Abstract
Background:
Despite being one of the most nephrotoxic drugs, gentamicin (GM) remains a mainstay as a first-choice agent in a vast variety of clinical situations owing to its superlative efficiency as a broad-spectrum antibiotic in treating several life-threatening bacterial infections. This urgently calls for the need for in-depth analysis of the mechanisms governing GM-induced nephrotoxicity and entails the necessity of presenting novel protective agents capable of ameliorating those renal deleterious effects. The reactive oxygen species and redox-sensitive transcription factors in GM-induced nephrotoxicity have recently called attention.
Purpose:
This study has been designed to shed light on the possible mechanisms of GM-induced nephrotoxicity and to provide a consensus set of histopathological, immunohistochemical, genetic and biochemical parameters elucidating the protective role of vitamin D against this nephrotoxicity.
Methods:
Twenty-four adult male albino rats were equally divided into four groups: group I (control group), group II (GM), group III (GM + vitamin D) and group IV (vitamin D only). Kidney function tests, histopathological examination, gene expression of nuclear factor 2, nuclear factor kappa beta (NF-κB) and western blot of NF-κB p65, assessment of glutathione peroxidase and nicotinamide adenine dinucleotide phosphate oxidase (NADPH) oxidase by ELISA, as well as immunohistochemical evaluation of inducible nitric oxide, malondialdehyde, 8-hydroxy 2 deoxyguanine and vitamin D receptor, have been carried out.
Results:
The kidney function deterioration, tissue oxidative stress development and the histopathological changes induced by GM were significantly attenuated by vitamin D administration.
Conclusion:
Vitamin D attenuates GM nephrotoxicity through its antioxidant properties and prevention of DNA damage.
Introduction
Gentamicin (GM) is one of the most important aminoglycoside antibiotics frequently described for the treatment of gram-negative infections. 1,2 The clinical usefulness of GM is limited by the development of nephrotoxicity. Recent studies have tailored a fairly coherent set of ideas about the role of reactive oxygen species (ROS) as crucial mediators of GM-induced nephrotoxicity. 3 –7
GM-induced nephrotoxicity was reported to chiefly affect the renal tubular cells causing direct tubular necrosis 1 accompanied by upregulation of oxidative stress markers 4 and apoptosis of proximal tubular cells. 8
Under normal physiological conditions, the excess ROS can be deactivated by the antioxidant enzyme system which detoxifies O· and H2O2 in cells. In oxidative stress conditions, there is a reduction in the antioxidants level due to excessive consumption by the oxygen free radicals. Previously, in vivo and in vitro studies have shown that GM enhances the generation of ROS metabolites, which may cause cellular necrosis via several mechanisms, including lipid peroxidation, protein oxidation and DNA damage. Administration of antioxidants can be used to prevent or ameliorate GM-induced nephrotoxicity. 9,10
One of the agents whose antioxidant properties have recently caught attention is vitamin D. In addition to its role in Ca++ homeostasis, vitamin D can participate in many other biological functions and crucial antioxidant pathways. 11 The antioxidant activity of vitamin D is achieved via controlling the genes involved in redox balance like glutathione, 12 glutathione peroxidase (GPx) 13 and NADPH oxidase. 14 Vitamin D can actually act through two different genomic and non-genomic pathways. The non-genomic pathway entails binding of vitamin D to membrane receptors with subsequent enhancement of calcium influx, phospholipase C, adenylate cyclase or mitogen-activated protein. 15 The genomic action occurs via a vitamin D receptor (VDR) which belongs to the nuclear receptors superfamily. VDR becomes activated by 1,25VD3 then dimerizes with the retinoid X receptor. 16 VDR gene expression regulation occurs at different levels and can be affected by environmental, genetic and epigenetic factors. 17
The current study has been designed to provide a cogent structural and molecular assessment of GM-induced nephrotoxicity and to elucidate the potential protective mechanisms of vitamin D on GM-induced renal damage through assessment of different histopathological, molecular and biochemical parameters.
Material and methods
Chemicals
Vitamin D was purchased from Medical Union Pharmaceuticals Company, Egypt. GM was supplied from Memphis Co. for Pharm. and Chem. Ind. (MEMCO), Egypt. Urea and creatinine assay kits were purchased from Diamond Company, Egypt. Polymerase chain reaction (PCR) kits were purchased from Qiagen, Germantown, Maryland, USA. Immunohistochemical kits were purchased from Aboot, USA. NADPH oxidase and GPx ELISA kits were supplied from the Cusabio Company, USA, and NF p65 antibodies for western blot analysis kits were provided from Santa Cruz Biotechnology, Inc., Santa Cruz, California, USA.
