Abstract
We evaluated EEG changes and pain relief in migraineurs with glyceryl trinitrate (GTN)-induced attacks treated with intravenous sodium valproate (iSVP). EEG frequency analysis was performed in 45 migraineurs without aura and in 19 controls at baseline, at the time of maximum GTN-induced headache, and 30 min after 300 mg iSVP. Nineteen migraineurs presented early specific headache (migraine identical to spontaneous attacks; MSp) and 26 presented early nonspecific headache (MnSp). During attacks in MSp there was an increase of theta [16.6% (14.8-19.3) to 19.4% (17.4-22.1), P = 0.02] and delta activity [3.6% (3.1-4.4) to 5.4% (3.9-6.5), P = 0.009], whereas there was no decrease in alpha [41.4% (36.2-45.1) to 39.7% (34.7-44.8)] or beta activity [37.6% (34.7-40.3) to 35.1% (33.5-38.8)]. iSVP reduced migraine from severe/moderate to mild/no pain in 17 (90%) MSp patients, and was associated with reversion of the slow rhythmic activity to baseline levels [theta 16.9% (14.6-18.9); delta 3.2% (3-4.1)]. There was no change in EEG frequency activity after administration of GTN or iSVP in controls and in MnSp. iSVP is well tolerated and effective in treating GTN-induced migraine in migraineurs without aura, and appears to restore the disturbances of cortical electrogenesis associated with these attacks.
Introduction
Valproate is an established agent for migraine prophylaxis (1) owing mainly to the potentiation of GABA-mediated responses (2). Several small studies using different doses and designs have evaluated the efficacy, safety and tolerability of per os (3) but mainly of intravenously administered valproate in the treatment of acute migraine attacks (4–7), status migrainosus (8) and chronic daily headache (9, 10), suggesting that valproate possesses significant analgesic properties. This is in accordance with experimental data documenting that valproate blocks neurogenic inflammation and directly suppresses nociceptive neurotransmission (11, 12). A study (13) has documented the efficacy of valproate per os in preventing glyceryl trinitrate (GTN)-induced migraine, but hitherto, to our knowledge, intravenous valproate has not been assessed in the treatment of acute GTN-induced migraine attacks.
In patients suffering from migraine without aura (MoA), EEG abnormalities are known to occur (14, 15). Although the relevant reports have often yielded contradictory results, probably owing to differences in study design and EEG interpretation, their data suggest that disturbances of cortical electrogenesis are frequent in migraine. In a previous study (16), we recorded EEG abnormalities, namely an increase of slower activity, during GTN-induced migraine attacks and have shown that these abnormalities were reversed after subcutaneous administration of sumatriptan. In the present study, we investigated the effect of intravenous valproate on EEG alterations associated with GTN-induced migraine. Whether valproate through its multiple modes of action on the central nervous system, in conjunction with its analgesic potential, could demonstrate an effect on EEG changes related to spontaneous migraine attacks or offer an alternative to the acute treatment of spontaneous migraine attacks remains to be investigated.
Methods
Subjects
The study included 45 patients (39 women, age 37.3 ± 3 years) suffering from MoA (mean duration of disease 7.8 ± 0.9 years) according to the International Headache Society (IHS) criteria (17) and 19 healthy age- and sex-matched controls. Patients were selected from the pool of patients referred to the headache out-patient clinic of our hospital between January 2004 and February 2006. All patients were interviewed by a physician using a standardized questionnaire, which included information about the presence and characteristics of headaches during the previous 12 months. Subsequently, headaches were classified according to the IHS criteria (17). All patients suffered at least one, and no more than six attacks of MoA per month, of moderate to severe intensity. Patients taking antiepileptics or other treatment that could possibly influence the appearance of headache or alter the EEG (e.g. β-blockers, calcium antagonists, selective serotonin reuptake inhibitors, tricyclics, benzodiazepines, major neuroleptics, oral contraceptives) were not included. Patients with ischemic heart failure or hypertension were excluded. Analgesics were not permitted for 72 h prior to the study. Caffeine, alcohol and smoking were not permitted for 12 h prior to the study. No patient was on prophylactic treatment for migraine for at least 1 month prior to the study, and no woman was pregnant.
