Abstract
The toxic effects of x-ray radiation on eye development was measured using zebrafish as a model organism. Zebrafish embryos at 8 h post-fertilization (hpf) were irradiated using X-rays at doses of 1, 2, 4, and 8 Gy. At 24 and 48 hpf, x-ray radiation induced a significant increase in reactive oxygen species (ROS) content and cell apoptotic signals. Both of these increases were dose dependent and there were significant positive relationships between them at 24 hpf. At 48 and 72 hpf, the increase of ROS concentration can be eliminated by increasing activities of superoxide dismutase and catalase. Although the ROS generated by x-ray radiation caused a significant increase in cell apoptosis at 24 and 48 hpf, the cellular layers of the retina and lens formation in the irradiated groups were not significantly disrupted at 144 hpf compared with the control group, with the exception of a heterogeneous distribution of the cells in inner nuclear cell layer and a significant decrease in the diameters of whole eyes after 8 Gy irradiation. X-Ray radiation at later stages of gastrulation may not cause distinct optic complications; however, there is still a risk of microophthalmia at high doses of irradiation.
Introduction
Ionizing radiation, such as X-rays, γ-rays, and heavy ions, are characterized by short wavelength and strong energy and penetrability, which make them more deleterious to animal tissues than nonionizing radiation. This type of radiation has been widely used in medicine for diagnosis, therapy, and in industrial applications. 1,2 However, a systematic toxicological study is still required to better understand the potential hazards of ionizing radiation, as it can induce various biological effects, including cellular death, mutations, carcinogenesis, and the after-effects of genetic toxicity. 3 Organisms exposed to ionizing radiation can generate a large number of reactive oxygen species (ROS), such as hydrogen peroxide (H2O2), lipid hydroperoxides, and peroxynitrite. H2O2 can react with various targets, thereby causing DNA and protein damage that can lead to cell damage or even cell death. 4,5 X-Rays are the most widely used ionizing radiation. Previous studies have proved that X-rays can induce toxic effects in various targets, such as lymphocytes, cerebellum, and the lens. 6 –8
The eye is one of the most important organs directly exposed to the environment. Many studies have focused on the effects of radiation on animal eyes. In vitro studies indicated that ultraviolet (UV) radiation can induce damage to human corneal, lens, and retinal pigment epithelial cells. 9 Moreover, in vivo studies showed that nonionizing UV radiation can cause oxidative damage, cell apoptosis, morphological changes, and visual defects in eyes. 10 –12 Compared with adults, embryos and larvae are more sensitive to radiation. Ionizing X-ray exposure can cause lens dysfunction and abnormal lens formation during embryonic development. 7,13 Another ionizing irradiation, γ-rays, can also cause cell apoptosis and disturb cell differentiation in the eyes. 14
Over the past decade, the zebrafish (Danio rerio) has been widely used as a vertebrate model in developmental study due to its small size, rapid embryonic development, the transparency of embryos, the large number of offspring, as well as several other advantages, enabling the continuous observation of its developing organs. In addition, many of the key genes involved in developmental processes, cell cycle progression, and proliferation as well as differentiation are highly conserved between zebrafish and humans. 15,16 More importantly, zebrafish are very visually orientated and their retina and lens show much the same morphology as other vertebrates including humans. Hence, they are well suited for examining ocular development, function, and disease. 17 In recent years, zebrafish have also been extensively investigated in an attempt to understand the DNA damage response to ionizing radiation, 3,18 –22 especially in the embryonic stage. Embryonic development is rapid in zebrafish. Major organ systems, including eyes are evident within 48 h post-fertilization (hpf). Moreover, embryogenesis occurs in vitro, and the effects of radiation exposure are easy to assess without parental killing. Thus, zebrafish embryos are often chosen to study the toxic effects of radiation on organ development.