Experimental animals
Twenty-four adult Wister male albino rats (180–240 g, 8–10 weeks old) were obtained from the animal house of El-Nahda University, Beni Suef, Egypt. All animals were acclimatized to the laboratory environment for 10 days. Animals were housed in well-ventilated cages with a room temperature of about 25° with 12-h light and 12-h dark cycle and free access to food and water ad libitum was allowed. The experiment was conducted according to the Institutional Animal Care and Use Committee (IACUC).
Experimental design
Animals were randomly divided into four groups, six rats/each group. Group I: Control group which received normal saline orally with the gastric tube. Group II: Rats received GM 100 mg/kg body weight by intraperitoneal (IP) injection as a single daily dose for seven successive days.
18,19
Group III: Rats received vitamin D3 1000 IU/kg/day orally by gastric tube for 2 weeks and then combined with gentamycin 100 mg/kg IP for 7 days.
20
–23
Group IV: Rats received vitamin D3 1000 IU/kg/day orally by gastric tube throughout the whole duration of the study.
Samples collection
Samples were obtained from overnight fasted animals through retro-orbital sinus, under ketamine and zylosin anaesthesia into non-heparinized tubes. The collected blood samples were kept for clotting for 30 min at 25°C, followed by centrifugation at 1000 ×
Serum urea and creatinine levels and NADPH oxidase and GPx
Serum urea and creatinine levels were investigated by colorimetric method using commercial Quanti Assay kits according to the manufacturer’s instructions (Bio Diagnostics, Egypt).
The levels of NADPH oxidase and GPx were detected in kidney tissues using ELISA kits (Cusabio, Inc., Houston, Texas, USA) according to the manufacturer’s instructions.
Histological analysis
Haematoxylin and eosin
The kidney tissues of rats were fixed in 10% buffered formalin solution for 24 h then embedded in a paraffin wax. Tissues were sectioned at thickness of 5-μm and stained with haematoxylin and eosin (H&E).
Acute tubular necrosis (ATN) scoring for estimating the percentage of tubules that displayed cell necrosis and cast formation was done as previously described 24 (0 = none, 1 = <10%, 2 = 10–25%, 3 = 26–45%, 4 = 46–75% and 5 = >75%). Area percentage of congestion was also carried out. All measurements were estimated under magnification 400 inside the standard measuring frame (85,550 µm2) in 10 non-overlapping random fields in a blinded fashion using the Leica Imaging System (Cambridge, England). Images were captured live on the screen under the light microscope (Olympus Bx-40, Olympus Optical Co. Ltd, Japan), then digitalized using ‘Lecia Qwin 500C’.
Electron microscopic examination
Kidney specimens were immersed in 2.5% glutaraldehyde (pH 7.4). Semithin sections were prepared at the 0.5-µm thickness and stained with 1% toluidine blue in 1% borax. Ultrathin sections were double stained with 4% uranyl acetate and 0.1% lead citrate, examined and photographed by JEOL JEM 1010 transmission electron microscope (Japan).
Immunohistochemical analysis
The deparaffinized sections on slides were used to detect malondialdehyde (MDA), inducible nitric oxide (iNOS), 8-hydroxy 2 deoxyguanine (8-OHDG) and VDR immunohistochemical expression using rabbit polyclonal antibodies (Aboot).
The area and optical density of immunohistochemically positive structures were estimated via the image analyser software program ‘Lecia Qwin 500C’ which is a Leica’s windows-based image analysis tool kit (Leica Imaging System Ltd) fitted to an IBM compatible personal computer (New York, USA) with a colour monitor attached to a Panasonic GP 210 video colour camera (Tokyo, Japan). The Leica Imaging System was first automatically calibrated to convert the measurement units (pixels) produced by the image analyser program into actual micrometre units. Using the measuring field menu, the parameters of interest (the area, area percentage and optical density) were selected. In each chosen field, the tissue was enclosed inside the standard measuring frame measuring (85,550 µm2) and then the brown stained immunohistochemically reactive structures were masked by a blue binary colour to be measured.
Quantitative real time-PCR (qRT-PCR) examination
Total RNA was isolated from kidney tissue homogenate using Qiagen tissue extraction kit (Qiagen) according to the manufacturer’s instructions. The purity and the concentration of RNA were confirmed using spectrophotometry at 260/280 nm ratio (dual wave length Beckman, Spectrophotometer, USA). One microgram of RNA was utilized for cDNA production for reverse transcription-PCR (RT-PCR) using the Super Script III First-Strand Synthesis System as described in the manufacturer’s protocol (Fermentas, Waltham, Massachusetts, USA). Real-time RT-PCR reactions were carried out using TaqMan® gene expression assays for nuclear factor 2 (Nrf2), nuclear factor kappa beta (NF-κB) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Applied Biosystems). The primer sequences used were as follows: for Nrf2: forward primer 5′-CAC ATT CCC AAA CAA GAT GC-3′, reverse primer 5′-TCT TTT TCC AGC GAG GAG AT-3′; for NF-κB: forward primer 5′-CATTGAGGTGTATTTCACGG-3′, reverse primer: 5′-GGCAAGTGGCCATTGTGTTC-3′; for GADPH: forward primer 5′-AGG TCG GAG TCA ACG GAT TTG GT-3′, reverse primer: 5′-GTT TGC TCT AGA ATG AAC GGT GGA AGG-3′.
