Abstract
Oral lichen planus is a chronic non-infectious mucosal inflammatory disease caused by an imbalance between reactive oxygen species homeostasis and antioxidant defense systems. Notably, excessive oxidative stress products result in related autoimmune reactions. Further, it activates signaling pathways related to the development of oral lichen planus, as evidenced by the detection of damage to deoxyribonucleic acid, protein, and lipid. Thus, the mechanisms of reactive oxygen species-mediated oxidative stress in the pathogenesis of oral lichen planus are numerous and complex. In this review, we first introduce oxidative stress and oxidative products. Then, we summarize the role and possible mechanisms of reactive oxygen species-mediated oxidative stress in the pathogenesis of oral lichen planus and present a clinical correlation between oxidative stress and oral lichen planus. Finally, we discuss the current challenges and future perspectives.
Introduction
Under normal conditions, the human body produces certain reactive oxygen species (ROS). The physiological concentration of ROS regulates physiological activities, such as intracellular homeostasis, signal transduction, and apoptosis, which plays a critical role in the elimination of bacteria and pathogens, host defense, and immune regulation. 1
In healthy cells, intracellular levels of ROS are balanced with intracellular biochemical antioxidants to prevent cell damage. When this critical balance is broken, oxidative stress(OS) occurs. 2 OS refers to the decreased generation of antioxidants and weakened antagonistic effect on ROS, or the increased production of ROS under certain external stimuli or case conditions, which exceeds the compensatory ability of antioxidants in the body, resulting in relatively excessive ROS. When an imbalance between oxidants and antioxidants in the body occurs, the cell cannot eliminate excessive reactive oxygen. The body is in a state of high OS, which would cause protein, lipid, and deoxyribonucleic acid(DNA) damage, thus leading to cell dysfunction in the body. The role of ROS in the pathogenesis of various diseases has been confirmed. 3
OLP is a chronic inflammatory disease with an incidence of 0.5%–2% in adults. 4 At present, it is currently believed that the pathogenesis of OLP is related to immune, genetic, and mental factors. 5 Some scholars have found that OS is involved in the occurrence and development of OLP, 6 which provides a new treatment direction. Related studies have shown that OS is present in patients with OLP and interacts with inflammation and the immune system. ROS and oxidative damage, together with increased lipid peroxidation and imbalance in the antioxidant defense system, are closely related to the occurrence and development of OLP. 7 A recent meta-analysis detailed oxidative stress markers but did not elucidate the possible mechanism of ROS in the pathogenesis of OLP. 8 In this review, we mainly focus on the role and mechanism of ROS in the immune and inflammatory pathogenesis of OLP.
OS and oxidative products
OS is a concept in redox biology and medicine; it is defined as the imbalance between oxidants and antioxidants, causing a disturbance in redox signaling and control and molecular damage. 9 The research fields of OS include chemistry, biochemistry, cell biology, physiology, pathophysiology, medicine, health, and disease research. 10 OS produces two main oxidation products: ROS and reactive nitrogen species (RNS).
The normal level of ROS has critical physiological functions. ROS can regulate growth, apoptosis, and signal transduction at the cellular level. Meanwhile, ROS can regulate blood pressure and cognitive and immune functions. 1 Medium and high concentrations of ROS induce apoptosis or even necrosis through OS. With the development of research on free radical biology, researchers have gradually realized that ROS can regulate the apoptosis and proliferation of specific tumor cells in both directions and discovered the internal correlation between free radicals and cell signal transduction. 11 ROS can produce cytotoxic damage to metabolic enzymes after reacting with superoxide and peroxynitrite, 12 thus causing oxidative damage to the body.
