Abstract
Serotonin (5-hydroxytrytamine (5-HT)) plays an important role in experimental seizures. Recently, we reported the depletion of 5-HT by parachlorophynylalanine (PCPA) in whole brain to enhance 6-Hz psychomotor seizures in mice. In the present work, we investigated the effect of 5-HT depletion in cortex and hippocampus, brain regions relevant for epilepsy, on behavioral and ultra-structural changes following 6-Hz psychomotor seizures in mice. In addition, we studied the effect of sodium valproate (SVP) on behavioral, biochemical, and ultra-structural effects induced by 6 Hz. Behavioral changes induced by 6 Hz stimulation were characterized as the increased duration of Straub’s tail, stun position, twitching of vibrissae, forelimb clonus, and increased rearing and grooming. PCPA administration further enhanced while SVP reduced these behaviors in mice. The 6-Hz psychomotor seizure induced ultra-structural changes in both cortex and hippocampus in mice treated with PCPA. Furthermore, PCPA administrations followed by 6Hz-induced seizures were accompanied by reduced hippocampal and cortical 5-HT. SVP attenuated the PCPA-induced ultra-structural changes and alterations of 5-HT content in the mouse brain. The study suggests the involvement of 5-HT in the 6 Hz psychomotor seizures and in the mechanisms of action of SVP against such seizures in mice.
Keywords
Introduction
Barton et al. reported the use of 6Hz-induced seizures as a model to screen antiepileptic drugs, which are useful in the therapy of pharmacoresistant epilepsy. 1 It is characterized by psychic manifestations (including grooming, rearing, behavioral arrest, etc.) and complex motor seizures characteristic of resistant partial seizures. A large body of evidence, both preclinical and clinical, has indicated the abnormalities in serotonergic system in the pathophysiology of epileptic seizures. 2 –6 Pharmacological studies have indicated that low serotonin (5-hydroxytrytamine (5-HT)) content in the brain leads to an intensification of seizures in genetically epilepsy prone rats (GEPRs). 2 5-HT depletion is reported to destabilize neuronal networks rendering the neurons more vulnerable to pathological changes leading to seizures. 6 Although there is no evidence for serotonin gene mutations in human epilepsy syndromes, however, deletion of 5-HT2c receptors has been demonstrated to cause audiogenic seizures in mice. 3,7 In humans, a decrease in 5-HT1A receptor binding in the brains of patients with epilepsy has been reported. 4,5
Parachlorophenylalanine (PCPA) acts as a selective and irreversible inhibitor of tryptophan hydroxylase, which is a rate-limiting enzyme in the biosynthesis of serotonin and hence leads to reduced 5HT levels. 8 In a recent study on genetically modified C57BL/6 N mice, reduction of 5-HT synthesis with PCPA was reported to increase the maximal electroshock seizure-induced seizure severity. The study concluded that serotonergic neurotransmission raises the seizure threshold and decreases seizure-related mortality. 9 These observations point toward a role of 5-HT in experimental seizures.
Recently, we reported the role of monoamines in 6Hz-induced psychomotor seizures and demonstrated that PCPA induced 5-HT depletion in whole brain and aggravated 6Hz-psychomotor seizures in mice. 10 In the present work, we aimed to investigate the effect of 5-HT depletion in cortex and hippocampus (brain regions relevant for epilepsy) by PCPA on 6 Hz psychomotor seizures in mice and possible protection by sodium valproate (SVP). The drug is reported to produce anticonvulsant effects against 6 Hz seizures in mice and to alter 5-HT levels in pentylenetetrazole-induced kindled mice. 11
Materials and methods
Animals
The study was carried out on male Swiss albino mice weighing 25–35 g procured from the Central Animal House Facility, Jamia Hamdard University. Mice were maintained under controlled temperature at 20°C and 50–55% humidity and light with natural light and dark cycle, with free access to food and water, and housed in an approved animal house facility of Jamia Hamdard. The experimental protocol was approved by the Institutional Animal Ethics Committee (protocol no.1010). Utmost care was taken to ensure that animals were treated in the most humane and ethically acceptable manner.
Drugs and reagents
SVP, PCPA, and the standards used for 5-HT estimations were purchased from Sigma (St Louis, Missouri, USA). PCPA (300 mg/kg/i.p) and SVP (200 mg/kg/p.o) were dissolved in sodium chloride and given in a volume of 10 ml/kg.
Experimental design
The treatment schedule is depicted in Table 1. Animals were randomly placed in each group with eight animals per group. The animals of each group were housed in different cages to ensure randomization. All experiments were conducted in a blinded fashion, the coding of animals in each group was carried out by another member of the laboratory.
