leads to the persistence of damaged DNA fragments resulting in an enhanced number of micronuclei or chromosomal aberrations. 52 Enhanced micronuclei formation in human peripheral blood lymphocytes is a reliable in vivo biomarker for evaluating the clastogenic damage caused by irradiation. 53,54 Whole-body X-ray exposure caused an increased number of micronuclei in spleen tissue and bone marrow cells of mice. 55,56 Zingiber officinale and Origanum vulgare extract reduced micronuclei formation induced by ionizing radiation in the bone marrow of mice. 57,58 Similarly, a significant decline in the micronuclei number in AV + X-ray-treated animals suggested the radioprotective ability of A. vera.
Oxidative stress induced by ionizing radiation is associated with increased oxidative DNA damage, which may contribute to apoptosis. 59 In the present study, splenic tissue sections from X-ray irradiated mice revealed numerous apoptotic nuclei when compared to control counterparts. These observations were in agreement with previous reports, which showed enhanced apoptosis after radiation exposure to animals. 60 However, apoptotic cells were found to be reduced upon the administration of A. vera extract to X-ray irradiated mice. Previously, we have reported that the apoptotic index was increased in testes of X-ray irradiated mice, which was decreased upon A. vera gel extract administration. 30
The alterations in hematological parameters are considered the most sensitive biological markers for the exposure to both internal and external irradiation. After whole-body gamma irradiation, injury to animal tissues was well reflected in the peripheral blood. 13,61,62 It has been reported that radiation therapy to hepatocellular carcinoma patients caused a reduction in peripheral blood lymphocyte counts. 63 The damaging effect of gamma radiation on RBC counts may be attributed to the loss of cells from the circulation by hemorrhage or leakage through capillary walls or direct destruction of mature circulating cells. 64 On the contrary, some reports indicate no change in RBCs and Hb levels due to gamma-ray exposure in mice. 65,66 In the present study, RBC count and Hb levels remained unaltered after X-ray exposure, which might be due to the remarkable regenerative capacity of blood cells to recover in short periods of time. 67 In the present study, elevation in WBC counts was observed in X-ray exposed mice when compared to control counterparts. Enhanced ROS generation results in the activation of macrophages due to the release of proinflammatory cytokines, which in turn contributes to neutrophil infiltration and inhibition of antioxidant pathways leading to apoptosis. However, the AV + X-ray group showed a decline in neutrophil counts when compared to the X-ray irradiated group.
Lymphocytes are also known to be radiosensitive and their depletion has been associated with a reduced immune response. 68 Some studies revealed a decline in splenic lymphocytes after X-ray exposure in mice. 48 Exposure to ionizing radiation can affect the immune system by suppressing or stimulating it, which contributes to neutrophil accumulation due to overproduction of ROS and plays a key role in the induction of apoptosis in the thymus. 69 In the present study, lymphocyte counts remained unaltered in all the treatment groups because radiosensitive phenotype has a tendency to induce apoptosis, while nonproliferative cells and activated lymphocytes are more radioresistant. 70 Thrall et al. 71 also reported that the platelet number did not change in a significant manner within 48 h after gamma irradiation. However, other studies reported significantly depressed platelet counts after radiation exposure to animals. 72 In the present study, blood platelet counts were significantly decreased in X-ray irradiated animals. These results are in concordance with Eshak and Osman 73 and Gharib et al., 74 which showed a reduction in platelet counts after radiation exposure to animals. However, the AV + X-ray group showed an improvement in platelet count, which revealed that A. vera could boost hematological parameters. These observations are supported by earlier investigations in which A. vera, Alstonia scholaris, and Emblica officinalis extract administration mitigated hematological alterations induced by ionizing radiations in mice. 9,75