Considering the wide use of X-rays in diagnosis and therapy and their toxic effects on different parts of the eye reported in previous studies, we investigated the effects of X-ray radiation on the integral eye, including retina and lens during development using zebrafish as a model organism in the present study. Several important biological endpoints including ROS generation, cell apoptosis, and morphological changes were used to evaluate the risks of ionizing radiation on eye development.
Material and methods
Zebrafish maintenance and embryo handling
Zebrafish maintenance and embryo collection were performed according to standard operating procedures as described elsewhere. 23 AB strain zebrafish (Danio rerio) were maintained under 14-h light/10-h dark cycles, with circulating freshwater aquaria at 28 ± 1°C. The fish were fed twice daily with decapsulated and freshly hatched brine shrimps (Brine Shrimp Direct, Ogden, Utah, USA). Zebrafish embryos obtained from the adults were segregated by sex and mated in tanks overnight. Spawning was induced in the next light cycle when the light was turned on. The embryos were collected and washed using standard zebrafish E3 culture medium (5 mmol/L sodium chloride (NaCl), 0.33 mmol/L calcium chloride, 0.33 mmol/L magnesium sulfate heptahydrate, 0.17 mmol/L potassium chloride (KCl)) at the one-cell to two-cell stage. 24 Then 50 embryos were transferred into 60-mm polystyrene cell culture dishes (Costar, Corning, New York, USA) with 20 mL of E3 culture medium. The embryos were incubated at 28.5°C. Zebrafish use and handling conformed to the Guide for the Care and Use of Laboratory Animals which was approved by the Institutional Animal Use and Care Committee (IAUCC) of Chinese Academy of Sciences. At 6–7 hpf, embryos were examined under a dissecting light microscope (Nanjing Jiangnan Novel Optics, China), and those embryos that had developed normally were selected for subsequent experiments, according to the standardized staging series set forth by Kimmel et al. 25
Embryo irradiation
Embryos were irradiated using an X-ray machine (Faxitron, Buffalo Grove, Illinois, USA) at 8 hpf (gastrula stage) which is before the beginning of eye morphogenesis (12 hpf), enabling the observation of apoptosis induced by radiation during the eye development. The beam energy was 100 keV with a dose rate of 1.750 Gy/min. The embryos exposed to the radiation were placed in a standard 225-cm2 polystyrene cell culture flasks with 20 mL of E3. The doses of x-ray radiation were 1, 2, 4, and 8 Gy. A control group that did not receive irradiation was also included. The embryos were washed with E3 culture medium immediately after the irradiation and transferred back to the 60-mm polystyrene cell culture dishes with 20 mL E3 culture medium. At 23 hpf, 300 embryos from each group were moved to 19.7 mmol/L 1-phenyl-2-thiourea (Sigma, St Louis, Missouri, USA) water to prevent pigment formation.
ROS measurement
The generation of ROS in the zebrafish embryos and larvae exposed to x-ray radiation until 72 hpf was measured using the fluorescent dye dichlorofluorescein–diacetate (DCFH-DA). Then, 15 embryos at 24 and 48 hpf and 15 larvae at 72 hpf from each group were incubated with 20 μg/mL DCFH-DA for 30 min in dark at 37°C. After washing with E3 culture medium and anesthetized using 0.01% MS 222 (Sigma), the fluorescence for each embryo and larva was observed and photographed immediately under a fluorescence microscope (Olympus BX51, Tokyo, Japan) at 100× magnification using blue filter with focus on the eyes.
Cell apoptosis assays
To determine whether in vivo X-ray exposures induced inappropriate cellular apoptosis, two independent cellular apoptosis assays were employed. Cell apoptosis was detected in live embryos using acridine orange (AO) staining. Since the quantification of apoptotic signals is widely used to investigate the effect of radiation on the whole embryos, 26 –28 in this experiment, we quantified apoptotic signals through staining with the vital dye AO at 24, 48, and 72 hpf, respectively. Fifteen samples from each group were assessed. The samples were incubated in 1 mL of 5 μg/mL AO for 40 min in the dark at 28°C and washed twice in the culture medium E3 thoroughly. They were then anesthetized using 0.01% MS 222 (Sigma). Fluorescence in each embryo and larva was photographed immediately using a fluorescence microscope (Olympus BX51) with focus on eyes at 100× magnification using blue filter.
The terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate nick end-labeling (TUNEL) assay was exploited to quantify cell apoptosis more specifically using ApopTag® Fluorescein In Situ Apoptosis Detection Kit (Oncor, Gathersburg, Maryland, USA). The TUNEL assay fluorescently labels the blunt ends of double-stranded DNA breaks that are indicative of programmed cell apoptosis. 26 Embryos at 24 and 48 hpf were fixed in 4% paraformaldehyde (PFA) at 4°C overnight. Then, the embryos were dehydrated by 30, 50, 70, and 100% ethanol successively and finally stored in 100% ethanol at −20°C until analysis. Thirty embryos from each group were evaluated. Before the TUNEL assay was performed, embryos were rehydrated and washed with 0.1% Tween 20 in phosphate buffer solution (PBT) twice. Next, the embryos were digested with proteinase K (10 μg/mL) for 15 min (24 hpf) and 30 min (48 hpf) and fixed again with 4% PFA for 30 min. After washing with PBT, the samples were incubated in 75 μL equilibration buffer for 1 h. Followed by removing the equilibration buffer, 100 μL TdT reaction mix was added into sample, in which the samples were incubated overnight at 37°C. In the next day, TdT reaction mix was cleared away and stop/wash buffer was added. After washing with PBT, the working strength anti-digoxigenin conjugate was applied, and the samples were incubated for 1 h in dark. Finally, the samples were photographed with a fluorescence microscope at 100× magnification using blue filter (Olympus BX51). The fluorescence within eyes was measured and quantified with ImagePro Plus software (Media Cybernetics, Silver Springs, Maryland, USA).
Enzyme extraction and biochemical assays
The embryos and larvae were homogenized on ice with 500 μL chilled phosphate-buffered saline buffer (137 mmol/L NaCl, 2.7 mmol/L KCl, 10 mmol/L disodium phosphate, 2 mmol/L monopotassium phosphate, pH 7.4). The homogenate was centrifuged at 3000g for 10 min at 4°C to obtain the supernatants. The amount of total protein in the supernatants was measured using the bicinchoninic acid (BCA) protein assay kit (Pierce, Rockford, Illinois, USA). The activities of superoxide dismutase (SOD) and catalase (CAT) were determined using diagnostic reagent kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the specified instructions of reagent kits. The measurements were implemented on a microplate reader (TECAN infinite M200, Switzerland) using 96-well plates.
Histological and morphological analysis
For histological evaluations, larvae at 144 hpf were fixed in 4% PFA and embedded in paraffin. Tissue slices (4 μm) were stained with H&E and photographed at 400× magnification under a fluorescence microscope (Olympus BX51) using white light. The pathological slices were evaluated according to Geiger et al. 14 Fifteen larvae from each group were measured. The diameters of the whole eyes and lens were measured to show the morphological changes of zebrafish eyes induced by x-ray radiation. The diameter of the whole eye was defined as the distance from the pigmented epithelium of one pole to the opposite pole paralleled with the spine. For the lens, the diameter was defined as the distance from the lens epithelium of one pole to the opposite pole paralleled with the spine.
Statistics
All values were expressed as means ± SD. The statistics were performed using Statistical Package for Social Sciences version 13.0 (SPSS, Chicago, Illinois, USA). One-way analysis of variance (ANOVA) was applied to detect significant differences between control and irradiated groups (p < 0.05). The figures were plotted using Origin 7.5 (OriginLab, Northampton, Massachusetts, USA).
