Abstract
1,3-dichloro-2-propanol is a food-borne contaminant reported to cause liver injury. In this study, we evaluated the protective influence of caffeic acid on 1,3-dichloro-2-propanol-induced hepatotoxicity in rats. Rats were randomized into five groups (A–E). Rats received distilled water or caffeic acid (10 or 20 mg/kg body weight) for 7 days. In addition, rats were challenged with 1,3-dichloro-2-propanol on day 7. Caffeic acid prevented 1,3-dichloro-2-propanol-mediated alterations in alkaline phosphatase, alanine and aspartate aminotransferases, albumin and total bilirubin in the serum of rats. Furthermore, caffeic acid lowered superoxide ion, hydrogen peroxide and cytochrome P2E1 while increasing the activities of superoxide dismutase, catalase and glutathione S-transferase in the liver of 1,3-dichloro-2-propanol-treated rats. Caffeic acid raised the levels of nuclear erythroid-related factor 2 (Nrf-2), protein kinase A and phosphoinositide 3-kinase. Caffeic acid pretreatment annulled 1,3-dichloro-2-propanol-mediated alterations in the oxidative stress biomarkers; caspase-3, glutathione, malondialdehyde, protein carbonyl and fragmented DNA, in the liver of rats. Contrastingly, caffeic acid lowered 1,3-dichloro-2-propanol-mediated increase in the levels of nuclear factor-kappa B (NF-κB), tumour necrosis factor-α, interleukin-1β (IL-1β) and IL-6. In addition, caffeic acid preserved the morphological features of 1,3-dichloro-2-propanol-treated rats. Results from this study revealed that caffeic acid protects against 1,3-dichloro-2-propanol-induced hepatotoxicity by enhancing the cytoprotective enzymes through Nrf-2 while lowering inflammation through NF-κB.
Introduction
1,3-Dichloro-2-propanol is a food-borne contaminant that has been detected during the production of foodstuff including infant formula. 1,2 In addition, it is used in the production of epichlorohydrin and epoxy resin. 3 Although, the mechanism of 1,3-dichloro-2-propanol formation under conditions such as food processing, cooking and storage has not been fully elucidated, chloride ions and lipid components are its important precursors. 3,4 Interestingly, the occurrence of this compound in some food products has been reported. 3,5 Indeed, studies have gone further to establish its toxicity. 6,7 Although, the occupational toxicity of 1,3-dichloro-2-propanol is not widely reported, there are reports of its moderate toxicity. 3 Indeed, industrial accident led to fatal hepatitis and necrosis in two middle-aged men who cleaned tank with epichlorohydrin and dichloropropanols. 3
In the liver, cytochrome p450 2E1 (CYP2E1) metabolized 1,3-dichloro-2-propanol to 1,3-dichloroacetone, a cytotoxic compound. 3 1,3-Dichloroacetone depletes the levels of glutathione (GSH), leading to the accumulation of free radicals and reactive oxygen species (ROS). 6 This accounted for its hepatotoxicity, with acute oral LD50 ranging from 110 mg/kg to 400 mg/kg body weight (BW) in rats. 4 These studies further established the involvement of apoptosis, inflammation and oxidative stress in the toxicity of 1,3-dichloro-2-propanol. 1,2,4 Other reported effects include developmental and reproductive toxicity, irritation of the mucous membranes, nausea and vomiting. 3 Indeed, 1,3-dichloro-2-propanol lowered the sperm count in the body and tails of epididymis of rats. 8 It was reported to induce neurological disorder in rats. 7
Caffeic acid is a cinnamic derivative of phenolic acids and is widely distributed in fruits and vegetables. 9 This phenolic acid is the functional component responsible for the antioxidant activity of fruits and dietary vegetables. 10,11 The catechol ring is the pharmacophore responsible for its ability to enhance protective enzymes. 12,13 Furthermore, the redox cycling activity of the catechol is responsible for its antibacterial activity. 14 In addition to these activities, caffeic acid has anticancer, antitumour and antidiabetic activities. 9 Despite the reported capability of caffeic acid to protect against oxidative stress-associated damages, its ability to protect against 1,3-dichloro-2-propanol-induced oxidative damage and hepatotoxicity has not been explored.
In this study, the protective influence of caffeic acid and associated role of redox transcription factors against 1,3-dichloro-2-propanol-induced liver damage was investigated.