In brief, real-time RT-PCR was performed in a 25-μl reaction volume consisting of 2X SYBR Green PCR Master Mix, 900 nM of each primer and 2μl of cDNA. Thermal cycling conditions were used, including a pre-amplification step of 95°C for 10 min, followed by amplification for 40 cycles of 95°C for15 s, 60°C for 1 min, and 72°C for1 min. Relative expression of the studied genes mRNA was calculated using the comparative threshold cycle method. All values were measured as the ratio of the specific genes to GAPDH which was used as an internal control gene and reported as fold change over background levels determined in the different groups where the relative value for the control group was established as one.
Western blot analysis
The methodology has been described previously. 25 The antibodies against NF kB p65 (NF p65) (Santa Cruz Biotechnology, Inc.) were used. The levels of NF p65 protein were detected against the house-keeping gene β-actin.
Statistical analysis
Data were summarized using mean and standard error of mean. One-way analysis of variance was used to analyse statistical differences followed by Tukey’s post hoc test for multiple comparisons. Results were expressed as mean ± SEM.
Results
Effect of vitamin D on renal functions
Table 1 shows a significant increase in serum urea and creatinine levels in GM group (79.98 ± 9.78 and 2.60 ± 0.329 mg/dl), respectively, when compared to control group (22.51 ± 1.78 and 0.89 ± 0.041;
Serum urea and creatinine among different groups.

a
b
Effect of vitamin D on NADPH oxidase and GPx activities
As shown in the Table 2, NADPH oxidase activity was significantly increased in GM group (687.8 ± 23.3) when compared to control rats (105.2 ± 5.6;
NADPH oxidase and GPx level among different groups.

NADPH: Nicotinamide adenine dinucleoide phosphate oxidase; GPx: glutathione peroxidase.
a
b
Histological results
Examination of H&E-stained sections
Kidneys of GM-treated rats displayed pathological glomerular, tubular and vascular abnormalities (Figure 1). The glomeruli appeared lobulated and were surrounded by widened urinary spaces. The renal tubules showed characteristic features of ATN which were displayed in the form of tubular vacuolation, rarefaction and intraluminal exfoliation of the tubular epithelial cells. The vascular changes were displayed in the form of marked thickening of the renal arterioles and obvious dilatation and congestion of renal blood vessels which were frequently surrounded by perivascular exudate. Preservation of glomerular and tubular architectural patterns with prevention of most of these deleterious changes was noticed with vitamin D administration. This was further elucidated by statistically significant improvement in vascular congestion and tubular necrosis scoring (