Controls were selected as follows: 171 employees of our institution consented to complete a detailed questionnaire focusing on the occurrence and characteristics of cephalalgia during the past 10 years. Based on their responses, 99 individuals were immediately excluded, 72 underwent a medical interview, and ultimately 54 individuals were judged suitable for inclusion. These individuals had experienced ≤ 15 episodes of cephalalgia of any kind during the past 10 years and two or fewer episodes per year for the past 2 years. Nineteen individuals were finally selected to create a group matched by age and sex to migraineurs. The study was approved by the ethics committee of our hospital and all individuals gave informed consent prior to their inclusion.
Study design
After an overnight fast, the subjects were lying comfortably in a temperature-controlled room (24 ± 2 °C) with eyes closed (care was taken to prevent the patients from falling asleep). Studies commenced between 10.00 and 10.30 h. An indwelling cannula was inserted into a forearm vein for the infusion of sodium valproate (SVP) and for the collection of blood samples. The cannula was kept patent with heparinized saline. EEG studies were performed at baseline, then individuals were administered 1 mg GTN sublingually and were closely monitored for the appearance of early GTN-induced headache. Based on our previous experience (15), we expected headache to peak within 30 ± 5 min after GTN administration. If headache developed, its characteristics were meticulously recorded and pain severity was reported on a 10-grade visual analogue scale (mild 1–3, moderate 4–6, severe 7–10). Headaches were classified into specific and non-specific according to Sances et al. (18). In particular, specific pertained to headache that: (i) fulfilled the IHS criteria (17) for any type of primary neurovascular headache and (ii) in the case of migraineurs, was characterized as identical to their spontaneous attacks by their own statements. Non-specific headache pertained to any headache that did not fulfill the above criteria (i–ii). A second EEG was performed at the time of maximal headache and immediately afterwards, 300 mg of SVP diluted in 100 ml of saline was infused at a rate of 50 mg/min, followed by a third EEG 30 min after the end of the infusion. Blood samples were collected at baseline, 30 and 60 min after the infusion of SVP. Within 24 h, all subjects underwent a telephone interview to investigate the potential appearance and characteristics of delayed GTN-induced headache.
EEG analysis
Electrodes were applied with collodion according to the 10–20 system. Data were acquired with a 16-channel electroencephalograph (Alvar Reega, Paris, France) connected to an Encefia store commercial system. One monopolar and three bipolar montages were used. Spectral analysis of the EEG was performed on 10-s epochs. At least 30 artefact-free epochs were recorded from each montage. Conventional EEG was recorded at the same time for the detection of possible artefacts and the exclusion of the corresponding epochs. The total recording time for each EEG was 14–16 min. The analysis was based on the average of all channels of the referential montage, according to our previous experience (16). In particular, the overall relative power of each frequency band was calculated for each EEG, and power changes of each frequency band among the three EEGs (baseline, during maximal GTN-induced headache and 30 min post SVP infusion) were assessed.
Plasma sodium valproate measurement
Blood samples were collected in heparinized tubes, were immediately centrifuged and plasma was stored at −20 °C until tested. Samples from each patient were tested in the same run. Plasma SVP was measured by a Cobas Integra analyser (Hoffmann La Roche, Basel, Switzerland) using the principle of fluorescence polarization.
Statistics
All results are presented as medians with quartiles. The Friedman test (non-parametric two-way
Results
Headache and headache relief
GTN induced headache of varying intensity in all subjects (Fig. 1). Nineteen out of 45 migraineurs (42.2%) presented specific headache, i.e. migraine with features identical to their spontaneous attacks (MSp). In 26 migraineurs GTN-induced headache somewhat differed from their spontaneous attacks and was classified as non-specific (MnSp). Seventeen individuals from the control group presented non-specific headache, whereas two complained only of ‘discomfort’ associated with a burning sensation in the head. Both these individuals received SVP. Two MSp patients reported delayed specific attacks 6 and 9 h after GTN administration, which were treated successfully with triptans. Three MnSp patients developed MoA 4, 8 and 9 h later. One subject from the control group presented a migraine-like headache 8 h after the study. The median intensity of headache in MSp patients was higher compared with MnSp or controls [6.6 (quartiles 4.9, 8.1) vs. 4.3 (1.7, 6.1) and 3.9 (1.9, 5.9); P < 0.007]. In the MSp subgroup, SVP infusion induced a reduction of headache from severe or moderate to mild or no pain in 17 (89.5%) patients at 30 min post infusion, and in 18 (95%) patients at 1 h post infusion (Fig. 2). In addition, two patients had a > 50% pain reduction at 30 min, and one patient at 1 h post treatment. Subjects with non-specific headache (controls and migraineurs) presented a > 50% reduction of their headache intensity 63 ± 11 min post valproate infusion. No adverse effects were recorded.