Materials and methods
Chemicals and solvents
Ethanol, epinephrine, 5,5′-dithiobis(2-nitrobenzoic acid), hydrogen peroxide (H2O2), 2,4-dinitrochlorobenzene (CDNB) and thiobarbituric acid were procured from Sigma-Aldrich Inc., St. Louis, Missouri, USA. Nuclear erythroid-related factor 2 (Nrf-2) was supplied by Abcam, Cambridge, Massachusetts, USA. All other reagents used were of analytical grade and supplied by Sigma-Aldrich Inc.
Animal groupings and treatments
Twenty-five rats (158.46 ± 3.66 g) obtained from the Animal Holding Unit, Department of Biochemistry, University of Ilorin, Ilorin, Nigeria, were randomized into five groups of five rats each. The rats were kept in metabolic cages, placed in well-ventilated house conditions and supplied with rat chow and water ad libitum for 2 weeks before treatment. Rats in groups A (control) and B orally received the vehicle for caffeic acid (CAS no. 331-39-5; purity ≥98%) administration for 7 days. Rats in groups C, D and E were orally gavaged with 20, 10 and 20 mg/kg (BW) of caffeic acid. In addition, groups B, D and E rats were challenged orally with 110 mg/kg BW 1,3-dichloro-2-propanol (CAS no. 96-23-1; purity 98%) on day 7. The doses of caffeic acid used were based on previous study by Ibitoye and Ajiboye. 9 Rats were euthanized under light diethyl-ether anaesthetization 24 h after last treatment. This study was approved by Nile University of Nigeria Committee on the Use of Laboratory Animals (NUN/BCH/ECULA/019/001). Liver homogenate and serum were prepared as described by Ajiboye et al. 15
Biochemical assays
Protein content of rat liver and serum were estimated as described by Olson and Markwell. 16 Alkaline phosphatase (ALP) activity in the liver of rats was estimated by reading the absorbance (405 nm) of the phenolate formed following the hydrolysis of ρ-nitrophenyl phosphate. 17 Alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin and total bilirubin in the serum of rats were determined as outlined in Randox Laboratories assay kits (Randox Laboratories Ltd., Antrim, UK). Superoxide anion radical was determined spectrophotometrically using nitroblue tetrazolium (NBT). Briefly, •O2 − generated reduced yellow NBT to form blue formazan particles. The absorbance of the blue formazan particles formed is read at 620 nm. 18 H2O2 in the liver of rats was quantified as described in the H2O2 assay kit (Sigma-Aldrich). The activity of superoxide dismutase (SOD) in the liver of rats was measured by the rate of epinephrine autoxidation to adrenochrome. 19 The absorbance of the adrenochrome formed was read at 480 nm. Catalase (CAT) and glutathione S-transferase (GST) in the liver were determined as described by Shangari and O’Brien 20 and Habig et al., 21 respectively. Liver Nrf-2 (Abcam) and nuclear factor-kappa B (NF-κB) were estimated in the liver nuclear fraction while protein kinase B (Akt pSer473, Abcam) and phosphoinositide 3-kinase (PI3K, Abcam) were determined in the cytosol by enzyme-linked immunosorbent assay. Tumour necrosis factor-α (TNF-α), interleukin-6 (IL-6) and IL-8 were determined as outlined in manufacturer’s assay kit manual (Sigma-Aldrich). The activity of caspase-3 was determined as outlined in caspase-3 kit (Sigma-Aldrich). Cytochrome P2E1 activity in the liver of rats was determined as described by Dicker et al. 22 Fragmented DNA was estimated as described by Burton. 23 Reduced GSH and glutathione disulphide were quantified as described by Ellman 24 and Rahman et al., 25 respectively. Extent of lipid peroxidation and protein oxidation was estimated by determining the end products malondialdehyde (MDA) 26 and protein carbonyl, 27 respectively.
Histopathology
Liver of rats was fixed in 10% formalin solution for 48 h then dehydrated using different grades of ethanol, xylene and embedded in paraffin. Section (3–4 μm) of the liver was stained with hematoxylin and eosin stains, and mounted on microscope (TP1020, Leica biosystems, Nussloch GmbH, Germany) for photomicrography as described by Ajiboye. 28
Statistical analysis
Data were expressed as the mean for five rats ± standard error of mean (SEM). Analysis of variance followed by Tukey–Kramer test for differences between means was used to detect any significant difference between the treatment groups using GraphPad Prism 6 for Windows, version 6.01 (GraphPad Software Inc., San Diego, California, USA). Differences were considered statistically significant at p < 0.05.