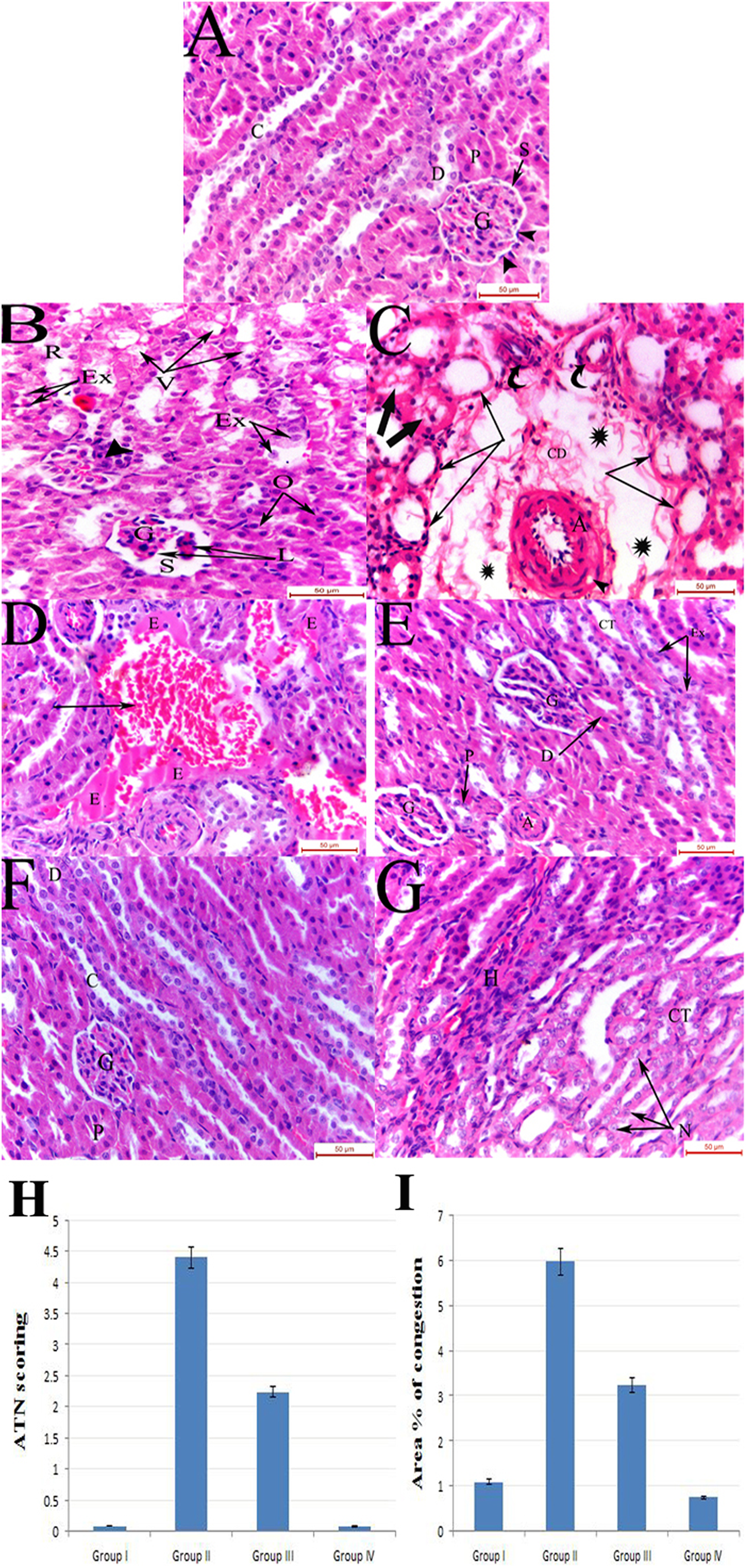
(a) Group I showing normal renal corpuscle composed of a normal glomerulus (G) formed of capillary tufts and surrounded by Bowman’s space (S). The parietal layer of Bowman’s capsule is lined by simple squamous epithelium (arrow heads), PCT (P), DCT (D) and collecting ducts (C). (b) Group II displaying glomerulus (G) with lobulated appearance (L) and widening of the urinary space (S). Renal tubules are seen exhibiting vacuolation (V), rarefaction (R) of their epithelial lining, others displaying obliteration (O) of their lumen and intraluminal exfoliation (Ex) of the tubular cells. (c) Group II exhibiting markedly thickened arteriole with intramuscular vacuolation (arrow head) in its wall. The arteriole is seen surrounded by wide spaces (asterisks) containing loose areolar connective tissue and cellular debris (CD). Dilated tubules lined by flattened epithelium (arrows) are observed with some tubules showing intraluminal casts (thick arrows). Normal sized arterioles are also observed (curved arrows). (d) Group II showing markedly dilated congested blood vessel (arrow) with irregular outline surrounded by areas of exudate (E). (e) Group III showing restoration of the normal architectural pattern of the PCT (P), DCT (D) and CT except for intraluminal epithelial exfoliation (Ex) in some CT. Lobulated glomeruli (G) are also observed. (f) Group IV showing normal glomerulus (G), PCT (P) lined by pyramidal cells with a narrow lumen, DCT (D) lined by cubical cells with a wider lumen and collecting ducts (C). (g) Group IV renal medulla showing normal Loop of Henle (H) and CT lined with simple cuboidal epithelium and exhibiting vesicular rounded nuclei (N) (H&E: ×400). CT: collecting tubules; H&E: haematoxylin and eosin. DCT: Distal convoluted tubules; PCT: Proximal convoluted tubules.
Examination of uranyl acetate and lead citrate-stained ultrathin sections
Ultrastructural examination of the proximal tubular cells of the gentamycin-treated group showed marked degenerative changes which were represented as cytoplasmic vacuolation, mitochondrial dissolution and margination of the nuclear chromatin (Figures 2 and 3). The vitamin D-treated group, on the other hand, displayed very few cytoplasmic vacuoles, preservation of mitochondrial cristae, euchromatic nuclei as well as intact microvilli (Figure 2).

An electron micrograph of PCT showed disturbed structure in GM group when compared to control one, while pretreatment with vitamin D prevented this disturbance. PCT: Proximal convoluted tubules; GM: gentamicin.