Study design with number of individuals by subgroup and timing of EEG investigations and blood sampling.
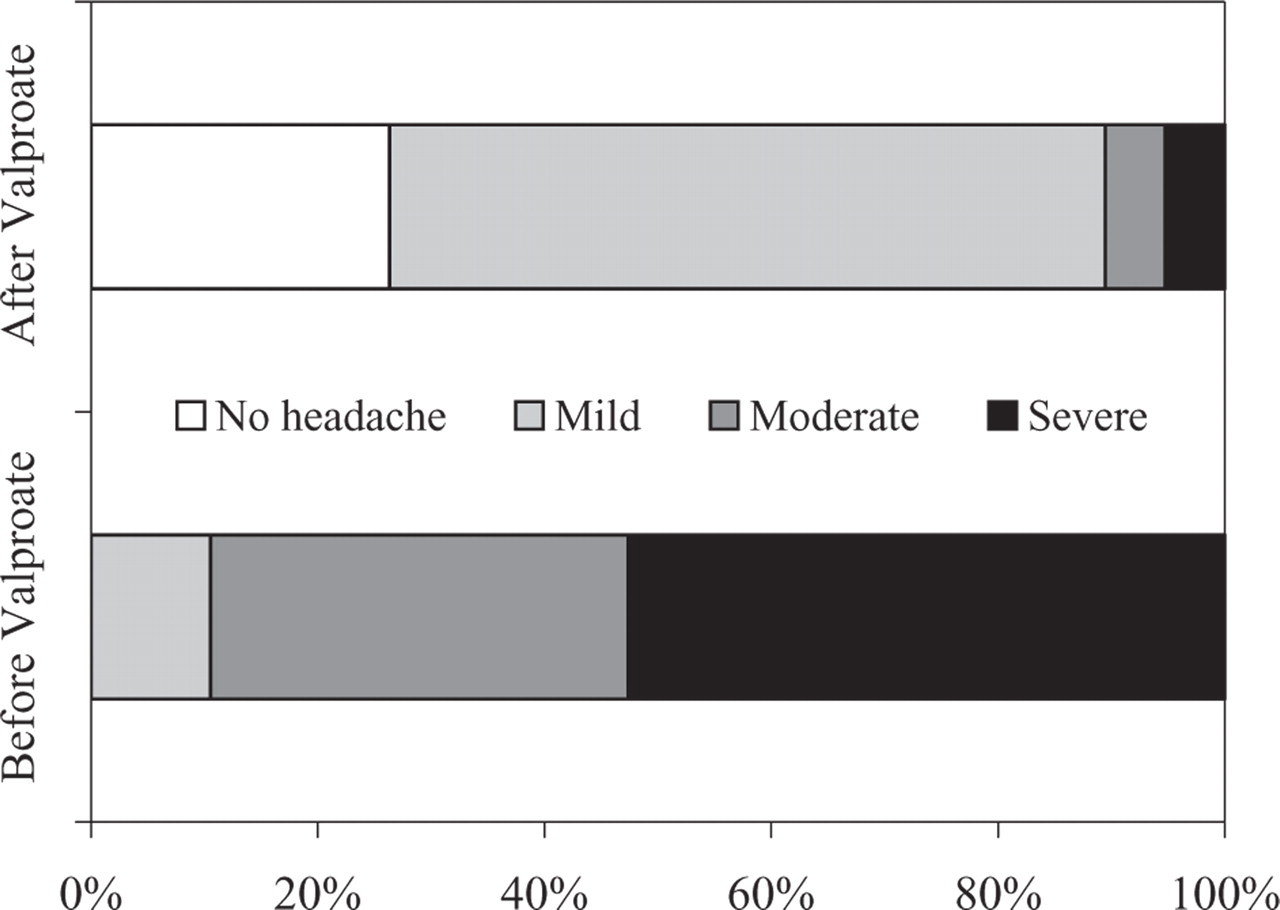
Response of migraineurs 30 min after sodium valproate infusion for the treatment of glyceryl trinitrate (GTN)-induced migraine without aura. Note the dramatic decrease in the percentage of patients with moderate and severe headache intensity after treatment.