Possible mechanism of ROS-mediated OS in OLP
OS and immunity
The etiology and pathogenesis of OLP have not been fully elucidated. However, previous studies have found that the pathogenesis of OLP may be related to genetic, immune, and infectious factors. 13 OLP is generally believed as a chronic inflammatory disease characterized by T cell-mediated autoimmune response. Its typical pathological manifestation is many lymphocytes in the lamina propria with dense zonal infiltration. In the process of OLP development, lymphocytes and cytokines constitute a complex immune response network, resulting in chronic destructive inflammation of local tissues. Dendritic cells (DCs) activation initiates cell/tissue damage and inflammatory response. DCs carry antigens to lymph nodes, produce cytokines and chemokines, and activate different lymphocytes, leading to cytotoxic responses in the basal epidermal cell layer. 14 As for triggering an immune response, OLP is considered to be caused by the body’s antigen. Scholars have made research achievements on the role of ROS in the pathogenesis of OLP. Sources of the oral OS include endogenous and exogenous factors, such as inflammation, food, smoking, and dental materials.15–16 In OS, excessive ROS was produced by the body’s cells to react with lipids and proteins in cells, change the antigen spectrum of proteins, and enhance antigenicity, leading to autoimmune diseases caused by oxidative modification of their proteins. 17 The pathogenesis of OLP includes antigen-specific and nonspecific mechanisms. 18
Antigen-specific mechanism
Antigen-specific mechanisms include the antigen presentation of basal keratinocytes and the killing of antigen-specific keratinocytes by CD8+ cytotoxic T cells. In patients with OLP, lipid peroxidation increases, and high concentrations of malondialdehyde (MDA) are involved in signal transmission and apoptosis by affecting the connection between Bcl-2 and Bax. Moreover, ROS are involved in regulating mitochondria-dependent on CD8+ lymphocytes by affecting the activity of NF-κB.
6
Therefore, ROS may be necessary for the pathogenesis of OLP and its complications, and its harmful effects result from its influence on the underlying mechanism of cell signal transmission and transduction, leading to keratinocyte dysfunction and apoptosis.
19
Otherwise, the subepithelial infiltration of T lymphocytes causes an increase in cytokines, which stimulate keratinocytes to produce ROS. ROS induce lipid peroxidation, and lipid peroxides cause severe damage to cell integrity, affecting membrane structure, fluidity, crosslinking, and function, leading to apoptosis. Keratinocyte apoptosis and epithelial basal cell liquefaction are significant pathological features of OLP, suggesting that ROS may play a vital role in the development of OLP (Figure 1).
20