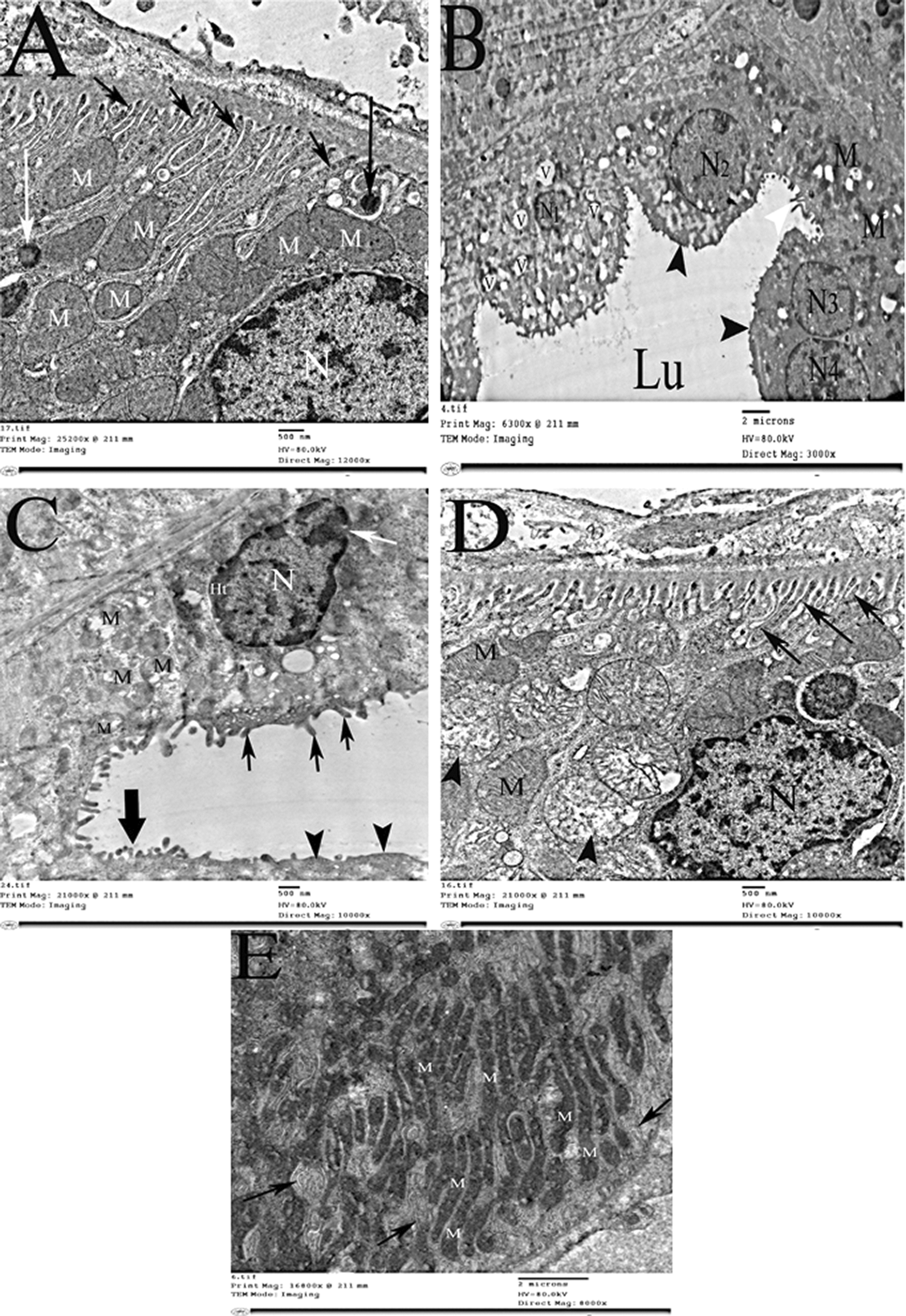
Electron photomicrograph of the distal convoluted tubule showed disturbed structure in GM group when compared to control one, while pretreatment with vitamin D prevented this disturbance. GM: gentamicin.
Cells lining the distal tubules of GM-treated rats showed swelling, vacuolation and loss of their apical microvilli. Their mitochondria displayed loss of the regular architectural pattern and the nuclei showed marginated heterochromatin and discontinuity of their nuclear envelope. Vitamin D markedly ameliorated these ultrastructural abnormalities where the distal tubular cells appeared healthy with numerous basal infoldings and intact mitochondria.