EEG analysis
All individuals who participated in the study had normal baseline EEGs without any evidence of paroxysmal activity. The main activity was alpha in 36 migraineurs and beta in the remaining nine patients. Results of the frequency analysis in the two subgroups of migraineurs (MSp and MnSp) and the controls at different time points are depicted in the Table 1. In MSp, the Friedman test showed that the percentage of alpha and beta activities did not differ among the three consecutive examinations (P = 0.3). Conversely, slow activity (theta and delta) exhibited significant differences (P = 0.002). In particular, analysis of the second EEG, conducted during maximal migraine intensity, showed an increase of theta (P = 0.02) and delta activity (P = 0.009), whereas alpha (P = 0.22) and beta activity (P = 0.1) were not significantly altered. The third EEG (30 min post valproate treatment) revealed that the previously observed increase of slow activity was restored to baseline levels (theta, P = 0.88; delta, P = 0.7 compared with baseline) (Fig. 3). In MnSp and in the control group, there was no significant change in EEG frequency activity after GTN administration or after SVP infusion (Table 1).
EEG frequency analysis in different subgroups and time points

MSp, migraineurs who presented specific glyceryl trinitrate (GTN)-induced headache; MnSp, migraineurs who presented non-specific GTN-induced headache; EEG 1, electroencephalogram at baseline; EEG 2, electroencephalogram during maximal headache intensity after GTN administration; EEG 3, electroencephalogram 30 min post sodium valproate infusion.

Percentage of the several frequency bands among the three consecutive EEGs (median with quartiles) in migraineurs who presented immediate glyceryl trinitrate (GTN)-induced attack of migraine without aura (MSp, n = 19). Slow activity (theta and delta) was increased during the attack and reverted to baseline values after sodium valproate (SVP) infusion. Conversely, alpha and beta activities did not differ among different time points. Only P-values (Friedman pairwise comparisons) < 0.05 are displayed.
Sodium valproate blood levels
These were stable between 30 and 60 min after its administration in migraineurs [30 min, 31.9 (28.8–34.5); 60 min, 30.6 (27.5–33.3); P = 0.6]. There were no differences between MSp and MnSp (P = 0.6), or between migraineurs and controls (P = 0.3).
Discussion
During the past decade, many studies have provided evidence supporting a key role for nitric oxide (NO) in the pathogenic cascade of migraine (19). Valproate demonstrates a plethora of in vivo and in vitro actions on the central nervous system, a combination of which is probably responsible for its proven efficacy in migraine prophylaxis (2), even in the GTN human migraine model (13). Although data also favour the use of valproate as an acute antimigraine agent (3–5, 7), its use in everyday practice remains to be established. Our study suggests that intravenous valproate is effective in early GTN-induced migraine attacks. It is rather surprising that all MSp patients in our study reported significant pain relief after valproate infusion. Previous studies have reported rates of response to intravenous valproate ranging from 25% to 73% (4, 5, 7, 10). However, these rates apply to patients with spontaneous migraine attacks severe enough to seek medical assistance in a hospital setting, who were treated at different time intervals from pain onset. All our patients were treated within 30 min after the onset of headache. Nevertheless, based on the results of our small study population, we can not hypothesize an increased effectiveness of intravenous valproate in GTN-induced compared with spontaneous migraine attacks.
In our cohort of migraineurs, 42% presented early GTN-induced migraine identical to their spontaneous attacks and only 11% presented a delayed attack of MoA. This rather contrasts to the literature, since migraineurs exposed to NO donors are known to present more often typical MoA late rather than immediately after exposure (17, 18). A short-term ‘prophylactic’ effect of intravenous valproate could be possible: valproate has an elimination half-time varying from 9 to 21 h, during which most of the delayed GTN-induced migraine attacks are expected to occur. Furthermore, experimental data document that even a single dose results in GABAA-mediated alterations in the neuronal long-term adaptive responses induced by noxious stimuli (11).