Results
Caffeic acid halts 1,3-dichloro-2-propanol-induced alterations in hepatocellular marker
The activities of ALP, ALT and AST in the serum of 1,3-dichloro-2-propanol-treated rats increased significantly (p < 0.05) by 3.65, 4.80 and 7.23 folds when compared to the control rats (Table 1). Administration of caffeic acid significantly reversed 1,3-dichloro-2-propanol-mediated increase, which compared significantly with the control (p < 0.05). In addition, the decrease in serum albumin with parallel increase in total bilirubin observed for 1,3-dichloro-2-propanol-treated rats was attenuated by caffeic acid (Table 1).
Caffeic acid pretreatment attenuates DCP-mediated alterations in the biomarkers of hepatocellular injury in the serum of rats.a

ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.
aValues are mean ± SEM for five rats. Values with different alphabetical superscripts (b, c, d) are considered significantly different at p < 0.05.
Caffeic acid mopped up ROS by increasing the cytoprotective enzymes through Nrf-2
1,3-Dichloro-2-propanol significantly (p < 0.05) raised the •O2 and H2O2 in the liver of rats when compared to the control rats (Figure 1(a) and (b)). Furthermore, a contrasting decrease in the antioxidant proteins SOD, CAT and GST was observed (Figure 1(c) to (e)). These significant (p < 0.05) alterations caused by 1,3-dichloro-2-propanol-mediated oxidative stress were reversed by caffeic acid in a dose dependent manner dose with the 20 mg/kg BW dose comparing significantly with the control rats (Figure 1). In addition, caffeic acid pretreatment decreases the activity of CYP2E1 in the liver of rats to a level comparable with the control rats (Figure 1(f)).

Caffeic acid annulled DCP-mediated increase in ROS by enhancing the cytoprotective enzymes. (a) Superoxide ion, (b) H2O2, (c) SOD, (d) CAT, (e) GST and (f) CYP2E1 in the liver of DCP-treated rats pretreated with caffeic acid. Values are mean ± SEM for five rats and are statistically significant at p < 0.05. ROS: reactive oxygen species. H2O2: hydrogen peroxide; SOD: superoxide dismutase; CAT: catalase; GST: glutathione S-transferase; CYP2E1: cytochrome P450 2E1; SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.
Furthermore, administration of caffeic acid to rats raised the levels of Nrf-2 in the liver of rats (Figure 2(a)). Interestingly, Akt and PI3K were equally enhanced when compared to the control rats (Figure 2(b) and (c)). However, Kelch-like ECH-associated protein 1 (Keap1) was significantly lowered by caffeic acid (Figure 2(d))

Caffeic acid enhanced Nrf-2 through upstream kinases (Akt and PI3K) and downregulation of Keap1. Levels of (a) Nrf-2 (b) Akt, (c) PI3K and (d) Keap1 in the liver of DCP-treated rats pretreated with caffeic acid. Values are mean ± SEM for five rats and are statistically significant at p < 0.05. Nrf-2: nuclear erythroid-related factor 2; Akt: protein kinase B; PI3K: phosphoinositide-3 kinase; Keap1: Kelch-like ECH-associated protein 1; SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.
Caffeic acid halts 1,3-dichloro-2-propanol-mediated changes in the biomarkers of apoptosis, lipid peroxidation and protein oxidation
1,3-Dichloro-2-propanol significantly raised the activity of caspase-3 in the liver of rats. Also, the level of fragmented DNA in liver of rats increased significantly when compared to the control rats (Figure 3(a) and (b)). Contrastingly, 1,3-dichloro-2-propanol depleted GSH level (Figure 4(a)) but induced lipid peroxidation and protein oxidation in the liver of rats, as evident from the increase in the levels of MDA and protein carbonyl (Figure 4(b) and (c)). Interestingly, pretreatment of rats with caffeic acid dose dependently annulled the increase and compared significantly (p > 0.05) with the control rats.

Caffeic acid halts DCP-induced apoptosis. (a) Caspase-3 and (b) fragmented DNA in the liver of DCP-treated rats pretreated with caffeic acid. Values are mean ± SEM for five rats and are statistically significant at p < 0.05. SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.

Caffeic acid restored GSH and prevents DCP-induced protein and lipid oxidation. Levels of (a) GSH, (b) MDA and (c) protein carbonyl in the liver of DCP-treated rats pretreated with caffeic acid. Values are mean ± SEM for five rats and are statistically significant at p < 0.05. GSH: glutathione; MDA: malondialdehyde; SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.