Possible mechanism of ROS-mediated OS in OLP. The pathogenesis of OLP in immunity includes antigen-specific and nonspecific mechanisms. Meanwhile, the products of tissue damage caused by ROS are effective damp-associated molecular patterns (DAMPs), which can trigger the innate immune response by combining multiple PRRs. DAMPs released under tissue injury can trigger inflammatory responses by activating immune defense mechanisms.
Antigen-nonspecific mechanism
Antigen-nonspecific mechanisms include mast cell degranulation and matrix metalloproteinase (MMP) activation in OLP lesions. ROS is involved in the nonspecific pathogenesis of OLP, such as p53, Bcl-2 family protein, TNF-α, Fas/FasL pathway, granzyme B, and MMP-9, which are related to apoptosis and lymphocyte infiltration of OLP lesions (Figure 1). 21
p53 can directly regulate approximately 500 target genes, thus controlling a series of processes, such as cell cycle arrest, cell aging, DNA repair, metabolic adaptation, and cell death. 20 Under mild stress, it may participate in antioxidant reactions to reduce ROS levels or participate in the DNA damage repair process while inducing the short cell cycle to stop to ensure cell survival and repair tissue damage. 22 It can inhibit the interaction between autophagy and Bcl-2 family members from promoting cell apoptosis and cell necrosis by causing changes in mitochondrial permeability. 23 Bcl-2 anti-apoptotic proteins maintain mitochondrial outer membrane integrity by inhibiting pro-apoptotic proteins, and the fate of cells depends on the balance of Bcl-2 family proteins. Cell signals can regulate the expression of pro-apoptotic or anti-apoptotic proteins so that the balance is tilted toward survival or apoptosis. When the balance is favorable to death, pro-apoptotic protein oligomers on the mitochondrial outer membrane cause mitochondrial outer membrane permeability and cytochrome c release. This phenomenon triggers a cascade of apoptotic proteases that cleave downstream substrates, leading to cell death. 24 Therefore, p53 can not only downregulate apoptosis inhibitors, such as PIG-3, Bcl-2, Apaf-1 but also directly stimulate mitochondria to release ROS and induce apoptosis. 21
High intracellular ROS concentrations cause cell death by activating cell death pathways. ROS are related to Fas-dependent pathways and pro-apoptotic signals of TNF-α. Intracellular tumor necrosis factor receptor-1 (TNFR-1) and FasR cause cell death by promoting intracellular ROS production. The TNF-α cytokine produced by activated macrophages inhibits osteoblasts and stimulates osteoclasts. After starting the TNFR-1 signaling pathway by TNFR-1-associated death domain protein (TRADD) antibody and TNF-α-receptor-related factor 2, TNF-α binds to TNFR-1, and TRADD further activates the Fas-associated death domain protein (FADD) antibody and caspase-8. Like TNFR-1, FasR recruits FADD to activate caspase-8, inducing apoptosis. 25