Treatment schedule.a

PCPA: parachlorophenylalanine; SVP: sodium valproate.
a Mice were subjected to 6 Hz stimulation after the respective treatments except in groups I, VII.and VIII. All the test drugs were administered for a period of 3 days. All observations were made 24 h after the last dose of PCPA.
6-Hz psychomotor seizures test
In the 6 Hz epilepsy model, investigational compounds are screened for their ability to block psychomotor seizures induced by a low-frequency (6 Hz), long-duration (3 s) stimulus delivered through corneal electrodes. These seizures are believed to model partial seizures observed in humans. Mice are challenged with a 44 mA current for 3 s delivered through corneal electrodes. 1,12
The animals were placed in a transparent Plexiglas chamber (24 × 17 × 13 cm3) and a camera was fixed on a vertical stand placed above it to record the epileptic behaviors in mice following 6 Hz stimulation. The 6 Hz psychomotor seizure was induced by corneal stimulation (6 Hz, 0.2 ms rectangular pulse width, 3 s duration at 44 mA) (ECT Unit, UgoBasile57800-001, Italy). Prior to the placement of corneal electrodes, a drop of 0.9% saline was administered to the eyes of mice. Animals were manually restrained and released immediately following the 6 Hz stimulation and observed for the presence or absence of seizure activity. 1
The total cumulative duration of behavioral parameters namely Straub’s tail, stun position, twitching of vibrissae, forelimb clonus, rearing, grooming, and distance traveled (velocity) were recorded over a period of 120 s. Animals were considered to be seizure protected if they resumed their normal exploratory behaviors within 10 s of 6 Hz stimulation. 13 Immediately after the recording of behavioral parameters (120 s), animals were anaesthetized with carbon dioxide inhalation then euthanized by cervical dislocation and hippocampus and cerebral cortex were removed and then stored at −80°C freezer for 5-HT estimation.
Transmission electron microscopy study
The 1 × 1 mm2 section of cerebral cortex and hippocampus were cut from each group (n = 3) and the tissue samples were fixed in 2.5% glutaraldehyde and 2% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.3) for 12 h at 4 °C. After washing in buffer, the samples were postfixed in 1% osmium tetraoxide (OsO4) for 1 h at 4°C . The samples were dehydrated in an ascending grade of acetone, infiltrated, and embedded in Araldite CY 212 (TAAB, UK). Ultrathin sections of grey-silver color interference (70–80 nm) were cut and mounted onto 300 mesh-copper grids. Sections were stained with alcoholic uranyl acetate and alkaline lead citrate, washed gently with distilled water, and observed under a Technai G2 transmission electron microscope (Fei Company, the Netherlands) at an operating voltage 200 kV. Images were digitally acquired using a CCD camera (Megaview III, Fei Company, Hillsboro, Oregon) attached to the microscope. The best representative pictures from each group are depicted in Figures 4 and 5.
Estimation of 5-HT using LCMS/MS
The 5-HT levels of the brain were estimated by liquid chromatography and mass spectrometry (LCMS/MS) as per previous method 14 with slight modifications. The brain samples (cortex and hippocampus) were homogenized in ice-cold methanol (10% w/v) and centrifuged at 14,000 rpm for 10 min at 0 °C. The supernatants were filtered through a 0.45 µm membrane filter and 500 µl was taken in a glass tube then dried at 20 °C in nitrogen evaporator. It was then reconstituted with 500 µl mobile phase and filtered again through a membrane filter in the vial. Standard solution and samples were injected into the column. The analytes were separated on a thermal C-18 column (5 µm, 50 × 4.6 mm) at ambient column temperature using mobile phase which consisted of methanol 5 mM ammonium formate buffer (55:45, v/v) at a flow rate of 0.6 ml/min. The contents of 5-HT were expressed in ng/mg wet tissue. The amount of 5-HT in standard and samples were quantified using area under the curve of corresponding sample using their straight line equation: 5-HT (y = 8803.8x + 22, 0145; R2= 0.999 for cortex); 5-HT (y = 5018x + 94,948; R2= 0.999 for hippocampus).
Statistical analysis
The data were expressed as the mean ± standard error of mean. The significance of differences among the groups was assessed using one-way ANOVA followed by Tukey-Kramer’s multiple comparison test except in the case of rearing and grooming where Kruskal Wallis ANOVA followed by Mann–Whitney as post hoc test was used and for number of animals protected, Fisher’s exact was used. The value of p < 0.05 was considered to be significant.