Results
ROS generation
The generation of ROS in zebrafish embryos and larvae was determined by DCFH-DA. Exposure to X-rays induced intracellular ROS generation in zebrafish eyes, and the ROS content increased in a dose-dependent manner at 24 hpf (Figures 1(a) to (e) and 2(a)). Statistical analysis showed that there was a significant difference in fluorescence signals among all five groups (ANOVA, F 4,70 = 86.885, p < 0.0001). A significant increase in fluorescence signals (Tukey’s test, p < 0.05) was observed in all irradiated groups. At 48 hpf, there was also a significant difference in fluorescence signals among the groups (ANOVA, F 4,70 = 61.479, p < 0.0001). All irradiated groups showed a significant increase in fluorescence signals (Tukey’s test, p < 0.05), with the exception of the 1 Gy-irradiated group. At 72 hpf, x-ray radiation at any dose did not induce a significant increase in fluorescence signals (ANOVA, F 4,70 = 1.704, p = 0. 159) (Figure 2(a)).

Detection of X-ray radiation-induced changes in the contents of ROS and cell apoptosis in eyes of live zebrafish embryos at 24 hpf. ROS content was measured using the fluorescent dye DCFH-DA. Cell apoptosis was determined using AO staining. ROS generation and apoptosis signals were indicated by white fluorescent spot on a black background, shown for (a) ROS content in the control group, (b) ROS content in the 1 Gy-irradiated group, (c) ROS content in the 2 Gy-irradiated group, (d) ROS content in the 4 Gy-irradiated group, (e) ROS content in the 8 Gy-irradiated group, (f) apoptotic signals in the control group, (g) apoptotic signals in the 1 Gy-irradiated group, (h) apoptotic signals in the 2 Gy-irradiated group, (i) apoptotic signals in the 4 Gy-irradiated group, and (j) apoptotic signals in the 8 Gy-irradiated group. ROS: reactive oxygen species; DCFH-DA: dichlorofluorescein–diacetate; AO: acridine orange; hpf: hours post-fertilization.

Concentration–response curves of ROS content and cell apoptosis measured as relative fluorescence within the eyes of zebrafish embryos and larvae. (a) ROS content and (b) cell apoptosis signals. *p < 0.05, N = 15: significant differences between the control and irradiated groups were determined using one-way ANOVA. ROS: reactive oxygen species; ANOVA: analysis of variance..
Cell apoptosis
Staining with AO indicated that the difference in apoptotic signals in all groups was significant (ANOVA, F 4,70 = 284.816, p < 0.0001) at 24 hpf. Compared to a few apoptotic signals within zebrafish eyes in the control group, a considerable increase in apoptotic signals (Tukey’s test, p < 0.05) was observed in all the irradiated groups. The apoptotic signals increased in a dose-dependent manner (Figures 1(f) to (j) and 2(b)). Apoptotic signals increased significantly with increasing ROS content at 24 hpf (Pearson’s correlation, r = 0.99, p = 0.001). At 48 hpf, a significant difference in fluorescence signals was also seen in all groups (ANOVA, F 4, 70 = 58.29, p < 0.0001). Irradiation (4 and 8 Gy) induced a significant increase in fluorescence signals (Tukey’s test, p < 0.05). At 72 hpf, there was no significant difference in apoptotic signals among the groups (ANOVA, F 4, 70 = 2.339, p = 0.064) and the apoptotic signals in all irradiation groups were significantly different from that of the control group (Figure 2(b)). The correlation between ROS content and apoptotic signals was not significant at both 48 hpf (Pearson’s correlation, r = 0.678, p = 0.087) and 72 hpf (Spearman’s correlation, r = 0.051, p = 0.935). In the TUNEL assay, similar results were observed at 24 hpf. X-Ray radiation irradiated at 4 Gy resulted in significant TUNEL labeling within zebrafish eyes (ANOVA, F 1, 28 = 419.846, p < 0.0001) and in the tissues outside the eye (Figure 3(a) and (c)). However, the differences in TUNEL labeling within eyes between the control and 4 Gy group were diminished at 48 hpf (ANOVA, F 1, 28 = 0.106, p = 0.748; Figure 3(b) and (d)).