Caffeic acid reversed 1,3-dichloro-2-propanol-mediated increase in biomarkers of inflammation
Single oral administration of 1,3-dichloro-2-propanol raised the levels of pro-inflammatory cytokines, TNF-α, IL-1β and IL-6, in the serum of rats (Figure 5(a) to (c)). Furthermore, the transcription factor, NF-κB, responsible for regulating inflammation and immune response was raised in 1,3-dichloro-2-propanol-treated rats (Figure 5(d)). Daily administration of caffeic acid for 7 days dose dependently annulled 1,3-dichloro-2-propanol-induced increase in these inflammation biomarkers (Figure 5). Interestingly, caffeic acid (20 mg/kg BW) compared significantly (p > 0.05) with the control rats (Figure 5).
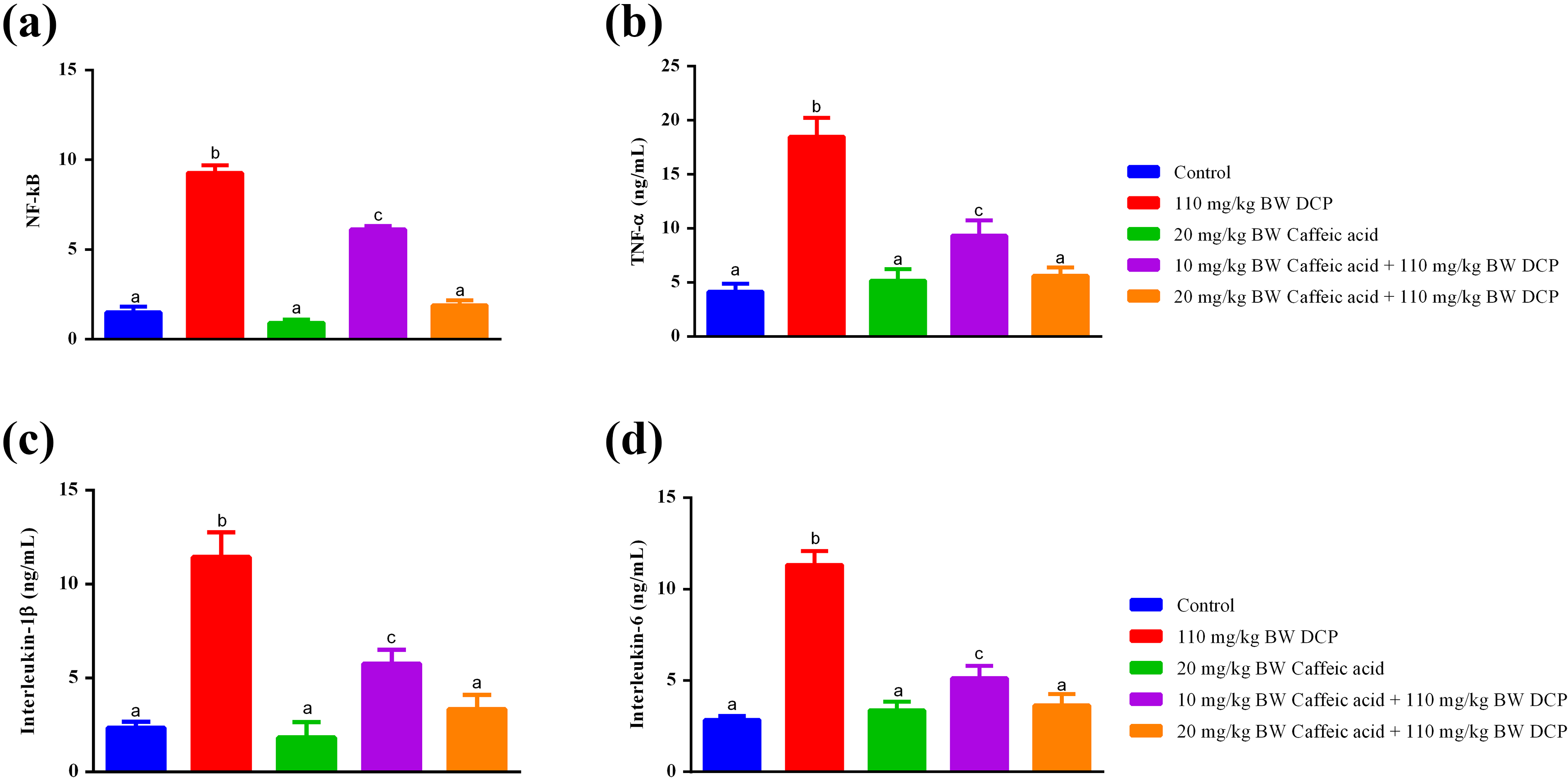
Caffeic acid halts DCP-induced inflammation by lowering the pro-inflammatory cytokines through NF-κB. (a) NF-κB, (b) TNF-α, (c) IL-1β and (d) IL-6 in the liver of DCP-treated rats pretreated with caffeic acid. Values are mean ± SEM for five rats and are statistically significant at p < 0.05. NF-κB: nuclear factor-kappa B; TNF-α: tumour necrosis factor-α; IL: interleukin; SEM: standard error of mean; DCP: 1,3-dichloro-2-propanol.