Results
Effect of SVP on protection in PCPA primed 6 Hz seizures in mice
6 Hz induced seizures in six of eight animals. Pretreatment with SVP (200 mg/kg/day, p.o.) significantly protected around seven mice against 6 Hz stimulation while PCPA (300 mg/kg/day, i.p.)-treated animals did not show any protection. Pretreatment and post-treatment with SVP in PCPA-treated mice protected five mice against 6 Hz (p < 0.05; Figure 1).

Effect of SVP on protection in PCPA primed 6 Hz seizures in mice. SVP was given pre and post PCPA for 3 days. Pretreatment with SVP significantly protected mice against 6 Hz stimulation whereas PCPA-treated animals did not show any protection. Pretreatment and post-treatment with SVP in PCPA-treated mice show protection against 6 Hz. The data represent the protection against 6 Hz seizures (n = 8). Statistical analyses were performed using Fisher’s exact test. ***p < 0.001 when compared with sham control, #p < 0.05, ###p < 0.001 when compared with Vehicle (saline) + 6 Hz, †††p < 0.001 when compared with PCPA+6 Hz. PCPA: parachlorophenylalanine; SVP: sodium valproate; n = 8.
Effect of SVP in the 6 Hz psychomotor seizure behavior in mice treated with PCPA
The results are depicted in Figure 2(a) to (d). Behavioral changes induced by 6 Hz stimulation were characterized as the increased duration of Straub’s tail (Figure 2(a)), stun position (Figure 2(b)), twitching of vibrissae (Figure 2(c)), and forelimb clonus (Figure 2(d)). This behavior was significantly attenuated by pretreatment with SVP (p < 0.001). PCPA, however, significantly increased the duration of Straub’s tail, stun position, twitching of vibrissae, and forelimb clonus as compared to 6 Hz. The PCPA-induced amplification of behavioral changes in mice was significantly attenuated by SVP