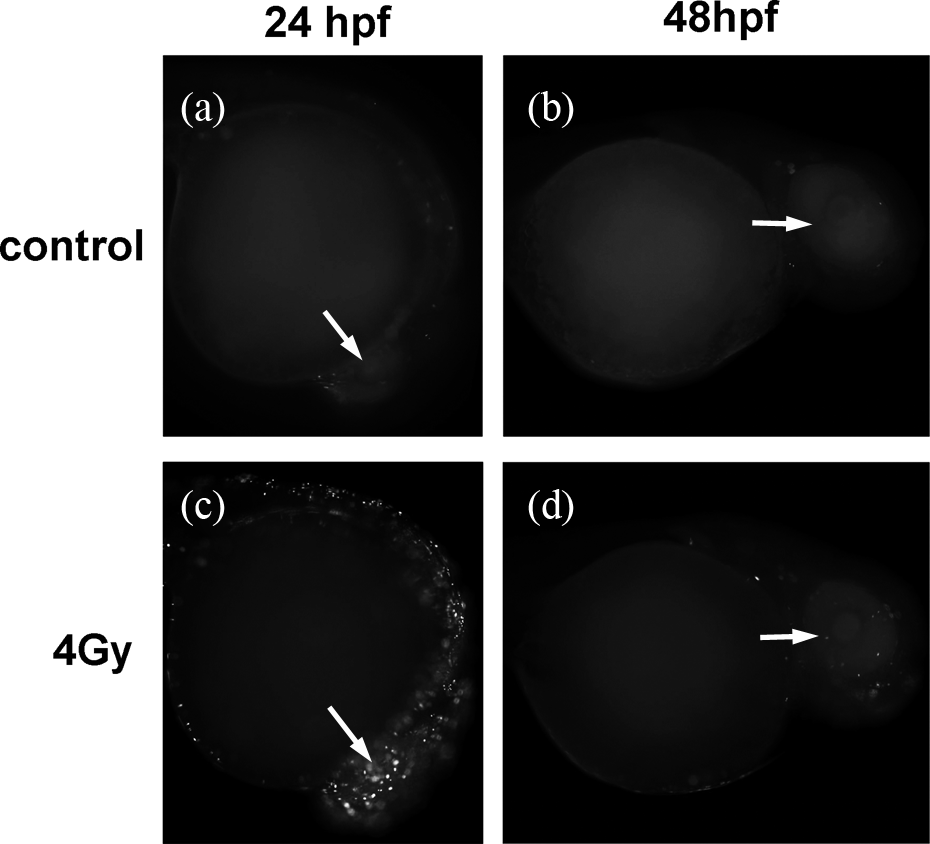
Detection of apoptosis in live zebrafish embryos using the TUNEL assay. TUNEL labeling was emitted from cells undergoing cell apoptosis indicated by a white fluorescence signal on a black background, shown for the eye (white arrow) for (a) TUNEL labeling in the control group at 24 hpf, (b) TUNEL labeling in the 4 Gy-irradiated group at 24 hpf, (c) TUNEL labeling in the control group at 48 hpf, and (d) TUNEL labeling in the 4 Gy-irradiated group at 48 hpf. TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end-labeling; hpf: hours post-fertilization.
Antioxidant enzyme activities analysis
To investigate the condition of the antioxidative system in the zebrafish embryos and larvae after irradiation, SOD and CAT activity was measured. As shown in Figure 4(a), x-ray radiation significantly increased with 2 and 4 Gy irradiation compared with the control (Tukey’s test, p < 0.05) at 24 hpf. Significant increases of SOD activities in 1 and 2 Gy-irradiated groups and significant decrease in 8 Gy-irradiated group (Tukey’s test, p < 0.05) was observed at 48 hpf. By 72 hpf, the activity of SOD significantly increased in all irradiated groups compared with the control group (Tukey’s test, p < 0.05). At 96 hpf, significant increase was still observed in 4 Gy-irradiated group but a significant decrease in 8 Gy-irradiated group (Tukey’s test, p < 0.05; Figure 4(a)).