Granzyme B (GB) triggers ROS-dependent apoptosis in mitosis by activating a biochemical pathway centered on the destruction of the respiratory chain. By directly attacking the mitochondria, GB divides NADH’s NDUFV1, NDUFS1, and NDUFS2 subunits, triggering the increase in ROS in target cells. This process results in ROS production in mitotic centers, loss of complex I and III activities, disordered respiratory chain, impaired mitochondrial respiratory function, and loss of mitochondrial ridge connections. 25
Studies have reported increased expression of MMP-1–3 in epithelial OLP cells and increased expression of MMP-9 in OLP inflammatory infiltrating cells. Thus, MMP is considered to play a role in OLP basilar membrane rupture, causing intraepithelial inflammatory cell migration. 26 The interaction of various nonspecific mechanisms aggravates the occurrence and development of OLP.
OS and inflammation
OLP is a chronic inflammatory disease involving the oral mucosa with unknown etiology. The abnormal expressions of inflammation-related cytokines are a specific reflection of immune dysregulation and may play an essential role in the occurrence and development of OLP. 14 Inflammation is an adaptive response to potentially dangerous phenomena in the host and aims to eliminate risk factors, induce tissue repair, and restore tissue homeostasis. 27 When the body is in an OS state, the damage in biomolecules exceeds its repairability, leading to cell death and extracellular matrix decomposition. Necrotic cells and damaged extracellular mediators release various internal and external molecules of cells, which trigger an inflammatory cascade reaction by identifying pattern recognition receptors (PRRs). 28
Moreover, OS conditions can induce lipid peroxidation and protein degeneration to produce oxidation-specific epitopes, which are effective damp-associated molecular patterns (DAMPs). DAMPs can trigger the innate immune response by combining multiple PRRs. 29 DAMPs released under tissue injury can trigger inflammatory responses by activating immune defense mechanisms, especially HMGB-1, TLR, and NLRP-3 receptors; inflammatory bodies; and transcriptional factors (Figure 1). Among them, OS is closely related to the activation of the NF-κB pathway. 30 In chronic inflammatory diseases, tissue repair cannot be carried out promptly; thus, continuous cellular stress amplifies the inflammatory response, further leading to significant changes in tissue function accompanied by the constant disorder of the systemic internal environment. 27
Clinical correlation between OS and OLP
Indicators for assessment of OS
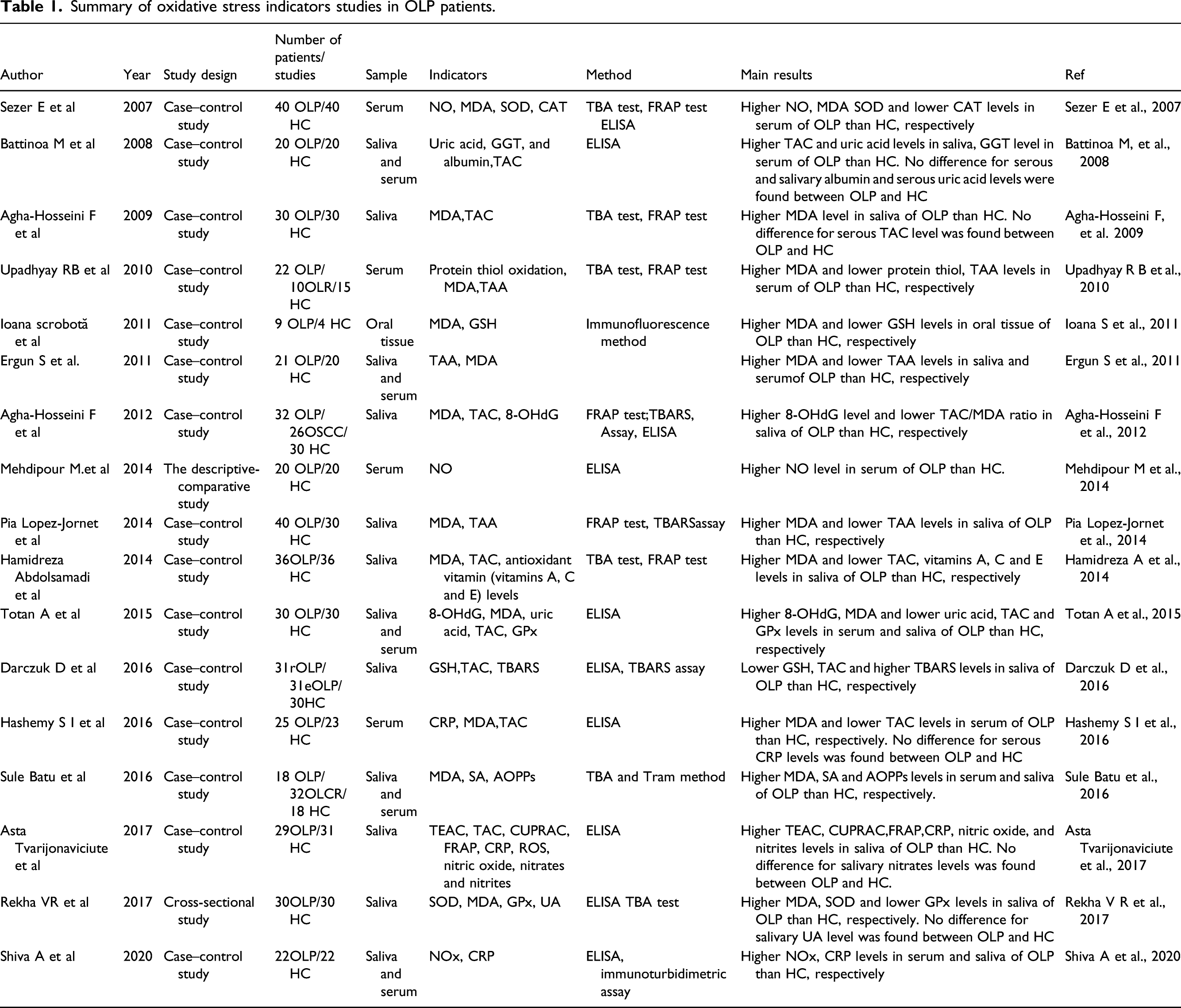
Summary of oxidative stress indicators studies in OLP patients.