Immunohistochemical results
Administration of vitamin D caused highly statistically significant reduction of iNOS (Figure 4), MDA (Figure 5) and 8-OHDG (Figure 6) immunohistochemical expression when compared to control group (
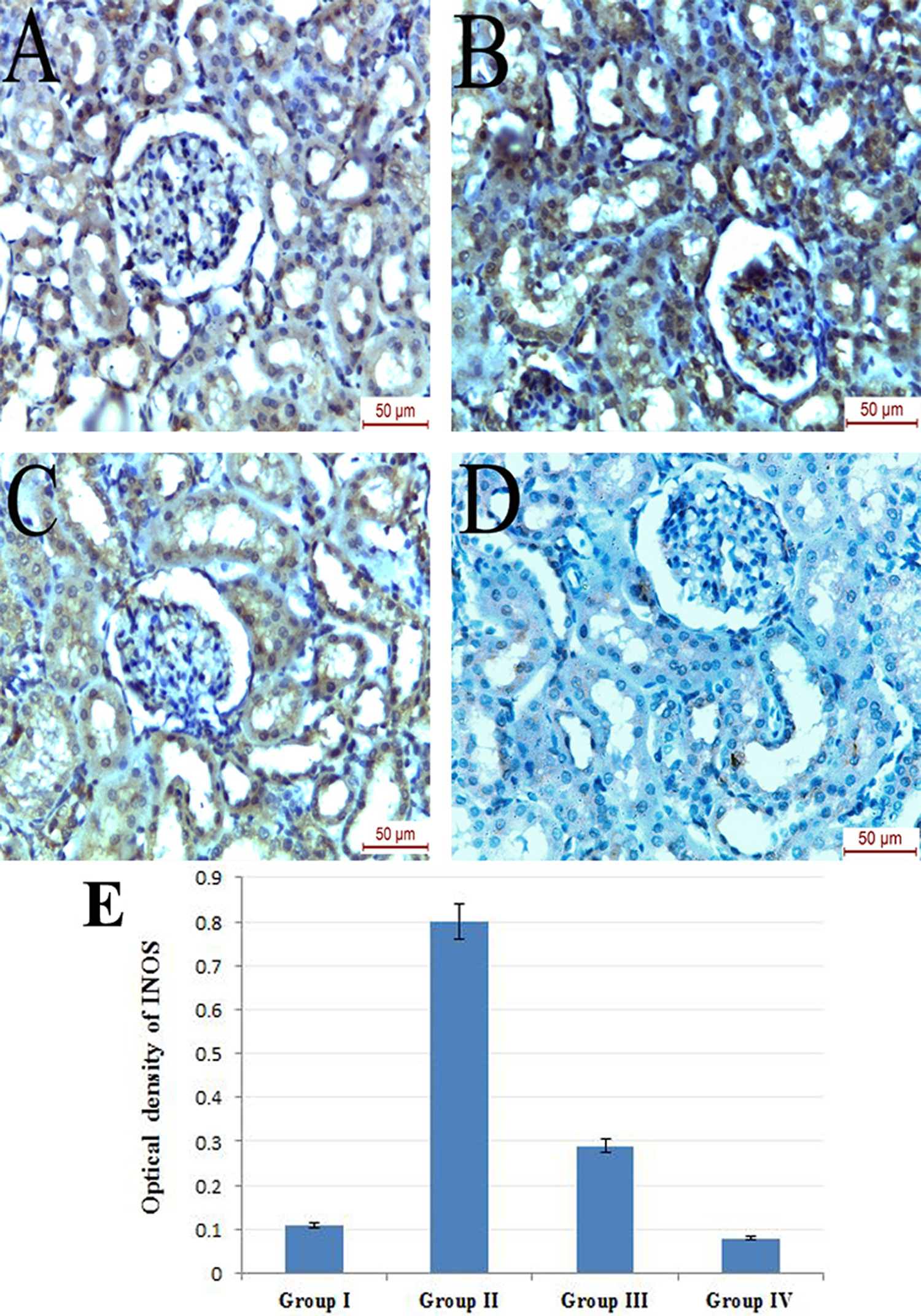
Photomicrograph of iNOS immunohistochemically stained kidney sections of (a) group I showing mild immunoreactivity, (b) group II displaying strong positive immunoreactivity with high cytoplasmic expression within the renal tubules and the glomeruli, (c) group III showing mild immunoreactivity and (d) group IV showing negative iNOS immunoreactivity. (e) Mean optical density of iNOS (group I vs. II,

Photomicrograph of MDA immunohistochemically stained kidney sections of (a) group I showing very mild immunoreactivity, (b) group II displaying strong positive MDA immunoreactivity within the renal tubules and the glomeruli denoting marked deposition of lipid peroxidation products. (c) group III showing moderate immunoreactivity and (d) group IV showing very mild MDA immunoreactivity. (e) Mean area percent of MDA immunoexpression (

Photomicrograph of 8-OHDG immunohistochemically stained kidney sections of (a) group I showing mild immunoreactivity, (b) group II displaying strong positive 8-OHDG immunoreactivity within the renal tubules and the glomeruli indicating marked oxidative DNA damage, (c) group III showing moderate immunoreactivity and (d) group IV showing very mild 8-OHDG immunoreactivity. (e) Mean area percent of 8-OHDG immunoexpression (