pretreatment and post-treatment except for the stun position in the post-treatment group. SVP per se didn’t induce any of these behavioral changes but PCPA per se significantly increased the stun position, twitching of vibrissae, and forelimb clonus with a maximum increase in the stun position (Figure 2).
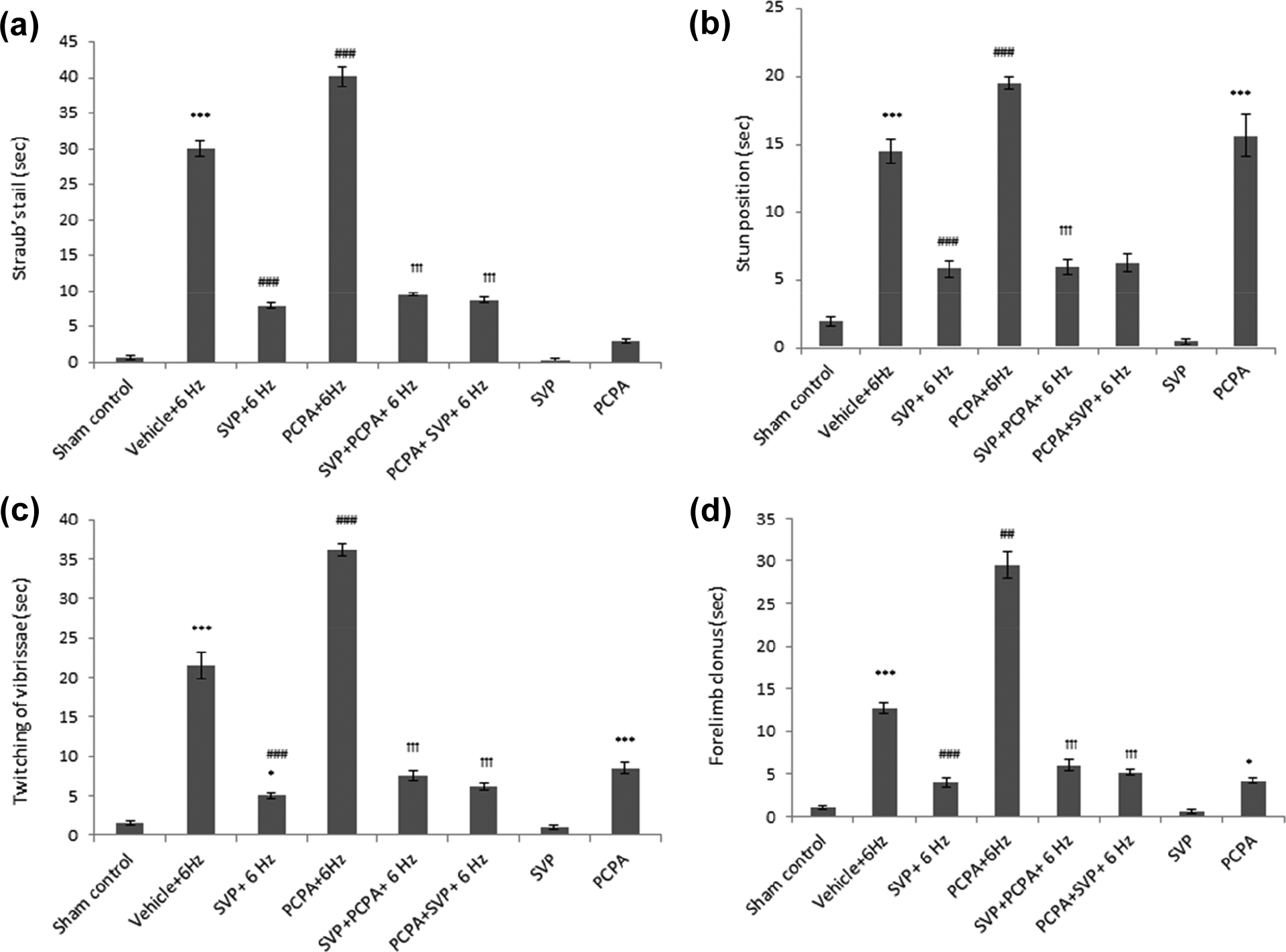
Effect of SVP in the 6 Hz psychomotor seizure behavior in mice treated with PCPA. SVP was given pre-PCPA and post-PCPA for 3 days. Behavioral changes induced by 6 Hz stimulation including increased duration of Straub’s tail (Figure 2(a)), stun position (Figure 2(b)), twitching of vibrissae (Figure 2(c)), and forelimb clonus (Figure 2(d)) are enhanced by PCPA. The PCPA-induced amplification of behavioral changes in attenuated by SVP pretreatment and post-treatment except for the stun position in post-treatment. Data were represented as mean ± SEM and analyzed by one-way ANOVA followed by Tukey-Kramer multiple comparison test, *p < 0.05, *** p < 0.001 when compared with sham control; ##p < 0.01, ###p < 0.001 when compared with Vehicle (saline) + 6 Hz; ††† p < 0.001 when compared with PCPA+6 Hz. PCPA: parachlorophenylalanine; SVP: sodium valproate; n = 8; ANOVA: analysis of variance.
Effect of SVP on locomotion in the 6-Hz psychomotor seizures in mice treated with PCPA
Locomotion changes after 6 Hz stimulation were characterized by a frequency of rearing and grooming. SVP pretreatment significantly reduced rearing (Figure 3(a)) and grooming (Figure 3(b)) and increased the velocity (Figure 3(c)). PCPA per se and when given 6 Hz, induced a remarkable increase in rearing, grooming, and velocity, which was attenuated by pretreatment and post-treatment with SVP. The latter alone did not produce any changes in such behavior (Figure 3).