Our spectral EEG analysis suggests that in migraineurs without aura, early GTN-induced migraine attacks are associated with increase of the slow rhythmic activity, which is abolished following intravenous administration of valproate (16). EEG studies in migraine are well known for several methodological weaknesses and diverse study designs that are mainly responsible for contradictory or inconclusive results (15). As we have shown before (16), by limiting the number of variables that are assessed by spectral EEG analysis only to the average relative power of the four frequency bands, the reliability and reproducibility of the analysis are maximized. In our study, important alterations of slow EEG activity were not observed in migraineurs who developed non-specific headache or in controls, implying that what has an impact on the electrical activity of the brain is the underlying NO-triggered mechanism of MoA and not GTN administration per se. This assumption is supported by the reversal of EEG changes shortly after the administration of valproate along with the significant relief of migraine. A previous study (20) has elegantly shown that lateralized dorsolateral pontine activation in unilateral GTN-induced migraine attacks persisted after pain control by sumatriptan. We were not able to document any lateralization of the EEG alterations either before or after administration of SVP. However, it should be noted that the majority (13/19) of our MSp patients suffered from (and developed during the study) poorly lateralized migraine. Furthermore, whereas other subcortical structures such as the thalamus are known to influence cortical electrogenesis, no data exist as to whether activation of the dorsolateral pons may interfere with EEG activity. It should be emphasized that the EEG alterations observed in our study in GTN-induced migraine attacks can not be extrapolated to spontaneous attacks of MoA. The EEG changes in spontaneous MoA attacks using the methodology of the present study are currently unknown.
The present study replicates the results of our previous study with sumatriptan, an agent with distinctly different mechanisms of action from valproate. However, both are efficacious antimigraine agents supporting further that blocking the pathophysiological cascade of migraine (albeit via different pathways) may be the main determinant of restoration of EEG activity to baseline. On the other hand, both agents may share a hitherto unknown common mechanism of action that could account for their influence on migraine-associated EEG alterations. Interestingly, sumatriptan and valproate have been shown to block c-fos antigen expression in the trigeminal nucleus caudalis of rodents after noxious meningeal stimulation (11).
Severe GTN-induced systemic hypotension may be associated with EEG changes; however, all EEG data were acquired in a supine position, thus minimizing the effect of venous vasodilation on arterial pressure. Furthermore, there were no differences in arterial pressure post GTN administration among the two subgroups of migraineurs and the controls.
In conclusion, our results indicate that acute intravenous SVP administration: (i) is safe, effective and well tolerated in early GTN-induced migraine attacks in patients suffering from MoA, and (ii) restores to baseline the EEG abnormalities, namely the increase in slow activity, associated with these attacks.
Study limitations
It is widely accepted that after GTN administration in migraineurs a bimodal response takes place: an early non-specific headache (IHS 8.1.1.1), and a late triggered migraine attack (IHS 8.1.1.2) with a mean latency of 130–150 min (18, 19, 21). In one study (18) the mean time to onset of the ‘specific headache response’, i.e. an attack of migraine identical to the patients' spontaneous attacks, was 136.6 ± 130.4 min post GTN administration. This is an enormous variation, implying that some migraine attacks occurred very early. Indeed, in this study (18) 34% of migraineurs without aura developed a specific headache response within 60 min post GTN. Accordingly, the authors suggested that the dichotomization of the headache response to nitroglycerin into immediate and delayed is superfluous and that the specific response to nitroglycerin, in many cases, appears within the first 60 min (18). In our study, 19/45 (42%) migraineurs developed migraine identical to their spontaneous attacks early after GTN administration; this is quite close to the rate reported previously (18). In another study (21), the latency of triggered migraine attacks was 140 min, ranging between 20 and 330 min, confirming once more that GTN-induced migraine can occur quite early, albeit this is not the rule. Knowing that the majority of specific GTN-induced migraine attacks occur late, the best study design would be to follow the patients for several hours post GTN for the appearance of migraine and then to treat with valproate and perform the EEG studies. Unfortunately, this was not applicable for practical reasons. Moreover, we wanted to provide the same stable and controlled conditions for all subjects for the acquisition of EEG data and we therefore opted for the present study design, knowing that the rate of migraineurs with an early specific response would be low. Nevertheless, data support that these early GTN-induced migraine attacks are not infrequent and are not different from the ‘classical’ delayed ones (18, 21).
An effort was made to exclude individuals with a family history of migraine from the control group. The only patient from the control group who presented a delayed migraine-like attack had a free family history at the initial interview. On subsequent interviews, after completion of the study, he claimed a vague recollection of rare ‘headaches’ from his mother's sister without any further details. Unfortunately, we were not able to construct a detailed genealogical tree of this patient.