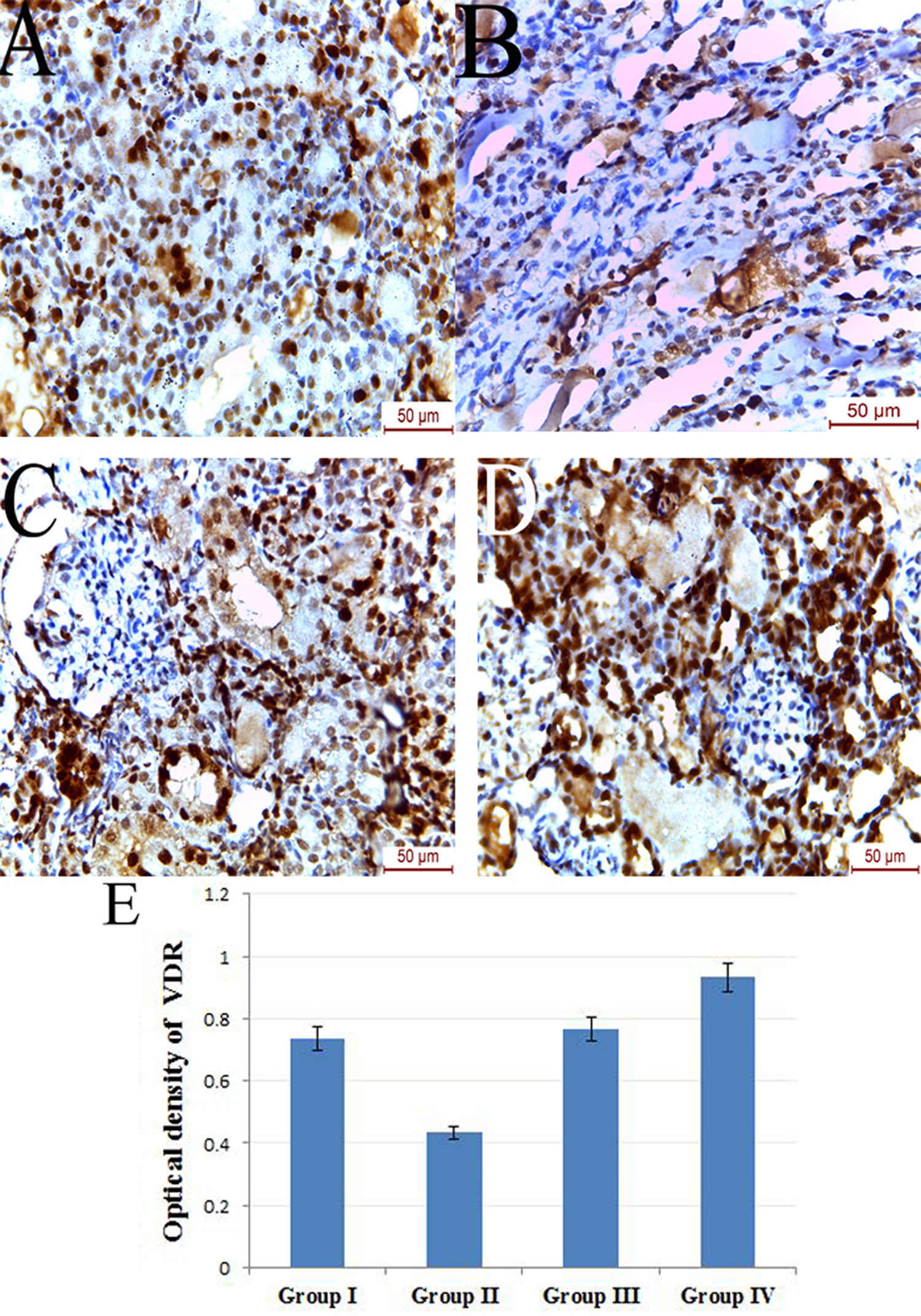
Photomicrograph of VDR immunohistochemically stained kidney sections of (a) group I showing moderate immunoreactivity, (b) group II displaying reduction in immunoreactivity, (c) group III showing restoration of immunoreactivity and (d) group IV showing strong positive immunoreactivity with high expression within the renal tubules and the glomeruli. (e) Mean optical density of VDR (group I vs. II,
RT-PCR results
Expression of NF-κB was significantly increased in GM group (11.8 ± 1.1) relative to control group(1.01 ± 0.0068;
Results of NF-κB and Nrf2 expression among different groups.

NF-κB: nuclear factor kappa beta; Nrf2: nuclear factor 2.
a
b
c
d
Western blot result
NF p65 protein expression was found to be upregulated under the effect of GM (3.58 ± 0.07) relative to control rats (1.67 ± 0.046;

Western blot analysis, nuclear NF p65 expression in GM group showed high significant increase relative to control group (
Discussion
GM is a broad-spectrum aminoglycoside antibiotic whose principal drawback is nephrotoxicity. So, GM-induced nephrotoxicity is a good experimental model of drug-induced renal injury. The purpose of this study is to evaluate the protective effect of vitamin D against the experimentally induced renal toxicity with GM in rats.