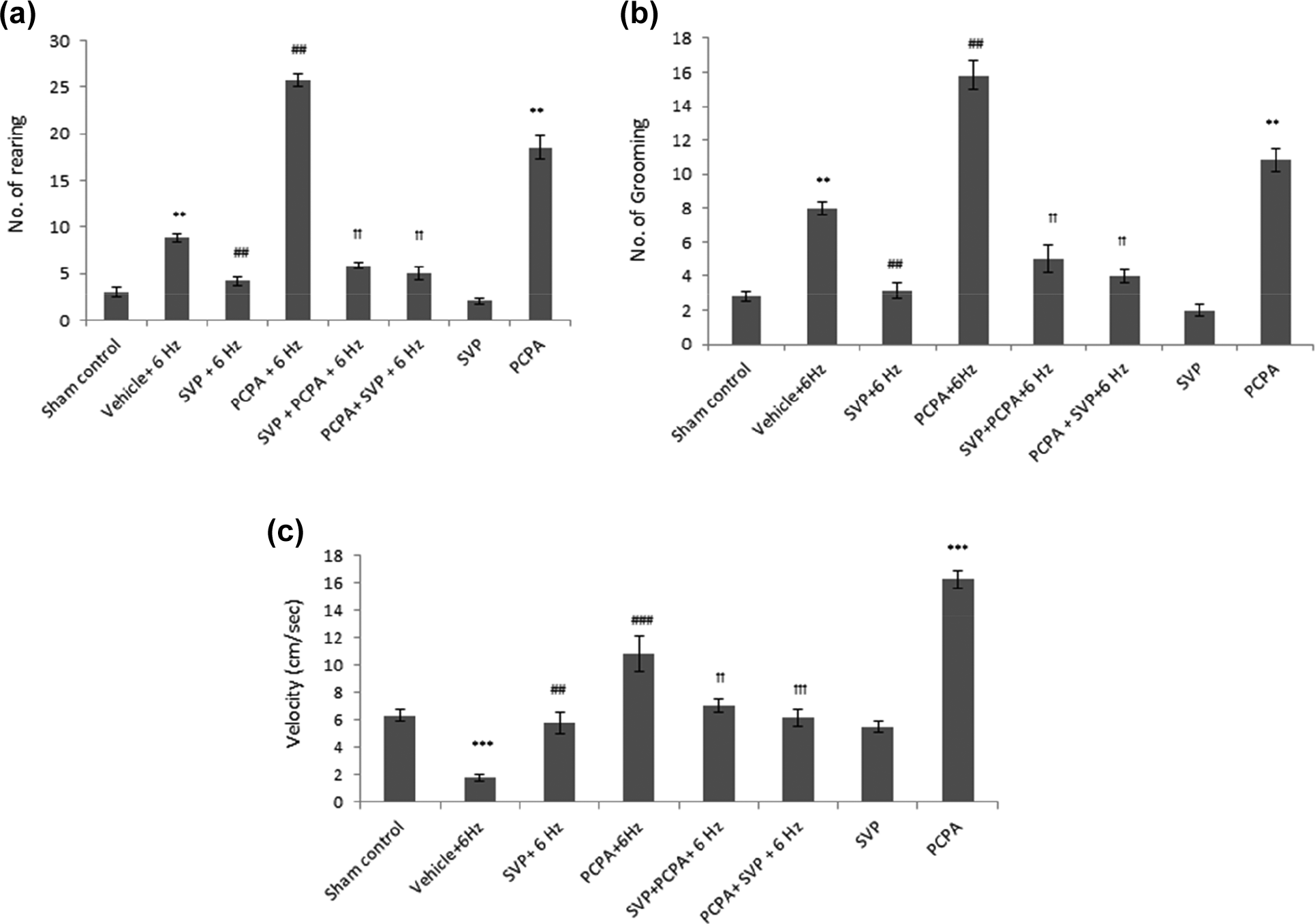
Effect of SVP in the 6 Hz psychomotor seizure behavior and locomotion in mice treated with PCPA. SVP was given pre- and post-PCPA for 3 days. Locomotion changes after 6 Hz stimulation were characterized by increased frequency of rearing and grooming. SVP pretreatment reduced rearing (Figure 3(a)) and grooming (Figure 3(b)) and increased the velocity (Figure 3(c)) while PCPA enhanced it. The enhancement by PCPA is attenuated by pretreatment and post-treatment with SVP. Data were represented as mean ± SEM and analyzed by Kruskal–Wallis ANOVA followed by Mann–Whitney as post hoc test for rearing and grooming and velocity analyzed by one-way ANOVA followed by Tukey-Kramer multiple comparison test, ***p < 0.001 when compared with sham control; ##p < 0.01, ###p < 0.001 when compared with vehicle (saline) + 6 Hz;††p < 0.01, †††p < 0.001 when compared with PCPA+6 Hz. PCPA: parachlorophynylalanine; SVP: sodium valproate; n = 8.
Effect of SVP in the 6-Hz psychomotor seizure-induced ultrastructural changes in cortex and hippocampus in mice treated with PCPA
Electron microscopic studies were performed on cortex and hippocampus in mice. In cortical and hippocampus cells of sham control mice, subcellular structures were normal, and there was no damage in mitochondrial and nuclear structure (Figures 4(a) and 5(a)). In contrast, the cytoplasm of 6 Hz mice was found disorganized with a swollen nucleus and chromatin loss with partly damaged axons in cortical cells while disorganized mitochondria in the hippocampus (Figures 4(b) and 5(b)), but these changes were reversed in both cortex and hippocampus by SVP (Figures 4(c) and 5(c)). In PCPA+6 Hz group, the degenerative changes were found in the nucleus and myelinated axon with many vacuoles and disorganized mitochondria in cortical cells, in contrast, this damage was more severe in the hippocampus with cytoplasm degeneration, organelles indistinct and nucleus appeared on the verge of death (Figures 4(d) and 5(d)). Furthermore, no ultrastructural changes were found in SVP per se but in PCPA per se group, mitochondria appeared normal in the cortex but disorganized in the hippocampus (Figures 4(g) and 5(h)). Pretreatment and post-treatment with SVP both were found effective in inhibiting cellular degeneration in cortical and hippocampal cells. SVP-treated epileptic mice exhibited normal subcellular structures; for example, the cytoplasm was homogeneous with abundant mitochondria. The chromatin was dispersed, the nucleus was normal (Figures 4(c), 5(c) and 4(e), 5(e)).