Imbalance between oxidation and antioxidation in OLP
Scholars have confirmed the increase of ROS and the imbalance between the oxidant and antioxidant systems in patients with OLP. Tissue homogenate, 43 serum, and saliva are often used as test samples when evaluating OS-related indicators in patients with OLP. They found that the MDA level in patients with OLP was significantly higher than that in the healthy control group. 44 The GSH level was reduced by evaluating the markers of OS MDA and GSH in oral tissue homogenate. 21 Abdolsamadi et al. 45 confirmed that patients with OLP had increased MDA levels and decreased TAC and antioxidant vitamin levels. Totan et al. 26 concluded that patients with OLP had significantly increased serum levels of MDA and 8-OHDG and significantly decreased TAC, GPx, and uric acid levels by detecting OS indicators in serum and saliva.
The role of ROS in EOLP and oral cancer
As a precancerous lesion, EOLP has a specific potential for malignant transformation, seriously affecting the physical and mental health of patients. 46 The risk of EOLP malignant transformation has long been controversial, ranging from 0.4% to 3.7%. 47 Vlkova et al. found increased lipid peroxidation and carbonyl stress markers in patients with oral precancerous lesions. The decreased antioxidant enzyme expression leads to the decline in antioxidant status, which may help to reveal the etiology or pathogenesis of oral precancerous lesions and the transformation mechanism to oral cancer. 48 Agha-Hosseini et al. found that patients with OSCC had significantly higher MDA and 8-OHDG levels and lower TAC levels than the control group. The TAC/MDA ratio of patients with OSCC was significantly lower than that of patients with OLP and the control group. Compared with the control group, patients with OLP had significantly lower TAC/MDA ratio and higher 8-OHDG levels. 49 These results indicated an OS imbalance in patients with OLP and OSCC and an increased risk of cancer in patients with OLP.
In cancer, most of the energy supply of cancer cells is obtained through the increase of glycolysis rate and the oxidation of pyruvate in the mitochondria, unlike that of nontumor cells. Glycolysis replaces oxidative phosphorylation to produce ATP in cancer cells. This metabolism is crucial for cancer cells to adapt to low-oxygen conditions and reduce mitochondrial defects and ROS production.
14