Caffeic acid improved the morphological features of the liver of 1,3-dichloro-2-propanol-treated rats
1,3-Dichloro-2-propanol administration triggered morphological alterations in the liver characterized by necrosis, haemorrhage and inflammatory cell infiltration (Figure 6). These alterations were reversed by caffeic acid in a dose dependent manner. In fact, pretreatment of rats with caffeic acid at the dose of 20 mg/kg BW improved the morphological features to similar to the control (Figure 6).

Caffeic acid preserved the morphological features of the liver of DCP-treated rats. Histoarchitecture of the liver of DCP-treated rats pretreated with caffeic acid (×300): (a) control rats, (b) DCP (110 mg/kg BW), (c) caffeic acid (20 mg/kg BW), (d) caffeic acid (10 mg/kg BW) + DCP (110 mg/kg BW), and (e) caffeic acid (20 mg/kg BW) + DCP (110 mg/kg BW). White arrows show normal hepatocyte; red arrows show dilated blood vessels and inflammatory cells; blue arrows show mild pyknotic nuclei; and black arrow shows necrotic cells. BW: body weight; DCP: 1,3-dichloro-2-propanol.
Discussion
Flavonoids, phenolic acids and polyphenols are the functional components responsible for the health benefits of functional foods. 29 –31 Indeed, recent studies demonstrated the impact of caffeic acid, a cinnamic derivative of phenolic acid, on oxidative stress. 9 Interestingly, oxidative stress is critical in 1,3-dichloro-2-propanol-induced liver damage. 1,4,6 This study demonstrated that caffeic acid lowered the activity of CYP2E1 and enhanced the activities of cytoprotective enzymes through longevity transcription factor, Nrf-2.
ALP, ALT, AST, albumin and bilirubin, important biomarkers for liver functional capacity, 28,32 are found to be elevated in the serum of 1,3-dicloro-2-propanol-treated rats. 1,4,6 Consistently, these biomarkers were elevated in the serum of 1,3-dicloro-2-propanol-treated rats, thus suggesting hepatic injury in rats. The caffeic acid-mediated reversal of 1,3-dichloro-2-propanol-induced alterations in these biomarkers shows hepatoprotection. Indeed, this is consistent with studies that have demonstrated the protective effects of caffeic acid using these biomarkers in hepatic injury model. 33 Interestingly, studies have reported that caffeic acid is one of the hepatoprotective compounds in Phyllanthus muellarianus leaves. 34
Antioxidant proteins (SOD, CAT and GSH) are important in counteracting the deleterious effects of ROS on cellular macromolecules. 35 –37 Consistent with previous studies, 1,4,6 1,3-dichloro-2-propanol lowered the activities of SOD and CAT in the liver of rats. Contrastingly, the levels of ·O2 − and H2O2 were raised in the liver of rats suggesting overwhelmed antioxidant enzymes. The ROS accumulation could be due to the decrease in antioxidant enzyme activities and may lead to oxidative degradation of cellular macromolecule. Interestingly, studies have demonstrated oxidative attack on carbohydrates, proteins, lipids and DNA. 1,6 Studies have reported the capability of caffeic acid to enhance antioxidant enzymes in different animal models of hepatotoxicity. 33,38 In this study, caffeic acid attenuated 1,3-dichloro-2-propanol-mediated decrease in SOD and CAT, and increase in ·O2 − and H2O2. Furthermore, the decreased activities of GST in the liver of 1,3-dichloro-2-propanol-treated rats were reversed by caffeic acid. Indeed, GSH depletion was reported to be important for 1,3-dichloro-2-propanol hepatotoxicity. 4 Also, CYP2E1 responsible for the activation of 1,3-dichloro-2-propanol was significantly lowered by caffeic acid. The effects on the cytoprotective enzymes suggest the modulatory role of caffeic acid.