Effect of SVP in the 6 Hz psychomotor seizure-induced ultrastructural changes in the cortex in mice treated with PCPA. ((a): Sham control) shows normal ultrastructure in cortex, nucleus (N) appears healthy and euchromatic (highly transcriptionally active), cytoplasm contains normal mitochondria (m), myelinated axons (a) appear normal, with normal mitochondria (m) in axoplasm. ((b): 6 Hz) shows myelinated axons (a1, a2), with partly damaged myelin. The nucleus (N) is swollen and shows chromatin loss. The cytoplasm is disorganized. ((c): SVP+6 Hz) Nucleus (N) with mild perinuclear heterochromatin, myelinated axons (a) appear normal in outlook, cytoplasm contains normal mitochondria (m). ((d): PCPA+6HZ) shows nucleus (N) with mild perinuclear heterochromatin, cytoplasm is degenerative, with many vacuoles (v), organelles are indistinct, mitochondria (m) are disorganized, myelinated axons (a) appear degenerative. ((e): SVP+PCPA+6 Hz) shows normal nucleus (N), cytoplasm with slightly disorganized organelles (arrows) with normal mitochondria. ((f): PCPA+SVP+6 Hz) shows cytoplasm of a glial cell containing normal mitochondria (m), the myelinated axon (a) appears slightly irregular, compressed in its myelin sheath thickness. ((g): SVP per se) shows an intact nucleus (N), which is euchromatic, the cytoplasm is normal, with healthy mitochondria and few inclusions (stars). ((h): PCPA per se) shows the thickness of myelin varies in axon number a1 and a2 (thin myelin sheath), compared to that in other axons (a3–a6, thick sheath), cytoplasm contains normal mitochondria (m). SVP was given pre and post PCPA for 3 days. PCPA: parachlorophenylalanine; SVP: sodium valproate; n = 3.

Effect of SVP in the 6 Hz psychomotor seizure-induced ultrastructural changes in hippocampus in mice treated with PCPA. ((a): sham control) shows normal ultrastructure in hippocampus, nucleus (N) appears healthy, cytoplasm contains normal mitochondria (m), myelinated axon appear normal, with normal axonal cytoskeleton (a). ((b): 6 Hz) shows nuclei (N) with mild heterochromatin, cytoplasm in one cell contains dilated rough endoplasmic reticulum (arrowheads) and disorganized mitochondria (arrows), the myelinated axon (a) appears abnormal in its myelin sheath. ((c): SVP+6 Hz) Nucleus (N) with mild perinuclear heterochromatin, myelinated axons (a) appear normal in outlook, cytoplasm contains normal mitochondria (m). ((d): PCPA+6HZ) shows nucleus (N) with dense accumulation of perinuclear heterochromatin (star), cytoplasm is degenerative, organelles are indistinct, the nucleus appeared as on the verge of death (dying). ((e): SVP+PCPA+6 Hz) shows slightly disorganized myelinated axons (a), normal mitochondria (m). ((f): PCPA+SVP+6 Hz) shows nuclei (N) with mild heterochromatin, cytoplasm in this cell contains slightly dilated endoplasmic reticulum (arrowheads). Mitochondria appear normal. ((g): SVP per se) shows an intact nucleus (N), which is euchromatic, the cytoplasm is normal, with healthy mitochondria (m). ((h): PCPA per se. The myelin sheath of the axons (a) is normal, few mitochondria (m) appear disorganized. SVP was given pre- and post-PCPA for 3 days. PCPA: parachlorophenylalanine; SVP: sodium valproate; n = 3.