Increased ROS in cancer cells may involve multiple mechanisms. The internal mechanism of intracellular ROS increase may be oncogene activation, tumor suppressor gene inactivation, high metabolism, and mitochondrial dysfunction (Figure 2). ROS can induce Wnt signaling, especially the Wnt/β-catenin pathway. Wnt activates the stability of downstream signal molecules and promotes the accumulation of β-catenin. Some scholars reported that many Wnt ligands could promote the epithelial-mesenchymal transition and cancer stem cell activity in various cancers, leading to the formation and development of tumors. The role of ROS in OLP and oral cancer. The course of OLP is chronic and even has a specific potential for malignant transformation. In OLP patients, steady-state levels of oxidative modifications in DNA can lead to DNA mutations, which can induce the occurrence of oral cancer.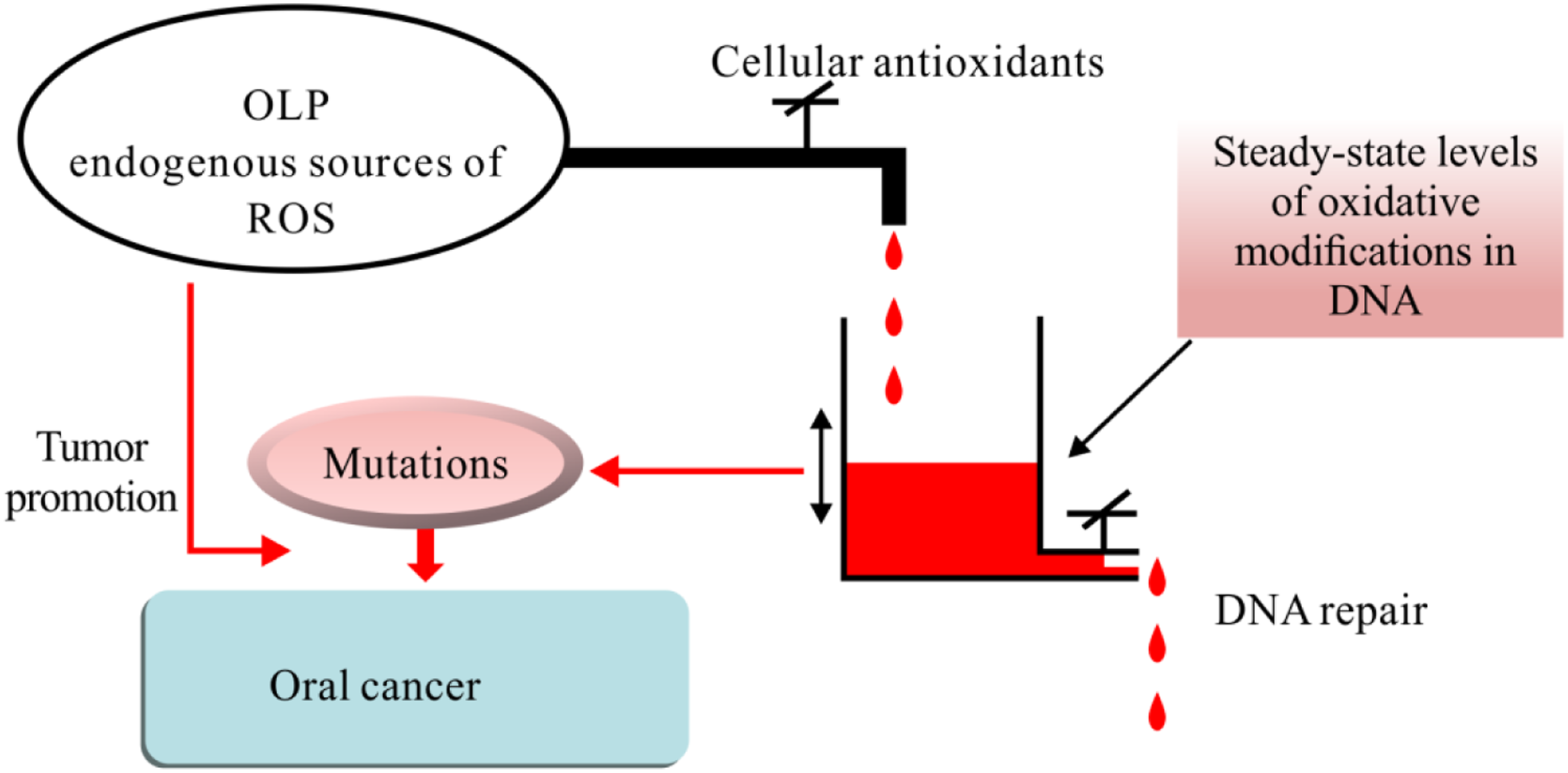
Conclusions
Nonspecific overproduction of ROS can mediate OS, cause-related autoimmune reactions, and activate signaling pathways related to the development of OLP. ROS-mediated OS is particularly critical in cellular metabolism and inflammatory signaling. Thus, it plays a pivotal role in OLP associated with metabolic dysregulation and inflammation. However, the specific mechanism of OS is still unclear and needs further study at the molecular level. Studying ROS signaling and toxic disease status is essential for discovering new therapeutic targets and developing innovative therapeutic strategies. The antioxidant therapy of OLP is expected to become a new adjuvant therapy. In addition, ROS-mediated OS could contribute to oral cancer via general mechanisms of DNA damage and protein modulation. In the future, it is challenging to develop therapies that do not compromise normal cellular functions that rely on ROS or redox pathways while achieving the abatement of OLP processes driven by ROS and redox. We expect the feasibility of combined approaches targeting ROS-mediated OS and metabolism for successful OLP therapy to be an exciting research topic. Future lines of pre-clinical research should focus on improving knowledge to understand better the molecular mechanisms involved in the pathogenesis of OLP and its relationship with ROS-mediated OS.
Footnotes
Acknowledgements
Thanks are due to Dr. Wang for assistance with the proofreading of this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