Ionizing radiation-induced changes in the activities of SOD (a) and CAT (b) in zebrafish embryos and larvae at 24, 48, 72, and 96 hpf. Each value is expressed as the mean ± SEM. *p < 0.05, N = 3: significant differences between the control and irradiated groups were determined using one-way ANOVA.SOD: superoxide dismutase; CAT: catalase; ANOVA: analysis of variance; hpf: hours post-fertilization.
In comparison with the control group, significant increase of CAT activities was only observed in 4 Gy-irradiated group at 24 hpf and significant decrease was only observed in 8 Gy-irradiated group at 48 hpf (Tukey’s test, p < 0.05). By 72 hpf, the activity of SOD only significantly increased in 2 Gy-irradiated group compared with the control group (Tukey’s test, p < 0.05). Similar to the activities of SOD, a significant decrease of CAT activities was observed in 8 Gy-irradiated group (Tukey’s test, p < 0.05) at 96 hpf (Figure 4(b)).
Histological and morphological analysis
The concentric cellular layers, including the ganglion cell layer, inner nuclear cell layer (INL), inner plexiform layer, optic nerve, outer nuclear layer, outer plexiform layer, outer segments of photoreceptors, and retinal pigment epithelium could be clearly distinguished both in the control and irradiated groups at 144 hpf. Compared with the control group, the layering of the retina after irradiation was not significantly disrupted. However, a heterogeneous distribution of the cells was observed in INL after 8 Gy irradiation. Similarly, no distinct changes could be detected in the lens (Figure 5).

Effects of ionizing radiation on the morphology of the developing eye in zebrafish larvae at 144 hpf detected using H&E staining. (a) Control group, (b) 1 Gy-irradiated group, (c) 2 Gy-irradiated group, (d) 4 Gy-irradiated group, and (e) 8 Gy-irradiated group. GCL: ganglion cell layer; IPL: inner plexiform layer; INL: inner nuclear cell layer; OPL: outer plexiform layer; ON: optic nerve; ONL: outer nuclear layer; OS: outer segments of photoreceptors; RPE: retinal pigment epithelium; H&E: hematoxylin and eosin; hpf: hours post-fertilization.
The mean diameter of whole eyes in the control group at 144 hpf was 306.87 ± 9.83 µm (Appendix 1), and significant differences were detected among the groups (ANOVA, F 4, 70 = 4.436, p = 0.003). The diameter of the whole eye in the 8 Gy-irradiated group was significantly reduced compared with the other groups (Tukey’s test, p < 0.05), whereas the differences between the other pairwise groups were not significant. The mean diameter of the lens in the control group at 144 hpf was 99.40 ± 3.66 µm (Appendix 1), and there were no significant differences among the groups (ANOVA, F 4, 70 = 0.277, p = 0.070) (Figure 6).

Ionizing radiation-induced changes in the diameters of whole eyes and lenses. *p < 0.05, N = 15: significant differences between the control and irradiated groups were determined using one-way ANOVA. ANOVA: analysis of variance.