Effect of SVP in the 6-Hz psychomotor seizure-induced changes in hippocampal and cortical serotonin levels in mice treated with PCPA
The results are presented in Figure 6. The stimulation of 6 Hz significantly reduced the endogenous 5-HT levels in both hippocampus and cortex of mice (p < 0.001). Pretreatment with SVP significantly caused reversal of this reduction. Administration of PCPA facilitated further reduction in endogenous 5-HT levels in both hippocampus and cortex of mice (p < 0.001) as compared to 6 Hz group. This reduced serotonin was significantly attenuated by the pretreatment and post-treatment of SVP with more significant attenuation in hippocampal as compared to cortical serotonin levels. Both SVP and PCPA per se increased and reduced serotonin levels, respectively, with highly significant effect in the case of PCPA (Figure 6).

Effect of SVP in the 6 Hz psychomotor seizure-induced changes in the brain 5-HT levels in mice treated with PCPA. SVP was given pre- and post-PCPA for 3 days. 6 Hz reduced the endogenous 5-HT levels in both hippocampus and cortex of mice. Administration of PCPA facilitated further reduction of endogenous 5-HT levels in both hippocampus and cortex of mice. This reduced serotonin was attenuated by the pretreatment and post-treatment of SVP. Data were represented as mean ± SEM and analyzed by one-way ANOVA followed by Tukey-Kramer multiple comparison test, *p < 0.05, **p < 0.01, ***p < 0.001 when compared with sham control; #p < 0.05, ##p < 0.01, ###p < 0.001 when compared with vehicle (saline) + 6 Hz; €p < 0.05, €€p < 0.01, €€€p < 0.001 when compared with SVP+6 Hz; †p < 0.05, ††p < 0.01, †††p < 0.001 when compared with PCPA+6 Hz. PCPA: parachlorophenylalanine; SVP: sodium valproate; 5-HT:5-Hydroxytryptamine; ng/mg; nano-gram/milligram; n = 8.
Discussion
Seizures as evidenced by studies showing the elevation of 5-HT levels in the brain following several important antiepileptic drugs. 15 In an earlier animal study, 5-hydroxytryptophan (5HTP), a serotonin precursor, was reported to have antiepileptic effects and later, anticonvulsant effects of serotonin reuptake inhibitors (SSRIs) including fluoxetine were reported in animal models. 16,17 However, SSRIs that increase synaptic 5-HT previously were also thought to reduce seizure threshold and promote seizure occurrence. 18,19 Majority of reports, however, points toward their anti-convulsant role and that SSRIs are well-tolerated in patients with epilepsy. 20 Thus, the depletion of serotonin concentrations in the brain was reported to increase seizure susceptibility and to decrease seizure threshold in several animal models of epilepsy. 21 Recently, studies have suggested that the potentiation of the serotonergic system could be useful in controlling the epileptic seizures especially in pharmacoresistant epilepsy 22 thus providing a link between 5-HT and pharmacoresistant epilepsy. The present work provided further evidence in this direction by proving the involvement of brain serotonin in the 6 Hz epilepsy model.
In our study, the application of 6 Hz stimulation induced significant behavioral changes in the vehicle-control cohort, which was characterized as the increase in the frequency of rearing and grooming and the duration of Straub’s tail, stun position, and reduced velocity (increased immobility) in mice. The behavioral changes in our findings were in line with the previously published reports characterizing 6 Hz seizures as the presence of reduced velocity of movement and increased the duration of the stun position and forelimb clonus in mice. 23,24 We found that in PCPA pretreated cohorts, mice showed greater behavioral changes such as significantly enhanced duration of Straub’s tail, stun position, forelimb clonus, twitching of vibrissae, and stereotype behavior like increased in frequency of rearing and grooming compared to the vehicle-control indicating that depletion of 5-HT enhances 6Hz-induced behavioral alterations. Thus, we report the pro-convulsant effect of PCPA in the 6 Hz epilepsy model.
Neurodegenerative changes have been described in several models of experimental epilepsy. 25,26 We report the ultra-structural changes in brain cortex and hippocampus in 6 Hz epilepsy model for the first time. These changes were characterized by disorganized cytoplasm with a swollen nucleus and chromatin loss in cortical cells and disorganized mitochondria in the hippocampus. Pretreatment with SVP effectively inhibited such changes at the ultra-structural level. PCPA, on the other hand, resulted in further neurodegeneration when given prior to 6 Hz. Both pretreatment and post-treatment with SVP reduced such changes with pretreatment showing more profound effect. Thus, ultra-structural changes further confirmed our findings on behavioral seizures and indicated the neuroprotective potential of SVP in ameliorating 6 Hz and PCPA-induced structural changes in cortex and hippocampus.