Concordant with previous literatures, 26,27 the current study demonstrated several histological and ultrastructural changes along with significant deterioration of renal functions upon GM administration. On the other hand, vitamin D prevented most of these functional and histological disturbances. The protective effect of vitamin D against several forms of renal injury and nephrotoxicity has been previously reported. 28 –30
However, Hur et al. reported that vitamin D could not exert a proper protective effect on gentamycin-induced nephrotoxicity. This could be due to the very small dose of vitamin D (0.5 µg) used in that study. 31 Another possible explanation to those contradictory results could be the fact that the authors used vitamin D concomitant with GM while in our study we used it as prophylaxis providing it as a protective agent for prevention of nephrotoxicity.
On the other hand, some studies have reported a nephrotoxic effect of vitamin D. Wang et al. documented that vitamin D caused nephrotoxicity in refractory rickets patients. 32 This finding may be considered as a special state for refractory rickets due to metabolic disturbance results in the development of rickets despite normal vitamin D concentration, while another study recorded the nephrotoxic effect of vitamin D without any explanation for the molecular mechanism of this toxicity. 33
Many studies have proved the important role of vitamin D as a potent antioxidant. 34 In our study, as documented by previous studies, 26 GM-treated rats showed significant increase in NADPH oxidase level and decrease in GPx level when compared to control rats and these findings were avoided by vitamin D. On the other hand, rats received GM showed strong positive immunoreactivity of iNOS and MDA as compared to control group. Administration of vitamin D resulted in formation of mild reaction. It is well-known that MDA and iNOS are biomarkers for excessive ROS and free radicals formation. Activation of iNOS has been reported to aggravate the renal injury in GM-induced nephrotoxicity. 35 Previous studies have also pointed out the role of vitamin D in the regulation of iNOS expression. 36
In the present study, the gentamycin-induced upregulation of 8-OHDG expression was ameliorated by vitamin D administration; 8-OHDG is an important biomarker of DNA damage accompanying oxidative stress consequences 11,37 and it was found that even small dose of vitamin D (800 IU/day) considerably helps to reduce of 8-OHDG. 38 On the other hand, rats received GM showed a significant decrease in immunoreactivity of VDR while administration of vitamin D showed positive immune reactivity for the receptor. The antioxidant effect of vitamin D depends mainly on VDR activity. 39,40 So, decreased VDR level may aggravate the renal damage induced by GM.
In the current study, we confirmed the modulatory effect of vitamin D on the gene expression of NF-κB and Nrf2. Also, vitamin D was found to downregulate NF p65 as observed by western blot analysis. NF-κB is an important biomarker for oxidative stress and inflammation. Previous studies reported that the reno-protective function of vitamin D is related to its regulation of NF-κB expression. 41, 42
An important negative regulator for NF-κB activation is the inhibitor of NF-κB (I κB) 43 which binds to p65 subunit in the cytosol causing blockage of the nuclear translocation of the p65/p50 heterodimer. An activated IκB kinase (IκK) causes phosphorylation of IκB leading to degradation of IκB and consequently upregulation of NF-κB. 44 Vitamin D was found to prevent degradation of IkB with inhibition of nuclear translocation of p65/p50 resulting in inhibition of NF-κB. 44 In addition, VDR knockout or VDR loss was found to be associated with high expression of 8-OHDG and NF-κB. 45,46
Nrf2 has a powerful cytoprotective effect through binding to the antioxidant response element (ARE), responsible for regulation of the expression of many cytoprotective genes concerned with the cellular antioxidant and anti-inflammatory actions. 47 The Nrf2/ARE signalling pathway contributes to regulation of about 200 genes involved in the cellular antioxidant and anti-inflammatory defence systems, 47,48 as catalase, superoxide dismutase, haeme oxygenase-1, GPx-2 and glutathione S-transferase. 49,50 Vitamin D was found to be involved in the regulation of Nrf2 expression. 51 It is also noteworthy that Nrf2 can regulate the genes important for vitamin D expression which acts as a positive feed back loop. 52
Conclusion
In this study, we detected the protective effect of vitamin D against GM-induced acute renal damage as rats received vitamin D showed significant decrease of urea and creatinine levels which is found to be elevated under the effect of GM. The protective effect of vitamin D is through antioxidant effect as vitamin D increased the expression of Nrf2 and inhibited the expression of NF-κB. Vitamin D also prevents DNA damage related to oxidative stress.
Vitamin D has demonstrated protection against GM-induced renal damage. The mechanism of this protection is through the anti-oxidant effect of vitamin D via activation of Nrf2 and inhibition of NF-κB expressions. Vitamin D can also prevent oxidative stress-induced DNA damage through reduction of 8- OHDG expression.
Footnotes
Acknowledgements
The authors would like to thank Dr Waffaa lecturer of pharmacology and toxicology, Faculty of Pharmacy, Beni Suef University, for her help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