Discussion
When organisms are exposed to ionizing radiation, the ray energy is absorbed by small molecules, primarily water surrounding the biomacromolecules, which can produce a large number of ROS. High levels of free radicals (such as
In our study, a significant increase in ROS content within the eyes was detected at both 24 and 48 hpf suggesting that x-ray radiation can induce marked oxidative stress in zebrafish eyes. However, there was no significant increase in ROS content induced by irradiation at 72 hpf, indicating that oxidative stress can diminish with time. This is because mechanisms have evolved to prevent ionizing radiation inducing oxidative stress by compartmentation, antioxidant compounds capable of forming stable free radicals, and the evolution of enzyme systems, which can diminish intracellular ROS content. Many studies have shown that environmental agents can induce toxic effects on enzyme activity and this effect is dose-dependent. 30 Environmental agents at low doses can cause increases in the activities of SOD and CAT, which can eliminate the increase in ROS concentration and maintain ROS production at a stable level in cells and tissues. 31 However, environmental agents at high doses can induce a decrease in the activities of SOD and CAT, and consequently the ROS concentration increases significantly, exceeding the antioxidative capacity of the cellular antioxidants. 3,32 The measurement of ROS generation showed that the ROS content increased in a dose-dependent manner. One gray irradiation induced a significant increase in ROS content at 24 hpf; however, this increase in ROS diminished at 48 hpf when the SOD activities significantly increased in 1 Gy-irradiated groups, suggesting that the increase in ROS content of 1 Gy-irradiated group was eliminated by antioxidant enzyme at 48 hpf. The other irradiation doses induced a significant increase in ROS content at both 24 and 48 hpf, but did not at 72 hpf when the SOD activities significantly increased in all of the irradiated groups and CAT activities significantly increased in 2 Gy-irradiated group, indicating that the increases in ROS content of 2, 4, and 8 Gy-irradiated groups were eliminated by antioxidant enzyme at 72 hpf. It is notable that only 8 Gy irradiation induced significant decrease in SOD and CAT activities and a significant increase in ROS content at 48 hpf, suggesting that x-ray radiation at high dose can induce a decrease in the activities of SOD and CAT, and then the ROS content increases significantly, exceeding the antioxidative capacity of the cellular antioxidants. In general, the ROS generated by lower dose (1 Gy) irradiation were reduced more quickly by cellular antioxidants than those generated by higher dose (2, 4, and 8 Gy) irradiation.
When the production of ROS exceeded the antioxidant ability of the organism, oxidative stress and even tissue damage can be induced. 33 A significant increase in apoptotic signals within zebrafish eyes was detected at 24 hpf when the ROS content also increased significantly in the irradiated groups. The number of apoptotic cells increased with increasing ROS content and was dose dependent indicating that at 24 hpf a large number of ROS was generated by x-ray radiation at all doses. Therefore, oxidative stress was induced and subsequently caused significant cell apoptosis within the eyes. At 48 hpf, only 4 and 8 Gy irradiation induced a significant increase in apoptotic signals within the eyes, which indicated that the toxic effect induced by oxidative stress can be relieved by antioxidative mechanisms, but this is dose dependent. The higher the dose, the longer the cell apoptosis induced by ROS was sustained. At 72 hpf, the significant increases in both ROS content and cell apoptosis disappeared in the irradiated groups, which further proved that cell apoptosis within the eyes after x-ray radiation was induced by excess ROS. It is worth noting that AO staining and TUNEL assays showed a consistent increase in apoptotic signals at 24 hpf. However, a significant increase in apoptotic signals in the 4 Gy irradiation group was detected by AO staining but not by TUNEL assay because the latter is more specific. The TUNEL assay does not detect all forms of double-strand DNA breaks (such as blunt-ended breaks). Even so, TUNEL labeling is still regarded as a standard marker of cell apoptosis. 14
A number of complications induced by radiation have been reported in mammals, including humans. Radiation may cause retinal and optic atrophy, lens opacification, microophthalmia, and microcephaly. 14 Feared optic complications such as cataract formation, retinal degeneration or atrophy, blindness, and microcephaly have been reported after negligent or therapeutic exposure to radiation. 34 –40