To confirm whether such effects of SVP were mediated via serotonin, we measured cortical and hippocampal serotonin levels. The neurochemical analysis of the brain sample revealed considerably higher depletion of 5-HT levels in PCPA and PCPA+6 Hz cohorts than vehicle control. This was expected as PCPA is a known depletor of brain 5-HT content. 8 However, 6 Hz stimulation further reduced the endogenous 5-HT levels in both hippocampus and cortex of mice. Whether this low 5-HT was a cause or consequence of seizures is not clearly known. Our results are in agreement with previous studies reporting 5-HT depletion in various other animal models of epilepsy including maximal electroshock seizures in mice and rats, 27,28 an atypical absence model of AY-9944-treated rats, 29 and sensory-induced seizures in El mice. 30 In addition to these reports, marked protection against kainic acid-induced seizures was reported in mice which have genetically high levels of 5-HT. 31 More recently, genetically modified mice lacking 5-HT neurons were reported to have lower seizure threshold and increased seizure-induced mortality in maximal electroshock and pilocarpine models of acute seizures. 9,32 Hence, increasing literature suggest a serotonergic mechanism in epilepsy pathophysiology and the low brain 5-HT content in increasing susceptibility to seizure induction. Thus, our finding of PCPA enhancing seizure activity is in agreement with the existing literature.
However, our results contrast with few previous reports suggesting anticonvulsant effects of PCPA. For instance, acute PCPA treatments reduced audiogenic seizures in mice and prevented seizure development by amygdala stimulation in rats, 19,33 and chronic treatment with PCPA retarded the development of bioelectrical and behavioral seizure phenomena in rabbits. 34 It is possible that 5-HT affects seizures differentially in various models that require investigations.
Our study found considerably higher 5-HT levels than vehicle-control in SVP treated mice, indicating that SVP might mediate protection against 6 Hz seizures via increasing the brain 5-HT content. We noted a significant protection of 6 Hz seizures in both groups of mice that were pretreated with SVP before PCPA (pretreatment cohort) and that were given SVP after PCPA (post-treatment cohort). Although we expected that post-PCPA administration of SVP may not restore 5-HT levels to the extent to be able to provide protection in 6 Hz seizures. However, we observed that SVP was able to restore 5-HT and provided protection in both pretreatment and post-treatment groups. Although the reasons for this are not clearly understood, there might be two reasons: (a) PCPA at a dose of 300mg/kg provided only partial depletion and (b) 6 Hz was also depleting serotonin levels irrespective of PCPA. Hence, it is possible that SVP restored the 6 Hz-induced depletion of 5-HT in the post-treatment groups. Nevertheless, the protective effect of SVP is in concordant with a recent study reporting that SVP was effective in blocking the seizure-induced behavioral changes and reducing the after discharge duration in the lamotrigine-resistant, amygdala-kindled pharmacoresistant epilepsy. 35 Therefore, we conclude that SVP protected against 6 Hz seizures and resisted 6 Hz evoked behavioral changes possibly by elevating hippocampal and cortical 5-HT levels, and our findings are consistent with the report that SVP elevates the brain serotonin in audiogenic seizure-prone balb/c mice, GEPRs, and in pentylenetetrazole kindled mice. 11,36,37 SVP elevated 5-HT despite PCPA administration, which could be due to the fact that the dose of PCPA used in the present study induced only 40–50% depletion. This dose permitted us to observe the combined effect of 6 Hz and PCPA for further reduction in 5-HT levels. This wouldn’t have been possible if we selected a dose that makes 5-HT completely absent.
To conclude, our study importantly implicates low brain 5-HT content to be one of the underlying causes of 6 Hz epilepsy model and that SVP-mediated protection against 6 Hz results possibly via modulation of 5-HT content though other mechanisms also contribute to the overall anticonvulsant action as reported previously for other monoamines and gamma amino butyric acid (GABA). More experiments with 5-HT receptor antagonists are needed to strengthen this hypothesis. Further investigations may be carried out using other antiepileptic drugs effective in this model.
Footnotes
Authors’ note
The part of the results presented in this article was based on a student’s thesis work. We confirm that we have read the journal’s position on issues involved in ethical publications and affirms that this reportis consistent with those guidelines.
Acknowledgments
The authors are thankful to the staff at the SAIF facility of AIIMS for help in electron microscopy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant provided by the Indian council of medical research (ICMR) India, grant no. (45/52/14-PHA/BMS). The work was carried out in the Neurobehavioral Pharmacology laboratory supported by University Grants Commission Special Assistance Program (UGC-SAP).