Catechol and Michaelis acceptor groups of plant chemicals including phenolic acids are important for chemoprevention. 13,15,39,40 These chemicals enhance the expression of Nrf-2 through Keap1 degradation pathway 15 or upstream kinases (Akt and PI3K) leading to increase in the expression and activities of cytoprotective enzymes. 41 –44 Lee et al. 6 reported transient increase in expression of Nrf-2, which further decreased. In this study, 1,3-dichloro-2-propanol lowered the level of Nrf-2 in the liver of rats. However, no change was observed in the levels of Keap1, Akt and PI3K. Caffeic acid mediated increase in Nrf-2, Akt and PI3K, and decrease in Keap1 could be responsible for the observed increase in SOD, CAT and GST. Indeed, caffeic acid was reported to enhance the expression of Nrf-2, 40 suggesting that the elevation observed in this study led to the observed increase in cytoprotective enzymes and attenuation of 1,3-dichloro-2-propanol-mediated increase in ·O2 − and H2O2.
GSH depletion is an important step in 1,3-dichloro-2-propanol hepatotoxicity. 3,6 Consistently, administration of 1,3-dichloro-2-propanol lowered the levels of GSH in the liver of rats. The parallel increase in oxidative stress biomarkers, MDA and protein carbonyl observed in this study, is consistent with previous studies. 4 These events signal oxidative attacks on cellular macromolecules such as membrane lipids and proteins, which could trigger membrane disorganization, functional loss and protein and DNA base modification. 45 Caffeic acid mediated increase in Nrf-2 and cytoprotective enzymes could have led to the attenuation of 1,3-dichloro-2-propanol-mediated depletion of GSH, and increase in MDA and protein carbonyl in the liver of rats. Indeed, it might have translated to detoxification of ROS, the culprit of oxidative attack on cellular macromolecules.
Apoptosis and inflammation are also reported in 1,3-dichloro-2-propanol-induced hepatotoxicity. 1,4 Consistent with these studies, the activity of caspase-3 increased significantly in the liver of rats. Furthermore, the levels of TNFα, IL-1β and IL-6 were raised. Caffeic acid reversed the 1,3-dichloro-2-propanol-induced alterations of apoptotic and inflammatory biomarkers. Interestingly, caffeic acid lowered these biomarkers in different disease model. 9 In addition, 1,3-dichloro-2-propanol-mediated increase in the fragmented DNA in the liver of rats was reversed, confirming the anti-apoptotic and anti-inflammatory activities of caffeic acid. This could be due to the enhanced antioxidant defence and ROS detoxification as observed in this study.
Consistent with previous studies, 1,6 we observed pyknotic nuclei, inflammatory cell infiltration and haemorrhages in the liver of 1,3-dicloro-2-propanol-treated rats, which indicate necrosis. Pretreatment of rats with caffeic acid prevented these morphological changes suggesting the modulatory effects. This could have resulted from the elevated antioxidant and cytoprotective enzymes that might have prevented oxidative assault on the liver cells.
In conclusion, we have shown that caffeic acid mediates its hepatoprotection by enhancing Nrf-2 in the liver of rats through Akt and PI3K pathway, leading to increased antioxidant and cytoprotective enzymes. Furthermore, caffeic acid lowered inflammatory transcription factor (NF-κB) and the pro-inflammatory cytokines. This prevented 1,3-dichloro-2-propanol-induced inflammation, ROS and oxidative stress (Figure 7). The consumption of fruits, foods and vegetables rich in this phenolic acid is encouraged to prevent liver toxicity.

Caffeic acid prevents DCP-induced hepatic injury by upregulating Nrf-2 through upstream kinases (Akt and PI3K) and Keap1 pathways leading to enhanced cytoprotective proteins. The cytoprotective enzymes annulled DCP-induced oxidation of cellular macromolecules. Furthermore, the cytoprotective enzymes lowered the levels of ROS leading to downregulation of NF-κB and inflammation. ROS: reactive oxygen species; Nrf-2: nuclear erythroid-related factor 2; Akt: protein kinase B; PI3K: phosphoinositide-3 kinase; Keap1: Kelch-like ECH-associated protein 1; NF-κB: nuclear factor-kappa B; DCP: 1,3-dichloro-2-propanol.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work in Antioxidants, Redox Biology and Toxicology Research Laboratory was supported in part by Academic Incentive Grants (NUN/RGC/007-010) awarded to TO Ajiboye by Nile University of Nigeria (NUN).