In this article, although significant cell apoptosis was detected within eyes before 48 hpf, we did not find obvious optic complications, such as retinal atrophy and lens opacification induced by x-ray radiation at different doses, with the exception of a heterogeneous distribution of the cells in INL and a significant decrease in the diameter of whole eyes in the 8 Gy-irradiated group. Our results are not consistent with those of Geiger who demonstrated that 10 Gy γ-ray irradiation caused marked abnormal eye development, including retinal atrophy with the loss of distinguished cellular layers, lens opacification, and microophthalmia. 14 Irradiation dose is one of the factors that result in these inconsistencies. However, a more important reason is that the irradiated developmental stage is different. The resistance of embryos to radiation increased substantially with embryonic age. In Geiger’s study, zebrafish embryos were irradiated at 4 hpf which is the blastula stage followed by the gastrula stage. The gastrula stage is the first definable point at which organs begin to develop and is generally described as occurring at about 5 to 10 hpf. 25 Geiger’s results support the suggestion that the susceptibility of embryos to the toxic effects of radiation appears to reduce after the early gastrula stage (5–7 hpf), which indicate that events occurring at or before this stage are particularly radiosensitive. 14,41 In our study, embryos were irradiated at 8 hpf, which is the later gastrula stage. The toxic effects induced by ionizing radiation at this stage seemed to be limited. The x-ray radiation-related DNA damage including single-strand breaks, can be repaired by the cellular DNA repair system, such as poly (adenosine diphosphate-ribose) polymerase, x-ray cross-complementing group 1 protein, and DNA ligase III. 42 –44 Moreover, the inner retinal neurons have regenerative capacity following damage. 6 Therefore, although significant cell apoptosis was observed, morphological changes were barely detected.
Although the cellular layering of the retina were not obviously affected by x-ray radiation, the statistical analysis showed that there was a significant decrease in the diameters of whole eyes in the 8 Gy-irradiated group at 144 hpf, indicating that the risk of microophthalmia induced by high dose irradiation requires further investigation. We measured the diameter of the lens simultaneously in the control and irradiated groups but no significant differences were detected. At 24 hpf, apoptotic cells were distributed in both the lens and retina, but at 48 hpf, they were almost all distributed in the retina. In addition, INL present a heterogeneous distribution of the cells after 8 Gy irradiation. Therefore, we assumed that 8 Gy x-ray irradiation can induce cell apoptosis and even cell death, which may cause the thinning of the retina cellular layer and then led to smaller whole eyes. However, more experiments are needed to determine the molecular mechanism of microophthalmia induced by ionizing radiation.
In conclusion, x-ray radiation at doses of 1, 2, 4, and 8 Gy at 8 hpf induced a significant increase in cell apoptosis within zebrafish eyes at 24 hpf; however, only 4 and 8 Gy irradiation induced significant cell apoptosis at 48 hpf. The number of apoptotic cells increased with increasing ROS content, indicating that ROS generated by x-ray radiation is a main factor to induce significant apoptosis in zebrafish eyes during development. However, the role of DNA damage (DNA double-strand breaks) in the induction of apoptosis should not be excluded. 45 Both cell apoptosis and ROS generation were dose-dependent. The increase of ROS concentration can be eliminated by the increasing activities of antioxidant enzymes system, especially SOD. The higher the dose, the longer the cell apoptosis induced by ROS was sustained. Although significant cell apoptosis was detected within 48 hpf, there was no marked changes in the layering of the retina observed at 144 hpf, but a heterogeneous distribution of the cells in INL and a significant decrease in the diameters of whole eyes were observed after 8 Gy irradiation. X-Ray radiation at the later stage of gastrulation may not cause distinct optic complications based on the pathological findings, but high dose irradiation may still increase the risk of microophthalmia.
Footnotes
Acknowledgment
The authors are grateful to the professional editing service “International Science Editing” for improving writing and language style.
Authors’ Note
The authors RZ and JS contributed equally to this work and should be considered co-first authors.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by grants from the National Basic Research Program of China [2010CB834202]; the National Natural Science Foundation of China [10835011]; and the Scientific Technology Research Projects of Gansu Province [0702NKDA045, 0806RJYA020].