Ionizing radiation induces enhanced ROS production, which leads to upregulation of inflammatory reaction as indicated by the production of proinflammatory cytokines such as IL-6 and TNF-α. 76,77 Inflammatory mediators like cytokines and chemokines result in the activation and recruitment of leucocytes to tissue from the blood circulation. 78 In response to ionizing radiation, inflammatory cells such as neutrophils are recruited to inflamed lesion via release of proinflammatory cytokines. 79 It was previously reported that transient inflammatory response is induced by neutrophil infiltration, excessive ROS, and inhibition of antioxidant pathways leading to apoptosis after whole-body X-ray irradiation. 68 TNF-α induces neutrophil apoptosis via death receptor signaling through TNF receptors, leading to enhanced ROS generation and caspase activation. 69 It has been reported that rapid and massive apoptosis may induce neutrophil accumulation. 69 In the present study, neutrophil counts increased in the X-ray exposed group, which may be due to increased levels of ROS that may induce apoptosis when compared to the control group. However, in the AV + X-ray group, neutrophil counts were reduced when compared to the X-ray irradiated group. Similarly, E. officinalis linn, A. scholaris, and A. vera extracts showed less severe radiolesions and an early onset of recovery in mice blood. 8,75
LPO is a well-established mechanism involved in the oxidative deterioration of the cell architecture which may eventually lead to cell death. 80 Radiation exposure caused elevated levels of LPO in blood, liver, kidney, testes, and spleen of animals. 30,31,81 In the present study, X-ray irradiated animals showed twofold elevation in ROS and LPO levels when compared to control and A. vera-administered animals. However, the AV + X-ray group revealed reduced ROS and LPO when compared to X-ray irradiated animals. Previously documented studies also exhibited enhanced LPO levels in hepatic and testicular tissues of animals upon gamma-ray exposure, which was improved by the administration of A. vera. 11,82,83
The amount of LDH enzyme in the serum reflects alterations in the plasma membrane integrity and/or permeability. 84 It has been reported by various authors that elevated activity of LDH was observed after irradiation of serum, thymus, liver, spleen, kidney, and testis of mice. 30,31,85 In the present study, an increase in the activity of LDH was observed in the serum, which is a clear indication of damage caused by the exposure to X-ray irradiation. 30,31,86 This observation is in concordance with reports, where Podophyllum hexandrum administration to gamma-irradiated animals exhibited significantly decreased LDH activity in Bronchoalveolar lavage fluid (BALF) of mice lung. 87 LDH activity in serum was found to be decreased in Ginseng and A. vera pretreated and irradiated mice. 88 Reduced LDH levels in AV + X-ray group showed the ameliorative potential of A. vera.30,31 It is speculated A. vera conferred protection by preserving the membrane integrity and reducing the leakage of these enzymes from hepatic, splenic, renal, and testicular tissues induced by X-ray irradiation.
Various endogenous and exogenous antioxidants are used to maintain redox balance in the cells. 89 Considering oxidative stress–mediated damage by ionizing radiation, it is expected that antioxidants would have the capability to combat the damaging effects by neutralizing ROS. 90 Due to the excessive ROS production, the host antioxidant defense mechanisms are overwhelmed, resulting in oxidative injury to cellular components. Previously documented studies showed a reduction in GSH content after radiation exposure in blood, hepatic, splenic, renal, and testicular tissues of mice. 30,31,91,92 In the present study, blood and spleen GSH concentration was found to be reduced in X-ray irradiated animals when compared to the control group. This could be due to enhanced utilization of antioxidant system during detoxification of the free radical generated by ionizing radiation. Previous studies showed the enhanced activity of CAT, GSH-Px, GR, and SOD in brain, liver, and kidney of animals after X-ray exposure. 93 –95 The administration of Panax ginseng and A. vera extract improved the endogenous antioxidant enzymes in the hepatic and renal tissues of X-ray irradiated animals. 96 –98
Conclusion
In the present study, A. vera pretreatment to X-ray irradiated animals reduced damage by protecting against radiation-induced histopathological, biochemical, histopathological, hematological, and DNA damage. These effects may be attributed to its antioxidant, anti-inflammatory, and anti-apoptotic potential.
Footnotes
Acknowledgements
The authors gratefully acknowledge the financial support provided by the University Grant Commission–Rajiv Gandhi National Fellowship, New Delhi, India.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the University Grant Commission-Rajiv Gandhi National Fellowship, New Delhi, India (RGNF-SC-CHA-3794 2011-12).
